Abstract
Partial anomalous pulmonary venous return (PAPVR) involving the left upper lobe pulmonary vein is an exceptionally rare congenital anomaly that, if untreated, can lead to pulmonary hypertension (PH). Its nonspecific clinical presentation often results in delayed diagnosis. We report the case of a 58-year-old woman who initially presented with progressive dyspnea and was diagnosed with group III pulmonary hypertension attributed to obstructive lung disease. Two years later, she returned with similar symptoms after discontinuing diuretic therapy. Further evaluation uncovered a previously undetected anomalous left pulmonary vein draining into the left innominate vein. This case highlights the diagnostic challenges of PAPVR, its role in contributing to PH, and the critical need for high clinical suspicion and comprehensive evaluation in patients with unexplained pulmonary hypertension.
Keywords
Introduction
Partial anomalous pulmonary venous return (PAPVR) is a congenital anomaly, often underdiagnosed due to its rarity, initial asymptomatic nature, and nonspecific presentation. The majority of PAPVR cases involve the right-sided pulmonary veins, with only 3% attributed to anomalous drainage of the left upper lobe pulmonary vein. 1 The abnormal drainage of one or more pulmonary veins into the systemic venous circulation creates a left-to-right shunt that can lead to right-sided heart dilation and vascular remodeling, resulting in pulmonary hypertension (PH). 2
We present the case of a 58-year-old female with PH, initially classified as group III PH, and later incidentally diagnosed with PAPVR involving the left upper lobe pulmonary vein. This report also highlights the diagnostic challenges associated with PAPVR.
Case presentation
A 58-year-old female presented to the Emergency Department with a 6-month history of worsening dyspnea. Over the preceding 10 days, her symptoms had acutely worsened, accompanied by mild chest discomfort and bilateral leg swelling. Her medical history included asthma and hypertension. On arrival, she was found to be hypoxemic with an oxygen saturation of 74%, which improved to 94% with 5 liters of supplemental oxygen. Physical examination revealed bilateral inspiratory crackles and pitting edema in the lower extremities.
Laboratory investigations demonstrated an elevated NT-proBNP (Brain natriuretic peptide) level of 2110 ng/mL (range <125 ng/mL) and normal troponin levels. Computed tomography (CT) chest angiography revealed mild bilateral patchy ground-glass infiltrates, an enlarged pulmonary artery (PA) trunk, and dilatation of the right atrium (RA) and right ventricle (RV) with no evidence of pulmonary embolism (Figure 1). Transthoracic echocardiography (TTE) showed a left ventricular ejection fraction (LVEF) of 75% to 80%, moderate RA and RV dilatation, systolic and diastolic flattening of the interventricular septum, reduced RV systolic function, and an estimated PA systolic pressure of 55 to 60 mm Hg (Figure 1). No intracardiac shunt was identified on color Doppler or agitated saline contrast injection.

(A) Contrast-enhanced computed tomography (CT) of chest (mediastinal window) with blue arrow showing an enlarged main pulmonary artery measuring 41 mm (normal <29 mm). (B) Contrast-enhanced CT of chest with (mediastinal window) demonstrating dilated right ventricle (RV) and flattening of interventricular septum (green arrow). (C) Echocardiogram in systolic phase showing dilated right ventricle (RV) and right atrium (RA) with normal left ventricle (LV) and left atrium (LA) and flattening of interventricular septum (green arrow). (D) Echocardiogram in diastolic phase showing dilated right ventricle (RV) and right atrium (RA) with normal left ventricle (LV) and left atrium (LA) and flattening of interventricular septum (green arrow).
The patient was initiated on intravenous furosemide. Right heart catheterization (RHC) performed the following day, while the patient was receiving 3 liters of supplemental oxygen via nasal cannula. The procedure confirmed PH, with a mean pulmonary artery pressure (mPAP) of 41 mm Hg and a high cardiac output of 8.46 L/min by the Fick method (Table 1). A vasoreactivity test using the standard adenosine protocol confirmed that the patient was non-vasoreactive, indicating calcium channel blocker therapy would not be beneficial. Further investigations, including complete blood count, thyroid-stimulating hormone, HIV screening, antinuclear antibody screen, rheumatoid factor, and thiamine levels, were unremarkable. Imaging studies, including CT angiography of the chest and abdomen and Doppler ultrasound of the liver, ruled out systemic shunts, pulmonary arteriovenous malformations, and splenomegaly. At the time of discharge, the patient’s hypoxemia had resolved with diuresis.
