Abstract
Thrombocytopenia 2 (THC2) is an autosomal dominant disorder characterized by ankyrin repeat domain 26 mutation and moderate thrombocytopenia. THC2 exposes patients to a low risk of bleeding and an increased likelihood of myelodysplastic syndrome/acute myeloid leukemia. Germline evaluation for a genetic disorder should be considered when a patient presents with isolated thrombocytopenia and associated dysmegakaryopoiesis. In this case report, we present a male patient who presented with isolated thrombocytopenia but was ultimately confirmed to have an inherited THC2 thrombocytopenia/myelodysplastic syndrome. Given the rarity of the disease, no clear guidelines on how to follow THC2 patients over the long term have been established. We recommend a monthly complete blood count and clinical visits every 3 months at a minimum.
Introduction
Myeloid neoplasms (MN) with familial occurrence are rarely reported. These entities are uncommon and are also underdiagnosed. The 2016 World Health Organization includes 3 categories of MN with germline predisposition: (1) neoplasms without preexisting conditions, (2) neoplasms with a history of preexisting platelet disorder (MN with germline
Thrombocytopenia 2 (THC2) is an autosomal dominant disorder characterized by ankyrin repeat domain 26 (
Case Presentation
A 49-year-old male with a past medical history of hypothyroidism, low testosterone, and hypertension was referred to our cancer center in April 2018 for evaluation of isolated thrombocytopenia and suspected MDS. He self-reported a long-standing history of thrombocytopenia, which had first been documented at the age of 17. During that time, his platelet count was 40 × 109/L, but hemoglobin concentration and leukocyte counts were normal. Evaluation by a hematologist was performed at that time along with a BM biopsy that was reportedly “normal.” Notably, current laboratory testing revealed the platelet count to be 21 × 109/L, hemoglobin 13.8 g/dL, and white blood cell count 5.5 × 109/L. He mentioned general fatigue only. Finally, he reported that members in his family were also affected by thrombocytopenia (Figure 1), and he was able to provide a detailed report of exactly which family members as well as their approximate platelet counts (almost all were <100). Furthermore, he also had a family history of breast and colon cancer in his mother and gastric cancer in his maternal grandfather. Social history was negative for alcohol use, and he was self-employed as a turkey farmer.

