Abstract
Aim:
This systematic review critically evaluates in vitro studies on the osteoblastic behavior of human mesenchymal stem cells (hMSCs) and human osteoblast-like cells on three-dimensional (3D) bone scaffold materials. The review identifies key osteogenic proliferation and differentiation determinants and explores their potential clinical implications in oral and periodontal surgery.
Materials and Methods:
A comprehensive electronic search was conducted across Medline/PubMed, Embase, Scopus, and Web of Science for articles published up to May 3, 2025. Inclusion criteria were limited to in vitro studies, written in English, investigating osteogenic proliferation and differentiation of hMSCs or human osteoblast-like cells on xenograft granule-shaped or block-formed scaffolds. The risk of bias in the included studies was assessed using established parameters for cell culture research.
Results:
The review included 22 studies out of 931 screened articles. Deproteinized bovine bone mineral (DBBM) was the most frequently investigated biomaterial. Alkaline phosphatase (ALP) activity, osteocalcin immunoassay, and morphological analysis were identified as key tools for assessing in vitro osteoblast differentiation. Significant heterogeneity in scaffold composition and experimental design precluded meta-analysis, and results were synthesized narratively.
Conclusions:
Xenogeneic bone substitutes, particularly DBBM, demonstrate optimal osteoconductivity in vitro. While these in vitro findings suggest potential for clinical application, the heterogeneity of existing research highlights the need for well-designed in vivo studies and clinical trials to validate these results and establish optimal bone scaffold materials and strategies for oral and periodontal surgery.
Clinical Relevance:
Biofunctionalization and cell-to-scaffold contact are essential clinical requirements for attachment and integration of bone grafts. Biologically, a deeper understanding of the specific roles, reliability, and optimal conditions for applying bone substitutes in clinical applications is required.
Introduction
Regenerative dentistry is a captivating frontier with aspects yet to be fully understood and promising potential in terms of clinical relevance. In bone tissue engineering, a scaffold is an osteoconductive structure designed to support three-dimensional (3D) tissue regeneration by creating a stable and bioactive microenvironment for neoangiogenesis and cellular proliferation and differentiation. Combining these aspects is crucial for effective bone repair and integration with the host tissue, allowing the scaffold to support the initial stages of bone formation and ensuring the long-term functionality and stability of the regenerated tissue.1,2 The promotion of bone healing at the recipient site occurs through different mechanisms, which include osteoconduction and osteoinduction. 3 Osteoconduction is the capacity of a bone graft material to form a bioactive environment for host cell growth, allowing the formation of new bone along its surface. 4 Osteoinduction refers to the recruitment of host-derived stem cells to the grafted area, where local signaling proteins trigger their differentiation into osteoblasts that form new bone tissue. 5
Therefore, an ideal scaffold for bone tissue engineering should exhibit several key features: excellent biocompatibility; ease of application to the defect site, along with osteoconductive and osteoinductive properties; a biodegradation rate that matches the natural bone formation rate; a porous structure that enhances cell attachment, proliferation and osteogenic differentiation by mimicking the extracellular matrix; mechanical strength and the ability to bear load comparable to natural bone; a design that improves vascularization and integration with the host tissue.6,7 Until now, to the best of the authors’ knowledge, an ideal scaffold having all these features has not yet been found for bone tissue engineering and the closest graft having all these properties is autologous bone.
Autologous bone grafts remain the gold standard in regenerative procedures due to their superior biocompatibility and intrinsic osteogenic properties. In fact, they are the only grafting material that supports all three pillars of the bone regeneration triad (osteogenesis, osteoinduction and osteoconduction). However, it does require an additional surgical procedure, which can lead to increased donor site morbidity and limited availability. 8 For this reason, other types of biomaterials (i.e., allografts and xenografts) have been well-established in reconstructive surgeries due to their biocompatibility and osteoconductive properties.9–12 Despite detailed histological comparisons through in vivo human or animal models are valuable, assessing the degree of osteoconductivity might be challenging. In vitro studies using human mesenchymal stem cells (hMSCs) or human osteoblast-like cells (hOLCs) tend to yield more consistent and clear results, offering precious insights into material performance than in vivo models. 13 Different studies used hMSCs or hOLCs in in vitro model to evaluate osteoblast activity, bone formation, differentiation and responses to various stimuli or treatments.13–34 Bone formation is related to the adherence of osteoblasts to the graft material, which depends on the morphology of the graft surface structure. 20 Osteogenic cells’ migration and proliferation are largely influenced by the interaction between the cell membrane and the scaffold’s surface characteristics. 19 Beyond the architectural structure, surface features like micro and macro-pore sizes, interconnecting system, in vitro solubility, and chemical composition of bone substitutes seem to have a significant impact on the target cells. Thus, when selecting an appropriate bone substitute for clinical use, it could be useful to consider not only the material’s biomechanical properties and degradation rates but also how it affects cellular processes (migration, proliferation, and differentiation). In this context, Mayr-Wohlfart et al. 35 established that the pore size required for the successful integration and ingrowth of bone cells in scaffolds must be at least 100 µm to promote proper nutrient exchange and cell migration. Furthermore, both the interval between osteoblast seeding and the addition of culture medium, as well as the cell seeding density, have a substantial impact on osteoblast proliferation. According to Al-Ahmad et al. 33 the best proliferation rate and attachment were achieved using a seeding density of 105 cells/mL and with two hours adding the culture medium after seeding the cells. Similarly, Petrovic et al. 28 suggested that the density of cell seeding onto biomaterials should be carefully estimated and exceed 2 × 104 cells/cm2. In light of this, Bobbert and Zadpoor 36 highlight that the success of bone substitute materials largely depends on the initial seeding density and the material’s architecture. Too low density can result in insufficient cell interaction, while very high densities may cause overcrowding and reduce cell proliferation and differentiation. Typically, an optimal seeding density of 5 × 105 to 1 × 106 cells/cm² is recommended for osteoblast cultures, as these levels allow for efficient attachment, growth and differentiation on 3D scaffolds. However, adjustments may be necessary based on the specific scaffold and experimental conditions.
