Abstract
Aim:
Recently, although studies have shown that biomaterials containing graphene oxide (GO) in biomedicine stand out for their positive effects, the effect of GO on dental tissues when used with dental materials is not well known. The aim of this study was an evaluation of the cytotoxic effects of GO on gingival fibroblasts when it is combined in two different ratios with Mineral Trioxide Aggregate (MTA).
Materials and Methods:
In this in-vitro study, a homogenous mixture of adding +0.1 weight (wt)% and +0.3 wt% GO to Angelus MTA was created (two experimental groups) and compared with pure Angelus MTA and negative control groups. The materials were mixed according to the manufacturer’s instructions, and Teflon molds were used to form 24 disc-shaped samples for each group. The samples were divided into groups according to the simple random sampling method. The cytotoxic effect of samples was determined on gingival fibroblast cells by using the MTT test, and total oxidant status (TOS) and total antioxidant capacity (TAC) kits in 24 and 72 hours. The data were statistically analyzed using one-way ANOVA and Tukey tests.
Results:
A significant difference was found between the material-applied groups and the control group at the TAC 24 and 72 hours and between the groups containing GO and the control group at the MTT 72 hours and TAC and TOS 24 and 72 hours (p < .05).
Conclusion:
The addition of GO to MTA increased the dose and time-based toxicity and oxidant amount, and decreased antioxidant capacity.
Introduction
Vital pulp treatments have been practiced in dentistry for more than 200 years. 1 This treatment utilizes several procedures, for instance; direct pulp capping, indirect pulp capping, and partial or complete pulpotomy. 2 Direct pulp capping is a technique for coating exposed dental pulp with a defensive material and stimulating reparative dentin formation. 3
Calcium hydroxide has been accepted and used as a gold standard material for pulp protection applications for many years. 4 However, it has disadvantages such as poor sealing ability, high solubility, microleakage that can occur over time, and shows tunnel defects in dentin tubules.5, 6 Because of these disadvantages, mineral trioxide aggregate (MTA), which is a highly biocompatible material, has been proposed as a pulp capping material. MTA provides thicker dentin bridges and causes less pulp necrosis compared to calcium hydroxide. Nevertheless, MTA also has disadvantages such as the potential to cause discoloration on the teeth, long setting time, and difficulty of use.6, 7 MTA is commercially available under different names. According to one of the manufacturers, that is, Angelus MTA, it contains 80% Portland cement and 20% bismuth oxide. 8 It does not have calcium sulfate in the structure of the material to shorten the curing time (10 min). 9
Graphene is at the thickness of a single atom, has a two-dimensional (2D) crystal structure, and has a hexagonal lattice of carbon atoms. 10 Due to its physicochemical and mechanical properties as well as its excellent electron transport properties, it is the focus of attention of scientific research. 11 In recent times, graphene has been used in numerous applications in biomedicine such as the diagnosis of diseases, antibacterial and antiviral materials, cancer targeting and photothermal treatment, electrical stimulation of cells, drug delivery, and tissue engineering studies.10, 12–15 It is known to contribute to the adhesion, growth, proliferation, and differentiation of osteoblasts. 16 Graphene-based materials consist of species for instance graphene oxide (GO), reduced graphene oxide (rGO), graphene nanoplatelet (GNP), and graphene quantum dots (GQDs).17, 18
GO is a unique material that can be seen as a single monomolecular graphite layer with various oxygen-containing functional groups, for example, epoxy, carbonyl, carboxyl, and hydroxyl groups. 19 It is the oxidized form of graphene and thanks to the functional groups in its structure, it can be dispersed into water and be a suitable material for biomedical studies. 20
Graphene has also become the subject of dentistry research. In the study conducted by He et al., GO was used as an antibacterial agent against dental pathogens and it was found to be antibacterial. 21 In another study, it has been observed that the mechanical properties of composites with added graphene have significantly improved. 22 In the study conducted by Guazzo et al., they stated that the use of GO alone or with other dental materials would increase bioactivity and other biomedical benefits, because the GO substrate increases the expression of DMP-1 and DSPP genes associated with odontogenic differentiation23, 24; however, high doses of graphene have been shown to create toxic effects. 25 Therefore, it is important to determine the optimum usage rate for graphene and its derivatives to show the expected effect of biomaterials used in dentistry.
