Abstract
Coccidioidomycosis (CM) is an endemic fungal disease that is prevalent in the Southwestern United States, Mexico, and Central and South America. The majority of cases are asymptomatic. Symptomatic cases primarily present with pneumonic disease, varying in severity from mild to severe. In a minority, extrapulmonary disease is known to occur. These disseminated cases typically manifest as meningitis, osteomyelitis, synovitis, and integumentary. Coccidioidomycosis has been identified in nearly every part of the body, including the soft tissues and cartilaginous tissues of the oropharynx. Disseminated CM to the larynx specifically in the pediatric populations is rarely documented. This is a case of persistent stridor misdiagnosed as viral croup in a 9-month-old male with protracted disease. Herein, diagnosis and treatment of laryngeal mass found to be CM are defined with a salutatory outcome.
Keywords
Introduction
Coccidioides immitis and Coccidioides posadasii exist in the environment as dimorphic fungi and are known to cause coccidioidomycosis (CM). 1 Annually, 150 000 to 300 000 infections occur in the United States due to these species. Both are endemic to the deserts of the Southwestern US, Latin America, and regions of Mexico. 2 Disturbance to the arid soil from heavy rains, dust storms, earthquakes or more commonly, due to farming, may lead to infectious fungi, known as arthroconidia, becoming airborne. Inhalation of arthroconidia is the typical acquisition of CM. Direct inoculation is the other possibility of CM although is uncommon. 3 Coccidioidomycosis is considered a noncommunicable infectious disease.1,4
