Abstract
In isolated partial anomalous pulmonary venous connections (PAPVCs), an abnormal vein connects venous blood from the pulmonary circulation to the systemic circulation, resulting in an extracardiac shunt. A single aberrant pulmonary vein (PV) is usually hemodynamically insignificant, and affected patients are generally asymptomatic. We describe a young Caribbean-Black woman with an isolated, singular PAPVC from the left inferior PV to the left innominate (brachiocephalic) vein that was hemodynamically significant, obfuscated by recurrent pleural effusions from catamenial pleural endometriosis.
Keywords
Introduction
Partial anomalous pulmonary venous connection (PAPVC) is characterized by incomplete pulmonary venous drainage into the right atrium (RA) or a systemic vein instead of the left atrium (LA). It was initially described by Winslow in 1739 and indicates the persistence of the embryonic anastomosis between the systemic and pulmonary vein (PV) plexus.1,2
PAPVC is uncovered in less than 1% of autopsies. 3 It typically involves an anomalous connection of the right PVs to either the superior vena cava (SVC) or the RA; however, it can include the inferior vena cava, the innominate (brachiocephalic) vein, or the coronary sinus. 4 PAPVC is also frequently associated with other congenital heart defects, most notably atrial septal defects (ASD), with an approximately 15% incidence in ostium secundum ASD and nearly 90% incidence in patients with sinus venosus ASD. 5
PAPVCs can result in maladaptive pathophysiological left-to-right shunts, which can be exacerbated by coincident ASDs. 6 In chronic, untreated cases, pulmonary hypertension (pHTN) and Eisenmenger syndrome can develop. 6 PAPVCs often go unnoticed when more established or recognized congenital heart disease (CHD) has been excluded. As such, we describe a young Caribbean-Black woman with an isolated singular PAPVC from the left inferior PV to the left innominate (brachiocephalic) vein with a hemodynamically significant shunt, obfuscated by recurrent pleural effusions from catamenial pleural endometriosis.
Case Report
A 38-year-old Caribbean-Black (Afro-Trinidadian) female with a medical history of endometriosis, intermittently adherent with the oral contraceptive pill, initially presented to the adult emergency department with acute dyspnea during the preceding 2 days. Her vital signs reflected systolic blood pressures of 142 mm Hg, heart rate of 112 beats per minute, and respiratory rate of 20 breaths per minute with an oxygen saturation of 92% on room air. Her physical examination revealed a mildly elevated jugular venous pulse (10 cm of water), right chest stony dullness, no peripheral edema, and active menses.
A 12-lead electrocardiogram revealed sinus tachycardia with an incomplete right bundle branch block, right ventricular hypertrophy, and subtle secondary ST-T segment changes (see Figure 1). A portable chest radiograph revealed a large-sized, right pleural effusion with mild perihilar congestion (see Figure 2). Pertinent diagnostic laboratory investigations included a D-dimer 208 ng/dL (normal ≤500 ng/mL), pro–brain natriuretic peptide (pro-BNP) 323 pg/mL (normal ≤300 pg/mL), cardiac troponin I (cTnI) 0.08 ng/mL (normal = 0.0-0.15 ng/mL). The patient’s arterial blood gas was consistent with mild hypoxia on 24% fractional inspiration of oxygen (FiO2) with an alveolar-arterial gradient (A-a gradient) of 14 mm Hg.