Furthermore, in vitro experiments are often carried out to test new potential biocompatible scaffolds for future clinical practice or to gain a better understanding of the effectiveness and the microscopical bio-interactions of widely-used commercially available biomaterials. In vitro outcomes showing an intimate interaction between osteoblasts and bone substitutes represent a necessary requirement to achieve positive clinical effects on the induction of vascularization and on bone regeneration and mineralization. Many studies investigated in vitro biocompatibility and osteogenic properties of different kinds of bone scaffolds, laying the groundwork for a more comprehensive understanding of osteoblasts’ growth on 3D biomaterials. Nevertheless, due to the heterogeneity of results, some clarity needs to be made.
Therefore, a systematic review was conducted on the existing literature about in vitro analysis of osteogenic differentiation of hMSCs and human osteoblast-like cells on widespread bone substitutes. In detail, the aim of this study was to systematically review the scientific literature on the in vitro osteoblasts’ behavior on the available 3D xenogeneic bone scaffolds, evaluating the cellular microenvironment and bio-interactions with biomaterials.
Methods
Protocol and Registration
This systematic review was conducted in accordance with the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines.
37
The methodology followed the JBI framework, and the protocol was registered on
The primary questions were formulated following the PICO criteria and were used to develop the search strategy: a) “What is the current state of knowledge regarding the biocompatibility and osteogenic properties of xenogeneic bone substitutes used in periodontal practice?”; b) “What is the in vitro evidence for the biocompatibility and osteogenic mechanisms of xenogeneic bone substitutes in periodontal applications?”; c) “How do xenogeneic bone substitutes influence osteogenic activity and tissue integration in dental bone regeneration procedures?”
Two reviewers (L.C. and F.R.) independently conducted an electronic search for English-language articles published in dental journals from January 1, 2000, until May 3, 2025, on the following databases: Medline/PubMed, Embase, Scopus and Web of Science. A manual search was also conducted among references of included studies. The search was based on a combination of medical subject headings (MeSH) terms and free text words as follows: “Mesenchymal Stem Cells,” “MSC,” “osteogenesis,” “human bone cell,” “osteoblasts,” “osteoblast-like cells,” “bone marrow cells,” “osteoblastic differentiation,” “bone regeneration,” “bone substitute,” “bone scaffold,” “bone graft,” “dental material,” “bone biomaterial,” “dentistry,” “dental practice,” “dental,” “In Vitro,” and “cell culture.”
All search strategies were initially based on the Medline/PubMed search strategy (Table 1) and subsequently adapted for each database to account for differences in controlled vocabulary and syntax conventions. References from various studies were included to identify relevant eligible studies. For study selection, two authors (L.C. and F.R.) independently reviewed the titles and abstracts of the studies according to the inclusion criteria. The final inclusion of studies was determined by screening and assessing the full texts, with decisions made by consensus between the two authors. Disagreements will be resolved by discussion and if necessary, a third reviewer (R.R.) will be consulted.
Search Conducted in Medline/PubMed Database.

Eligibility Criteria
In vitro studies investigating osteogenic proliferation and differentiation of hMSCs or hOLCs on xenogeneic bone substitutes.
In vitro studies focused on granule-shaped or block-formed scaffold.
In vitro studies using mesenchymal stem cells or osteoblast-like cells derived from human.
Studies written in the English language and published after 2000.
In vitro studies focused on granular scaffolds not loaded with osteogenic cells targeting bone regeneration.
Exclusion Criteria
Clinical trials, case reports, reviews, or animal studies.
In vitro studies using animal stem cells or animal osteoblast-like cells.
In vitro studies focused only on gel-hydrogel/fibrous/coralline scaffolds or composite and glass biomaterials.
Studies without details regarding stem cell origin, cell culture medium, bone substitute characteristics, time points appraisals, evaluation methods (proliferation and differentiation), or those that did not meet the aforementioned inclusion criteria.
