Abstract
Aim:
The purpose of the current investigation was to determine by radiographic means if a calcific dentinal bridge had formed following direct pulp capping (DPC) with calcium hydroxide (CH), mineral trioxide aggregate (MTA), or a combination of platelet-rich fibrin (PRF) and MTA.
Materials and Methods:
In the beginning, 45 systemically healthy patients who needed DPC and had pulpal exposure were enrolled in the trial. They were then randomly assigned to one of three groups: CH (control, n = 15), MTA (n = 15), or PRF + MTA (n = 15). To determine the radiographic density of the dentin immediately underneath the location of pulpal exposure, pre-operative intraoral digital radiographs of the afflicted teeth were acquired. Through sequential imaging, the radiographic evaluation of dentinal bridge formation was performed. All images were studied carefully, keeping the reference point constant.
Results:
Out of the 45 patients, finally 10 per group were analysed. According to an analysis of variance, difference in mean density values among the various groups was found to be significant [(p = .001). On bi-group comparison (using post-Tukey HSD), difference between CH and PRF + MTA was significant (p = .001), between MTA and PRF + MTA was significant (p = .022), and between CH and MTA not significant (p = .347)]. The PRF + MTA group showed the highest increase in mean density values.
Conclusion:
The results of the present study suggest that PRF and MTA when used in combination have better radiographic outcomes compared to MTA alone when used as DPC agents.
Keywords
Introduction
Vital pulp therapy (VPT) is described as “the techniques which aim to treat reversible pulpal injury, whenever dentin and pulp are affected by caries, restorative procedures, or trauma” by the American Association of Endodontics. Using the dentinogenic potential of the pulpal cells, pulp capping is one such treatment strategy that protects pulp vitality in cases of reversible pulp injuries brought on by trauma, iatrogenic exposure, or mechanical exposure during the removal of deep caries.1,2 Direct pulp capping (DPC) involves covering exposed pulp with a bioactive material with the goal of causing differentiation of odontoblast-like cells that would eventually produce reparative dentin at the pulp-capping material interface by enhancing the biosynthetic activity of adjoining primary odontoblasts, 3 and, the reconstruction of the dentinal defect with a bridge made of reparative dentin that is directly continuous with reactionary dentin around the pulp exposure is the most desirable result. 4
Since its inception from the use of gold foil and calcium hydroxide (CH), the concept of DPC has come a long way.5,6 Today, scientists are discussing bioactive and biomimetic materials, which have conductive qualities that encourage the creation of hard tissues. The idea of tailored pulp capping materials is even more appealing than innovative materials like premixed bioceramics 7 or melatonin 8 are under research, even more welcoming is the concept of targeted pulp capping materials. 9
Mineral trioxide aggregate (MTA), a bioactive, calcium silicate-based cement, brought a revolution in the field of repair and regeneration cementum and dentin and has generated widespread acceptance as a DPC material owing to properties like superior sealing ability, low solubility, and initiation of reparative dentin formation through the hard tissue forming cells. 10 Numerous DPC studies have shown that MTA induces dentin bridge building with minimal to no inflammation and improved success rates in sustaining pulp vitality for extended periods of time.11–13
Like in other aspects of dentistry, there is a paradigm shift toward the use of regenerative biomaterials for management of pulp exposures, namely, growth factors, Emdogain, and matrix-derived proteins. 14 Platelet-rich fibrin (PRF), a second-generation platelet concentrate, is an autologous bioactive material consisting of leukocytes, platelets, and an array of crucial healing proteins in a compact fibrinous milieu. 15 The PRF membrane releases a sizable number of cytokines, including platelet-derived growth factor (PDGF), transforming growth factor b1 (TGF-b1), and vascular endothelial growth factor (VEGF). 16 Though it has used extensively in pulpotomy and has shown successful results, its efficacy as a DPC agent is a comparatively recent area of research.17,18
The present clinic-radiographic study aims to study the effect of combined use of MTA and PRF, as compared with MTA alone, as depicted by an increase in radiographic density beneath the point of pulpal exposure the null hypothesis stated was that there would be no increase is radiographic density when using PRF in combination with MTA compared to MTA alone. To the best of the authors’ information, no clinical trial comparing outcomes of PRF in combination with MTA for direct pulp capping has yet been published.
Materials and Methods
Setting and Design
After being approved by the institution’s ethical committee, the study was carried out in conformity with the Declaration of Helsinki’s ethical principles for medical research involving human beings (King George's Medical University, Lucknow, India; Reference no. ECM II B-Thesis/P-4). Systemically healthy patients in the age group between 20 and 35 years, and having iatrogenic mechanical pulpal exposure or a deep carious lesion where pulpal exposure was anticipated during caries excavation were recruited for the study. Teeth where carious exposure did not occur after removal of soft caries, or where the exposure area was contaminated, teeth which were tender to percussion, having a negative or a very delayed response to electric and thermal tests when compared to healthy contralateral tooth, or radiographic evidence of any periapical lesion were excluded from the study.
