Abstract
Retroperitoneal fibrosis is caused by the replacement of normal retroperitoneal tissue with fibrosis. The majority of the cases are idiopathic, but some secondary causes include malignancy, infection, drugs, and radiotherapy. Immunoglobulin G-4 (IgG-4) related disease is a relatively newer disease and one of the rarer causes of retroperitoneal fibrosis. It usually involves the pancreas, lungs, kidneys, aorta lacrimal and salivary glands, or extrapancreatic bile duct. Elevated serum IgG-4 is the biomarker of the disease and its levels correlate with disease activity. High-dose glucocorticoid is the treatment of choice.
Introduction
Retroperitoneal fibrosis (RPF) is a rare disease caused by the replacement of normal retroperitoneal tissue with fibrosis. 1 The rate of incidence is 0.1 to 1.3 cases/100 000 persons per year. It is more prevalent in men with an average age of onset between 55 and 60 years. It can be divided into idiopathic form (>75% of the cases) or secondary form due to underlying malignancy, infection, drugs, radiotherapy, infection, and other causes. 2 We present a case of immunoglobulin G-4 (IgG-4)-related disease, which is a rare cause of idiopathic RPF.
Case Presentation
A 61-year-old Caucasian male with a history of hypertension, diabetes mellitus, and chronic obstructive pulmonary disease presented to the hospital with a 1-week history of generalized weakness, low oral intake, and abdominal pain. Physical examination was significant for tenderness in the right upper quadrant of the abdomen without any rebound or guarding.
Pertinent laboratory findings (Table 1) included potassium 8 mmol/L (3.6-5.2 mmol/L), chloride 116 mmol/L (100-108 mmol/L), urea nitrogen 86 mg/dL (7-24 mg/dL), creatinine 8.10 mg/dL (0.8-1.3 mg/dL), aspartate aminotransferase 5 U/L (11-39 U/L), alanine transaminase 10 U/L (12-78 U/L), total bilirubin 0.6 mg/dL (0-1 mg/dL), alkaline phosphatase 78 U/L (45-117 U/L), IgG subclass-4 27 mg/dL (1-123 mg/dL), C-reactive protein 9.5 mg/dL (0-0.5 mg/dL), erythrocyte sedimentation rate 93 mm/h (0-20 mm/h), white blood cell count 9700/µL (4000-11 000/µL), and hemoglobin 9.4 g/dL (13.5-18 g/dL). Computed tomography (CT) of the abdomen (Figure 1A) showed lobular soft tissue surrounding the aorta and the iliac arteries with mild medial deviation of the ureters. There was also bilateral hydronephrosis due to encasement of the ureters with intraperitoneal soft tissue.
Serial Laboratory Values With Reference Range.

Abbreviations: BUN, blood urea nitrogen; AST, aspartate aminotransferase; NA, not available; ALT, alanine aminotransferase; ALP, alkaline phosphatase; WBC, white blood cell count; Hb, hemoglobin; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; IgG, immunoglobulin G.
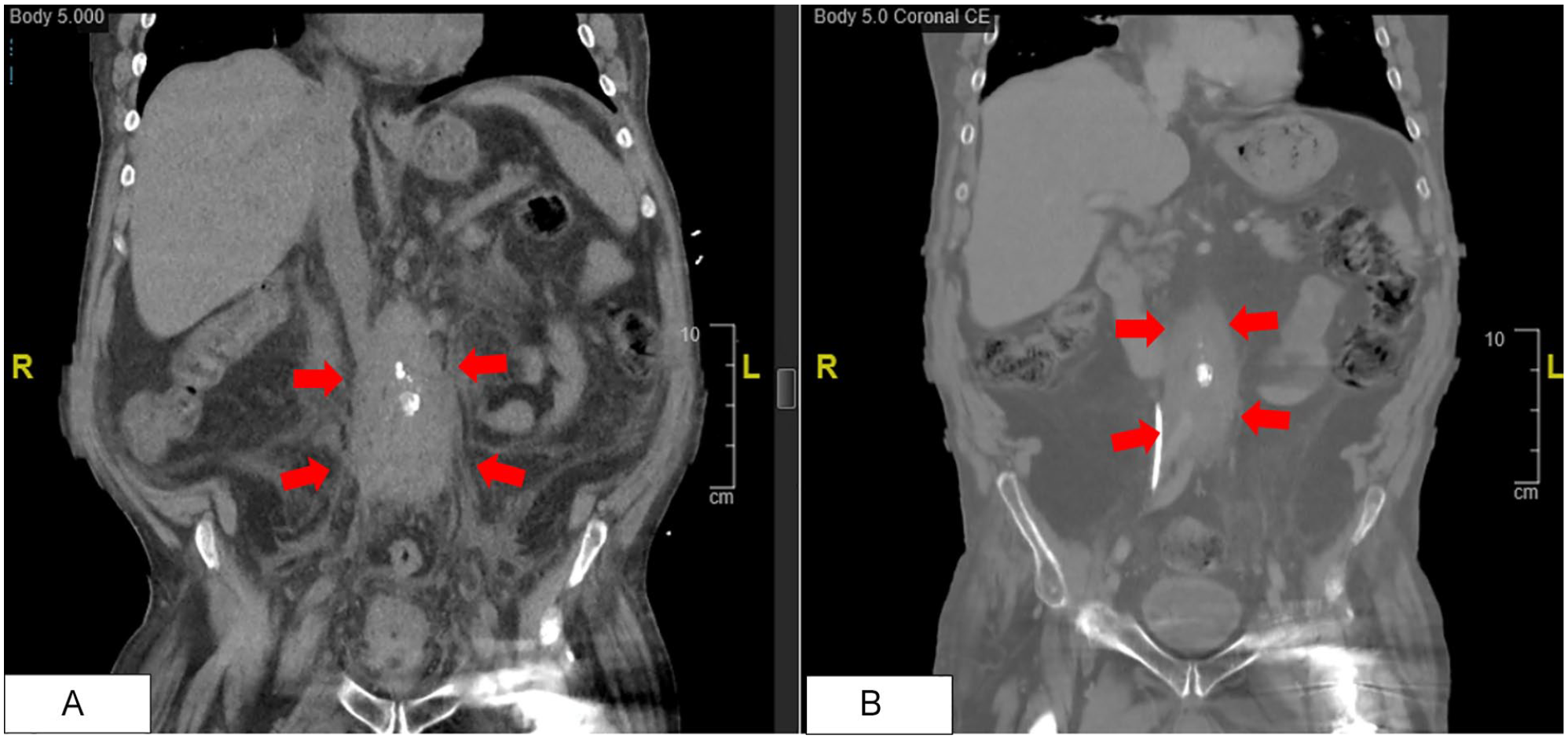
(A) Coronal section of computed tomography of the abdomen without contrast done on initial presentation shows retroperitoneal fibrosis (red arrows). (B) Coronal section of computed tomography of the abdomen with contrast done 45 days later shows decreased size of retroperitoneal fibrosis (red arrows).
The patient was anuric and emergently dialyzed since there was no improvement in hyperkalemia with intravenous insulin and dextrose solution. Serum creatinine and electrolyte abnormalities improved after the insertion of bilateral nephrostomy tubes. Core biopsy of the retroperitoneal tissue (Figures 2-4) revealed fragments of connective tissue with a bland loose spindle cell proliferation admixed plasma cells and lymphocytes, scattered eosinophils, interspersed hyalinized stroma, and suggestion of trapped fat. Staining for CD-138 highlighted a prominent population of plasma cells with variable cellularity. IgG-4 immunostaining showed positive staining plasma cells scattered throughout with variable cellularity with 20 positive staining cells per high-power field.

Plasma cells (black arrow) rich infiltrate with scattered lymphocytes and eosinophils in the background of fibroblastic connective tissue (hemoglobin and eosin, 10×).
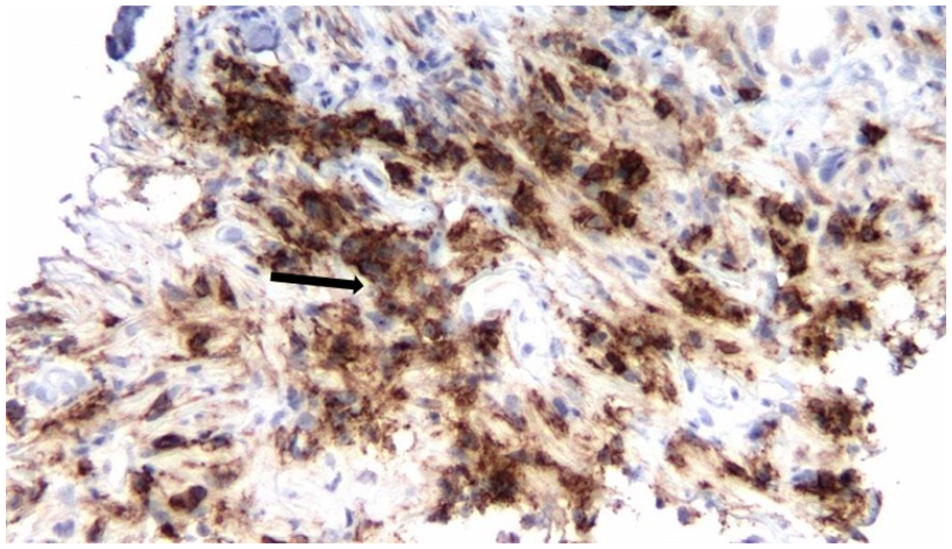
CD-138 immunostaining (black arrow) highlighting plasma cell infiltrate. CD-138 is a highly specific marker for plasma cells (40×).

