Abstract
The most common subtype of colon cancer is colorectal adenocarcinoma. Compared with other subtypes, such as signet-ring and mucinous, colorectal adenocarcinoma has been found to have lower rates of metastasis. Approximately 20% of colorectal cancer cases present with metastatic disease on initial evaluation. The most common locations for metastasis are the liver, lung, peritoneum, bone, and extra-regional lymph nodes. Metastatic disease to the skeletal muscle, however, is considerably rare. We present a clinical case of a 52-year-old female found to have a cystic iliopsoas muscle metastasis from rectosigmoid adenocarcinoma, initially classified as an infected fluid collection.
Introduction
Colorectal cancer (CRC) commonly arises in the context of genomic instability. 1 It is often due to alterations in the Wnt signaling pathway that heighten intracellular signaling involved in carcinogenesis, cell proliferation, division, and migration. It is the misregulation of these processes that lead to tumor development via constitutive cell proliferation. Symptoms of CRC are affected by the location of the tumor as well as whether or not it has metastasized to other locations in the body. 2 Alarm symptoms include worsening constipation, blood in the feces, decrease in stool caliper, loss of appetite, weight loss, nausea, vomiting, rectal bleeding, and/or anemia. 3 Risk factors include increasing age; male sex; high consumption of fat, sugar, alcohol, red meat, and processed foods; obesity; smoking; and lack of physical exercise. Many of these risk factors are associated with affluent Western societies, where poor diet, lack of exercise, and obesity rates are increased. 1 A total of 20% of CRC cases have metastatic disease at the time of initial diagnosis. Of the 3 major subtypes (signet ring cells, mucinous carcinoma, and adenocarcinoma) adenocarcinoma is reported to have the fewest metastases. In this article, we describe a case of an incidental finding of cystic ileus-psoas muscle metastasis from rectosigmoid adenocarcinoma.
Case Report
A 52-year-old female with a history of stage IA endometrioid adenocarcinoma, which was diagnosed and treated 5 years prior to this presentation, underwent a routine screening colonoscopy for CRC. The routine screening revealed an 8 mm flat, umbilicated, and superficially ulcerated rectal polyp. Hot snare technique was used for polypectomy. Pathologic evaluation diagnosed a low-grade, well-to-moderately differentiated invasive adenocarcinoma, with intact expression of all tested microsatellite instability genes. Subsequently, the patient underwent rigid sigmoidoscopy with endoscopic rectal ultrasound, which was unremarkable. No residual tumor was visualized. An additional endoscopic rectal ultrasound was performed at an outside hospital, which revealed a 5 mm sessile rectal polyp, positive pathologically for adenocarcinoma. A subepithelial lesion originating in the muscularis propria (T2 N0) was also visualized endoscopically.
The patient then underwent a low anterior resection of the primary rectosigmoid mass. Preoperative staging computed tomography (CT) scan of the abdomen and pelvis had noted right common iliac and left para-aortic lymphadenopathy, as well as a 5.7 × 4.2 cm nonenhancing hypodense left psoas cystic lesion with scalloping of the adjacent L4 vertebral body. This was addressed intraoperatively, but due to the inability to palpate the cyst, an aspiration was not performed. In addition to the primary resection specimen, multiple biopsies were taken of the omentum, regional lymph nodes, and the proximal and distal margins of the resection. Of those biopsies the omental, sigmoid colon, and the proximal and distal margins were all negative for malignancy. Primary rectosigmoid specimen showed a mucin-producing, moderately differentiated adenocarcinoma with invasion of the muscularis propria. The pathological staging after biopsy of the lymph nodes was a Stage III pT2 pN1a adenocarcinoma of the rectosigmoid. One out of the 10 lymph nodes tested were positive. Genetic testing revealed stability in microsatellite repair genes.
Postoperatively the patient received adjuvant combination oxaliplatin, capecitabine, and pelvic external beam radiation therapy. One month later a follow-up CT of the abdomen revealed a slight increase in the size of the mildly complex cystic lesion/fluid collection in the left psoas muscle, now eroding the L4 vertebral body (Figure 1). Magnetic resonance imaging of the abdomen revealed a well-defined cystic appearing lesion within the psoas muscle medially and anteriorly with considerable enhancement and swelling of the psoas muscle surrounding the lesion, with enhancement on T2-weighted sequences. CT-guided percutaneous aspiration of the cystic lesion in the left psoas muscle was performed (Figure 2), revealing clusters and sheets of atypical epithelial cells (Figures 3-5). Immunohistochemistry profile of these cells confirmed adenocarcinoma consistent with a colorectal primary malignancy. Pan-keratin, CDX-2, and CK 20 were positive. BRAF and KRAS exon 2 and 3 mutational tests were negative. The patient is currently maintained on platinum doublet chemotherapy with control of metastatic disease.

Follow-up computed tomography of the abdomen revealing a mildly complex cystic lesion/fluid collection in the left psoas muscle.

Computed tomography–guided percutaneous aspiration of the cystic lesion in the left psoas muscle.

Representing low-grade colonic adenocarcinoma, with mucin pool.