Those with CM are asymptomatic 60% of the time. Of the 40% who are symptomatic, CM most commonly presents as a primary pulmonary disease.1,4,5 One percent progress to extrapulmonary disease via lymphohematogenous spread. Dissemination is most likely to occur in immunocompromised individuals and is associated with higher mortality rates (Figure 1).1,2,4
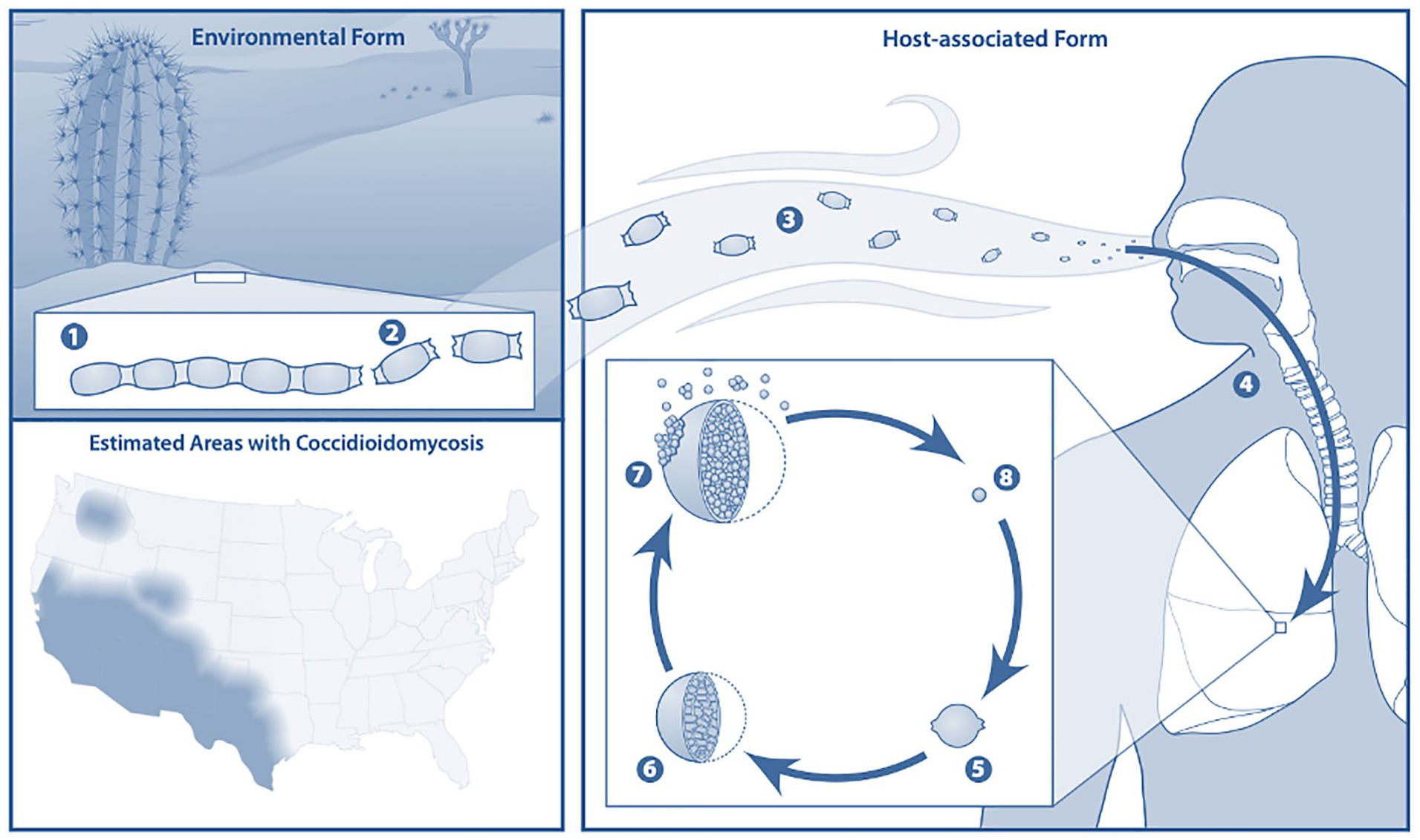
Coccidioidomycosis, a dimorphic fungi. Endemic regions of coccidioidomycosis in the United States. 6
Dissemination to the bones, joints, and skin are more common sites of disseminated disease.1,4 Laryngeal CM is considered rare.7-9 Disseminated subglottic CM may present as laryngotracheobronchitis (croup), with symptoms of upper respiratory illness and/or lower respiratory illness. 7
Clinical manifestations of laryngeal CM largely depend on the location and amount of tissue destruction.
1
Croup spectrum illness includes any pathologic involvement of the larynx, trachea, and bronchi, including laryngotracheitis, laryngotracheobronchitis, and laryngotracheobronchopneumonitis.7,10 The severity is defined based on the Westley croup score (score range of 0-17): Mild croup: Wesley croup score 0-2; intermittent barking-like cough, mild to absent suprasternal and intercostal retractions.
11
Moderate croup: Westley croup score 3-5; frequent barking-like cough, audible stridor at rest and with exertion, suprasternal and sternal retractions at rest, minimal distress and/or agitation.
11
Severe croup: Westley croup score 6-11; frequent barking-like cough, significant inspiratory stridor with intermittent expiratory stridor, marked sternal retractions, significant distress and agitation.
11
Impending respiratory failure: Westley croup score greater than 11; frequent barking-like cough, audible stridor at rest, sternal wall retractions, lethargy, drowsy, often dusky complexion on room air, paradoxical breathing.
