Abstract
A 41-year-old female with a past medical history significant only for right retinal vein occlusion presented with chest pain, cough, and shortness of breath. After being found to have a large right-sided pleural effusion and undergoing a nondiagnostic thoracentesis, a noncontrast chest computed tomography scan revealed multiple diffuse nodules in the right lung with irregular paraspinal pleural thickening. An extensive workup followed, with computed tomography–guided biopsy ultimately revealing the diagnosis. The following report describes the patient presentation, laboratory findings, and extensive clinical investigation, and provides a discussion of the epidemiology, imaging findings, prognosis, and differential diagnoses for the illness in question.
Clinical History and Presentation
A 41-year-old female with a past medical history significant only for right retinal vein occlusion presented with chest pain, cough, and shortness of breath for a duration of approximately 1 month, worsening over 2 weeks. The pain was described as pressure-like, intermittent, nonexertional, nonradiating, and worse with leaning forward. Her symptoms also included intermittent palpitations, poor appetite, decreased exercise tolerance, and an inability to sleep on her right side. The retinal vein thrombosis had occurred 9 years prior in the setting of oral contraceptive use, and led her to use lovenox during her most recent pregnancy in 2014. A thrombophilia workup at that time was negative. Originally from Mexico, the patient had moved to the United States 19 years prior. She worked at a restaurant, and did not smoke, drink, or use recreational drugs. She denied any personal or family history of autoimmune disorders. She also denied any family history of cancer, and at the time of presentation was up-to-date on age-appropriate cancer screening.
The patient was a mildly overweight Hispanic female who presented in no acute distress. With the exception of an oxygen saturation of 94%, vitals on admission were unremarkable. The only pertinent physical examination finding was decreased breath sounds in the right middle and right lower lung field.
A chest X-ray revealed opacification of the right middle and lower lung fields (Figure 1). The patient was placed on airborne precautions while 3 sputum samples were obtained in order to rule out tuberculosis infection; all 3 were negative. Follow-up computed tomography (CT) angiography imaging of the chest demonstrated a large right-sided pleural effusion with right-sided compressive atelectasis and mediastinal shift to the left, as well as slightly lobular posterior pleural thickening on the right (Figure 2).