This clinical pilot study was conducted over a period of two years. Power analysis was conducted and a minimum sample of seven patients in each group was required to ensure adequate sample size at 80% power. Study subjects were divided into three treatment groups according to the DPC protocol used; namely, Group I control group, CH, and Group II MTA, Group III PRF + MTA. Allocation concealment was done to allocate the subjects into various treatment groups. Sequentially numbered, opaque, sealed envelopes were prepared by an endodontist blind to the study and kept safely. Group information notes about the treatment to be given were packed into these sequentially numbered opaque envelopes and were opened only at the time of treatment. A single operator performed all the steps of the DPC procedure to remove inter-operator bias. However, outcome assessment on follow-up was performed by another endodontist blind to the intervention given to the patient and was not a part of the study. Subjects agreeing to participate in the study were explained about the treatment protocol and alternate treatment options and written informed consent was obtained.
Intervention
After a pre-rinse with 0.12% chlorehexidene mouthwash (Clohex Mouthwash, Dr Reddy’s Lab, India), administration of 2% lignocaine with 1:100,000 epinephrine (Lignox; Indoco Remedies Ltd, Mumbai, India), and rubber dam isolation (Hygenic Dental Dam; Coltene Whaledent, Langenau, Germany), caries was removed initially with a large round bur (number 2, diameter 1.0 mm, Mani Incorp., India) and then with a sharp spoon excavator to prevent a large exposure. Caries excavation was continued till dentin felt hard on excavation. Homeostasis was achieved by pressing the exposed pulp with 3% sodium hypochlorite (Parcan, Septodont, India) soaked cotton for 2–3 minutes. Cases where homeostasis was not achieved even after 5 minutes were not included in the trial.
For control group (CH), after achieving homeostasis, a CH liner (Dycal Ivory, Dentsply) was placed over the exposure site and some approximating healthy dentin. After setting of CH, tooth was restored temporarily with Cavit (3M, ESPE, Seefeld, Germany). If the patient was found to be asymptomatic after 24 hours, a GIC liner (Fuji Type 1, GC Dental India) was placed over the Dycal, and tooth was restored with a restorative resin.
After combining cement powder with distilled water in the MTA Group at a 3:1 powder-to-liquid ratio, the exposure site was coated with white MTA (ProRoot MTA, Dentsply). An MTA applicator (Angelus, Londrina, Brazil), the mixture was used to apply MTA to the exposure site and softly condensed with a moistened sterile cotton pellet to assure making direct contact with the exposed pulp tissue. The patient was called back the following day to check that the MTA was set permanently, and the tooth was rebuilt as for the CH group.