11
Viral, bacterial, or fungal infection of the larynx, trachea, or bronchi may lead to the onset of croup. Etiology cannot be clinically deciphered.10,12 Viral croup is responsible for >75% of airway obstructions in children from ages 6 months to 6 years. Adults may also have viral disease, although it is less common.10,13 Paramyxoviridae family is the most likely etiology in children. Fungal croup and bacterial croup are considered less common, especially in pediatrics.14,15 Reported cases of laryngeal CM are predominantly seen in adults with less than 10 reported cases in the pediatric population. Bacterial croup in the immunoprophylaxed is nearly eradicated since diphtheria and haemophilus influenza type b (Hib) immunizations.13,16 For nonimmunized pediatric patients with rapidly decompensating croup, diphtheria and HiB are viable threats. Additional considerations for bacterial causes include Staphylococcus aureus, Streptococcus pneumonia, and Moraxella catarrhalis. Immunoprophylaxis against parainfluenza or fungal croup is not indicated.12-14,16 Epiglottitis, foreign object, retropharyngeal abscess, and bacterial tracheitis should also be ruled out. 15
Residing in or recent travel to endemic regions with C immitis and C posadasii may be the clinical clue for subglottic CM diagnosis.1,5 Protracted disease beyond 7 days should also raise the level of suspicion, as most viral cases resolve within 48 hours to 7 days. Croup due to CM may demonstrate unique progressive features that wax and wane over many months to years.13,17 Time course, severity, and refractory cases are relevant features that guide diagnosis and management decisions. Rarely, there is complete resolution of disseminated CM without medical intervention for CM. 1
Diagnosis of laryngeal CM is based on epidemiological, clinical, laboratory, radiological, and histopathological findings. Gold standard is to identify the coccidioides spherules with endosporulation from biopsy or identification of CM after fungal growth from tissue sample. 1 A positive immunodiffusion antibody IgM and IgG for CM with or without elevated complement fixation (CF) in a reliable reference laboratory are also paramount. This is usually performed after negative work up for viral etiology, such as sputum polymerase chain reaction (PCR) of viral genome, viral antigen direct assay, and serum human parainfluenza virus (HPIV)-specific IgG antibodies.1,2 Coccidioidal serologies (IgM, IgG) are often delayed and repeat testing may be appropriate. Other characteristic features on blood analyses may demonstrate an elevated absolute eosinophil count (≥350 cells/µL).1,4 Elevated inflammatory markers such as C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR) have been seen but they are not specific.1,4
Chest imaging in CM with recent or concomitant pneumonic disease may reveal air space disease, consolidations, nodules, pleural effusion, cavity, or miliary pattern. 1 Hilar adenopathy and mediastinal widening may also occur. Cavitation’s and miliary pattern are representative of more severe disease. 1 An underlying immunocompromised state may be suspected. 1
Guidance for management of laryngeal CM in the pediatric population is based on published data for adults. This is because little studies are performed, and data are limited for the pediatric population. The guidelines from American Academy of Pediatrics (AAP), Infectious Diseases Society of America (IDSA), and American Academy of Family Physicians (AAFP) remain nonspecific.1,13 For both adults and children at Valley Fever Institute, we recommend antifungal therapy over observation. The goal of therapy is to reduce illness duration and severity of complications. The duration of therapy is recommended for a minimum of 1 year considering this as a form of soft tissue dissemination. 1 This is to achieve resolution of symptoms with clinical and serological response. Extension of treatment beyond 1 year might be needed for severe disseminated disease, concomitant dissemination to other tissues such as osseous, and persistent clinical manifestations and/or sero-reactivation. 1
Follow-up visits every 1 to 3 months up to 2 years is recommended after antifungal therapy is discontinued. 1 Repeat serologies and imaging is typical in posttreatment monitoring to correlate clinical improvement and/or deficits and to document radiographic resolution of disease. 1 In general, for disseminated CM including laryngeal not involving the central nervous system (CNS), oral fluconazole or itraconazole is the preferred initial therapy. Liposomal amphotericin B (L-AmB) might be needed for progressive and/or fulminant disease. Other antifungal therapies such as posaconazole or isavuconazole have been successfully used, but limited information is published within the pediatric population. In adults, they are typically considered for salvage therapies.1,18 Consultation to an infectious disease specialist and otolaryngology is highly suggested at the time of diagnosis with disseminated laryngeal CM. 19 Surgical intervention may be required for refractory cases.1,3,9,20-22
Methods
Approval was obtained from the Institutional Review Board of Kern Medical. A retrospective review of the patient’s record was performed. A literature search was conducted on PubMed, Research Gate, Google Scholar, Centers for Disease Control and Prevention, IDSA’s Clinical Infectious Diseases Journal database, the AAFP’s American Family Physician, and The American Academy of Pediatrics’ Pediatrics Journal database. The following search terms were applied: croup, subglottic mass, CM, persistent stridor, disseminated CM, laryngeal CM, laryngeal mass, and pediatric croup.