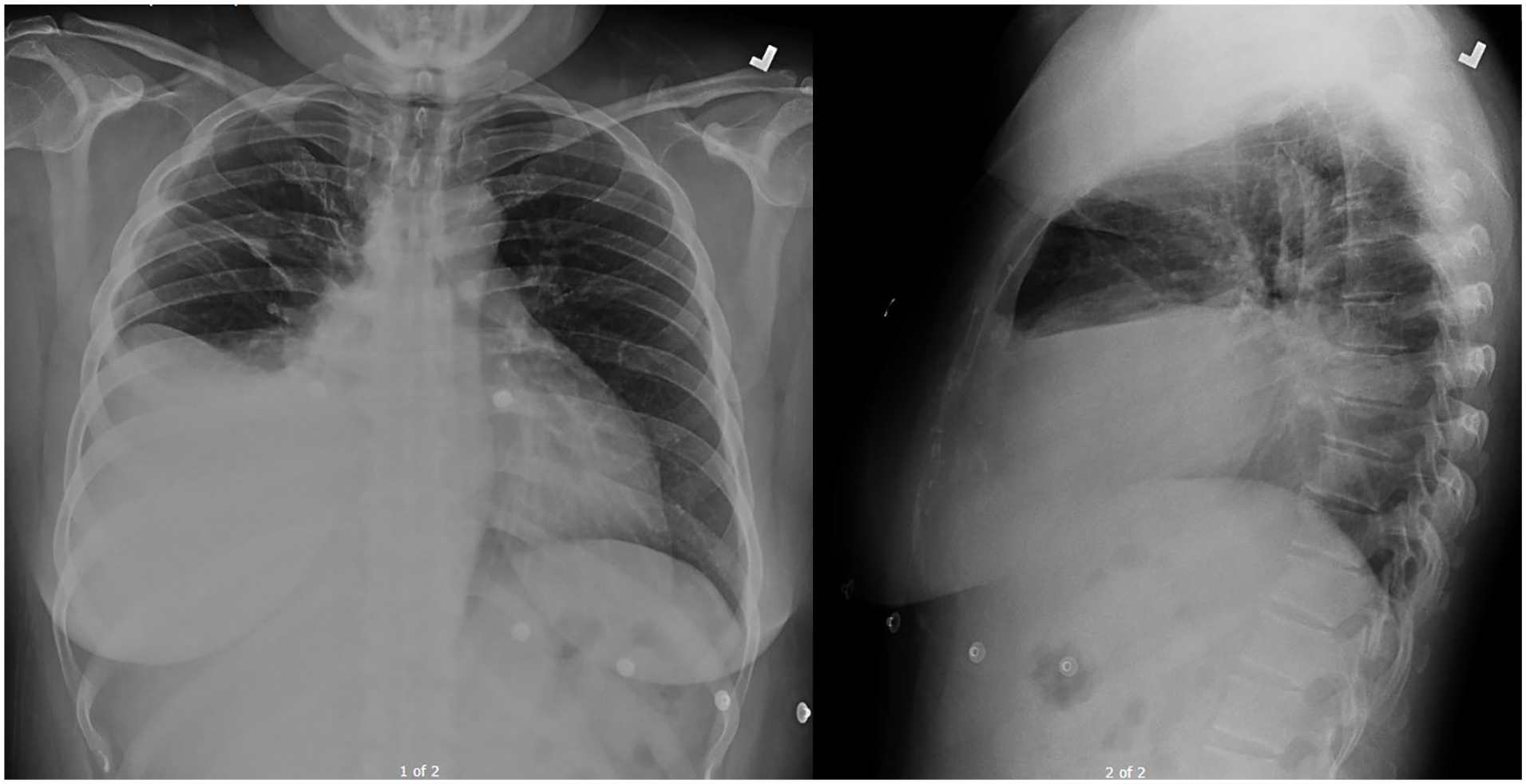
Frontal and lateral chest radiographs demonstrate large right-sided pleural effusion.

Computed tomography angiography of the chest, from superior to inferior, demonstrates a large right pleural effusion with right-sided compressive atelectasis and mediastinal shift to the left. Slightly lobular posterior pleural thickening on the right is also present.
A chest tube was placed, draining approximately 2400 mL of serosanguinous fluid over a 72-hour period. Cell count in the pleural fluid sample revealed 1200 total white blood cells, 95% lymphocytes. Pleural fluid studies revealed a pH of 7.413, creatinine 0.66, glucose 93, amylase 38, lactate dehydrogenase 358, protein 5.0, triglycerides 41, and adenosine deaminase of 4.1 (within normal limits). Pleural fluid culture revealed few polymorphonuclear leukocytes, no organisms, and no fungal growth. No acid-fast bacilli (AFB) were isolated.
A noncontrast chest CT scan after drainage demonstrated resolution of the pleural effusion, and revealed multiple diffuse nodules in the right lung, parenchymal versus pleural-based, with irregular paraspinal pleural thickening (Figure 3).

Computed tomography scan of the chest without contrast, from superior to inferior, demonstrates near resolution of the large pleural effusion following insertion of a pigtail catheter, which is visualized near the right hemidiaphragm. Multiple diffuse nodules, which appear to emerge primarily from the pleura, are now visible throughout the right lung, the largest measuring 2 cm abutting the right hemidiaphragm. Slightly irregular paraspinal pleural thickening in the right lower lobe is also present, along with a small right pneumo-ex-vacuo and a small right pleural effusion.
CT scan of the abdomen and pelvis failed to identify a clear primary; however, it did note a partially exophytic anterior lesion in the lower uterine segment, along with some free fluid and endometrial thickening. Given this finding and a mildly elevated CA-125, a pelvic/transvaginal ultrasound (US) was performed and Gynecology was consulted; however, the US failed to demonstrate any abnormality and Gynecology endorsed low suspicion for gynecological origin of the cancer.