Hemodynamic and Shunt Parameters From Right Heart Catheterization in Initial and Follow-up Evaluations.
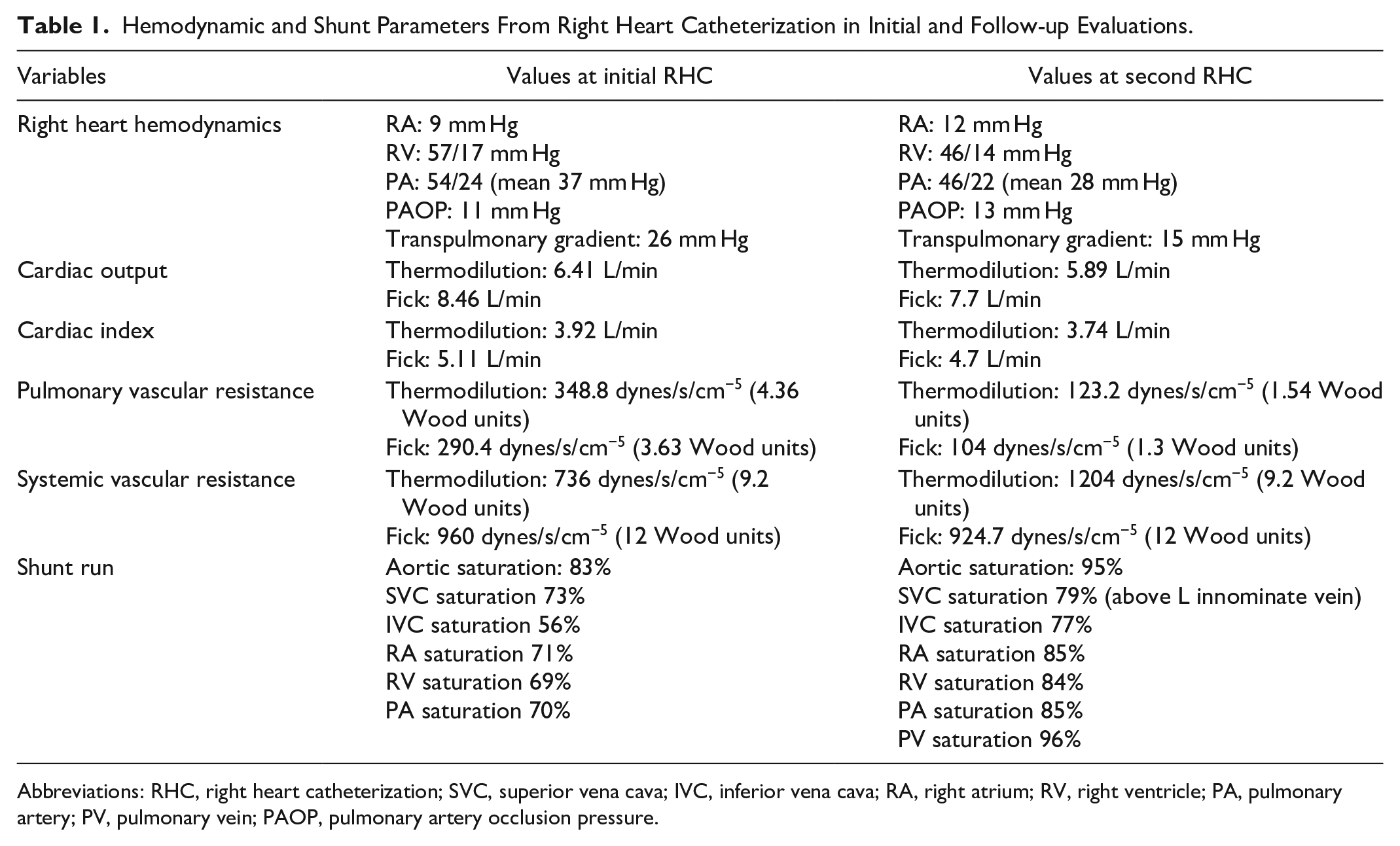
Abbreviations: RHC, right heart catheterization; SVC, superior vena cava; IVC, inferior vena cava; RA, right atrium; RV, right ventricle; PA, pulmonary artery; PV, pulmonary vein; PAOP, pulmonary artery occlusion pressure.