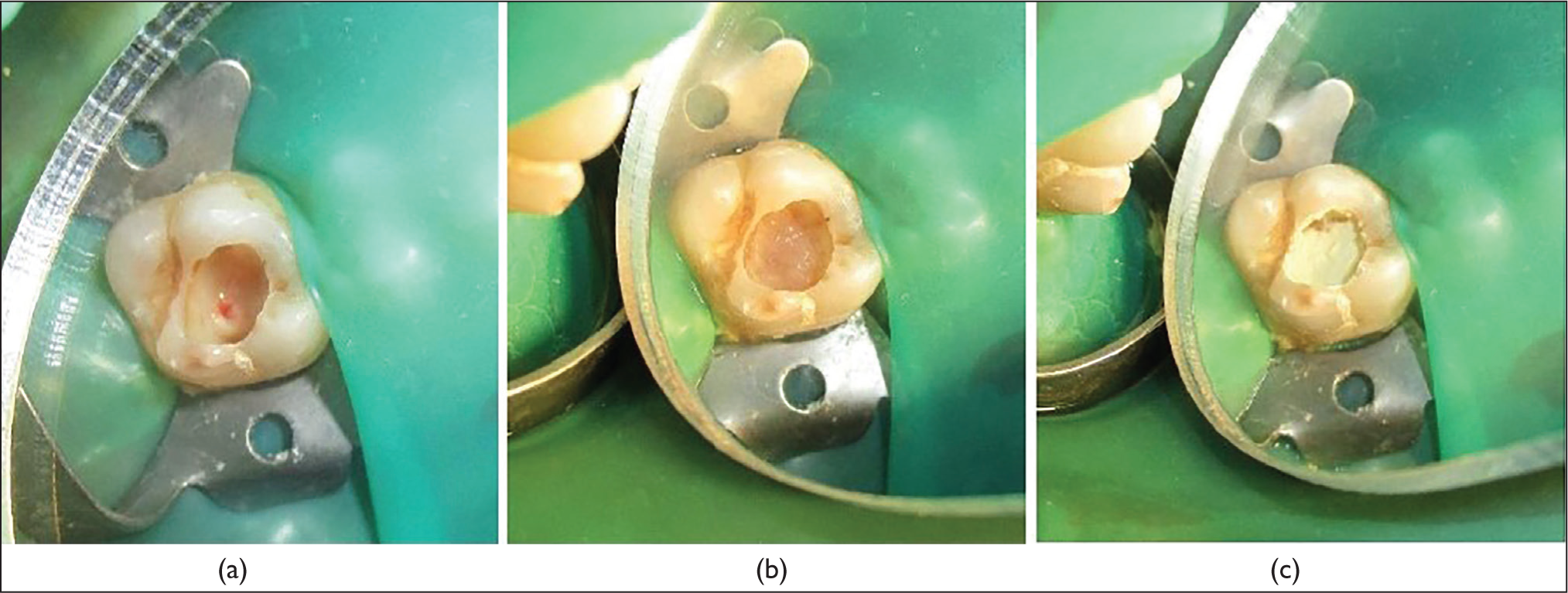
For (PRF + MTA) group, after initial caries removal bleeding control, the exposure site and surrounding dentin were covered with PRF followed by a layer of MTA. A volume of 5 mL of blood was collected from the patient’s forearm and placed into a glass test tube without the use of an anticoagulant in order to get PRF. A tabletop centrifuge (REMI Laboratories, Mumbai, India) was used to quickly centrifuge a blood sample for 10 minutes at 3,000 revolutions per minute. The resulting material consisted of three layers: red blood cells at the bottom, PRF in the middle, and acellular platelet-poor plasma at the top. A segment of the membrane from which the PRF was squeezed was then cut, and it was applied to the exposed area in direct contact with the exposed pulpal tissue (Figure 1a and b). The tooth was temporarily repaired by covering the PRF with a 3 mm thick coating of MTA (Figure 1c). The following day, permanent restoration using glass ionomer cement was carried out in order to provide MTA adequate time to complete final setting and restore like the other groups. Patients were given an anti-inflammatory medication on an S.O.S. basis for all the groups and were instructed to report back in case of any adverse event of pain, tenderness, discomfort following the pulp capping procedures.
(a) Pin-point Exposure in Maxillary Molar, (b) PRF Covering the Exposure Site, (c) Layer of MTA Placed Over the PRF.
Assessment
For all three groups, patients were evaluated once in a month till six months. On each appointment, the tooth was evaluated clinically for any change in color in the coronal area of tooth, pulpal status, any clinical signs of palpation or tenderness on percussion, the presence of any sinus, swelling, or mobility.
The method of radiographic assessment of calcific bridge formation beneath the area of pulpal exposure in the present study was based on clinical research by Lee et al., 19 where the outcome was measured by noting the dentin bridge thickness underneath the area of pulpal exposure. In the present study, an intraoral digital radiograph was taken for each participant using Intraoral digital imaging plate system (Soredex, Digora Optime, Finland) and an intraoral X-ray unit (ACTEON X-mind, Norwich, UK) at 50 Kvp and 10 mA. The Digora software for Windows, version 2.9 has a tool for density measurement, which automatically calculates the mean grey shade values at the selected points. Radiographic assessment of any dentinal bridge formation was done through sequential imaging. All images were studied carefully, keeping the reference point constant.