IgG-4 immunostaining (black arrow) with variable density of cells with >10/high-power field (40×).
The patient was started on prednisone 60 mg daily for induction therapy. Due to difficulties with glycemic control, steroid-sparing therapy was initiated early in the course of illness with rituximab 1 g biweekly. The clinical course included 2 hospital admissions with sepsis of urinary origin due to nephrostomy tube dislodgment. Rituximab infusions had to be temporarily stopped due to the same. However, imaging (Figure 1B) done 45 days later during one of the hospitalizations did show decreased size of the RPF. Given this improvement, the management plan included tapering the prednisone dose while resuming rituximab infusions.
Discussion
The IgG-4 related disease was first defined as a systemic disease in 2003 and got its present name in 2010. 3 The exact pathogenesis is unknown but it is believed to be an autoimmune proinflammatory condition mediated by both B-cells and T-cells, particularly CD4+ and T-follicular helper cells. 4 It characteristically involves the pancreas, lungs, kidneys, aorta, lacrimal and salivary glands, or extrapancreatic bile duct but has also been described in the thyroid gland, prostate gland, pachymeninges, and skin.5,6 About one quarter of patients with IgG-4-related disease develop RPF. 7
The Japanese Comprehensive Clinical Diagnostic criteria for IgG-4-related disease 8 includes the following: (a) presence of diffuse/localized swelling or masses in single or multiple organs; (b) elevated serum IgG-4 concentrations (>1350 mg/L); and (c) histopathologic examination showing marked lymphocyte and plasmacyte infiltration and fibrosis, as well as infiltration of IgG-4+ plasma cells at a ratio of IgG-4/IgG cells >40% and >10 IgG-4+ plasma cells/high-power field (definite: a + b + c; probable: a + c; possible: a + b).
Elevated serum IgG-4 is a marker of IgG-4-related disease. Its level is usually more than 1350 mg/L with multi-organ involvement associated with higher IgG-4 concentration. However, elevated IgG-4 levels alone cannot be used to make the diagnosis or exclude it as the levels can be normal in 3% to 30% 8 individuals as was the case with our patient. IgG-4 levels are also used to monitor treatment responsiveness as well as disease recurrence. Other laboratory parameters including white blood cell count, antinuclear antibodies, serum IgG and IgE, and inflammatory markers such as erythrocyte sedimentation rate and C-reactive proteins are also elevated but are not reliable for monitoring disease activity.
Imaging typically cannot distinguish between malignancy and disease in the affected organs. Ultrasonography is the preferred imaging modality of choice as it is cheap and readily available, and lesions appear as hypoechoic to isoechoic. On noncontract CT, the density of lesions is similar to that of muscles, so they are reported as soft tissue masses. CT is also used to monitor treatment and disease activity. Magnetic resonance imaging is a suitable choice in patients with renal insufficiency. Fluorine-18-labeled fluorodeoxyglucose positron emission tomography can identify active inflammation and can be used to find lesions in the retroperitoneum and other organs. 9
Histopathological examination is an important tool for diagnosis and is characterized by lymphoplasmacytic inflammation, fibrosis with a storiform pattern, and obliterative venulitis. However, the absence of these findings does not exclude the disease as these lesions are usually asymmetric, and accessing the retroperitoneum is difficult.
High-dose glucocorticoids is the treatment of choice. Although most patients initially respond to glucocorticoids, up to 33% have a relapse of the disease. 10 Steroid sparing therapy with rituximab is recommended for maintaining remission.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient for their anonymized information to be published in this article.