Data Collection and Analysis
After implementing the search strategy in each database, the results were processed using spreadsheet software and systematically organized into folders to facilitate the screening process. Quantitative data were then extracted from the selected folders, with the primary objective of addressing the research question. Two researchers (L.C. and F.R.) were responsible for extracting data from all the included studies, while the results were reviewed, and any discrepancies were resolved by a third researcher (R.R.). Retrieved variables were: “cell type,” “culture medium,” “differentiation medium,” “cell density,” “bone substitutes,” “objectives,” “read-out.” To further ensure consistency in study selection, an additional analysis was conducted using the Cohen kappa coefficient on
Risk of Bias Assessment
Using the QUIN tool 38 for risk-of-bias assessment for in vitro studies as a reference, the methodological quality of the studies included in this review was evaluated based on the framework provided by Marques et al. 39 which considers essential parameters for conducting cell culture studies. Each study was assessed for the presence of specific methodological details, including cell type, culture medium, cell passage number, culture conditions, number of experimental replicates, and description of methodology to evaluate the outcome. Two independent reviewers (L.C. and F.R.) conducted the assessment, marking each item as “yes” if the information was available or “NR” (not reported) if absent. Articles that reported one to three of referred information were classified as having a high risk of bias; four or five parameters were classified as having a medium risk of bias; and six or seven parameters were classified as having a low risk of bias.
Results
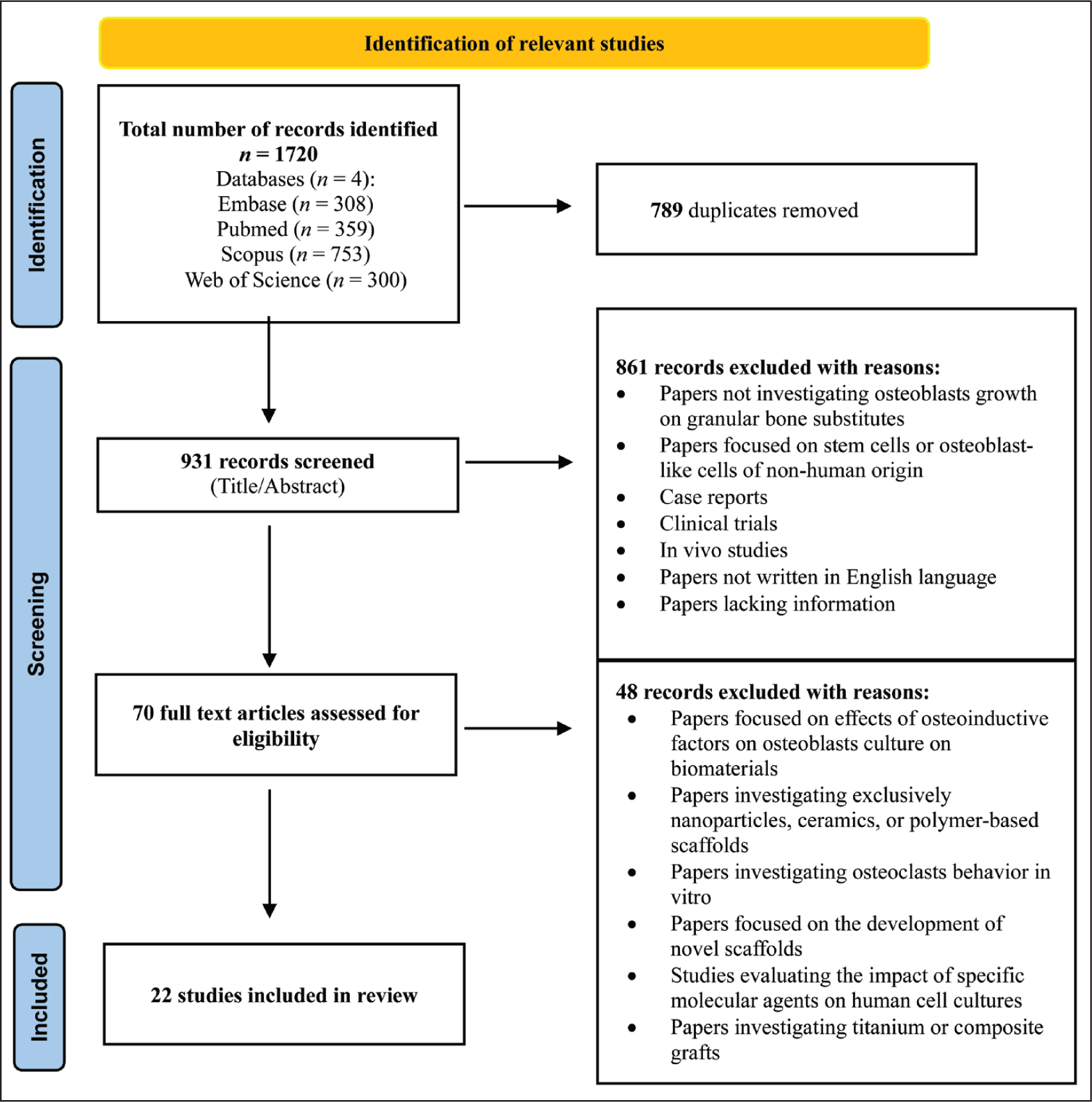
The flowchart according to the PRISMA checklist is reported in Figure 1. Based on the eligibility criteria and kappa coefficient calculations, the inter-examiner proportions of observed agreement between the two examiners were consistently high across all databases: 98.4% for PubMed, 98.3% for Embase, 96.2% for Web of Science, and 93.9% for Scopus. The initial database search produced 1,720 results. After duplicate removal, 931 articles were screened by title and abstract. Following this first assessment, 861 records were excluded as not relevant to the subject and 70 articles were selected for full-text analysis. After full reading, 48 articles were excluded because did not meet the established eligibility criteria, as reported in Figure 1. Finally, 22 studies13–34 were selected for the qualitative analysis and so included in the systematic review. Studies included in the systematic review and their variables of interest are reported in Table 2.