Observational Parameters
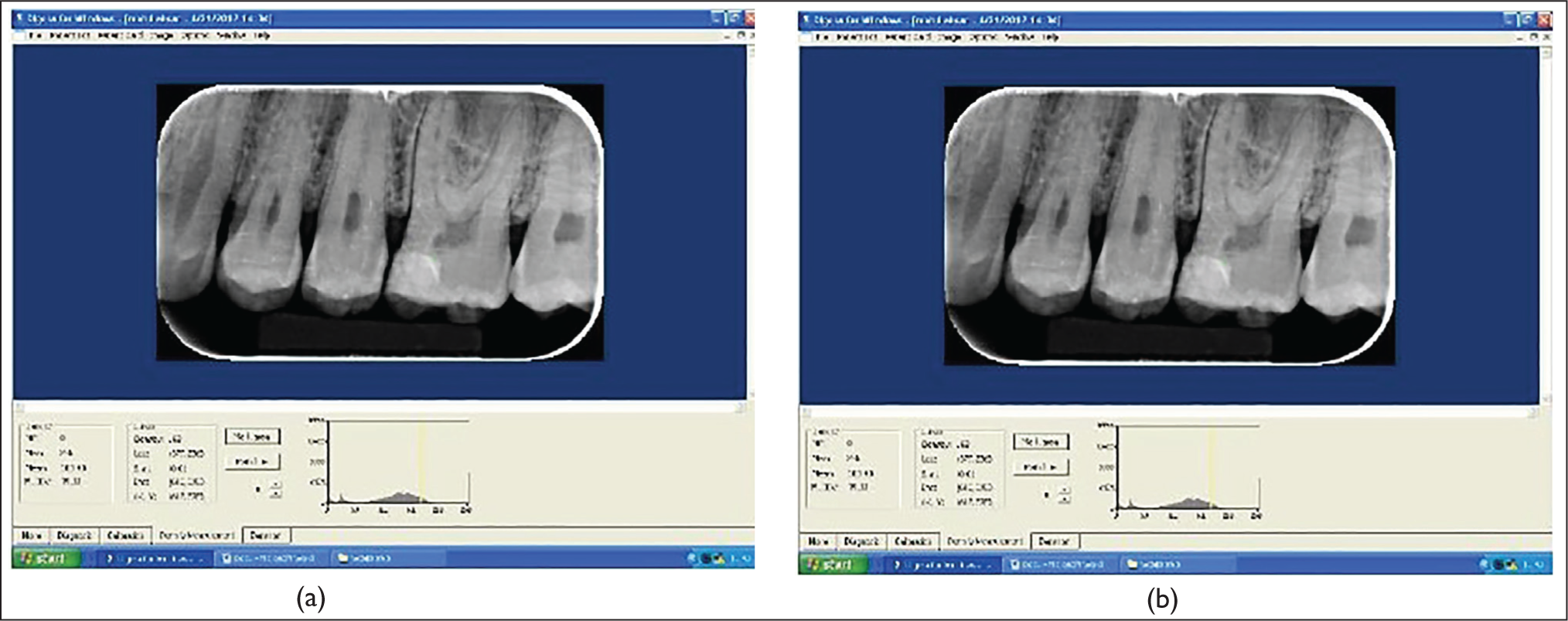
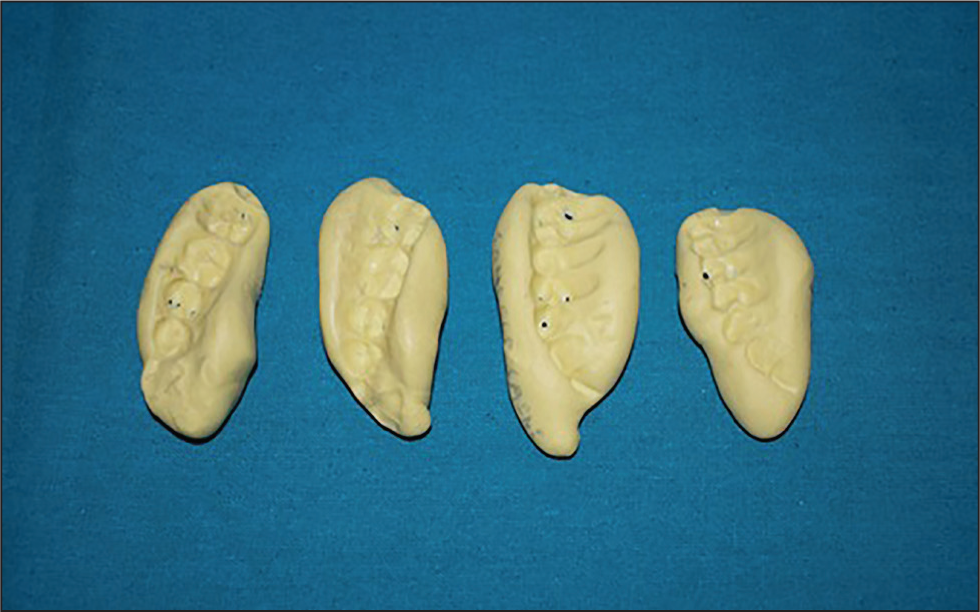
Co-ordinates of the location below the exposed portion of pulp were marked and recorded for density measurement using the SOREDEX software for all pre-operative, postoperative, and follow-up radiographs (Figure 2a and b). The position was kept constant on each follow-up appointment with the help of an IOPA positioning device (Rinn XCP, Dentsply Sirona), and a patient’s bite position recorded in silica for replication in follow-up appointments (Figure 3). To reduce error, three points were selected below the exposure area on pre and postoperative radiographs. These three points were most mesial (d1), middle (d2), and most distal (d3) points of the exposure area. Mean of readings of these three points was calculated and compared with six months follow-up radiographs at the same points.
(a and b) Screen Shot of the Digital Radiography Software Showing Density Measurement at Baseline and After Six Months.