Cytology smear slides representing columnar epithelial cells, which are adenocarcinoma cells in psoas muscle biopsy specimen.
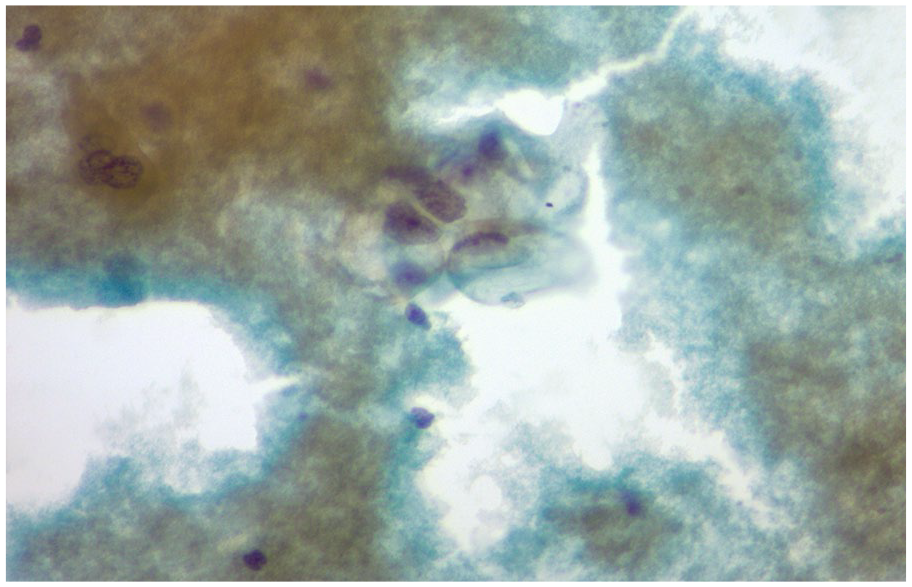
A cell block demonstrating the neoplastic columnar cells with abundant cytoplasm containing intracellular mucin.
Discussion
Although skeletal muscle comprises approximately 40% of body weight and is considered one of the largest organs of the human body, metastasis from solid organ malignancy is relatively rare. The histologic structure of skeletal muscle sarcolemma serves as a physical barrier against metastasizing tumor cells.
Of primary solid organ malignancies, the most common to metastasize to skeletal muscle are lung, gastric, and genitourinary cancers. Metastasis from CRC to skeletal muscle is a very rare manifestation. 4 Observationally, rectosigmoid CRC spreads significantly more to alternative sites such as the lungs, bones, and nervous system. Direct invasion to skeletal muscle is not described in the literature because most cases remain asymptomatic, and difficult to characterize by physical examination. Most reported cases involve autopsy series, highlighting the significant proportion that are missed during the course of disease management. Hasegawa and colleagues analyzed 194 autopsies, in which 11% had macroscopic metastases and 16% had microscopic invasion. 5 Another review of 52 patients with skeletal muscle metastases showed that the most commonly affected muscles are gluteus, psoas, and erector spinae. 6 According to a database from the Japanese Registry of Autopsies, the incidence of skeletal muscle metastases is 0.028%. 5 Other authors consider the prevalence of them between 0.03% and 5.6%.7,8 Ninety percent of patients with skeletal muscle metastases were found to have widespread metastasis lesions such as brain and lungs. 7
The 3 main routes of metastatic spread of colorectal cancer are hematogenous, lymphatic, or direct extension of primary disease. Surgery for colorectal cancer can cause direct local spread to skeletal muscle if not adequately resected. 4 A prior case of metastasis of the sigmoid colon cancer to the iliopsoas has been reported post-colectomy. 9 The patient reported in our clinical case had undergone numerous surgical procedures, including total abdominal hysterectomy, salpingo-oophrectomy, and myomectomy. These along with an exploratory laparotomy for CRC may have contributed to cancer seeding in a local region to the left psoas muscle.
Diagnosis of psoas muscle metastases can be challenging, as one of the differential diagnoses is psoas abscess. Malignancy should be suspected in any patient diagnosed with psoas abscess that does not respond to antibiotic therapy after 3 to 4 weeks. 1 As in our case, CT-guided fine needle aspirate is useful to differentiate abscess from psoas metastases. 6 In terms of diagnostic imaging, magnetic resonance imaging has more than 90% sensitivity for the detection of sarcoma and metastatic lesions in muscle,7,10 when compared with CT.
There are various theories that explain the rarity of skeletal muscle metastases. These include variability in blood flow, intermittent muscular contractions, lactic acid metabolism, pH, the presence of diffusible proteases, and other inhibitors that may block enzyme-dependent processes of invasion or tumor growth. Malignant cells are found more often in denervated muscle, which is unable to contract and induce mechanical trauma, as well as not being able to expel cells from the microvasculature compared with stimulated or innervated muscles. 11 There are several mediators such as tumor necrosis factor-α, transforming growth factor-β, lymphocyte infiltrating factor, interferon-γ, lactic acid, and plasminogen activator inhibitor that are released by muscle cells and exert an inhibitor effect on the growth of several tumor cell lines. 12
Skeletal muscle metastasis often means there is an underlying widespread pathology, which correlates to a poorer prognosis. The prognosis of patients with skeletal muscle metastases is variable from benign to poor depending on the primary tumor and disease stage. There are no data regarding muscle metastases from CRC, but information from lung cancer metastases indicates a mean overall survival of 8 months after resection of primary carcinoma. 5 By contrast, we reviewed 2 case reports of good outcomes after resection of solitary muscle metastases from cervical cancer.6,13
In conclusion, we report an interesting and unusual case of CRC metastasizing to the left psoas muscle initially diagnosed as a cystic lesion. Unlike other adenocarcinomas of the rectosigmoid junction, which normally metastasize to the liver or bone. We hope this case highlights the need to maintain a high level of suspicion for any abnormal clinicopathologic findings in patients with malignancy, which could potentially have significant consequences to morbidity and/or mortality.
Footnotes
Authors’ Note
The study was accepted for the Virtual American College of Gastroenterology Conference, October 2020.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
