Abstract
Bladder cancer is the most common urinary tract malignancy. Platinum-based chemotherapy is the first line of treatment in locally advanced or metastatic bladder cancer. Immunotherapy has become a novel therapy option in a broad variety of malignancies including bladder cancer. Immunotherapy is approved as first line of treatment in patients who are ineligible for platinum-based chemotherapy and second-line treatment for metastatic urothelial cancer who progressed after platinum-based treatments. We present the case of an 83-year-old female with metastatic bladder cancer who was chemotherapy ineligible and had complete response with immune checkpoint inhibitor pembrolizumab.
Keywords
Introduction
Bladder cancer is the most common urinary tract neoplasm. In the United States, approximately 80 000 new cases and 17 000 deaths occur each year due to bladder cancer. 1 In Western Europe and the United States, urothelial carcinoma is the common histologic type. 2 Muscle-invasive bladder cancer is defined when malignancy involves the detrusor muscle and have a higher metastatic potential. The most common presenting symptom in patients with bladder cancer is painless hematuria (grossly visible or microscopic), although irritative voiding symptoms (frequency, urgency, dysuria) can also be the presenting manifestation. 3 Depending on the age, comorbidities, functional status of the patient, and stage of the malignancy, treatment options include surgery, radiation, chemotherapy, and most recently immunotherapy and targeted therapies.
Case Presentation