Custom Silicon Index for Adjusting the X-ray Holder.
To remove inter-observer bias, the pre and post-radiographic assessment was done by a single observer, who was blinded for the group allocation.
Data were summarized as mean ± SD. Groups were compared by two-way analysis of variance (ANOVA) and the significance of mean difference within (intra) and between (inter) the groups was done by Tukey’s HSD post hoc test. Statistical analysis was done using SPSS version 19.0 (SPSS Inc., Chicago, IL, USA).
Results
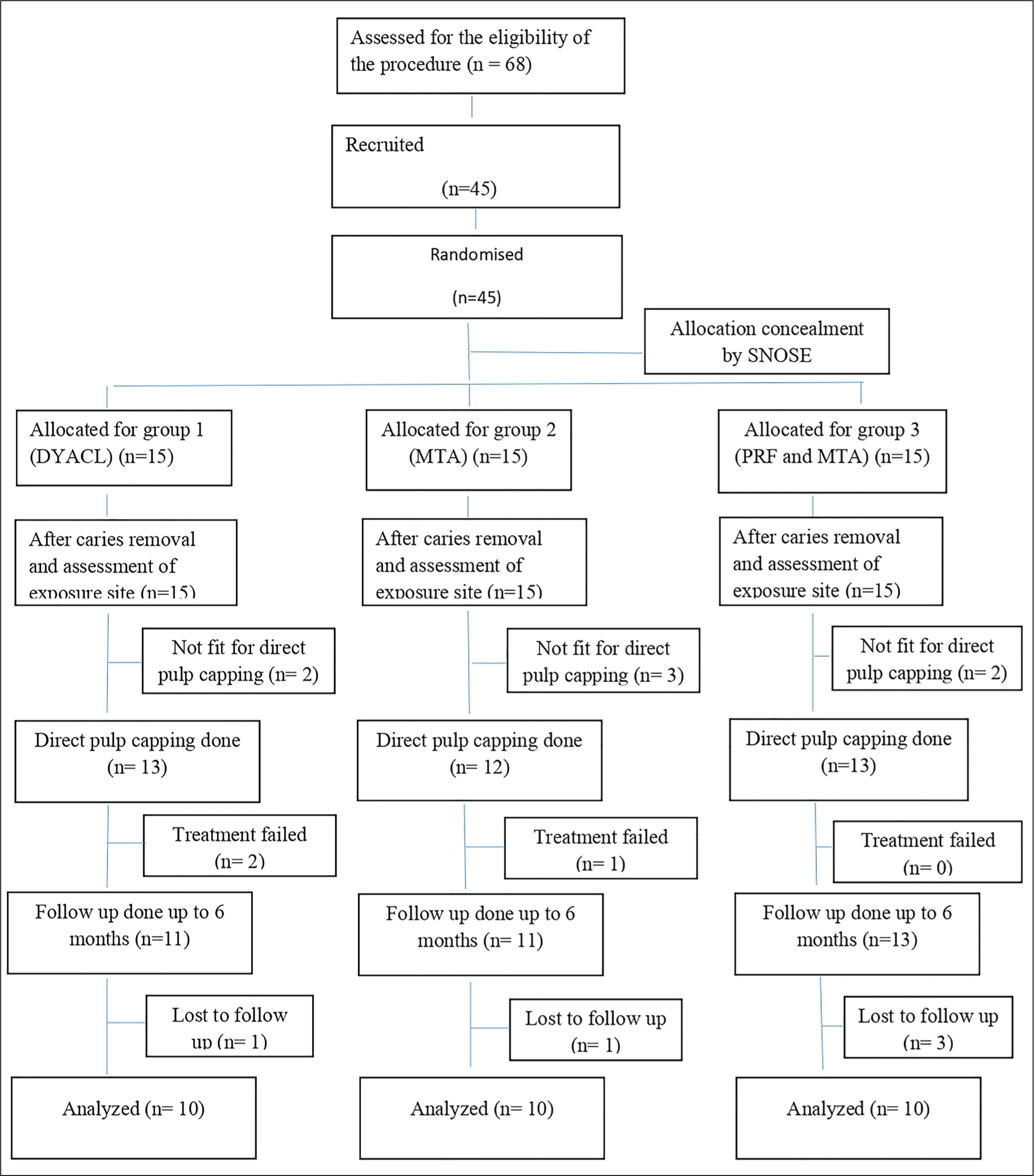
Out of 68 patients evaluated for eligibility, 45 patients conformed to the inclusion criteria and were enrolled in the study, leading to 15 patients per group. Few cases were excluded after initial caries excavation (2 for CH group, 3 for MTA group, and 2 for MTA + PRF group). Treatment failed in two patients in CH group and in one patient in MTA group. No DPC failed in the PRF–MTA group. A total of five patients were lost to follow-up. Finally, 10 patients per group were analysed per group. The flow of patients recruited, excluded, and finally analysed is depicted in Figure 4.