Case Presentation
A 9-month-old Latino boy was diagnosed with upper respiratory infection at another institution. Three months after his initial diagnosis, he underwent several visits at urgent cares and emergency departments (ED). He was initiated on inhaled steroids and received systemic steroids and was prescribed a course of antibiotics. He presented to our institution’s ED for 3 months of progressive waxing and waning of a nonproductive barky cough, inspiratory stridor on exertion and fever.
On physical examination, his vital signs showed temperature of 38.0°C, heart rate of 76 beats per minute, respiratory rate of 26 breaths per minute, and blood pressure of 100/90 mmHg. His oxygen saturation was 87% on room air. He was in acute distress due to difficulty breathing. The physical examination was positive for barking cough, inspiratory stridor, costovertebral retractions, and decreased breath sounds on the right lower lung field. Nasal cannula at 2 L of oxygen improved oxygen saturation to above 92%. Wesley croup score was 5, moderate severity. 11
Initial laboratory examination revealed an absolute eosinophil count of 375 cells/µL without lymphocytopenia, anemia, or thrombocytopenia. Chemistry was unremarkable. Inflammatory markers ESR 30 mm/h and CRP 8 mg/L (≤0.3 mg/L) were elevated. 1
Pneumonia was initially diagnosed by clinical findings. Anterior-posterior (AP) chest X-ray revealed right lower lobe (RLL) consolidation with mediastinal widening (Figure 2).
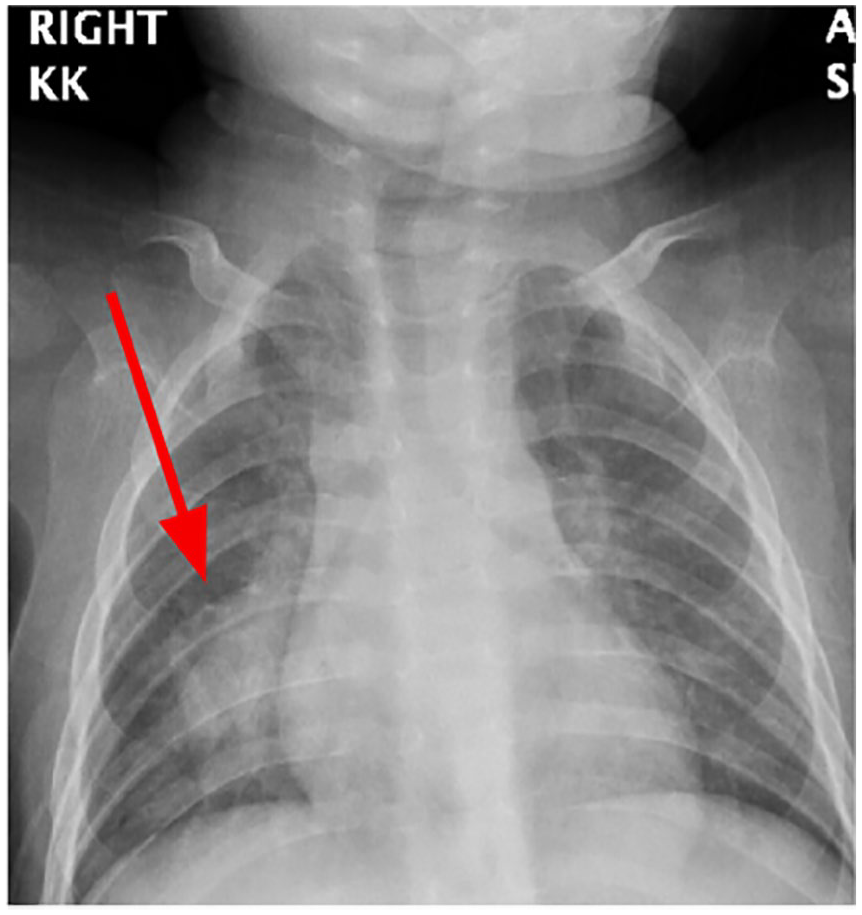
Chest X-ray anterior-posterior view with right lower lobe consolidation.