Other tumor markers (CA 19-9, CEA, CA 15-3, and CA 27-29) were negative. Positron emission tomography (PET)-CT demonstrated metabolically active pleural-based masses in the right hemithorax and nodular masses in the right lung, with mediastinal and retrocrural and retrocaval lymphadenopathy concerning for malignancy/metastatic disease. The PET scan also noted a mildly enlarged right tonsil; however, subsequent laryngoscopy showed no evidence of malignancy of the tonsils or head and neck.
Cytological studies of the pleural fluid revealed what were thought to be gland-forming malignant cells negative for Ber-EP4, Napsin-A, p63, and desmin (favoring adenocarcinoma), positive for CK5/6 and negative for CK7, CK20, TTF-1, CEA, calretinin, GATA-3, and mammaglobin, an unusual immunostaining profile that prevented classification of the primary tumor. Tissue biopsy was recommended.
The patient subsequently underwent bronchoscopy with US-guided transtracheal/transbronchial sampling. The biopsy showed fragments of fibrous tissue and fragments of cytologically bland cells positive for calretinin and CK5/6, and focally positive for CK7, while negative for TTF-1, Napsin A, p63, and p53. While these findings were considered consistent with mesothelial origin, further evaluation/characterization was not possible and re-biopsy was recommended.
A CT-guided biopsy of right-sided pleural tissue demonstrated poorly differentiated carcinoma morphology with necrosis and an immunostaining profile consistent with a diagnosis of malignant pleural mesothelioma (MPM; Figure 4).
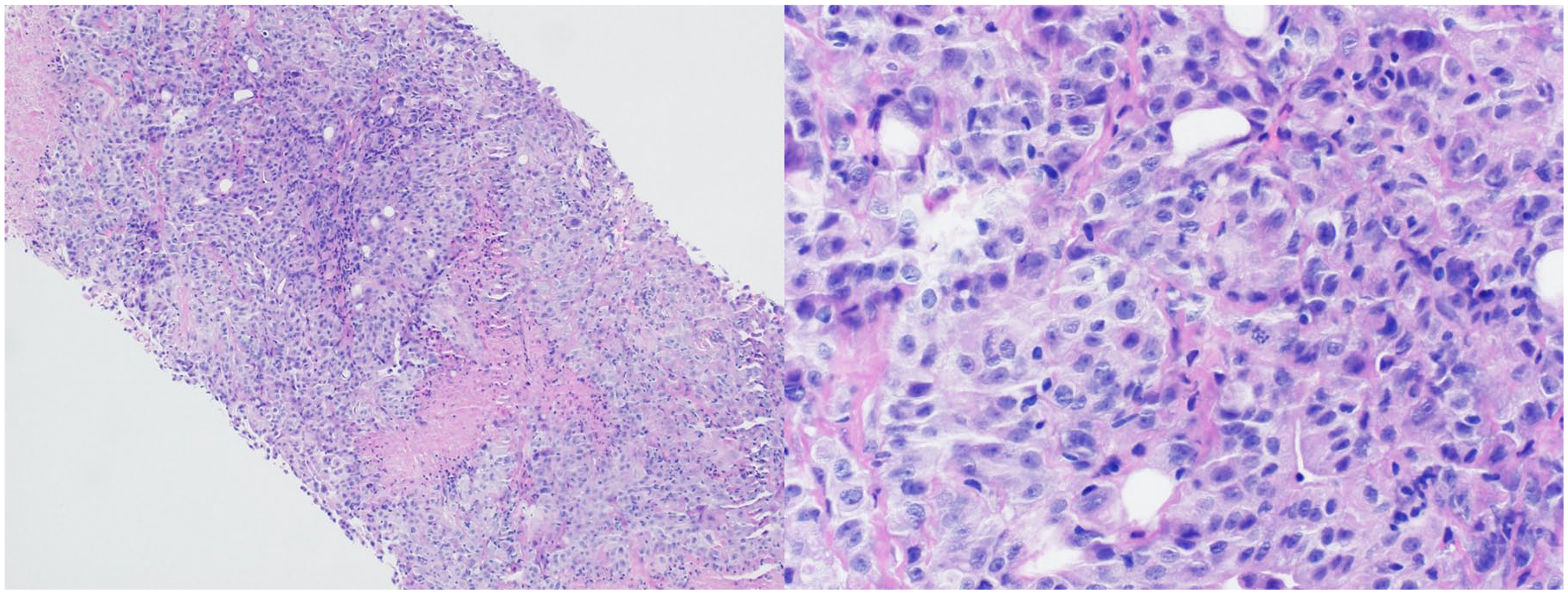
100×(left) and 400× (right) magnifications of right-sided pleural tissue obtained via core needle biopsy, stained with hematoxylin and eosin, reveal a malignant neoplasm—primarily solid growth with poorly differentiated carcinoma morphology—with regions of necrosis.
