Abstract
Aim:
To investigate the effects of selected alcohol-free mouthwashes with different formulations (zinc-hydroxyapatite, hydrogen peroxide, and sodium fluoride) on the surface microhardness of a single-shade universal resin composite.
Materials and Methods:
Forty disc-shaped specimens (8 × 2 mm) from the universal resin composite (Omnichroma®), and a nano-hybrid composite (Tetric® N-Ceram) were prepared. After polymerization, baseline surface microhardness values were recorded using Vickers microhardness tester. The samples from each material were randomly assigned to 4 groups (
Results:
Significant reduction in microhardness was observed in all resin composite groups after immersion in the mouthwashes compared to baseline values (
Conclusion:
In vitro simulated use of the investigated mouthwashes negatively affected the surface microhardness of both tested resin composites. The observed effects were both mouthwash and material dependent.
Keywords
Introduction
Plaque control is vital for the maintenance of clean and healthy teeth and oral tissues. Various mechanical (e.g., toothbrush, floss) and chemical (e.g., mouthwashes, gels) methods have been advocated for effective dental biofilm control. 1
Mouthwashes are one of the most frequently used oral care products. 2 They are utilized as an adjunct to mechanical tooth brushing and as an aid to reach difficult intra-oral areas, with reported evidence of reducing the risk of dental plaque, gingivitis, and dental caries. 3
Early types of mouthwashes were limited to therapeutic formulations with antibacterial or remineralizing agents (e.g., chlorohexidine gluconate or fluoride), some of which may require a prescription from the dentist. Today, however, mouthwashes are widely available over the counter (OTC), while their formulations and indications have also expanded remarkably, with high versatility to accommodate almost every oral health need.
Many mouthwashes used to incorporate alcohol (e.g., ethanol), at varying concentrations, into their formulations as both a preservative and a solvent for other active agents, mainly due to its low cost and easy production. Nevertheless, the use of alcohol as a main ingredient (>25%) in mouthwashes is greatly diminished nowadays, and various alcohol-free mouthwash formulations were increasingly offered by many manufacturers. This trend can be attributed to several factors including the possible association between the use of alcoholic mouthwashes and increased oral cancer risk, albeit still controversial. 4 Another reason could be the limited efficacy of alcohol as an antiseptic agent compared to other active ingredients; while the shift could also have been driven—at least in part—by cultural and religious beliefs that favor non-alcoholic formulations.3–5
New formulations of mouthwashes incorporate other active ingredients with biomimetic properties such as hydroxyapatite (HA) in its various forms. 6 These formulations provide a promising alternative as preventive and remineralizing agents. 7 HA is a naturally occurring mineral that makes up the majority of natural tooth enamel. It can be sourced naturally, for instance from bovine bone and marine structures, but it can also be produced chemically for biomedical application, with the desired properties determined by a number of factors, including the source, chemical purity, and crystalline morphology of the HA crystals. 8
Currently, several commercial oral care products have included HA as the main ingredient that exhibits biomimetic remineralizing potential. 6 In fact, incorporating HA into oral care products can be traced back to the 1970s when a toothpaste with synthetic HA was introduced by the National Aeronautics and Space Administration (NASA). This was specifically designed for astronauts who experienced a reduction in mineral density of teeth and bone due to the absence of gravity while in space.6, 9
Other types of mouthwashes with various formulations and active ingredients are also available. For example, mouthwashes may incorporate potassium nitrates, triclosan, zinc salt, amorphous calcium phosphate, hydrogen peroxide, or fluoride compounds (e.g., sodium fluoride, stannous fluoride).
The wide array of commercially available OTC mouthwashes is overwhelming, and their production rate is fast and not always matched by empirical research into their effectiveness and potential side effects. The daily use of mouthwashes may result in several potential effects not only on sound teeth and oral soft tissues but also on existing dental restorations.