The patient’s 12-lead electrocardiogram—sinus tachycardia (R-R interval underscored with the black line) with an incomplete right bundle branch block, right ventricular hypertrophy (identified by the black arrows), and subtle secondary ST-T segment changes.
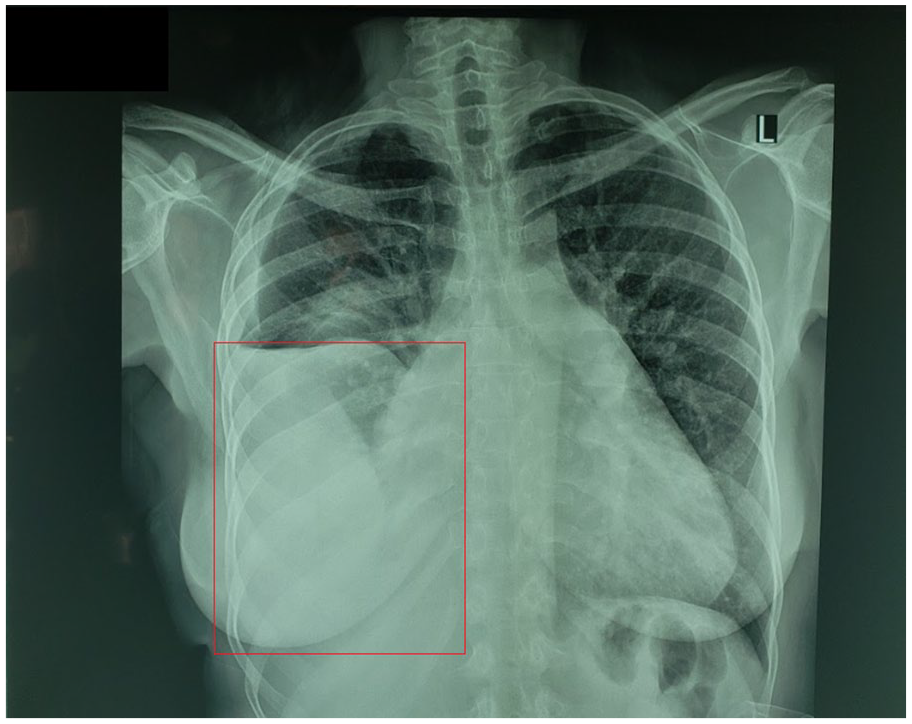
The patient’s chest radiograph (CXR)—large-sized, right pleural effusion (enclosed by the red box) with mild perihilar congestion.
The patient was subsequently admitted for an inpatient hospitalization, and computed tomography (CT)-guided therapeutic thoracentesis was performed with a resultant hemorrhagic exudative effusion. Despite this intervention, the pleural effusion recurred within 48 hours, and cardiothoracic surgery was consulted for a video-assisted thoracoscopic surgery (VATS) pleural biopsy, which later affirmed the presence of endometrial tissue on clinicopathological examination. The tentative diagnosis at this juncture was recurrent catamenial pleural effusions. On further interview, the patient acknowledged that she often experienced dyspneic episodes during menstruation.
During the ensuing hospitalization, she continued to experience persistent shortness of breath with an episode of paroxysmal atrial fibrillation (PAF). A 2-dimensional transthoracic echocardiogram (TTE) revealed preserved left ventricular function with moderate right heart enlargement, septal flattening, and mild pHTN with estimated right ventricular systolic pressures of approximately 45 mm Hg. A 2-dimensional and 3-dimensional transesophageal echocardiogram (2D/3D-TEE) also revealed an intact interatrial septum with agitated saline “bubble” study. The PVs were incompletely visualized and hence the rationale for further advanced imaging (see Figure 3). A cardiac CT angiogram (CCTA) affirmed the presence of the suspected PAPVC of the left inferior PV to the left brachiocephalic (innominate) vein with angiographically normal coronary arteries (see Figure 4). The patient also underwent cardiac magnetic resonance imaging (CMRI), which demonstrated the same aberrant connection with the remaining pulmonary venous vasculature appearing dilated in addition to the RA, right ventricle, and main pulmonary artery (see Figures 5 and 6, respectively). Left heart catheterization established angiographically normal coronary arteries with right heart catheterization revealing a significant hemodynamic left-to-right shunt with Qp:Qs ratio of 3:1, mild pHTN, and elevated pulmonary vascular resistance (see Table 1 for a summary of the pertinent cardiovascular investigations).

The patient’s 2-dimensional transesophageal echocardiogram. (A) Intact interatrial septum with agitated saline “bubble” study. (B) Incomplete visualization of the left and right pulmonary veins (PVs)—the left and right inferior PVs could not be imaged.

The patient’s cardiac computed tomography angiogram. (A) Cross-sectional view of the location of the anomalous left inferior pulmonary vein (PV) in relation to other cardiac structures. (B) Coronal section of the course of the left inferior PV with its relation to the pulmonary artery.

The patient’s cardiac magnetic resonance imaging scan. (A) Cross-sectional view of the location of the anomalous left inferior pulmonary vein (PV) in relation to other cardiac structures. (B) Sagittal section showing the connection of the left inferior PV to the innominate (brachiocephalic) vein. (C) Sagittal section showing the engorged, dilated pulmonary venous vasculature, reflecting the hemodynamically significant left-to-right shunt and increased transpulmonary flow.

The patient’s cardiac magnetic resonance imaging scan. (A) Right ventricular (RV) outflow tract view indicating the moderately dilated main pulmonary artery (PA). (B) Coronal section of the course of the left inferior pulmonary vein with its relation to the PA. (C) Apical 4-chamber view indicating the moderately dilated right atrium (RA). (D) Apical 4-chamber view indicating the moderately dilated RV.
Summary of the Pertinent Cardiovascular Investigations.