Thrombocytopenia pedigree. Proband marked by arrowhead. “D” stands for patients with known thrombocytopenia. Numbers between parentheses stand for platelets count (103/mm3). Subjects with history of gastrointestinal malignancy marked by green cross.
Results and Discussion
A BM aspirate and biopsy at the time of consultation at our institution revealed a hypercellular BM, thrombocytopenia, and prominent dysmegakaryopoiesis showing micromegakaryocytes with hypolobated or bilobated nuclei concerning for single lineage MDS (Figure 2A-C). BM cytogenetics detected an abnormal clone: 46,XY,inv(12)(p13q21)[7]/46,XY[13]. Myeloid malignancy mutation panel by next-generation sequencing (NGS), using 37 genes, was performed on peripheral blood specimen at GenPath Laboratories. Comprehensive thrombocytopenia panel by sequence analysis, using 43 genes, and deletion/duplication analysis by high-density array comparative genomic hybridization was performed on peripheral blood specimen at the University of Chicago Genetic Services Laboratories. Complete sequence analysis of the coding regions of the included genes in this panel and the 5′UTRs of
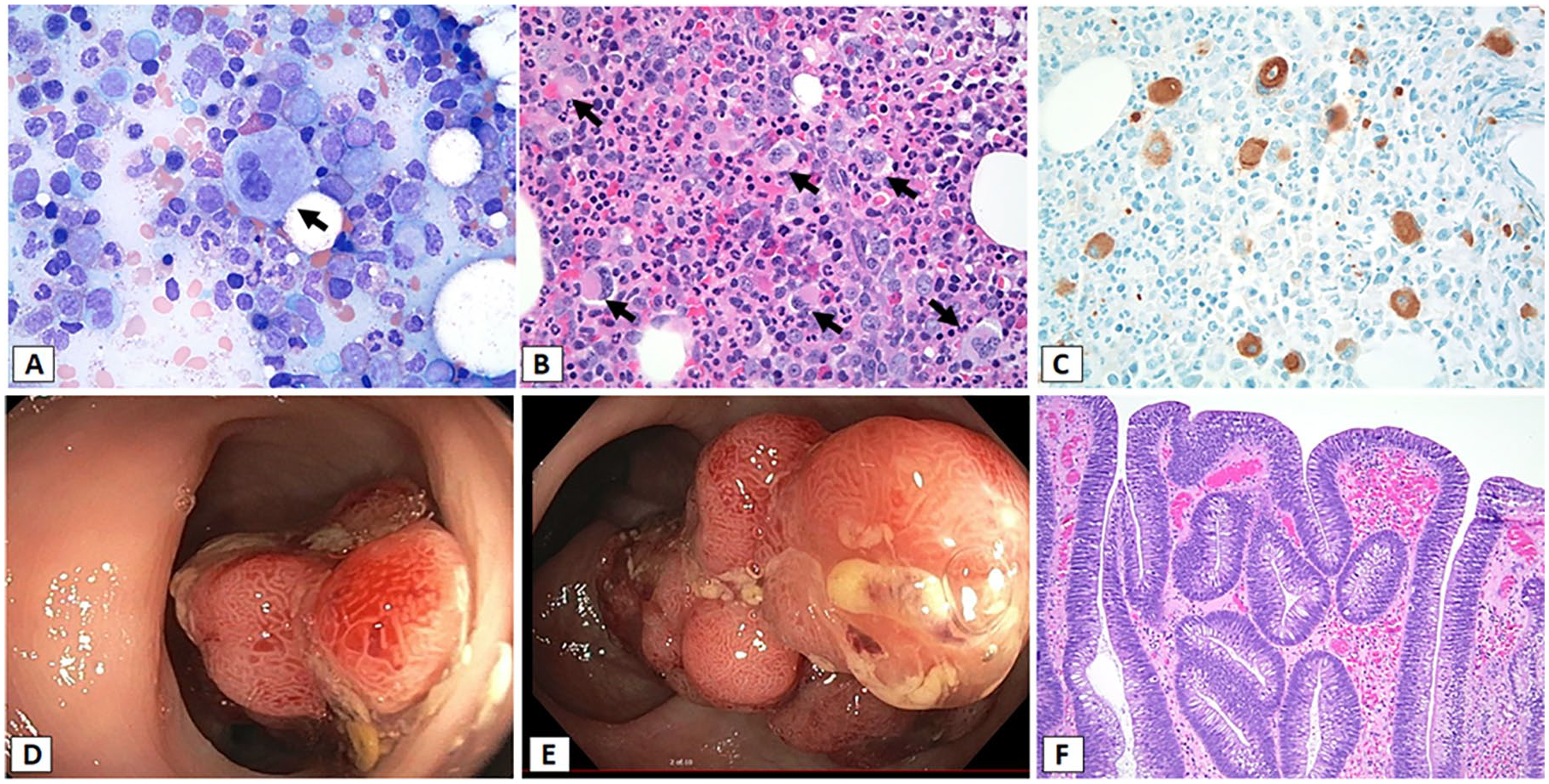
(A) Bone marrow aspirate smear showing trilineage hematopoietic maturation with dysplastic megakaryocytes with small size and bilobted nuclei (arrow; Wright stain, ×1000). (B) Bone marrow core biopsy showing hypercellularity (90%) with trilineage maturation and several dysplastic megakaryocytes with hypolobated nuclei (arrows; hematoxylin-eosin [H&E], ×400). (C) CD61 immunohistochemistry on the core biopsy highlights increased numbers of megakaryocytes with dysplastic small megakaryocytes with hypolobated nuclei (×400). (D and E) Large multiple polyps with the largest in size 2.4 × 1.5 × 1.2 cm in the rectosigmoid colon. (F) Tubulovillus adenoma of polyp (H&E, ×100).