Available evidence from the current literature showed that daily use of some mouthwashes, especially those with low pH and certain active ingredients (such as alcohol or essential oils) can negatively affect the physical, optical and mechanical properties of resin-based restorative materials.10–14
Recently, a universal single-shade dental composite (Omnichroma®) has been introduced by Tokuyama Dental. This resin composite material has uniformly sized supra-nano spherical fillers and utilizes smart chromatic technology that allows the use of a single-shade resin composite that matches all 16 VITA classical shades. 15 The wide shade matching ability of this composite is expected to facilitate the clinical application steps, minimize shade matching variability, and reduce the resin composite inventory.16–18 Omnichroma has been shown to exhibit a broad reflection spectrum that can contribute to improved color adjustment potential when restoring human incisors of various shades. 18
Most of the currently available research on universal dental composites focuses on the optical properties and the color-adjusting potential of the material.19–22 As a result, there is a paucity of evidence on other properties of this type of resin composite (e.g., surface microhardness, compressive and flexural strength).
Surface hardness refers to a material’s resistance to permanent surface indentation or penetration, and it is an important surface mechanical property of resin composite. The Vickers microhardness test is one of the methods used to evaluate the microhardness of resin-based composites. 23 It measures the material’s resistance to a diamond (pyramid) indenter, which is expressed as a Vickers hardness number (VHN). The microhardness of resin composite surface has, significantly, been associated with other important properties (e.g., compressive strength, abrasion resistance) and its reduction may indicate the materials’ low wear resistance. 24
Research to date has not yet determined how the daily use of some oral care products such as mouthwashes would affect the surface properties of the universal resin composite, particularly the surface microhardness.
Therefore, this in vitro investigation was set out to explore the effects of selected alcohol-free mouthwashes with different formulations (zinc-HA, hydrogen peroxide, and sodium fluoride) on the surface microhardness of the universal single-shade resin composite (Omnichroma®) compared to a conventional nano-hybrid resin composite (Tetric® N-Ceram).
The following null hypotheses will be examined:
There is no significant difference in the microhardness values of the universal dental composite before and after immersion in each of the tested mouthwashes. There is no significant difference in the microhardness values between the two tested resin composites after immersion in the tested mouthwashes. The composition of the tested mouthwashes has no significant effect on the post-immersion microhardness values of the resin composites.
Materials and Methods
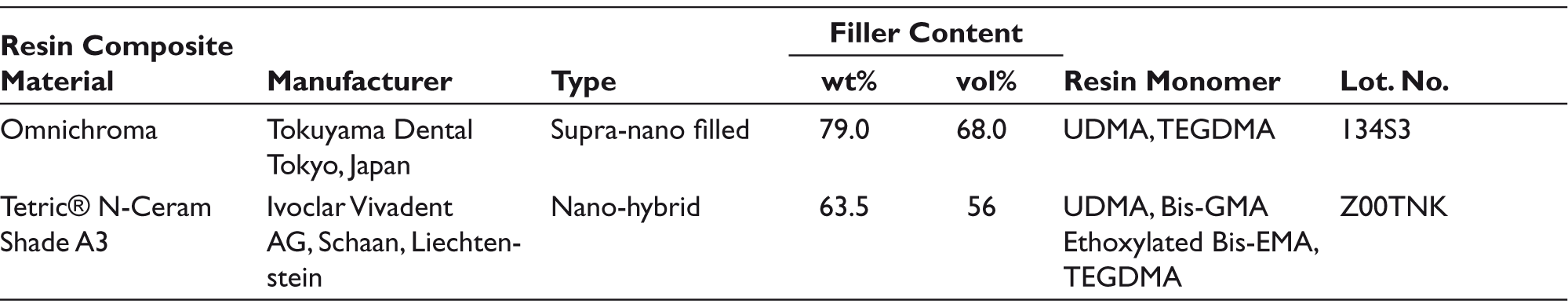
The resin composite materials used in the current study are Omnichroma® single-shade composite with supra-nano spherical fillers; and Tetric® N-Ceram, which is a nano-hybrid used as a control; their detailed composition is presented in Table 1.
Composition of the Evaluated Resin Composites Materials.