The cells found in the largest proportion of the pulp and periodontal ligament connective tissue are observed as fibroblasts. For this reason, pulp capping materials come into contact with these cells during vital pulp therapy. 26 Human dental pulp stem cells and human gingival fibroblast cells show similar characteristics and have the ability to give similar responses to external factors when used in regenerative procedures. 27 Both cell groups are used in cytotoxicity tests of dental materials.28, 29
Although graphene has been used successfully in many areas of biomedicine due to its high mechanical properties, electrical conductivity and osteogenic potential, its use in dentistry is just developing.10, 12–15,23,24 More studies are needed to determine the advantages and disadvantages of the use of graphene and its derivatives in dentistry. Therefore, in this study, GO, a graphene derivative, which is likely to contribute positively to the dentin repair mechanism, 23 was added to Angelus MTA in 0.1 wt% and 0.3 wt% ratios. The experimental capping materials were formed and their cytotoxic effects on gingival fibroblast cells were evaluated by using 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyl tetrazolium bromide (MTT) test and total oxidant status (TOS) and total antioxidant capacity (TAC) kits. The null hypothesis of this study is that GO added to Angelus MTA has toxic effects on gingival fibroblast cells in all ratios.
Methods
Setting and Design
An ethical committee approval statement is not required for this in vitro cytotoxicity assessment study. This study was conducted at Bilecik Seyh Edebali University, Medical Pharmacology Department. The study was carried out in two periods within one month. In the first period, the experimental materials were prepared by a dentist. In the second period, a medical pharmacologist was involved in performing the cytotoxicity tests. In this periods, GO (Nanografi, Ankara, Turkey), Angelus MTA (Angelus, Londrina, PR, Brazil), Dulbecco modified eagle’s medium (DMEM), Fetal calf serum (FCS), Roswell Park Memorial Institute (RPMI) 1640, phosphate buffer solution (PBS), antibiotic/antimycotic solution (100×), and trypsin–EDTA got from Sigma-Aldrich (St. Louis, MO, USA) materials were used.
Material Preparation and Sampling Criteria
Firstly, precision scales weighed 0.1 g (0.1 wt%) and 0.3 g (0.3 wt%) GO (Nanografi, Ankara, Turkey) for 1 g Angelus MTA (Angelus, Londrina, PR, Brazil), and were thoroughly grounded in an agate mortar to obtain a homogeneous mixture. The prepared Angelus MTA + 0.1 wt% GO and Angelus MTA + 0.3 wt% GO mixtures and pure Angelus MTA were mixed according to the manufacturer’s instructions and placed in standard cylindrical Teflon molds of 4 mm diameter and 2 mm height. The samples were kept in an incubator (Nuve ES 110, Turkey) at 37°C and 90% ± 5% relative humidity for 48 hours until they reached sufficient hardness and were then removed from the Teflon mold. The samples were examined with an optical stereomicroscope at 30× magnification and those with defects or cracks on the surface were eliminated. Only the samples with smooth surface continuity were accepted. The samples were randomly selected with a simple random sampling method and divided into groups.
Group 1: Angelus MTA + 0.1 wt% GO
Group 2: Angelus MTA + 0.3 wt% GO
Group 3: Pure Angelus MTA
Group 4: Control
The minimum sample size required to detect a significant difference was calculated at least three in each group (nine in total), considering type I error (alpha) of 0.05, power (1-beta) of 0.8, and effect size of 2.35. Twelve of the samples were used for the MTT analysis (24 and 72 hours; n = 6), six of the samples were used in the TAC analysis (24 and 72 hours; n = 3), and the remaining six samples were used in the TOS analysis (24 and 72 hours; n = 3) for each group. The materials and their chemical contents are given in Table 1, and the physical properties of GO are listed in Table 2.
Components of Angelus MTA and Graphene Oxide.