No mutations were detected in the 37 gene myeloid malignancy mutation panel by NGS. A DNA sequence analysis of the
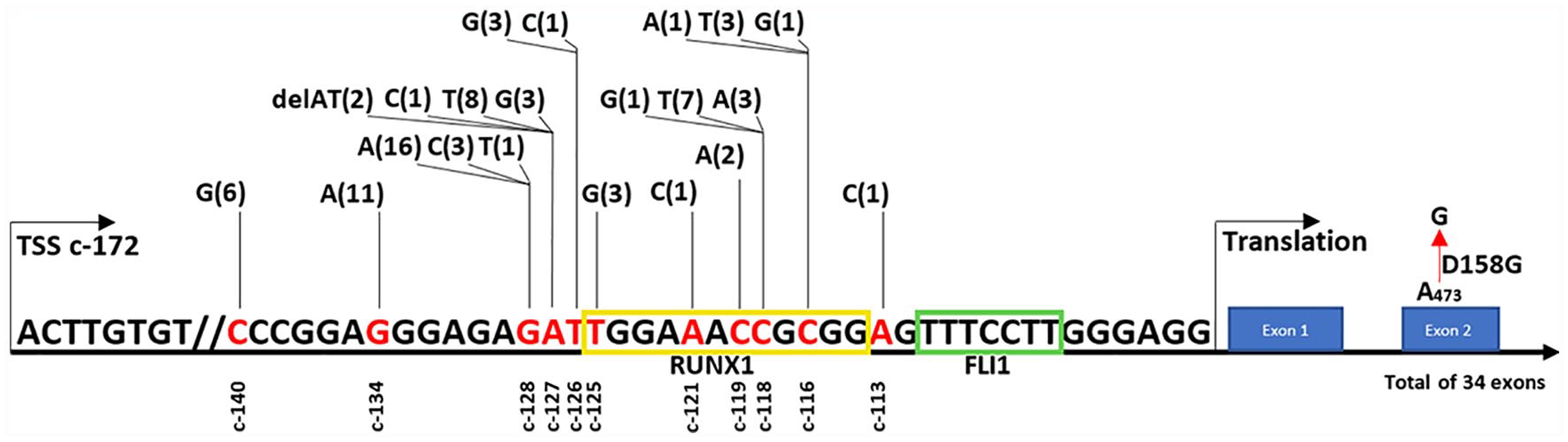
5′UR of ANKRD26 gene with different mutations linked to THC2. Position c-172 indicates transcription start site (TSS). Yellow and green boxes represent RUNX1 and FLI1 binding sites, respectively. Numbers between parentheses represent total number of families affected by each mutation. Red cross indicates position of sequence change in our patient.
The patient was followed with monthly compete blood counts and clinical visits every 3 months. His white blood cell count, platelet, and hemoglobin levels were stable for 6 months, and then his hemoglobin level started to decrease. Ultimately, his anemia workup revealed an iron deficiency anemia. A repeat BM biopsy was performed and revealed hypercellularity, thrombocytopenia, and similar dysmegakaryopoiesis. Blast cells remained at 2%, and the cytogenetic analysis detected the same abnormal clone with no major changes.
Colonoscopy was recommended given new iron deficiency anemia. He was started on aminocaproic acid 500 mg once daily due to low platelet count of 20 × 109L−1. Colonoscopy revealed large polyps (2.4 × 1.5 × 1.2 cm) in the rectosigmoid colon that could not be excised in the first attempt (Figure 2D and E). Also, multiple small polyps were found in the colon and resected successfully. The large rectosigmoid polyp was resected by second-attempt colonoscopy after platelet transfusion. Subsequent pathological examination demonstrated a tubulovillous adenoma (Figure 2F). Given his high risk, he will need to repeat the colonoscopy after 3 years according to colon cancer screening guidelines. 19 The patient currently is stable with anemia and thrombocytopenia. A BM transplant physician was consulted, and future planning for a matched unrelated donor was recommended.
Pathogenic variants in the 5′UTR and protein coding regions of
Furthermore, the severity of bleeding diathesis in patients with THC2 is variable. Although life-threatening events were reported, no patient died from hemorrhage among the 78 patients from all 21 families.
7
The 62-year-old patient with
Although THC2 is rare disease, isolated thrombocytopenia and BM dysplasia limited to the megakaryocyte lineage are very rare22,23 and carry a higher likelihood of progression to MN. Thus, when dysmegakaryopoiesis is found in a patient having thrombocytopenia, the differential diagnosis of MDS and hereditary thrombocytopenia should be investigated.
In conclusion, THC2 exposes patients to a low risk of bleeding and an increased likelihood MDS/AML. Our patient had colon polyps and a tubulovillous adenoma although gastrointestinal malignancy is not known to be associated with ANKRD26 mutation(s). Germline evaluation for a genetic disorder should be considered when a patient presents with isolated thrombocytopenia and associated dysmegakaryopoiesis. Current platforms of NGS testing do not typically have the
Footnotes
Author Contributions
TK and HEC designed the research. TK, RN, LAG, HJR, and HEC preformed research, analyzed data, and wrote the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Cleveland Clinic Committee on Human Research approved the study.
Informed Consent
Written informed consent was obtained. However, our institution does not require informed consent for individual case reports.