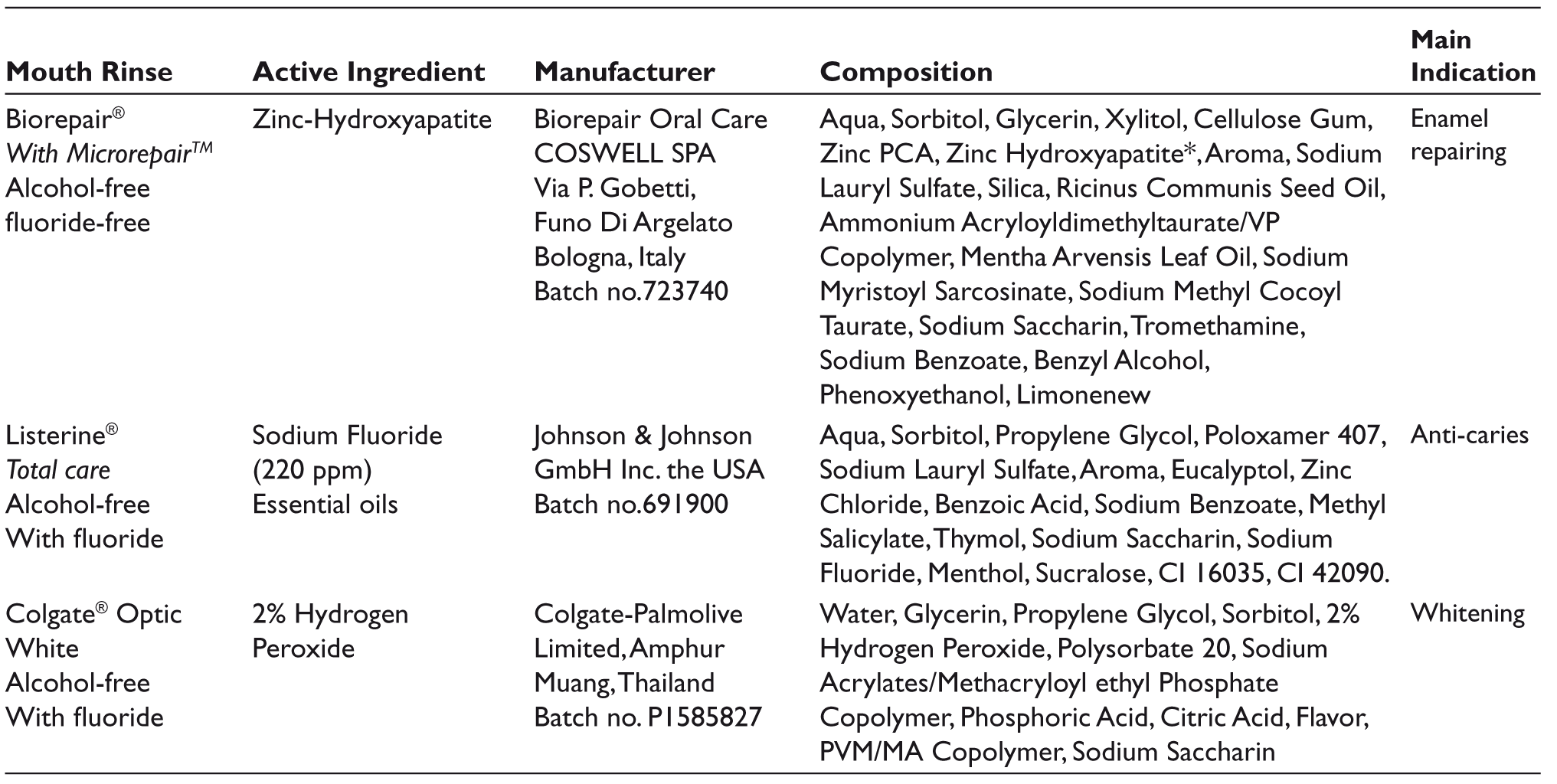
The immersion solutions included: Three alcohol-free mouthwashes, in addition to distilled water which is the negative control. The three tested mouthwashes are: Biorepair® (zinc-HA-based mouthwash), Listerine® (sodium fluoride-based mouthwash), and Colgate® Optic White (hydrogen peroxide-based mouthwash); their detailed composition is presented in Table 2.