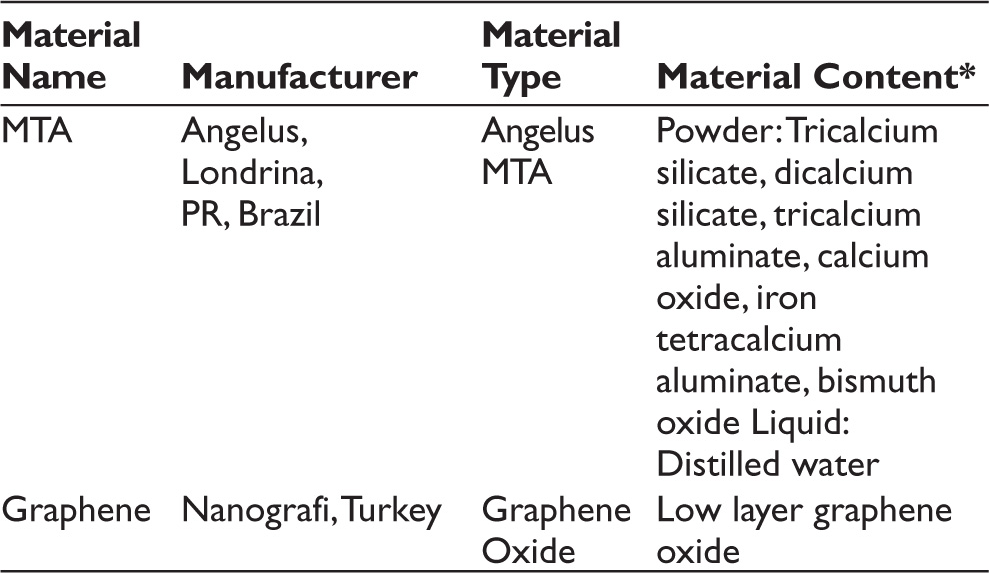
*The content of the materials is tabulated in accordance with producers.
Physical Properties of Graphene Oxide.*
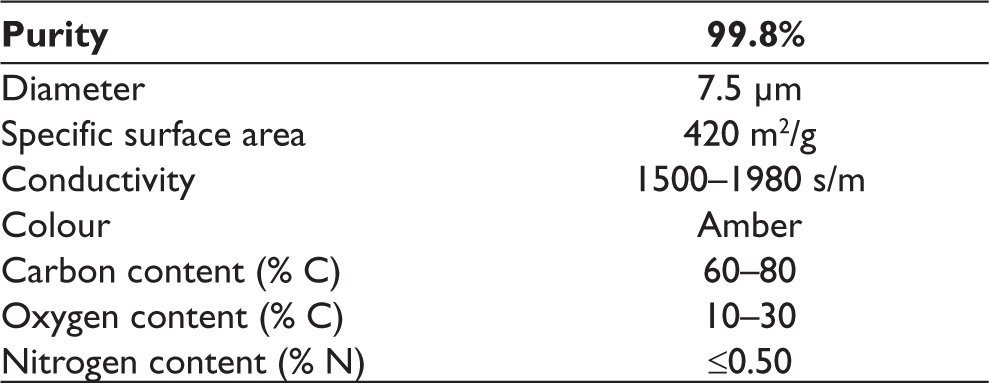
Observational Parameters
In vitro evaluation of the cytotoxic effects of the prepared samples on gingival fibroblast cell line done by MTT (primary variable), TAC, and TOS analysis (secondary variables) at the 24th and 72nd hours. The gingival fibroblast cell line (ATCC® PCS-201-018™) was obtained from Seyh Edebali University and Medical Pharmacology laboratory. Initially, the cells were dissolved and then centrifuged to obtain pellets. The pellet was re-suspended in growth media (DMEM, 10% FBS, and 1% antibiotic) and the cells were seeded into a 24-well culture plate at the density of 1×10 5 cells/mL. An insert membrane was placed in the wells, and direct contact between cells and dental material was blocked. The cells were treated with Angelus MTA + 0.1 wt% GO, Angelus MTA + 0.3 wt% GO, and Angelus MTA and incubated for 24 and 72 hours (5% CO2; 37°C). As a control, 100 μL DMEM only was supplemented to one set of wells for 24 and 72 hours.
MTT Analysis
Gingival fibroblast viability was evaluated by MTT assay (Cayman Chemical, MI, USA) following 24 and 72 hours. Briefly, at the end of the experiment (24 and 72 hours), 10 μL of MTT reagent was supplemented to each well. After 4 hours of incubation (5% CO2; 37°C) periods, formazan crystal formation occurred. About 100 μL of dimethyl sulfoxide was added to dissolve formazan crystals. The plate was read at 570 nm using an ELx808 Biotek Microplate reader.