Flowchart of Literature Search and Study Selection.
Included Studies and Assessed Variables.






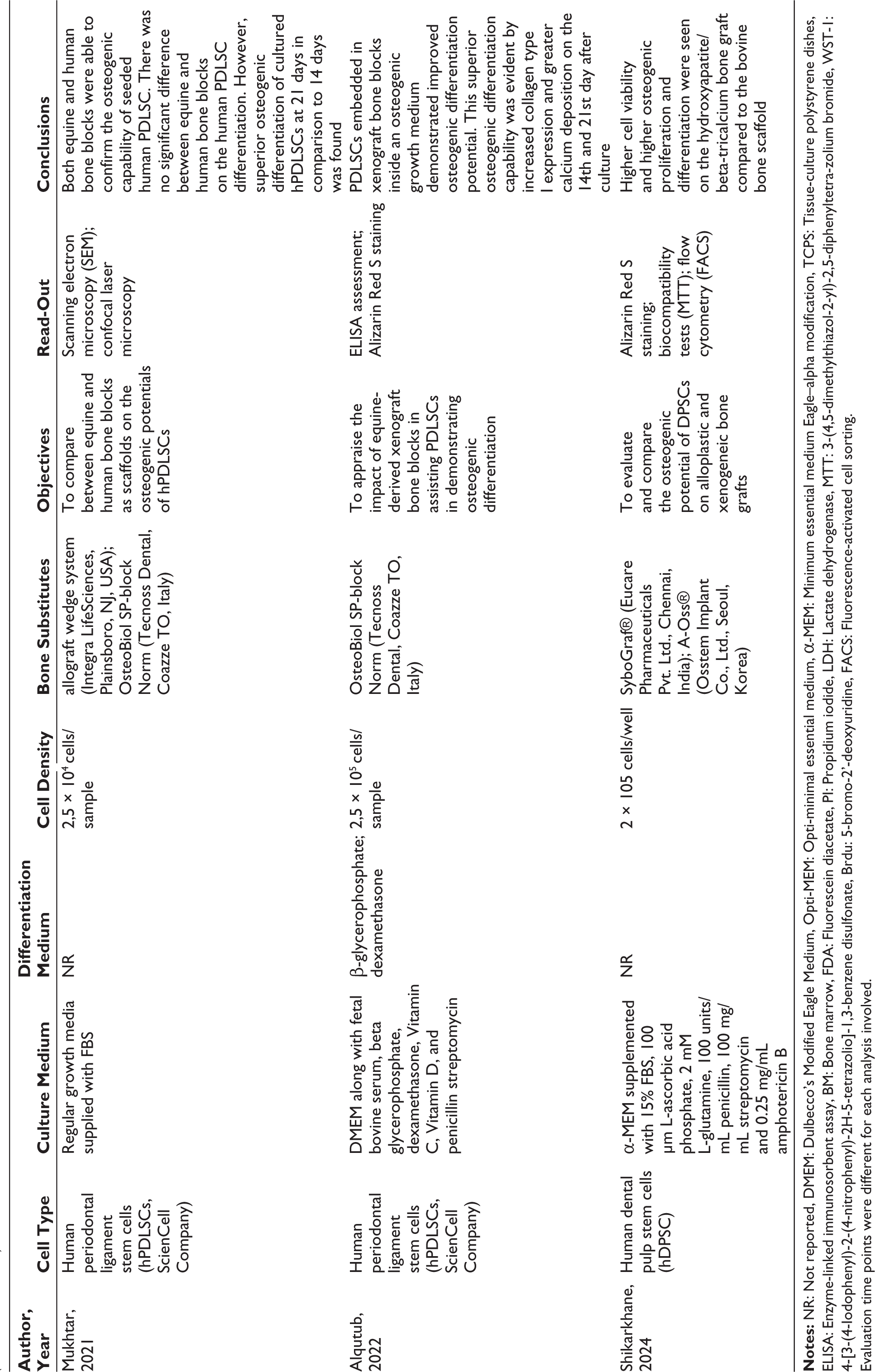
Evaluation time points were different for each analysis involved.
Among the included studies, different stem cell sources were used in the in vitro experiments such as ex vivo human bone cells,13,14,19–21,26,33, osteoblast-like cells,16–18,25,28,32, human periodontal ligament stem cells,15,23,24,30,31 ex vivo human bone marrow mesenchymal stem cells,19,27 and human dental pulp stem cells.22,29,34
A great variability of scaffold composition was found and the most investigated one was the deproteinized bovine bone mineral (DBBM) (n = 18), followed by b-tricalcium phosphate (n = 5), composite bone grafting material (an anorganic bovine-derived hydroxyapatite matrix combined with a synthetic peptide that mimics a specific sequence, P-15, found in collagen type I) (n = 3), equine xenograft material (n = 3), a-tricalcium phosphate (n = 3) and porcine xenograft material (n = 1).