Mouthwashes Investigated in the Current Study.
Sample Size Estimation
The G*Power 3 software 25 was used to estimate the sample size with a power of 90% and a significance level of .05. For the four experimental groups, a sample size of 10 samples per group was determined.
Specimen Preparation
Forty disc-shaped specimens were made for each resin composite material using a Teflon mold with an internal diameter of 8 mm and 2 mm thickness. The specimens were made carefully to avoid any air entrapment during the placement of the material. The mold was sandwiched between two Mylar strips and microscopic slides (1 mm thickness) on each side to prevent the formation of an oxygen inhibition layer and to achieve a smooth surface of specimens. The samples were polymerized from the top surface for 20s with a Polywave LED curing unit (Bluephase®, Ivoclar Vivadent, Schaan/Liechtenstein) under standard curing mode. The light-curing unit had an output irradiance of 1,200 mW/cm 2 and wavelength range of 430–480 nm. The curing distance was standardized to 1 mm by using a 1-mm glass slide. A calibrated radiometer (Bluephase® Meter, Ivoclar Vivadent, Schaan/Liechtenstein) was used to verify the irradiance before each use. Immediately after cure, each sample was gently pushed out from the mold and the excess flash of the material was removed using a sharp blade.
Microhardness Test
The microhardness was measured with a Vickers micro-Hardness Tester (NOVA 130 Vickers hardness testing instrument, INNOVATEST Europe BV, Maastricht, The Netherlands) using a diamond pyramid micro-indenter. After polymerization, each specimen was positioned centrally beneath the indenter of the microhardness tester to obtain the baseline microhardness measurements. The test was conducted at room temperature (23 ± 1°C) under a load of 300 g and dwell time of 15 seconds. Three indentations, 1 mm apart from the specimen margins and from each other, were made and averaged to yield a single micro-hardness number (VHN).
After baseline measurements, the samples from each material were randomly assigned to four groups (
Statistical Analysis
Data will be analyzed using SPSS (v.20, IBM Corp, Armonk, NY). The outcome variable is the microhardness values, while the independent variables were the mouthwashes and the resin composites. One-way analysis of variance (ANOVA) test and paired sample
Preliminary analysis revealed that the data were normally distributed as assessed by Shapiro–Wilk test (
Results
Omnichroma Group
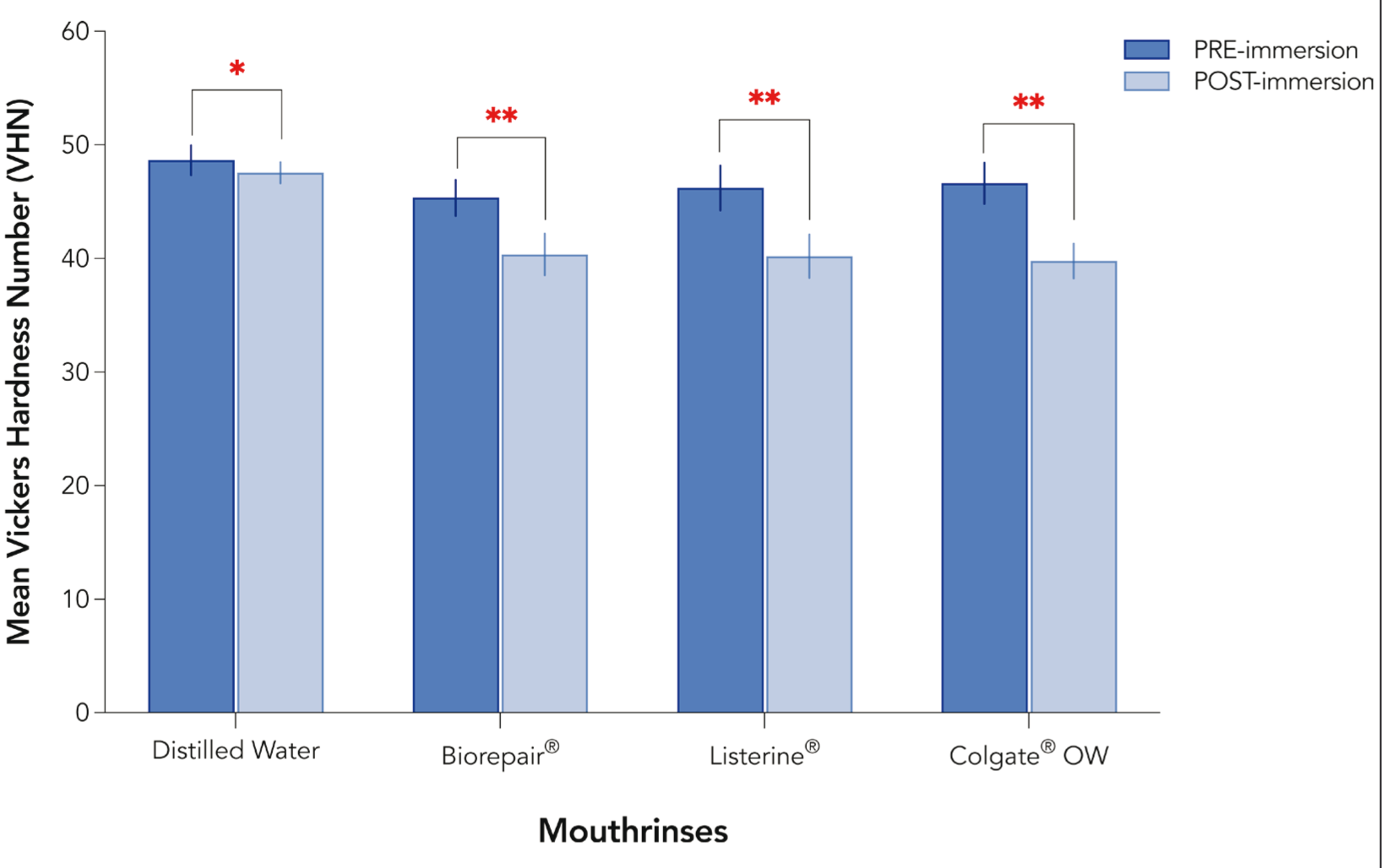
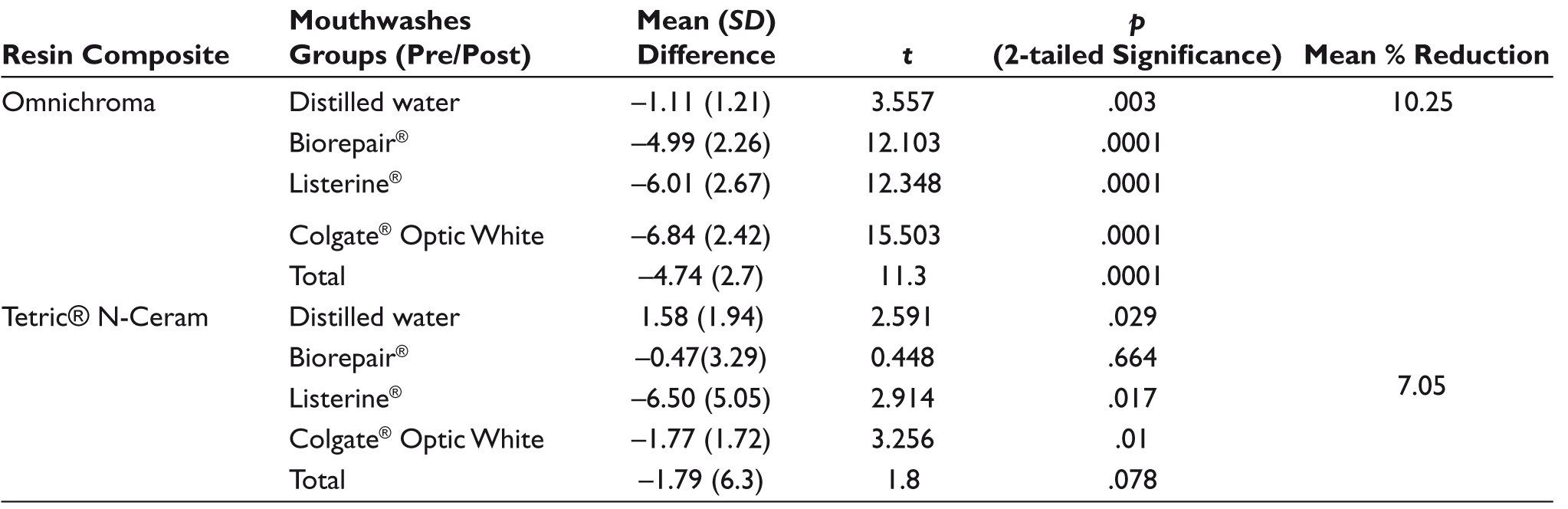
Significant reduction in the microhardness of Omnichroma was observed in all the groups after immersion in the mouthwashes compared to baseline values (
Pre- and Post-immersion Microhardness Values for Omnichroma Resin Composite Group. Single Asterisk Denotes. Significant Difference at (P < .01), While Double Asterisks Denote Statistically Significant Difference at (P <.0001).