TAC and TOS Assays
TAC and TOS assays are called the evaluation of color density with spectrophotometric properties (from Rel Assay Diagnostics® Company, Gaziantep, Turkey). All procedures were done according to manufacturing protocol.TAC values were calculated as Trolox Equiv mmol/L−1 and TOS values were calculated as H2O2 Equiv mmol/L−1.
Statistical Analysis
The normality of the data was assessed using the Kolmogorov-Smirnov test. One-way analysis of variance (one-way ANOVA) was used to compare variables among the groups. After a significant one-way ANOVA test, the Tukey test was applied for multiple comparisons. Research results were analyzed with IBM SPSS Statistics V. 25 for Windows (SPSS Inc., Chicago, IL). Significance was evaluated at p < .05.
Results
MTT Analysis Results
The data of the control group were equalized to 100 and the data of the other groups were compared with the control group. MTT test values are given as ‘%’. The percentage values of cell viability of the MTT test are given in Table 3 for 24 and 72 hours. In the 24th-hour evaluation, the highest cell viability rate was 94% in the Angelus MTA group and the lowest cell viability rate was 90% in the Angelus MTA + 0.3 wt% GO group. In the 72nd-hour evaluation, the highest cell viability rate was 92% in the Angelus MTA group and the lowest cell viability rate was 75% in the Angelus MTA + 0.3 wt% GO group.
MTT, TAC, and TOS 24th and 72nd Hours Results.
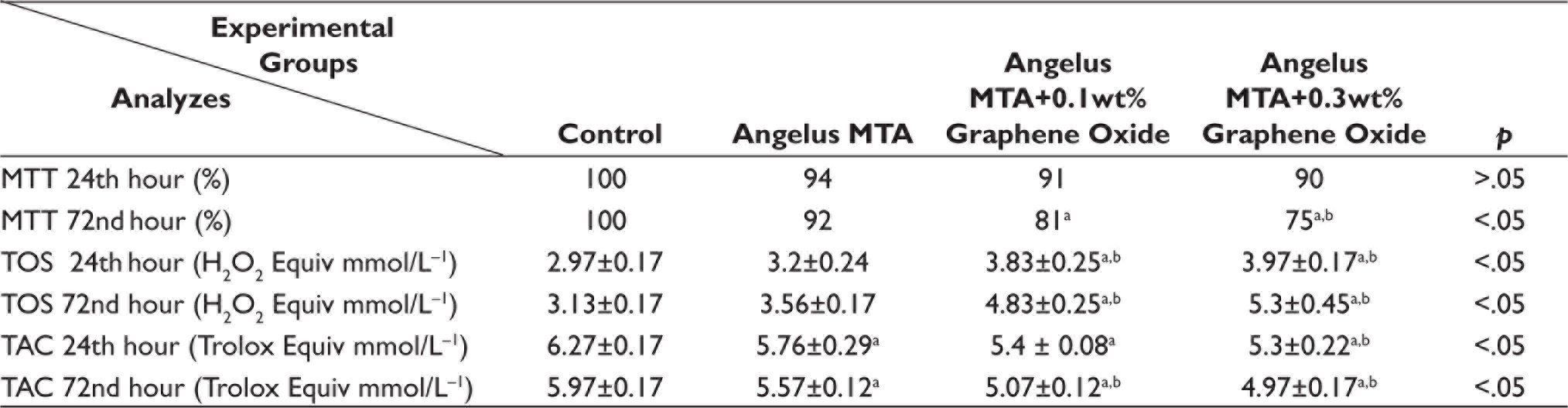
TOS and TAC values are given as ‘mean ± standard deviation’.
aSignificantly different from control group (p < .05; Tukey test).
bSignificantly different from Angelus MTA group (p < .05; Tukey test).
Angelus MTA: Angelus mineral trioxide aggregate; MTT: 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyl tetrazolium bromide; TOS: Total oxidant status; TAC: Total antioxidant capacity.
For the 24th-hour results, there was no statistically significant difference between the control and material-applied groups (p > .05). For the 72nd-hour results, a significant difference was found between the GO added to Angelus MTA groups and the control group and between the Angelus MTA + 0.3 wt% GO group and the Angelus MTA group (p < .05).