There was a significant heterogeneity in applied biotechnology and evaluation periods, which were changed according to the specific type of biochemical or biological analysis being conducted.
In terms of differentiation evaluation, different methods were used such as alkaline phosphatase (ALP) activity (n = 10), reverse transcriptase polymerase chain reaction (RT-PCR) (n = 6) or quantitative reverse transcriptase polymerase chain reaction (qRT-PCR) (n = 1), osteocalcin synthesis immunoassay (enzyme-linked immunosorbent assay [ELISA]) (n = 5) and morphological cell analysis through scanning electron microscopy (SEM) (n = 14) and confocal laser scanning microscopy (CLSM) (n = 2). For the differentiation evaluation, considering the ALP activity (an early marker of osteo-differentiation), the shortest time-point found was 3 days 14 and the longest was 28 days, 31 both on DBBM material.
As a mineralization assay, a marker of advanced osteogenic cell differentiation, the most popular technique was Alizarin Red S staining (n = 6). Within a period of time of 5–40 days, all cell lineages showed osteogenic differentiation on DBBM, hydroxyapatite, and equine-derived bone material.
For the proliferation/viability evaluation, which stands also as biocompatibility tests, three studies20,21,28 employed the WST-1 assay (4-[3-(4-iodophenyl)-2-(4-nitrophenyl)-2H-5-tetrazolio]-1,3-benzene disulfonate), four studies16,21,32,34 employed the MTT assay (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetra-zolium bromide), two studies16,21 employed the LDH (lactate dehydrogenase) assay, two studies31,33 used the trypan blue exclusion technique, two studies21,26 used the Brdu assay (5-bromo-2′-deoxyuridine) and one study 26 used Crystal Violet staining. Additionally, two studies22,32 investigated a specific protein profile (Western Blot analysis) in human osteoblasts, while one study 17 exclusively focused on the genetic effects of a bone substitute on osteoblast-like cells. For proliferation/viability evaluation, the shortest period found was 10 minutes 21 for DBBM and nano-hydroxyapatite and the longest was 14 days 33 for all the tested materials.
In the context of osteogenic differentiation, the most frequently investigated genes were collagen type-I (Col1a1),18,26 runt-related transcription factor 2 (Runx2), 26 ALP,16,18,22,26 osteocalcin (or bone gamma-carboxyglutamate protein 2, Bglap2),13,18,19,22,26 osteopontin (Op), 26 osteonectin (On), 16 MEPE (matrix extracellular phosphoglycoprotein), 22 bone sialoprotein II (BspII), 16 b-actin 26 , and transforming growth factor-beta (TGF-β) 18 In all these studies, RT-PCR experiments normalized gene expression to the housekeeping gene GAPDH (glyceraldehyde 3-phosphate dehydrogenase). Data from the six studies analyzing genes expression showed controversial results.
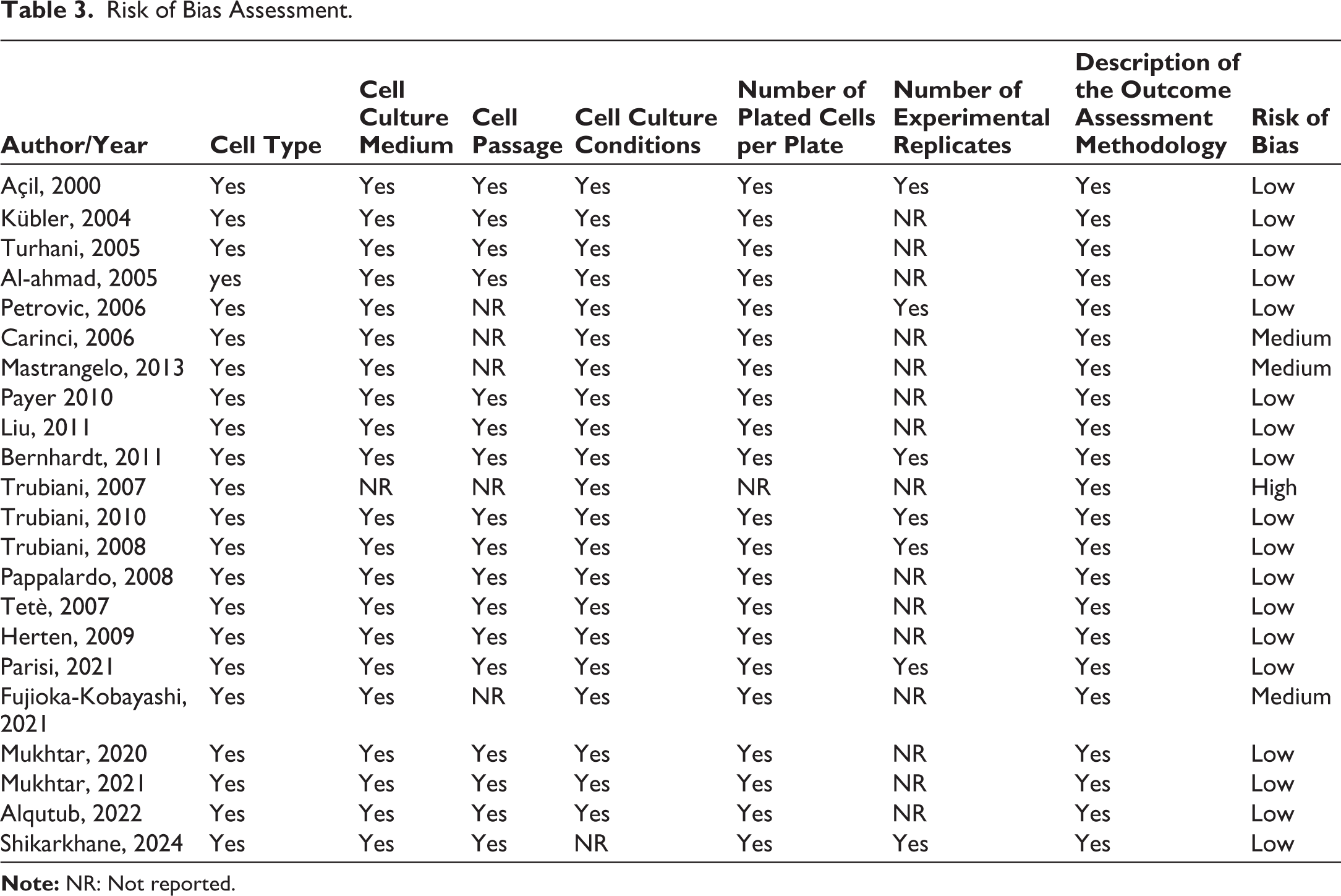
Regarding to bias risk assessment, only one study resulted at high risk, 30 while most of them showed a low score of bias. In particular, some of the included studies did not report information about the amount of cell passage (n = 5) and number of replicates (n = 15) (Table 3).