Discussion
Based on the pathological findings, the patient was diagnosed with stage IV MPM, epithelioid type, with malignant effusion, multiple pleural nodules, and mediastinal and retrocrural lymphadenopathy.
In light of these findings, the patient was questioned further regarding potential risk factors/exposures. She reported that 20 years ago she worked in a sewing factory for about 6 months, but denied any dust/occupational exposures during that time period. Around 18 years ago, she reported that her apartment had construction in 3 of the 5 rooms and she would clean up after the workers were done. She had asked the landlord at that time to be relocated, but the landlord denied her request. A similar situation occurred 2 years ago, but she was again denied relocation.
Although it occurs rarely overall (with an incidence of ~2500 cases annually in the United States), MPM is the most common primary malignancy arising from pleural mesothelial cells. 1 MPM has a median survival time of less than 18 months, or as short as 4 to 8 months if left untreated.1-3 While 2-year survival time is 46% for stage I disease, it drops to 17% for stage IV disease; unfortunately, diagnosis typically occurs when patients have already reached stage III or IV. 2 Some studies suggest that screening for and monitoring certain biomarkers, including mesothelin, osteopontin, and fibulin-3, may allow for earlier detection of MPM and, therefore, improve patient survival. 2
Asbestos exposure is reported in 40% to 80% of patients with MPM, and risk of developing the disease increases as the intensity and duration of exposure increase. 1 While MPM has been classically considered an occupational disease—given that asbestos exposure tends to occur in certain workplace environments—exposure can also occur indirectly for those in close proximity to those with occupational exposure, or it can occur entirely without patients’ knowledge. 4 For example, one study identified exposure to talcum powder as a risk factor in a group of women who developed MPM, with asbestos identified in the talcum powder in several cases. 5
The latency period between asbestos exposure and MPM onset ranges from 20 to 40 years, 6 with patients typically presenting after the age of 50 years, and death occurring at around the age of 70 years. 7 Consistent with historical patterns of occupational exposure, males tend to be affected more frequently (male-to-female ratio of approximately 3-4:1). 7 Common presenting symptoms include non-pleuritic chest wall pain (unilateral or bilateral) and progressive dyspnea, typically secondary to pleural effusion.1,7
While most laboratory tests are relatively nonspecific, elevated serum levels of soluble mesothelin-related protein have been associated with MPM, with 64% sensitivity and 89% specificity for the diagnosis. 1
Imaging plays a critical role in the diagnosis, staging, surveillance, and treatment response of MPM.3,4 Chest radiography is often the first imaging study to be performed and the first to demonstrate abnormalities associated with the disease, including unilateral pleural effusion (~80% of cases), diffuse pleural thickening (~60% of cases), and pleural masses (45% to 60% of cases).1,3 Other potential findings include intrathoracic lymphadenopathy and bony destruction secondary to tumor spread. 3 If encasement of the lung occurs, pulmonary volume loss may result in elevation of the ipsilateral hemidiaphragm, mediastinal shift, and/or narrowing of the intercostal spaces.1,3
As the preferred imaging modality for evaluating MPM, chest CT can shed light on the nature of the primary tumor, the presence and/or extent of local invasion and lymphatic involvement, and spread outside of the thoracic cavity. 1 It is also vital for intervention planning, such as image-guided biopsy. Pleural thickening (82% to 92% of cases), which can range from focal nodular thickening to complete peripheral encasement of the lung, and unilateral pleural effusion (74% to 89% of cases) are the most common features detected.1,3 Calcified pleural plaques (~20% of cases) are representative of asbestos-related disease. 1 CT scan may also reveal invasion of the chest wall, pericardium, mediastinum, or diaphragm, and detect lymph node involvement. Pulmonary metastasis, which may present as nodules, masses, or lymphangitic carcinomatosis (nodular thickening of the interlobular septa), may also be seen. 1