TOS Analysis Results
Descriptive statistics for the TOS test 24- and 72-hour variables are summarized in Table 3. Data were calculated based on the hydrogen peroxide (H2O2 Equiv mmol/L−1) level. TOS values are given as ‘mean ± standard deviation’. Increased concentration of GO caused an increase in oxidant levels. The lowest TOS values after 24 and 72 hours were calculated in the control groups, and the highest TOS values after 24 and 72 hours were in the Angelus MTA + 0.3 wt% GO groups. As a result of statistical evaluation for the TOS test 24 and 72 hours, a significant difference was found between the GO added to Angelus MTA groups and the control groups and between the GO added to Angelus MTA groups and the Angelus MTA groups (p < .05).
TAC Analysis Results
Descriptive statistics for the TAC test 24- and 72-hour variables are summarized in Table 3. Data were calculated in Trolox Equiv mmol/L−1. TAC values are given as ‘mean ± standard deviation’. Increased concentration of GO caused a decrease in antioxidant levels. The highest 24th hours TAC values were detected in the control group. The lowest TAC values were calculated in the Angelus MTA + 0.3 wt% GO group. As a consequence of statistical evaluation, there was a significant difference between the material applied groups and the control group and between the Angelus MTA + 0.3 wt % GO group and the Angelus MTA group (p <0.05). The highest 72nd hours TAC values were calculated in the control group and the lowest TAC values were in the Angelus MTA + 0.3 wt % GO group. As a consequence of statistical evaluation, there was a significant difference between the material applied groups and the control group and between GO added to Angelus MTA groups and the Angelus MTA group (p < .05).
Discussion
The purpose of vital pulp treatment is to prevent the progression of infection by removing the factors that irritate the pulp and to stimulate the formation of tertiary dentin and thus keep the tooth active in the mouth.30, 31 One of the most important properties of pulp capping materials is their ability to stimulate the formation of quality mineralized tissue. 32 For this reason, it is necessary to increase the biocompatibility of pulp-capping materials to promote the biological response of pulp tissues, idealize pulp-capping techniques, and expand tertiary dentin formation. 33 In a review, it was stated that graphene seems to be biocompatible with dental pulp stem cells. 23 When we look at the literature, it is seen that there are conflicting results regarding the biocompatibility of graphene. At the same time, studies on its use in dentistry are very limited. To contribute to the literature on the usability of graphene, which has the potential to enhance the formation of hard tissue in vital pulp treatments, GO was added to Angelus MTA at the rate of 0.1 wt% and 0.3 wt%, and its biocompatibility was evaluated.
MTA is a topical material that has been increasing in clinical use in recent years and has been proven to be more successful in vital pulp treatments than calcium hydroxide in many studies.34, 35 Therefore, MTA (Angelus, Londrina, PR, Brazil) was preferred as the basic material in our study.
Graphene-based materials have been suggested as fresh formation biologically active nano-materials that can provide an environment for the adhesion, proliferation, and differentiation of stem cells to osteogenic origins. 36 Adding graphene-based materials to the content of materials used in bone regeneration can increase osteoconductivity by stimulating biomineralization and cellular osteogenic differentiation. Ilyas et al. recently reported that a mixture of calcined GO-bioactive glass ceramics had supported the formation of hydroxyapatite in an in vitro study. 37 In the study conducted by Park et al., 38 titanium membranes coated with GO greatly increased the formation of new bone in calvarial defects of rats.
Since GO increases the expression of two genes associated with odontogenic differentiation from dental pulp stem cells, such as DMP-1 and DSPP, using it alone or in combination with other dental materials may increase bioactivity and biomedical benefits.23, 24 Various studies have been conducted in dentistry using graphene-based materials. It has been shown to have a positive effect on teeth whitening, 39 an inhibitory effect on S. Mutans when added to the dental adhesive material, 40 and when collagen membranes are coated with GO, it increases cell viability on dental pulp stem cells and causes osteoblastic differentiation. 41 In a study by Sun et al., 42 porous titanium was coated with GO at three different concentrations (0.1 mg/mL, 0.5 mg/mL, and 1.0 mg/mL) and used as pulp sealing material. The adhesion of human dental pulp stem cells to the titanium surface coated with 1.0 mg/mL GO increased, odontogenic differentiation occurred, and antibacterial activity was increased. In our study, we added 0.1 wt% and 0.3 wt% of GO to Angelus MTA at rates similar to the studies in the literature. The results of the present study showed that GO which was added to Angelus MTA at two different rates (0.1 wt% and 0.3 wt%) decreased Angelus MTA’s biocompatibility and therefore the null hypothesis of the study was accepted.