Risk of Bias Assessment.
Discussion
Numerous experimental studies have investigated different bone graft materials to identify an appropriate bone substitute which should be biocompatible and capable of supporting cell growth and differentiation.15,31 In a laboratory setting, scaffolds or matrices provide structural support to mimic the complexity and organization of natural tissue, offering a more accurate representation of cellular behavior in a structured environment. 40 However, despite their advantages, 3D culture systems are often limited by high costs, the need for specialized equipment, and challenges in replicating long-term bone remodeling processes. 41
The successful osseointegration of a bone substitute material relies on the activity of nearby bone cells or their precursors. Since cellular adhesion is a prerequisite for the proliferation of adherent cells, in vitro studies can be effective in assessing the biocompatibility of bone substitutes.18,23 Among the numerous commercially available bone graft materials, according to the current literature,18,23,25,26,31 DBBM is one of the most widely studied and clinically applied xenografts for regenerative purposes.2,42–46 This popularity is attributed to its chemical and structural similarity to human bone, high osteoconductivity, and slow resorption rate, which provides a long-term scaffold for new bone formation. Despite its advantages, DBBM is not without limitations. Unlike autografts, it lacks osteogenic and osteoinductive properties, relying entirely on host cells for bone regeneration. 5 Studies have reported variable results in terms of cellular response, biointegration, and long-term remodeling. 41 Some concerns include delayed resorption, which may impair complete bone regeneration, and foreign body reactions, potentially affecting clinical outcomes. 47 In this context, it ought to be mentioned that most of the materials evaluated show good clinical results in a high number of clinical studies.48–55 Nevertheless, more experimental studies13–33 have been conducted to test and compare the in vitro osteoblasts’ behavior on xenograft granule or block-type bone substitute biomaterial with a huge heterogeneity of results.
However, a meta-analysis was not conducted due to substantial heterogeneity among the included studies. Specifically, notable variations were identified in scaffold composition and structural properties, cell seeding densities, experimental protocols (such as culture duration and osteogenic induction methods), and the outcome measures employed. This methodological and biological variability precluded the possibility of performing a meaningful quantitative synthesis. Consequently, a narrative synthesis was undertaken to integrate and interpret the findings across studies qualitatively.
To fully understand the complex process of osteogenic differentiation, molecular pathways should be taken into consideration. The differentiation of osteoblasts is a complex and orderly process orchestrated by signaling molecules operating at both the local and systemic levels. Numerous signaling pathways have been identified as key regulators of osteoblast specification and differentiation.54,55 According to our review, all authors who reported differentiation media consistently used three key factors, either individually or in combination, to promote osteoblast differentiation: β-glycerophosphate (β-Gly), dexamethasone (Dex), and ascorbic acid (Asc). Usually, DAG (how the combination of Dex, Asc and β-Gly is called) is used to create an osteogenic differentiation medium because it can mimic the natural bone microenvironment.54–56 These products facilitate osteogenic differentiation by enhancing the activation of distinct intracellular signaling pathways in the expression of Runx2 (a key transcription factor in osteoblast differentiation and bone formation).