Outpatient evaluation included a pulmonary function testing that showed mild obstructive ventilatory defect. The sleep study and ventilation-perfusion study were unremarkable. As RHC demonstrated a pulmonary artery occlusion pressure (wedge pressure) of 11 mm Hg (<15 mm Hg) and a high transpulmonary gradient of 26 mm Hg (>12 mm Hg), Group II PH due to left heart disease was excluded. She was prescribed a corticosteroid inhaler, and oral furosemide (20 mg daily) was continued. Pulmonary hypertension-specific therapy was not initiated, given her PH was categorized as group III, attributed to obstructive lung disease. During follow-up appointments, the patient reported an initial improvement in her perceived dyspnea, which later stabilized. She consistently remained in the intermediate-low risk category according to the 4-strata risk assessment tool for PH follow-up.
Two years later, she returned with worsening dyspnea and leg swelling after discontinuing furosemide. Repeat CT pulmonary angiography revealed findings similar to her initial imaging but additionally identified an anomalous left pulmonary vein draining into the left brachiocephalic trunk (Figure 2). A retrospective review of her initial CT confirmed the missed anomaly. The TTE showed LVEF of 60%, moderate RA and RV dilation, reduced RV systolic function, and an estimated pulmonary artery systolic pressure of 50 to 55 mm Hg. In contrast to previous RHC, the shunt evaluation this time demonstrated a 6% step-up in oxygen saturation between the superior vena cava (SVC) and RA, confirming a left-to-right shunt (Table 1). This discrepancy was due to the SVC saturation measurement being appropriately obtained above the drainage of the left brachiocephalic trunk, now guided by the knowledge of PAPVR from the CT findings. The shunt fraction (Qp/Qs) was calculated to be 1.52. Consequently, her PH was reclassified as group I due to adult congenital heart disease.

Computed tomography (CT) of chest: Axial views (A, B) and coronal views (C, D) displaying an anomalous left pulmonary artery vein (blue arrows) draining into the left innominate vein (yellow arrows). Green arrows indicate superior vena cava.
The patient received treatment with intravenous diuretics. Due to her mild PH and intermediate-low risk classification based on the 4-strata risk assessment tool (WHO-FC III, NT-proBNP 81 pg/mL, and a 6-min walk distance of 460 m), PH-specific therapy was not advised. She declined additional evaluations, including cardiac magnetic resonance imaging (MRI) and referral to an adult congenital heart disease center, choosing instead to continue with close outpatient monitoring.
Discussion
The estimated prevalence of PAPVR is between 0.1% and 0.7%. Left-sided PAPVR is observed in only about 10% of cases, with anomalous drainage of the left upper lobe pulmonary vein being even rarer, accounting for just 3% of cases. 1 Data on this condition are extremely limited. In 1 study, Anika and Loren 3 reviewed the literature and identified only 9 articles meeting their criteria for left-sided PAPVR, with just 6 discussing cases involving the left upper lobe type. They described a case of a left upper lobe pulmonary vein draining into a vertical vein, which subsequently drained into the left innominate vein, similar to the presentation in our case.