Broad spectrum antibiotics were administered. He received azithromycin, intravenous (IV) fluids, dexamethasone, and nebulized racemic epinephrine breathing treatments. After a 5-day hospital course, his respiratory status improved, and he was sent home on room air to follow up with his pediatrician. After his discharge, his coccidioidal serology resulted as immunodiffusion IgM weakly reactive, IgG nonreactive with CM CF titers of <1:2.
For the next 4 weeks, the patient had waxing and waning of cough and inspiratory stridor with exertion and fever. He returned to the ED with severe acute respiratory distress and inspiratory stridor on exertion. Wesley croup score was 5. He initially had slight improvement with nebulized epinephrine treatments, but after 3 hours, he progressed to stridor at rest, which was refractory to treatments. Wesley croup score increased to >6.
Radiographic imaging with a repeat chest X-ray revealed subglottic narrowing with worsening of his perihilar and peritracheal lymphadenopathy (Figure 3).

Chest X-ray anterior-posterior view with subglottic narrowing, mediastinal widening, and lymphadenopathy.
Given findings of subglottic narrowing, the patient was transferred to a tertiary pediatric center. At the tertiary facility, magnetic resonance imaging (MRI) revealed a laryngeal mass encompassing most of his true vocal cords (TVC). Diagnostic laryngoscopy redemonstrated gross mass findings with partial airway encroachment (Figure 4).

Laryngoscopy demonstrating a laryngeal mass encompassing true vocal cord.
Tissue biopsy of the laryngeal mass showed coccidioidal spherules with endosporulation and grew C immitis later. Findings were pathognomonic for CM (Figure 5).

Histologic tissue evaluation revealed a coccidioidal spherule with endosporulation.
Repeat serologies revealed coccidioidal immunodiffusion IgM (reactive) and IgG (reactive) and CF titers of 1:32. His condition progressed to a Wesley croup score of 11. Tracheostomy was required. A peripheral intravenous central catheter (PICC) line was placed. Antifungal induction therapy with L-AmB was initiated via PICC line. The decision for surgical intervention of the laryngeal mass was originally considered but deferred to allow time for clinical response to therapy. MRI of the head, neck, chest, and abdomen ruled out additional disseminated foci. Whole body bone scan was also unremarkable. Lumbar puncture was completed which confirmed absence of CNS CM. Treatment was deescalated after 1 month of L-AmB to oral fluconazole 120 mg once daily. Repeat laryngoscopy showed adequate TVC movement with complete resolution of the laryngeal mass. The patient was discharged home with a tracheostomy. He was monitored closely outpatient (Figure 6).
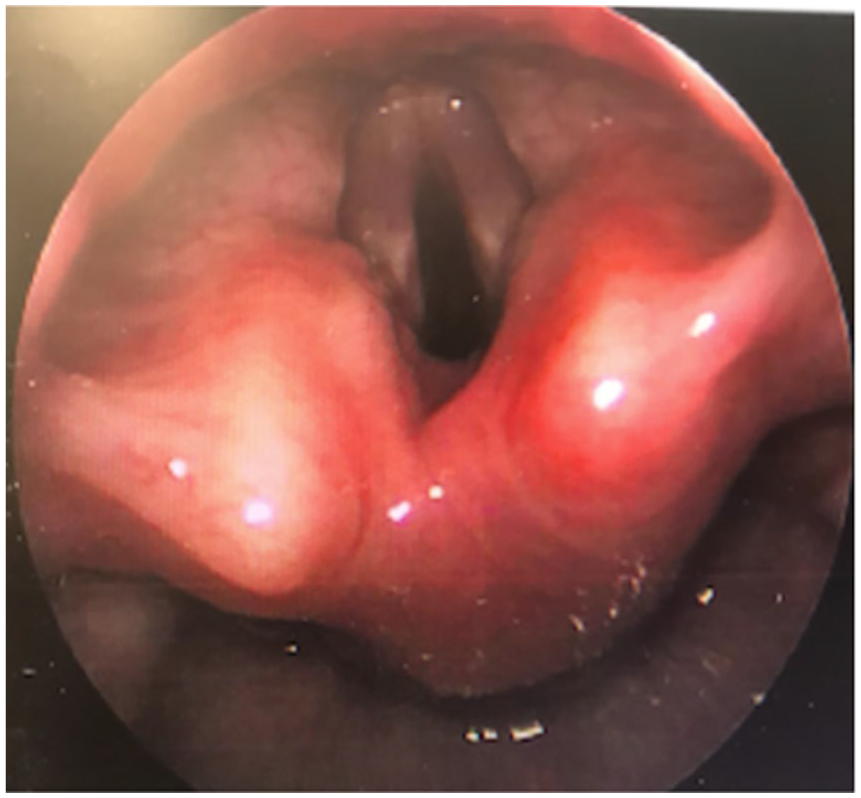
Repeat laryngoscopy status/post 1 month of liposomal amphotericin B, revealing complete resolution of mass.