For these reasons, in the context of differentiation evaluation, the Runx2 gene marker should be included in RT-PCR analysis as an evaluation methodology of osteoblast differentiation because Runx2 upregulation is the evidence that osteo-differentiation factors (DAG) are effectively influencing the cell culture. Among the included articles, three authors18,22,26 have involved Runx2 into the RT-PCR gene marker profile. Runx2 has an activation domain that turns on the genes for osteocalcin (OC) and COL1A1 in addition to its conserved DNA-binding domain. 55 Additionally, osteoblasts alter their gene expression and produce a specific protein pattern depending on their functional requirements and their distinct stage of differentiation. Runx2 plays a prominent role in osteogenesis when pluripotent mesenchymal stem cells are committed to the bone cell lineage before expression of the osteoblast phenotype. Col1, OPN, and ALP represent the early stages of osteoblastic differentiation, characterized by the proliferation, synthesis, and maturation of the cellular matrix. Subsequently, OC indicates the onset of mineralization, while bone sialoprotein (BSP) serves as a late-stage marker of bone formation.54,55 RT-PCR has proven to be a reliable experimental method for evaluating the expression of gene markers involved in osteoblastic differentiation. According to more articles included in the current systematic review,18,22,26 ALP and OC mRNAs increased in a time-dependent manner with the development of the osteoblast phenotype and differentiation, proliferation, and growth rates on DBBM samples were greater when compared with the other experimented biomaterials. Specifically, Fujioka-Kobayashi et al. 18 observed significantly higher mRNA levels for Runx2 and OC at 14 days in cells cultured on DBBM small-size granules compared to those on DBBM large-size granules. This suggests that the increased surface area of the smaller DBBM granules may promote differentiation more effectively than the larger granule scaffolds. Furthermore, as reported by Parisi et al. 26 the biological pre-activation of DBBM using porcine bone-conditioned medium (a combination of blood-derived growth factors and bone substitute) did not result in significant changes in the expression levels of osteogenic differentiation markers at the mRNA level. Instead, according to other authors,13,16,19 genetic marker expression in human osteoblast cells in the presence of DBBM was significantly lower compared to HA+P15 and hydroxyapatite. Nonetheless, Turhani et al. 13 did not report any cytotoxic effects of DBBM but showed reduced activity in cells grown on this type of biomaterial in comparison to HA+P15. The authors attributed these differences, contrasting with other in vitro and clinical findings, respectively to the distinct compositional characteristics of the compared composite biomaterials and the direct involvement of osteoblasts in bone metabolism and calcium/phosphate homeostasis. Conversely, Bernhardt et al. 16 were unable to perform gene expression analysis of osteogenic markers on DBBM due to insufficient RNA extraction, caused by the very low number of attached cells at all time point investigations. To understand the drastic outcome of this study compared to the other included articles, it is essential to consider the experimental setup, which enabled the quantitative evaluation of only the cells that adhered directly to the material’s surface. Regarding the genetic effects of DBBM on osteoblast-like cultures, Carinci et al. 17 showed the ability of granule-shaped bone substitute materials to upregulate certain functional activities of osteoblast-like cells, including cell cycle regulation, signal transduction, immunity, apoptosis, and vesicular transport. This significant influence on gene expression may stem from the inherent ability of these biomaterials to mimic the natural bone matrix during the graft osteointegration, supporting cell migration, attachment and osteoblastic differentiation.
In addition to gene expression analysis of osteogenic markers, successful osteoblastic differentiation and bone tissue formation can be evaluated in vitro by assessing ALP activity and performing Alizarin Red S staining. Osteoblast-like cells are known to exhibit maximal proliferation and (ALP) activity between days 7 and 10. Thereafter, these parameters tend to decrease while osteocalcin and osteonectin expression rises, along with enhanced nodule formation. 57 For these reasons, ALP analysis should not be performed beyond 10 days in 3D culture system experiments, as a shift of the cells to a secretory phase is physiologically expected due to cultivation under osteogenic differentiation conditions, where proliferation decreases. Instead, for long-term investigations, it is preferable to use other outcome assessment methods, such as osteocalcin synthesis immunoassays and viability/proliferation assays. Although unequivocal cytocompatibility, some articles included in this review did not report favorable results regarding ALP activity on the DBBM scaffold due to excessively long time-point evaluations (over 10 days)13,28 or found no statistically significant differences in ALP activity between bone mineral and other tested biomaterials.16,20,27 A decrease in ALP activity could indicate either a regression to osteoprogenitor cells or progression to osteocytes, which typically express low levels of this enzyme.58,59 From a biochemical standpoint, the reduced ALP activity might also be linked to phagocytosis, as an increase in intracellular calcium could exert an inhibitory effect. 13
In this context, where mineralization is considered a functional endpoint in in vitro research reflecting advanced osteogenic differentiation, Alizarin Red S staining represents a useful and effective method to assess calcium deposition levels. Its relevance lies in the dye’s ability to selectively bind calcium-rich extracellular matrix, thereby indicating active bone matrix formation.60,61 More studies included in this review19,22,24,30–32,34 proposed this technique as a mineralization assay to demonstrate the ability of biomaterials to support the formation and long-term maintenance of organized and structured bone tissue.