Some studies have shown that gingival fibroblasts can release many types of cytokines in return for various stimuli in the same way as odontoblast, endothelial cells, and immune cells.43, 44 In this study, gingival fibroblast cells were used in MTT, TAC, and TOS tests to evaluate the biocompatibility of the materials. Similar to our study, Luczaj-Cepowicz et al. 29 and Samiei et al. 26 used gingival fibroblast cells to measure the cytotoxicity of pulp capping materials by MTT analysis in their studies.
It has also been shown in the literature that when carbon-based materials are added to calcium silicate-based materials, they significantly improve the mechanical properties and cell compatibility of calcium silicate cement.45–47 In a research, GO was added to Portland cement at the rates of 0.01%–0.05% by weight, and the highest increase in compressive strength was observed with the addition of 0.02 wt% and 0.03 wt% GO (28.5% and 46.4% increase, respectively). 48 In our previous study, adding GNP, a graphene-derived material at the rates of 0.1% and 0.3% to MTA; It has been observed that GNP added to MTA causes particle size reduction and increases microhardness. As a result, GNP contributed to the physical and mechanical properties of MTA. 49 Based on our previous research, where we saw the positive results of adding graphene to Angelus MTA on microhardness, we conducted this study to evaluate the biological effects of adding graphene to Angelus MTA. In the present study, we aimed to see the favorable effect of GO on gingival fibroblast cells with the investigational capping materials obtained by adding 0.1 wt% and 0.3 wt% GO to Angelus MTA.
rGO 0.25 wt%, 0.5 wt%, 0.75 wt%, 1 wt%, and 1.5 wt% were added to the calcium silicate (CaSiO3) material in one of the studies conducted by Mehrali et al. 46 According to the MTT test performed in human osteoblast cells, on the first, third, and fifth days, the number of cells rose extensively with an increasing rGO concentration. In a similar study by Mehrali et al., 50 human fetal osteoblastic cell line (hFOB) MTT analysis was performed by adding 0.5, 1, 1.5, and 2 wt% GNP to calcium silicate. MTT results showed that cell viability on 1 wt% GNP was higher than the other groups; but also, in all concentrations of GNP had an effect on cell proliferation in the hFOB cell line. In our study, GO was added to Angelus MTA, which is a calcium silicate-based material; however, it was found that the toxicity grade had extended as the amount of GO increased. In this respect, the results of our study and those of Mehrali et al. 46, 50 contradict each other.
In a study by Qutieshat et al., 51 graphene oxide nanoplatelets (GONPs) were added to 1 wt% and 3 wt% to Portland cement. Balb/C3T3 fibroblast cells were performed with MTT analysis on days 1, 3, and 7. According to the result, cell viability decreased in all groups on the first day, cell viability increased to the control level in the group with 3 wt% GONPs on the third day, and a significant increase in cell proliferation was observed in the group with 1 wt% GONPs, and cell viability decreased in the control group. On the seventh day, positive biocompatibility was observed in the group where only 1 wt% GONPs was added, while cell viability decreased in the other groups. This study contradicts our study where GO was added to Angelus MTA at much lower rates (0.1 wt% and 0.3 wt%) and showed toxicity. However, in this study, similar to our study, GONPs, which were highly added, showed a more toxic effect, and an increasing toxic effect was observed depending on dose and time.
Park et al., 52 investigated the toxicity of GNP in vivo and in vitro, where GNP was applied to human bronchial epithelial cell lines in vitro at rates of 2.5, 5, 10, and 20 μg/mL. According to the MTT results, cytotoxicity was increased in 24-hour and 72-hour evaluations, depending on the dose. The study conducted by Park et al., 52 showed similarity to our study in which the addition of GO to Angelus MTA had a dose-dependent toxic effect. In a study evaluating the concentration-dependent cytotoxicity of rGO-chitosan substrate on human mesenchymal stromal cells (hMSCs), it was observed that hMSCs did not show any signs of cytotoxicity when exposed to rGO at a concentration of less than 0.1 mg/mL. However, rGO has been observed to be cytotoxic to hMSCs at a concentration higher than 0.1 mg/mL. 25 In our study using similar ratios, the supplementation of 0.3 wt% GO was found to be more cytotoxic than 0.1 wt% GO, depending on the dose.