Cardiothoracic surgery was reconsulted regarding the surgical repair of the PAPVC. In the interim, the patient was instituted on an optimal cardiovascular regimen that included apixaban for her PAF, angiotensin receptor blocker with a neprilysin inhibitor (ARNI), β-blocker (BB), mineralocorticoid receptor antagonist (MRA) for her heart failure and nitrates, tadalafil, and calcium channel blockade (CCB) for her pHTN. The rest of her hospital course was uneventful, and she was discharged home with scheduled corrective surgery in 2 months (due to our limited resource setting pending consultation from an international specialist CHD cardiothoracic surgeon).
Discussion
PAPVC encompasses a subgroup of CHD, resulting in incomplete pulmonary venous drainage to the right heart, and left-to-right shunting. Shunt hemodynamics is predicated on the quantity and caliber of PVs and the presence of other associated cardiopulmonary defects such as ASD in nearly 90% of cases. 7 Usually, significant shunting occurs with the presence of at least 2 aberrant connecting veins culminating in right atrial, ventricular, and pulmonary artery dilatation. Generally, each PV contributes approximately one quarter of the pulmonary blood flow. However, in the setting of PAPVC, shunt flow is increased due to a relatively lower pressure system in the RA and SVC. 6
Clinically, patients can experience a wide spectrum of clinical presentations, which include heart failure, arrhythmias, pHTN, and possibly, Eisenmenger physiology.1,8 Asymptomatic PAPVC without coexistent cardiac defects is rare. To date, there are no identified risk factors for its development. 9 Our patient experienced a constellation of these heart failure symptoms, PAF, and pHTN. Initially, the differential diagnoses included pulmonary embolism due to the oral contraceptive pill, which was quickly refuted with a normal D-dimer and pleural endometriosis with the histological evidence; however, there were no other convincing elements to explain her right heart dilatation and pHTN.
The embryo-pathological basis is that initially, drainage occurs into the cardinal and umbilical vitelline via the splanchnic plexus. A craniocaudal diverticulum forms in the superior aspect with subsequent extension to lung buds. In the inferior region, there is regression, and the cranial portion forms the common PV, integrating into the left atrial wall. PAPVC arises due to an inadequate or incomplete fusion between the common PV and the splanchnic plexus. 9
This condition is typically diagnosed by TTE and TEE. It should be considered as a tentative diagnosis when an echocardiogram demonstrates unexpected right heart dilation in the absence of overt CHD such as ASD or fewer than 4 PVs connecting to the LA. 10 Although TTE is less sensitive and may not detect PAPVC in one third of cases, it remains the first-line diagnostic test as it can provide useful hemodynamic and structural information such as the presence of pHTN and right-sided heart enlargement, respectively. 11 TEE possesses better sensitivity and accuracy in experienced operators; however, complete anatomical delineation is occasionally difficult, for example, visualizing the superior aspect of the SVC. 2
While right heart catheterization may provide critical information with regard to oxygen saturations, pressures, and pulmonary vascular resistance, pulmonary angiography may not provide a complete overview of minor accessory, abnormal vessels. There is also the added caveat that the acquired information may be compromised as the true SVC saturation may not be accurate or reliable due to venous mixing. This diagnostic test has been supplanted by less invasive complementary imaging techniques such as TEE, CCTA, and CMRI. There is still a niche role for delivering transcatheter therapies, for example, small vessel occlusion.
Electrocardiographic-gated CCTA offers a rapid, accessible noninvasive imaging modality to detect the course of anomalous veins and associated CHD; however, radiation exposure remains a concern. 12 High-definition, multidimensional reconstructed images with additional anatomic detail and enhanced vasculature are crucial in corrective surgery planning.
CMRI is also gaining traction in complex and challenging cases that require surgical intervention. It can provide comprehensive information on precise anatomical architecture, shunt hemodynamics, and valvular function with PAPVC. 13 Additionally, it can be utilized with patients with chronic kidney disease, albeit with suboptimal diagnostic accuracy, and can employ phase-contrast velocity mapping for a detailed assessment of shunt function.
Generally, asymptomatic patients with hemodynamically insignificant shunts do not require intervention as there is no clinically significant impact on morbidity. 14 With regard to patients with symptoms and those with clinically significant left-to-right shunts, surgery is the definitive treatment. Transcatheter interventions for CHD are continually improving and may be attempted in selected cases where feasible. 15
There are several surgical interventions for PAPVC repair, which are contingent on the anatomy and coexistent cardiac anomalies. An anomalous left-sided PV is usually implanted in the LA or LA appendage. Postoperative complications include PV stenosis and arrhythmias. Surgical repair usually comprises complete closure of any septal defect and reorientation of any aberrant PVs into the LA. 16
The phrase “great imitator” is attributed to medical conditions that feature nonspecific, symptomatology that can easily be confused as a differential diagnosis, for example, oncologic, rheumatologic, and chronic infectious diseases. We hereby propose that PAPVC be added to this list of culprits given its enigmatic nature and challenging diagnosis.
In summary, there must be a high degree of clinical suspicion of PAPVC when dyspneic symptoms persist despite the apparent resolution of the initially diagnosed condition with incidental right heart enlargement. An armamentarium of novel, advanced imaging techniques such as TEE, CCTA, and CMRI can be used in tandem to identify notoriously elusive CHD such as PAPVC.
Conclusion
We detail a case of a young Caribbean-Black woman with an isolated PAPVC from the left inferior PV to the left innominate (brachiocephalic) vein that was hemodynamically significant, obfuscated by recurrent pleural effusions from catamenial pleural endometriosis. PAPVC should be considered as a differential diagnosis in challenging cases of refractory dyspnea and may require complementary imaging modalities to ascertain its diagnosis.
Footnotes
Authors’ Note
All pertinent, de-identified patient data can be obtained by contacting the corresponding author.
Author Contributions
RVS was involved with the conception of work and data collection. TD, VKS, SAP, BLM, FR, and PM were involved in interpretation. RVS, VKS, and SAP drafted the article. NAS critically revised the article. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Institutional and National Research Committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed Consent
The patient provided express written (and verbal) informed consent to have the details of her case published. UWI and NCRHA institutional approvals were not required for publication.