We report the case of an 83-year-old female who presented to the emergency department for lower abdominal pain. The pain started 1 month prior to the presentation and progressively got worse. A computed tomography (CT) scan of the abdomen and pelvis showed significant left-sided hydronephrosis, hydroureter, mass in left side of the base of the bladder (Figure 1), and 4.6 × 4.7 cm mass medial to the left common femoral artery (Figure 2). The patient underwent transurethral resection of the bladder tumor, which showed large necrotic mass involving left posterior bladder/trigone and left side of bladder wall (likely cause of left-sided hydronephrosis, hydroureter), and pathology was consistent with high-grade urothelial carcinoma with invasion of muscularis propria. The patient was diagnosed with metastatic bladder cancer. The patient was not a chemotherapy candidate based on the age and performance status and immunotherapy was started with pembrolizumab. The patient got 7 cycles of pembrolizumab (every 3 weeks) and then presented to hospital for breathing difficulty and CT scan of the chest showed pneumonitis. The patient was initiated on steroids, pembrolizumab permanently stopped, and eventually discharged home. The patient continued to follow-up as outpatient off therapy and repeat CT chest, abdomen, and pelvis 6 month post last treatment showed no evidence of metastatic disease in chest, abdomen, and pelvis–complete response (Figure 3).

Computed tomography scan of the abdomen and pelvis showing significant left-sided hydronephrosis, hydroureter, and mass in left side of the base of the bladder.
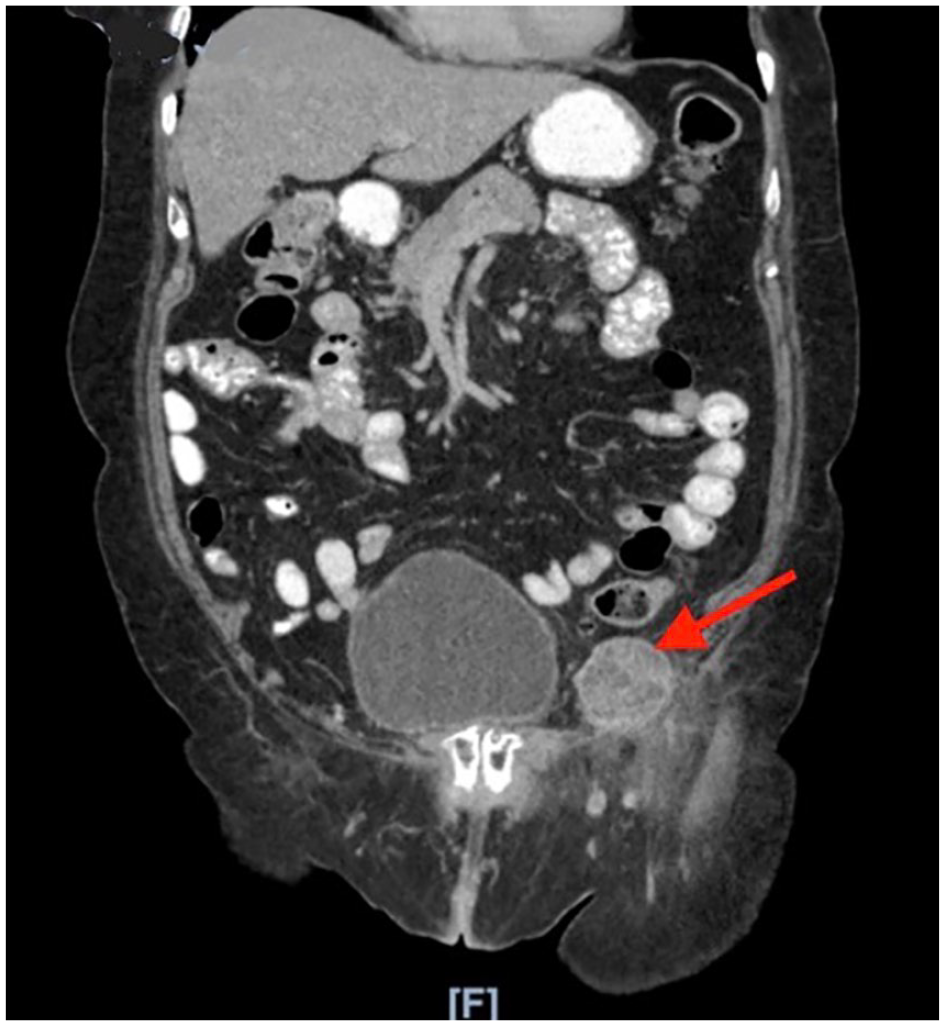
Computed tomography scan of the abdomen and pelvis showing 4.6 × 4.7 cm mass medial to the left common femoral artery.
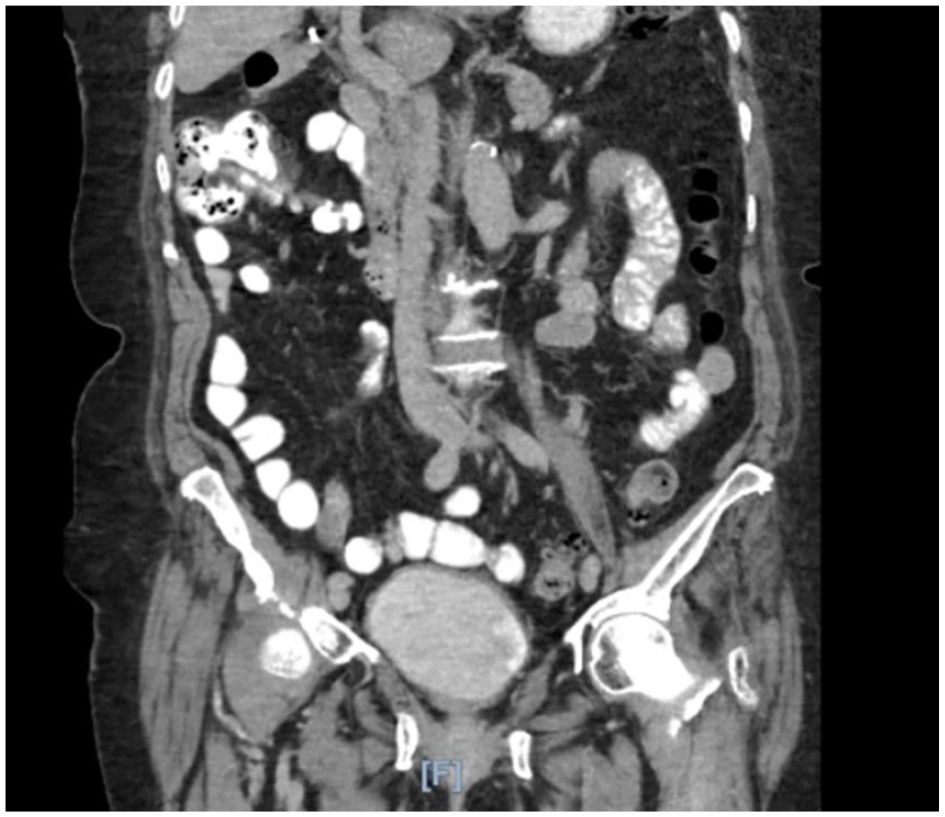
Computed tomography scan of the chest, abdomen, and pelvis showing no evidence of metastatic disease–complete response.