Oxidative stress is a term used to describe oxidative damage to organs, tissues, and cells caused by reactive oxygen species (ROS). 53 It is a pathological condition that occurs when the oxidative–antioxidative balance in our body is disrupted in favor of oxidants. 54 It has been found that the formation of ROS that induce oxidative stress is a mechanism that causes the toxicity of carbon nanotubes and GO, a graphene derivative. 55 It has been reported that the formation and destruction of ROS are dynamically stabilized within the cells, and if this equilibrium is disturbed, it can trigger intracellular protein inactivation, lipid peroxidation, mitochondrial dysfunction, and ultimately apoptosis or necrosis. 55 For this reason, in our study, we investigated the 24-hour and 72-hour TAC and TOS values of gingival fibroblast cells formed by the materials used.
Having investigated the groups, the highest TAC values were found in the Angelus MTA group and the lowest TAC values in the Angelus MTA +0.3 wt% GO group; whereas the highest TOS values were found in the Angelus MTA +0.3 wt% GO group and the lowest TOS values in the Angelus MTA group. Having examined the groups to which were added GO, a decrease in antioxidant capacity and an increase in oxidant capacity were observed based on dose. TAC and TOS data correlate with MTT test results and TAC values decrease as the amount of GO added to Angelus MTA increases. Therefore, the formation of free radicals could not be prevented, and cellular destruction and consequently cellular death occurred. On the other hand, it has been observed that as the amount of GO additions to Angelus MTA increases, the oxidative stress increases. In a study by Ding et al., 56 low doses of GO did not have a toxic effect on T lymphocytes; but it was observed that at doses above 100 μg/mL, GO caused oxidative stress, which led to apoptosis in T lymphocytes. In our study, similar to this study, as the rate of GO increased, the oxidative stress increased and this may have caused apoptosis in gingival fibroblasts.
The strength of this study is that it is one of the limited numbers of studies investigating the use of GO, an oxidized form of graphene, which is being used in biomedicine, in vital pulp treatments in dentistry. The limitations of this study are, first, that no cell lines other than gingival fibroblasts were used. The effect of GO and other derivatives of graphene on dental pulp stem cells should be considered when used with pulp-capping materials. Second, the effect of GO in only two different ratios was evaluated. The 0.1 wt% and 0.3 wt% rates determined in the present study may be cytotoxic rates on gingival fibroblast cells for GO. The effect, when used at different rates (especially at rates lower than 0.1%), should also be evaluated. Third, this study was only built on the effect of GO and MTA. The use of GO or other derivatives of graphene with other pulp-capping materials may have positive effects on pulp capping. The effects of the use of other derivatives of graphene with MTA or different capping materials should also be revealed in future studies. As Yang et al. 55 have summarized, physicochemical properties (functional groups of surface and structural defects) can determine the toxicity of graphene. It is currently not possible to make a general conclusion about the toxic effects of graphene-based materials.
As shown in previous studies, MTA is a biocompatible material and can be used as an ideal capping material. The addition of GO to MTA increases the toxicity and amount of oxidant and decreases the antioxidant capacity depending on the dose and time. However, the use of graphene and its derivatives at different rates with different pulp capping materials may have effects that will increase the success of pulp capping. Besides, to benefit from the superior physical and mechanical properties of graphene and its derivatives, its use in dentistry where biocompatibility is not at the forefront may be advantageous. This in vitro study should be supported by in vivo studies, and the effect of GO and other derivatives of graphene on the pulp tissue in direct pulp capping treatment of live organisms, and the potential of hard tissue formation, should be investigated. This in vitro study may serve as a reference for studies with GO and graphene derivatives in dentistry and may be developed and applied in other studies.
Footnotes
Declaration of Conflicting Interests
The authors received no financial support for the research, authorship and/or publication of this article.
Ethical Statement
Not applicable
Funding
This research was supported by the Research Fund of the Inonu University [Project Number: TCD-2018-1271].
Informed Consent
Not applicable