Flowchart Showing the Recruitment of Patients.
On comparing the post-pulp capping and six-month follow-up density differences, it was observed that the density difference was maximum for PRF + MTA group (mean 28.90 ± 12.15) followed by the MTA (18.13 ± 6.91). The minimum density change was observed for CH with a mean of 12.77 ± 4.47. The ANOVA test revealed that there was a significant difference in the mean density changes between the groups (p = .001) (Table 1).
Mean Density Change After Six Months Follow-up.
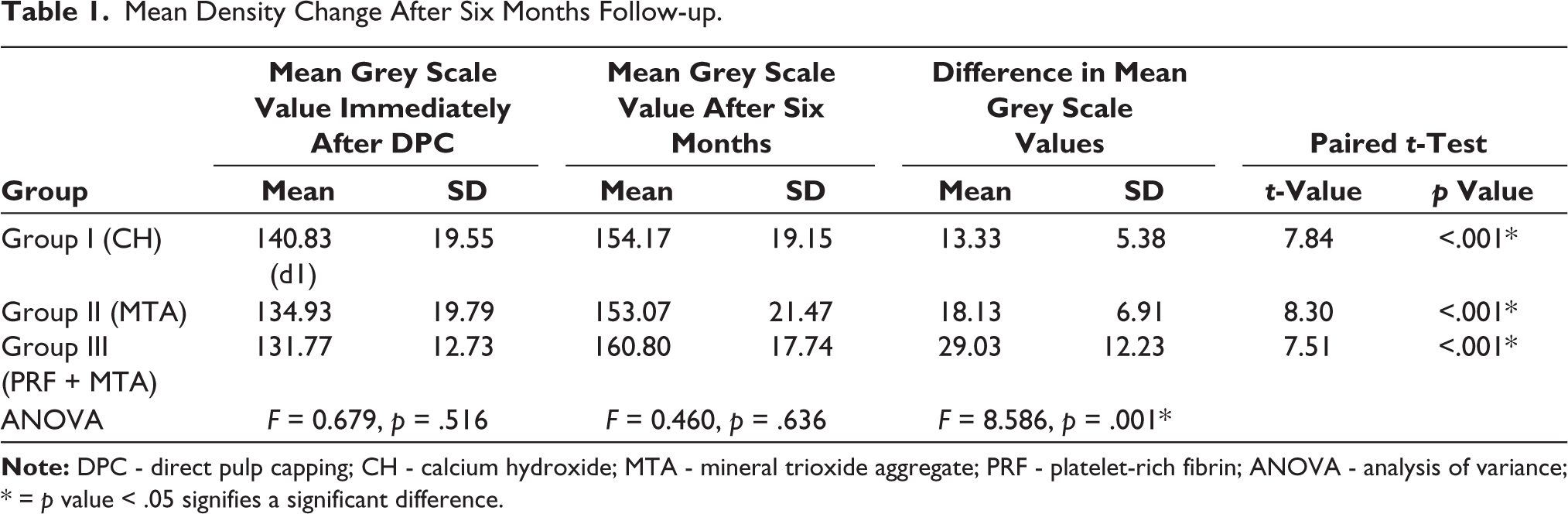
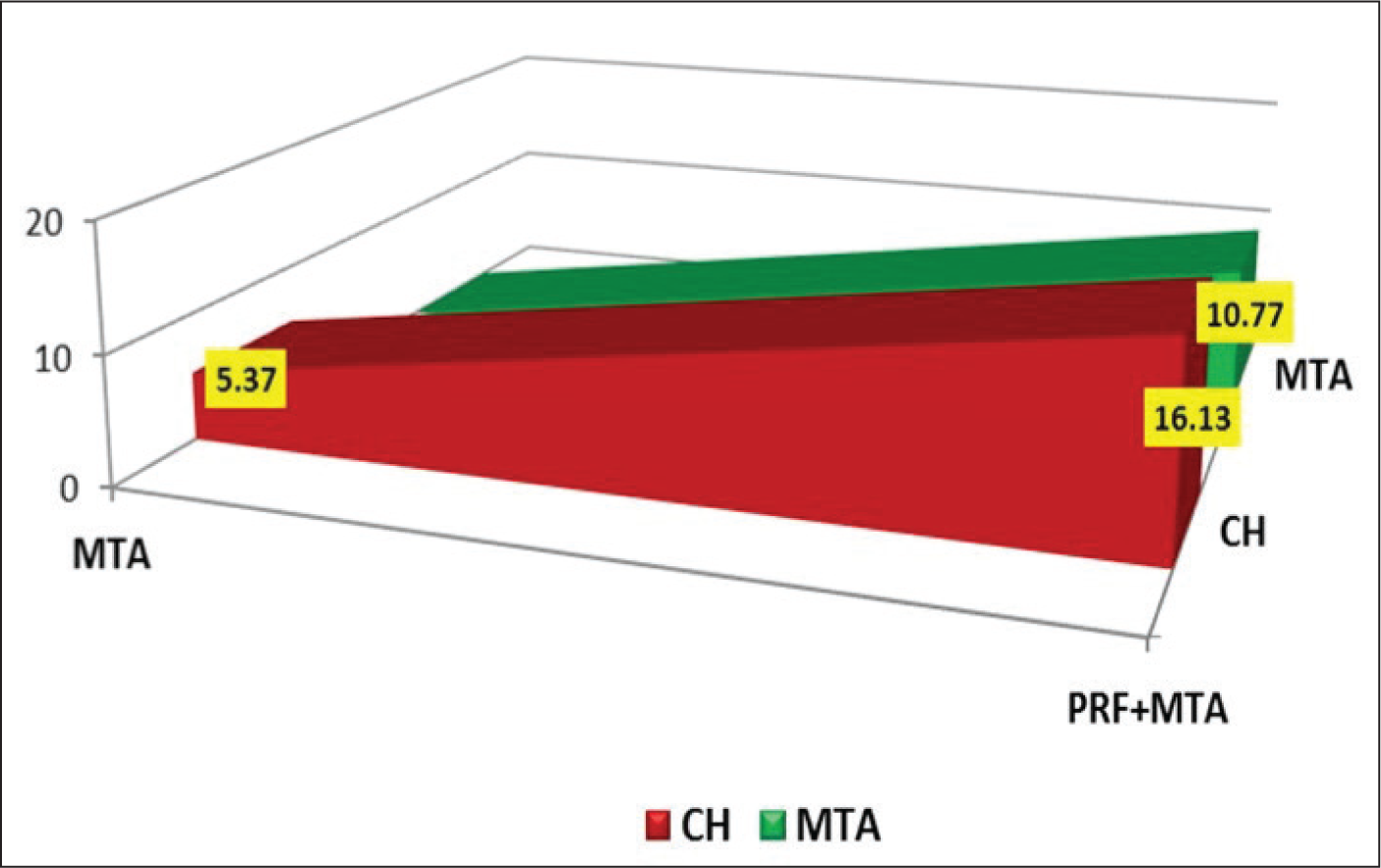
Intergroup Bi-comparison of radiographic density difference at six months (using the Tukey HSD) test showed that density increase difference between CH and PRF–MTA was 16.3 (p = .001); between MTA and PRF–MTA was 10.77 (p = .022); while difference between CH and MTA was not significant (5.37, p = .347) (Figure 5). The PRF–MTA group showed a significant increase in radiographic density, thus the null hypotheses were rejected.
Intergroup Bi-comparisons of Postoperative and After Six-month Density Differences of Dentine Below Various Materials.
Discussion
Based on the results of the present research, a combined use of PRF and MTA seems to have a synergistic effect in formation of calcific dentinal bridge in cases requiring DPC.
PRF was used in the present study owing to its potential to act as a scaffold to MTA and both used together will have synergistic effects on pulp healing. Up-regulation of various types of cytokines and biologic markers has been reported in the presence of MTA, and this property is thought to have helped in an enhanced release of signaling molecules from the PRF in the present study. 20 Although histological evidence is considered as the most confirmative indication of dentin bridge formation, it would involve destruction of the tooth being tested. Previous researchers studying the outcomes of VPT have used IOPAs to study the thickness of dentin bridge formation using a dial caliper, 19 or change in radiographic density assessed by independent observers. The present study used the tool that gives the grey scale value at a point on the IOPA X-ray, thus giving a quantifiable measurement of the radiographic density, and attempts to achieve a fair and measurable result aligning with the ALARA (as low as reasonably achievable) principle of radiation exposure.