Finally, one of the most direct and widely used methods for studying osteoblastic differentiation is the morphological analysis of the cell/scaffold complex using SEM, transmission electron microscopy (TEM) or CLSM. Bone tissue engineering’s success is dependent on the synergistic interaction between the progenitor cells and the grafts and cellular morphology has traditionally served as a crucial indicator for identifying the formation of new cells and assessing their overall quality. Furthermore, changes in cell morphology, such as a shift from a spindle-like to a more cuboidal shape, accompany the differentiation process, further indicating the transition of MSCs towards an osteoblastic lineage. Matsuoka et al. showed that cell geometry is highly correlated with the differentiation into osteogenic lineages and so SEM images can be a valid method for the assessment of cellular morphology. Several reports showed that cell geometry is highly correlated with the differentiation into osteogenic lineages.62,63 In fact, most studies included in this systematic review have considered and characterized osteoblast morphology on biomaterials as an indirect measure of cell health, vitality, and function within its microenvironment. From a morphological perspective, cells observed after 14–21 days of experimentation consistently exhibit a flattened, spread-out, polygonal shape, with extended cytoplasmic processes indicative of their osteoblastic differentiation. According to Mukhtar et al. 24 osteoblast cells displayed extending filopodia and fiber-like processes that connect the newly formed cells and the scaffold, forming intercellular bridging and anchorage. Similarly, Tetè et al. 29 demonstrated that cells seeded onto DBBM formed a multilayer culture that gradually spread across the biomaterial granules. Confirming the high osteoconductive capacity of the granular biomaterial, “bridges” were formed at the base of the particles, enabling other cells to move upward along the micro rough surface of the biomaterial, defying the force of gravity. In this context, as described by Pappalardo et al. 25 scaffolds became progressively colonized by cells interconnected through fine, delicate filaments. Over time, the granules’ surface was fully covered by cells, creating a “tent effect,” where the spaces within the biomaterial’s structure were sealed off by overlapping cells rather than being filled (perhaps to be filled later). Additionally, the star-like shape of the cells in close contact with the bovine bone, membrane specialization, exocytotic vesicles, the formation of a multilayered cell “shell: around the granules and the appearance of calcium as a result of ongoing mineralization, all suggest a differentiation effect induced by the deproteinized bovine bone.
Nevertheless, cell adhesion on xenogeneic bone minerals remains a controversial topic in the literature, as some authors have not observed direct contact between osteoblasts and the tested biomaterials. In a comparative study of 16 different biomaterials, Al-Ahmad et al. 15 reported that anorganic bovine bone substitute shows the lowest proliferation rate because of the smooth surface of this biomaterial which affects the adherence of the cell. These findings were consistent with those reported by Kübler et al. 20 who attribute the lack of effective cell adhesion to the smoothness of the material’s surface. Anyway, images of the SEM can be successfully used to identify the morphological characteristics of the newly formed cells.
Understanding the physical and biochemical interactions between human cells and xenogeneic bone substitutes could guide clinical engineering in the development of biomaterials capable of influencing and promoting osteogenic processes. In this context, in vitro observations provide a valuable reference for future research in cell therapy.
One of the main advantages of in vitro studies is the enhanced reproducibility of results and the adhesion to the essential parameters is fundamental for its achievement. Indeed, different in vitro aspects could influence cell viability, proliferation and osteodifferentiation such as cell density, concentration of the bone substitute material, scaffold morphology, and evaluated time points after reaching confluence. All these factors can interfere with experimental outcomes and may help to explain the varying results obtained by different research on this topic. Therefore, a risk of bias assessment table has been generated in this systematic review (Table 3). All the included articles used a negative control group and reported the description of the outcome assessment methodology, but most of them did not report data about experimental replicates. This information is crucial for validating the reliability of results and for supporting the inclusions of the studies for a systematic review, allowing an accurate comparison.
The main limitation of this review is that it takes into account in vitro experimental models. These kinds of models are unable to fully replicate the real conditions within the body, regardless of the cell line selected or the bone substitutes used. To gain a more comprehensive understanding of osteoblast biofunctionalization on xenogeneic bone substitutes, as well as molecular interactions and modifications in the cellular environment, the findings of this systematic review should be supplemented with reviews of clinical and pre-clinical studies, including in vivo and animal model research. However, this systematic review has shed light on the field of xenogeneic bone substitutes in dentistry, where the economic interests of companies often result in the commercialization of new products without appropriate pre-clinical studies. Due to the variability in study designs, scaffold types and assessment methods, it remains challenging to draw definitive conclusions regarding the clinical characteristics of these materials. Standardizing in vitro protocols in future research could help to enhance data consistency and provide more comprehensive insights for their potential clinical applications in dental practice.
Conclusions
The findings of this systematic review support the following conclusions:
Xenogeneic bone substitutes have demonstrated good osteoconductive properties, promoting cellular attachment and proliferation. However, their direct osteoinductive capabilities remain limited. Among xenogeneic biomaterials, DBBM stands out as the most widely used and studied bone scaffold, compared to equine and porcine-derived alternatives. In vitro studies are valuable tools for enhancing our understanding of the mechanisms underlying bone regeneration and for exploring how biomaterials influence cell fate, organization and interactions within their microenvironment. In vitro observations serve as a critical foundation for advancing future research in cell therapy. ALP activity, gene expression analysis, Alizarin Red S staining, and morphological evaluation are effective methods for assessing osteoblast differentiation and overall culture health in biocompatibility studies. It is crucial to standardize laboratory procedures to ensure the comparability and reproducibility of results across different studies.
Footnotes
Authors’ Contributions
All authors have read and approved the final version of the manuscript.
Data Availability Statement
The data sets generated and analyzed during the current study have been provided and are available within the submitted manuscript.
Declaration of Conflicting Interests
The authors do not have any financial interest in the companies whose materials are included in this article.
Ethical Approval
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