Chronic high flow and pressure within the pulmonary arterial vasculature can lead to endothelial damage, disrupting the endothelial barrier function and causing an imbalance in vasoactive mediators. This can trigger pulmonary vascular remodeling, ultimately resulting in irreversible pulmonary arterial hypertension (PAH) and right heart failure. 4
The clinical manifestations of PAPVR vary widely depending on the degree of shunting and associated cardiac anomalies. Symptoms range from asymptomatic cases to severe dyspnea, fatigue, or signs of right heart failure. The condition may remain undiagnosed for years, as its usual presenting symptoms such as dyspnea, right heart failure, and PH are not exclusive to PAPVR. Furthermore, it is not routinely investigated as a cause of PH, leading to frequent misclassification of PH as idiopathic or attributing it to coexisting lung diseases. 5
Transthoracic echocardiography is often the initial diagnostic tool for patients with suspected PH. However, the diagnosis of PAPVR is rarely made using TTE because standard imaging windows inadequately visualize pulmonary veins. 6 Although CT angiography is highly sensitive for diagnosing PAPVR, it is often overlooked during initial radiological evaluations due to its low incidence and a lack of clinical suspicion. 2 Consistent with other cases reported in the literature, the diagnosis in our case was made after obtaining a CT pulmonary angiogram to evaluate for pulmonary embolism. 7
Right heart catheterization (RHC), while essential for diagnosing PH, may not always detect the presence of a shunt. 8 Unless high SVC saturation above the innominate vein is measured, a step-up in oxygen saturation may go unnoticed, thereby missing the shunt. 9 In our case, the initial RHC did not detect a step-up in saturation because the high SVC saturation was not measured. At that time, PAPVR was not suspected, and the reported SVC saturation appeared falsely elevated due to the left-to-right shunt. However, subtle clues during RHC, such as discrepancies between cardiac output measurements, can raise suspicion. For instance, a higher cardiac output estimated by the Fick method compared to thermodilution may suggest the presence of a left-to-right shunt, as observed in our case. The calculation of systemic cardiac output by the Fick method is based on mixed venous oxygen saturation obtained from the pulmonary artery (PA). In PAPVR, the PA saturation will be higher due to left-to-right shunt leading to overestimation of cardiac output. However, this finding is not specific to PAPVR and can occur with other left-to-right shunts. 6
Diagnosing PAPVR presents unique challenges. The TTE is often inadequate, as the superior posterior atrial septum is poorly visualized, making the diagnosis rarely achievable through this modality. The gold standard for diagnosis is CT venography or cardiac MRI; however, these are not routinely performed in the evaluation of PH. While CT pulmonary angiography is highly sensitive, the rarity of PAPVR and lack of familiarity with the condition can lead to missed diagnoses.
Ancillary findings, such as the presence of a left-to-right shunt and a higher cardiac output measurement by the Fick method compared to the thermodilution method on RHC, may suggest the diagnosis. However, RHC can fail to detect a shunt if oxygen saturation measurements are not obtained above the level of the anomaly. Our case illustrates this challenge: on the initial RHC, SVC saturation was not measured above the shunt level, resulting in the failure to identify a step-up in saturation and, consequently, a missed left-to-right shunt.
In patients with unexplained PH, CT pulmonary angiography should be carefully re-reviewed with careful scrutiny of the pulmonary veins. Cardiac MRI is particularly valuable in detecting subtle anomalies and should be considered when PH remains unexplained or when a shunt is suspected but not confirmed through initial evaluations. 10
The management of PAPVR depends on the severity of symptoms, the magnitude of the shunt, and associated complications. Surgical correction is recommended in cases of right ventricular (RV) volume overload and when the pulmonary vascular resistance (PVR) is less than 3 Wood units (WU), and when the pulmonary-to-systemic blood flow ratio (Qp/Qs) exceeds 1.5. These criteria, based on data from atrial septal defect (ASD) studies and limited natural history series of PAPVR, aim to prevent irreversible pulmonary vascular remodeling, PH, and right heart failure. 11 The literature on PAH-specific therapy in PAPVR is limited. However, appropriately chosen patients, including those who are inoperable, may benefit from targeted therapy. 2
In conclusion, PAPVR involving the left upper lobe pulmonary vein is rare and difficult to diagnose. Echocardiography alone is insufficient for diagnosis, and the condition often goes unrecognized on CT angiography. 12 The RHC can provide valuable diagnostic clues, while early consideration of cardiac MRI and thorough review of CT angiograms are critical for timely diagnosis. Prompt surgical correction, when appropriate, can prevent irreversible pulmonary vascular remodeling and the progression to PH.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