Paired sample
Paired Sample
Pre- and Post-immersion Microhardness Values for Tetric® N-Ceram Resin Composite Group. Single Asterisk Denotes a significant difference at p < .05.
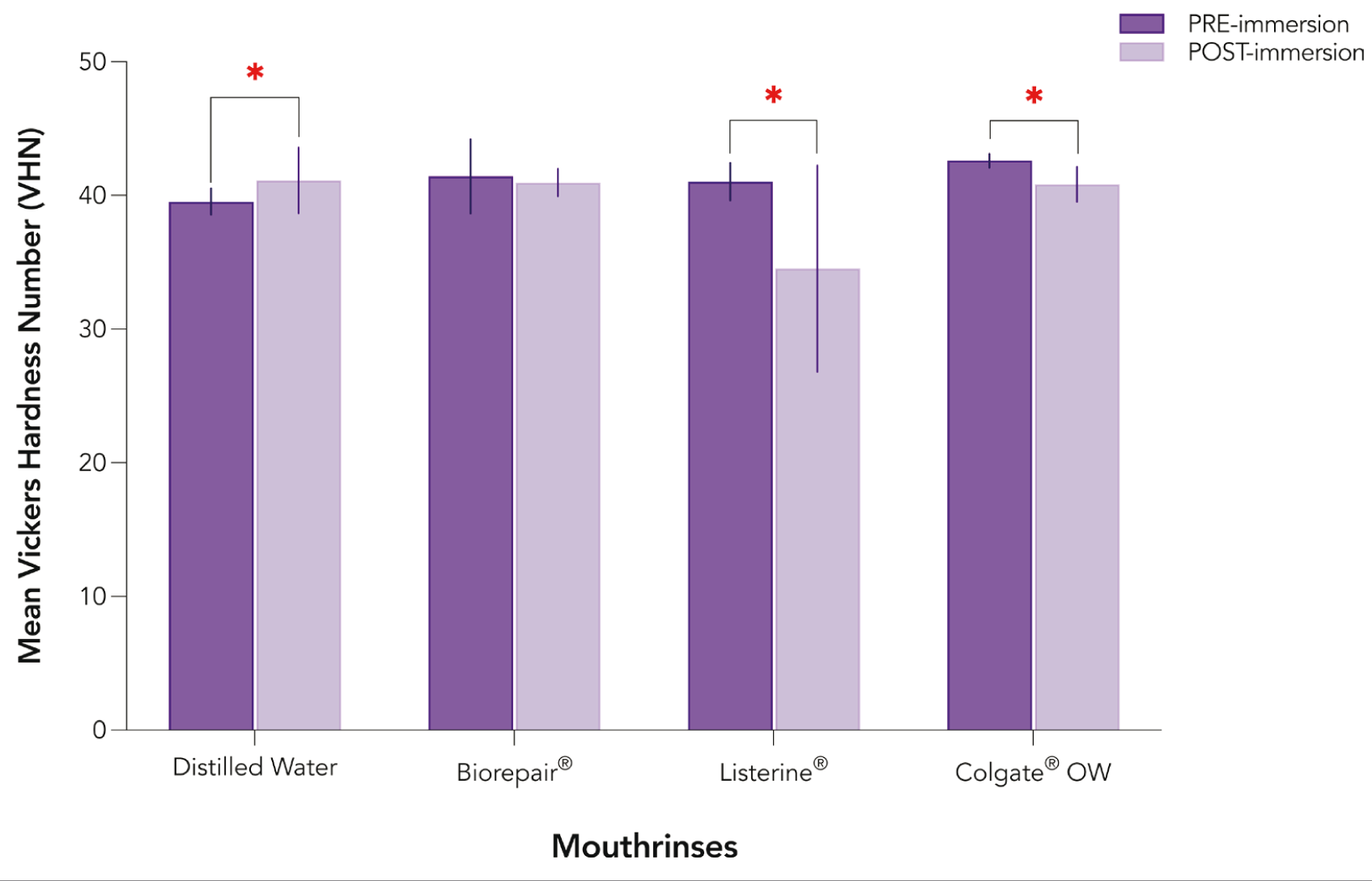
Tetric® N-Ceram Group
Differences between pre- and post-immersion microhardness values were significantly highest for specimens immersed Listerine® (
Inter-group Differences
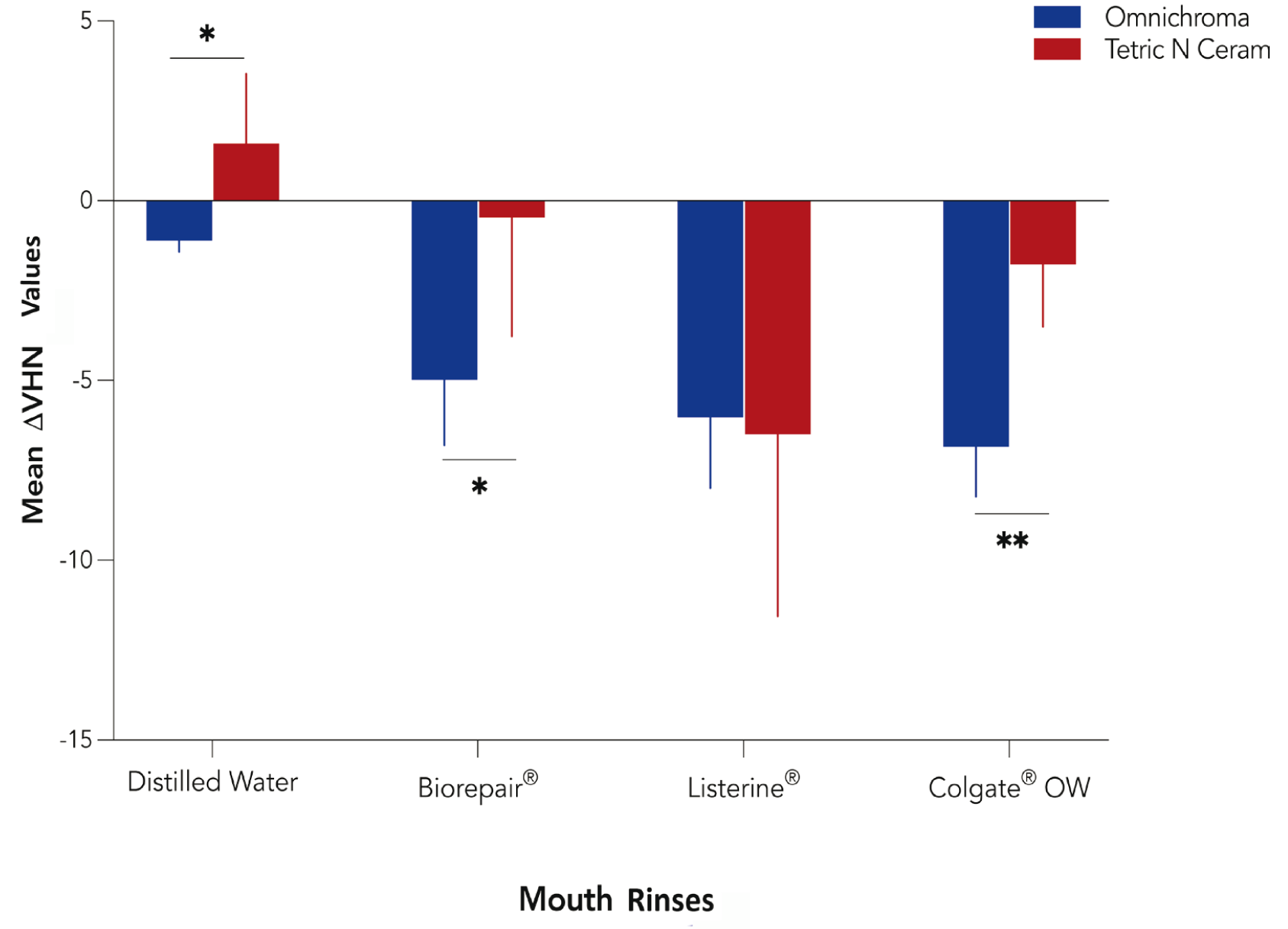
Two-way ANOVA (2×4 ANOVA) revealed a statistically significant interaction between the effects of resin composite type and mouthwashes on the post-immersion microhardness values,
Mean Difference (∆VHN) in Post-immersion Vickers Microhardness Values Among the Tested Resin Composites.
Discussion
This in vitro study explored the effect of simulated use of three alcohol-free mouthwashes on the surface microhardness of a recently introduced universal supra-nano-filled resin composite compared to a conventional nano-hybrid resin composite.
All the mouthwashes examined in the current study were found to significantly reduce the surface microhardness of both tested resin composites to varying degrees. Therefore, the first null hypothesis was rejected. This finding was in agreement with several studies.26, 28–31 Significant differences