Serologies revealed immunodiffusion IgM (weakly reactive), IgG (reactive), and CF titers downtrended from 1:32 to 1:16. Decannulation of tracheostomy was performed after 2 months of antifungal therapy. The patient recovered to his baseline of normal swallowing, regular speech, and normal breathing without stridor at rest and with exertion. All developmental milestones were met at follow-up visits except for mild speech delays due to tracheostomy. He was referred to speech therapy.
His antifungal therapy was continued for total of 13 months with down trending coccidioidal immunodiffusion IgM (nonreactive), IgG (weakly reactive), and CF titers (1:8) with completer resolution of symptoms. The decision was made to discontinue antifungal therapy with continued close monitoring every 1 to 3 months for the next 2 years (Table 1).
Summary of Clinical Course.

Outcome: Patient returned to baseline: No swallowing difficulties, speech impairment, stridor, or respiratory distress. Meeting all developmental milestones except for speech. Referral for speech therapy was required.
Abbreviations: HPIVs, human parainfluenza viruses; CM, coccidioidomycosis; CXR, chest X-ray; RLL PNA, right lower lobe pneumonia; MRI, magnetic resonance imaging; TVC, true vocal cords; WR, weakly reactive; R, reactive; NR, nonreactive; CF, coccidioidal compliment fixation titers; ICS, inhaled corticosteroids; L-AmB, liposomal amphotericin B.
Discussion
Disseminated CM to the subglottic structure and larynx is rarely reported in pediatric population. The true statistical incidence is unknown and due to lack of clinical trials, the management is a case-by-case basis. 2 We found 10 publications prior to this report.
Race and ethnic predilection identified in these cases were synonymous with adult cases with higher incidence in males, Hispanic, African American, and Filipinos.1,17 L-AmB was the most common initial therapy. Complete resolution was achieved in most of these cases after antifungal therapy. Some required surgical excision. 1 For severe swelling around the vocal cords, corticosteroids were beneficial for improving respiratory symptoms according to these case reviews. It was noticed that there was no consensus among the treatment course and dosing among these case series indicating a lack of data to support unified recommendations (Table 2).
Abbreviations: Unk, Unknown; AmB, Amphotericin B: Used prior to 1990; L-AmB, Liposomal Amphotericin B: Approved formulation after 1990; s/p, Status/post; Flu, Fluconazole; Sx, Surgery; DOT, Duration of Therapy.
Conclusions
Disseminated Laryngeal CM may present with nonspecific symptoms and should be in the differential diagnosis of unrelenting croup in endemic regions. 17 Laryngoscopy and biopsy are key to make the definitive diagnosis. Due to lack of clinical trials and definitive guidelines particularly in pediatric cases, consultation with experts in the field is necessary. Antifungal therapy is always indicated and should not be delayed. Duration of therapy is prolonged and is based on clinical response. 1
Footnotes
Acknowledgements
This case has been presented at the American Federation of Medical Research (AFMR)’s Western Conference (2019), University of California Los Angeles (UCLA) Solomon Scholar (2019), and the Coccidioidomycosis Study Group (CSG; 2019).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval to report this case was obtained from the Kern Medical Institutional Review Board; approval ID: 18050.
Informed Consent
Informed consent was obtained from the patient’s mother who granted permission to publish unidentified patient information.