Discussion
Platinum-based chemotherapy regimen remains the first-line treatment standard for locally advanced or metastatic bladder cancer. Immunotherapy is approved as first line of treatment in patients who are ineligible for platinum-based chemotherapy and second-line treatment for metastatic urothelial cancer who progressed after platinum-based treatments. 4 The programmed cell death 1 (PD-1) receptor is expressed predominantly by T-cells. Binding of PD-1 to its ligands (PD-L1/PD-L2) leads to inhibition of proliferation and immune response of T-cells. Tumors cells can avoid the immune system through the PD-1 mechanism. Pembrolizumab blocks the PD-1 receptor and inhibits both ligands (PD-L1/PD-L2) from interacting with PD-1 receptor resulting in revamping of the T-cell response and immune response. 5 The Food and Drug Administration has approved the PD-1 inhibitors nivolumab and pembrolizumab as well as the PD-L1 inhibitors atezolizumab and avelumab for patients with urothelial carcinoma.
Immunotherapy drugs can cause immune-related adverse events. The most commonly reported side effects are colitis, hepatitis, pneumonitis, endocrinopathies (hyperthyroidism, thyroiditis, primary adrenal insufficiency, hypothyroidism, diabetes), dermatologic events, acute kidney injury, rheumatologic events, and myocarditis. 6 Cessation of the immune checkpoint inhibitor and initiation of steroids is the most common treatment used for the adverse events.
In the KEYNOTE-052 study, 370 patients with advanced urothelial cancer who were cisplatin-ineligible and treatment naive were enrolled, 6% of patient’s achieved a complete response. 7 Abdelhakam et al reported a case of 53-year-old male with metastatic urothelial carcinoma who progressed on platinum-based chemotherapy with metastasis to the bone and achieved a durable complete response with checkpoint inhibitor—atezolizumab. 8 The patient in the report got 2 years of treatment with atezolizumab, and imaging with magnetic resonance and bone scan showed no evidence of disease. In our case, the patient received only 7-month treatment due to immunotherapy-induced pneumonitis, and CT chest, abdomen, and pelvis 6 month post last treatment showed no evidence of metastatic disease. Our case adds to the literature regarding complete response of a metastatic bladder cancer in an otherwise chemotherapy ineligible patient and side effect profile of immunotherapy.
Recently pembrolizumab was approved by the Food and Drug Administration for the treatment of patients with non-muscle-invasive bladder cancer with carcinoma in situ with or without papillary tumors. The patients were ineligible for or have elected not to undergo cystectomy and the tumor was bacillus Calmette-Guérin unresponsive. A total of 148 patients were enrolled in the study. In this study, complete response rate of 41% and median response duration of 16.2 months was achieved in the 96 patients with carcinoma in situ that was bacillus Calmette-Guérin unresponsive. Complete response lasting at least 12 months was achieved in 46% of responding patients. 9 Complete response has been observed in other types of malignancies. Based on analysis of data from 5 clinical trials of pembrolizumab, a total of 149 patients with microsatellite instability-high/mismatch repair deficient tumors (15 different tumor types—60% of patients having metastatic colorectal cancer), an overall response rate of 39.6% and complete response rate of 7% was achieved. 10
Conclusion
We present the case of an elderly female with metastatic bladder cancer who was chemotherapy ineligible and hence was given immune checkpoint inhibitor pembrolizumab and complete response was achieved.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our hospital does not require ethics committee approval for case reports.
Informed Consent
Verbal informed consent was obtained from the patient for the publication.