When compared to the CH group, the MTA group’s mean radiographic density was higher, although this difference was not statistically significant (p = .347). In recent years, interest in MTA, as a DPC agent has increased significantly. Tricalcium silicate, dicalcium silicate, and tricalcium aluminate are the main calcium oxide compounds that make up unset MTA. According to research, MTA can convert human dental pulp cells into odontoblast-like cells when it comes into contact with them directly. When used for pulp capping and in direct touch with pulp tissue, MTA does not build the layer of necrosis that is visible in the pulp and instead has a greater potential to produce a calcified dentin bridge.21,22 Yet, a few studies have highlighted the disadvantages of MTA. Fridland and Rosado 23 demonstrated that pulpal cells are damaged by a high pH of 12.5 that was established in the vicinity of the MTA and lasts for at least eight weeks. Garcia et al. 24 performed a study to evaluate the cytotoxic effect of different formulations of MTA on fibroblast using the cell viability test (MTT) and cell morphology analysis. In the seven-day period, different formulations of MTA showed a cytotoxic effect of fibroblasts. However, the combined effective outcomes of numerous recent meta-analyses demonstrate that MTA is an acceptable capping agent and a trustworthy therapy of choice for exposed pulp with DPC.25,26
More recently, a biological approach toward VPT has gained momentum, as the molecular and cellular activities underlying dental tissue regeneration and the induction of tertiary dentin production are now better understood. One such biologically active material is PRF which is classified as a second-generation platelet concentrate. Leukocytes, platelets, and cytokines such as interleukins, TGF-b, and VEGF are abundant in this autologous bioactive material, which reaches a high level in about two weeks. PRF has been used successfully in regenerative endodontics, surgical endodontics, and perforation repair. Recently, this biomaterial has shown promising outcomes in VPT cases as a pulpotomy agent. 27 In a recently published randomized control trial conducted by Shobana et al. 28 in 2022, the quality of dentin bridge formed under the site of exposure was assessed using a CBCT which gave a 3-D reconstructed image of the dentine bridge. According to this study, the platelet concentrates PRF and PRP (platelet-rich plasma) formed a significantly higher volume of dentine bridge than MTA.
According to Huang et al., 29 PRF had no cytotoxic effects on dental pulp stem cells and promoted pulpal healing by releasing growth factors like PDGF and TGF-b, which are crucial for the proliferation and differentiation of stem cells. In response to a subpopulation of T cells being activated, PRF produces anti-inflammatory cytokines including IL-4. In addition to inhibiting IL-1b-mediated activation of MMP-1, MMP-3, and prostaglandin E-2 synthesis, this IL-4 promotes healing by reducing inflammation. Additionally, MTA exhibits increased cytotoxicity when it is freshly combined, which may be related to its high pH 12. 30
Lee et al. 19 researched on the action of electrospun poly (ε-caprolactone) fiber meshes when applied under MTA in DPC. They found that teeth thus treated showed three times thicker bridge formation in a lesser duration of time compared with those treated with MTA alone. This was explained by the fact that this fiber mesh helped in segregating unset MTA-released noxious components from reaching the exposed pulp tissue thus providing an environment conducive for attachment and differentiation of dental pulp stem cells. 31 Therefore, it is possible that in a similar way, in the present study, PRF also acts as a protective barrier to prevent the direct contact of freshly mixed MTA with pulp thus explaining the greater density changes for this group. The increase in radiographic density for MTA + PRF being significantly higher than that of MTA alone.
After applying the GIC liner, the final restoration resin was added. MTA and GIC are suggested as effective coronal sealing materials to stop microleakage. This amalgamation of (MTA + GIC) creates a double coronal seal that prevents the entry of germs and maintains a stable environment. 32 When using CH, a defensive antibacterial seal that CH alone cannot offer is provided by the GIC liner. When restorative resin is employed as the permanent restorative material, it also prevents the pulling out of pulp capping material from direct contact with exposed pulp owing to polymerization shrinkage generated by the resin. 33
The present study had some limitations, including limited sample size, the use of a 2-dimensional digital radiography method for assessing results, and the lack of a correlation between the area of pulp exposure and the outcome studied.
Conclusion
According to the current investigation, PRF and MTA together appear to have a synergistic impact in cases requiring DPC, as evidenced by an increase in radiographic density below the pulp exposure. PRF being a product of patient’s own blood, is a safer, readily available, and less expensive scaffold with established repair and regenerative properties. A biologic approach may pave a way toward preserving dental pulp and avoiding more invasive endodontic therapy. This pilot study could be used as the foundation for larger-scale, longer-follow-up randomized control trials.
Footnotes
Acknowledgements
The authors would like to acknowledge the King George's Medical University Lucknow India for providing infrastructure and clinical material for this study.
Authors’ Contribution
Rhythm Bains: Conceptualization, designing the study,drafting of manuscript
Meena Singh: Clinical Procedures, documentation
romila Verma: Documentation & manuscript drafting
A P Tikku: Manuscript proof reading
Rakesh K Yadav: Documentation & Manuscript editing
Ramesh Bharti: Documentation & Manuscript editing
Data Availability Statement
All raw data pertaining to the manuscript can be available on request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved the Institutional ethics Committee of King George’s Medical University, Lucknow. Reference number ECM II B-Thesis/P-4.
Funding
The authors received no financial support for this article’s research, authorship, and/or publication.
Informed Consent
A written, informed consent form was taken from all the patients recruited in the study.