While thoracic magnetic resonance imaging is not routinely used, it is more sensitive and therefore may provide more specific detail than CT in terms of chest wall, mediastinal, and diaphragmatic invasion. 1 While typically adjunctive, PET-CT is also more sensitive than CT in identifying local invasion and better demonstrates both intra- and extrathoracic lymphadenopathy and metastasis. 1
The diagnosis of MPM is confirmed by cytology in only 20% to 30% of cases, and by percutaneous pleural biopsy in 20% to 23% of cases. 6 While thoracentesis is rarely diagnostic, high levels of hyaluronic acid in the pleural fluid are highly suggestive of MPM and, if present, are associated with a good prognosis. 8 Image-guided core and surgical biopsies demonstrate much higher sensitivities of 86% and 94% to -100% respectively. 1
In this case, diagnosis was confirmed by the presence of nodular pleural thickening and masses on imaging, and the results of US-guided transbronchial biopsy, CT-guided pleural biopsy, and immunohistochemistry.
Treatment of MPM depends on the extent of disease and relies on a combination of surgery, chemotherapy, and radiotherapy. 3
Differential Diagnoses
Several differential diagnoses were considered over the course of the clinical investigation, including primary lung cancer, solid pleural metastases, asbestos-related diffuse pleural thickening, reactive mesothelial hyperplasia, thoracic endometriosis, and pulmonary tuberculosis.
Primary lung cancer was less likely in this case given (1) the identification of several small masses rather than a singular large or larger mass that would appear consistent with a primary tumor and (2) that the masses were located primarily along the pleura instead of within the parenchyma. Besides several lymph nodes, no other tumor was identified outside of the lung region, making solid tumor metastases less likely.
The sparse nodular appearance of the lesions as opposed to the presence of pleural plaques or diffuse pleural thickening, combined with the lack of obvious history of exposure to asbestos, in this patient made asbestos-related diffuse pleural thickening less likely. 9 Furthermore, asbestosis is typically associated with parenchymal changes reflecting interstitial lung disease, which were not seen in this patient.
Epithelioid MPM, the most common histological subtype, can be particularly difficult to differentiate from reactive mesothelial hyperplasia, but MPM can be differentiated from other pleural lesions, including mesothelial hyperplasia, based on certain biomarkers, most notably BRCA1 associated protein 1 and p16 deletion. 10 Although both features have high specificity, their sensitivity tends to be limited even when used together. 10 Other biomarkers, like mesothelin and fibulin-3, may also help differentiate MPM from other malignant and benign causes of pleural effusion. 2
While thoracic endometriosis is considered uncommon, the thorax is the most frequent location for extra-abdominopelvic disease. While this female patient was of reproductive age, she had no prior presentations of catamenial pneumothorax or hemothorax, and no history of catamenial chest pain. Indeed, there was no history of endometriosis or its signs or symptoms (ie, cyclic pain with periods), making thoracic endometriosis unlikely. 11
Pulmonary tuberculosis was also considered given its ability to present in a variety of patterns and that the patient had lived in an endemic region, it was ultimately ruled out with 3 sputum samples negative for AFB, negative pleural fluid adenosine deaminase, pleural fluid negative, or AFB.
Conclusions
MPM is an aggressive malignancy with a wide differential that may be difficult to diagnose, particularly when the diagnosis is unexpected. While many markers and imaging characteristics may suggest the diagnosis, biopsy may be required in difficult cases to provide definitive diagnosis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Institutional Review Board approval is not required for case reports at the institution where this patient was treated.
Informed Consent
Our institution does not require informed consent for individual case reports.