in post-immersion microhardness values were observed among the two resin composites, when immersed in different types of mouthwashes, and thus, both second and third null hypotheses were also rejected.
The two resin composites reacted differently to the immersion solutions. The Omnichroma group showed the highest degree of vulnerability to Colgate® Optic White mouthwash, which is based on 2% hydrogen peroxide, and thus exhibited the greatest reduction in microhardness values (14.67%). By contrast, Tetric® N-Ceram was most vulnerable to immersion in Listerine® mouthwash with resultant significant reduction in microhardness values (19.06%). For both resin composites, the least reduction in microhardness was observed when samples were immersed in Biorepair® mouthwash, which is based on zinc-HA. Although Tetric N-Ceram (
In contrast to the current findings, Kwon et al. found that the reduction in microhardness of nano-filled composites subjected to 35% hydrogen peroxide, 15% carbamide peroxide, or distilled water were not significantly different after three weeks of immersion. 32
Surface degradation of resin composites with a subsequent reduction in surface properties (e.g., microhardness) can occur due to mechanical or chemical factors affecting the oral environment. Mechanical wear can be associated with occlusal forces, while chemical softening can occur upon exposure to various chemical agents found in saliva, food, beverages, and oral care products.33, 34 This degradation would subsequently affect the restoration esthetics, dimensional stability, physical, and mechanical properties, leading to other detrimental changes (e.g., increased plaque buildup, inflammation, or recurrent caries). Ultimately, all these factors would negatively influence the longevity and durability of the restoration.29, 33
Since the resin composites investigated in this study were not exposed to any mechanical forces, the observed changes in microhardness can be attributed solely to chemical reactions and dissolution in the immersion media. Liquid sorption and solubility are known to have negative effects on the structural and functional properties of resins, such as swelling, softening, plasticization, and hydrolysis. 35
Moreover, it is evident that the extent to which the tested resin composites were affected by the mouthwashes was dependent on the type of the material and its chemical composition (e.g., the polymer matrix, filler load, and type), as well as the rate of solution uptake by the resin matrix. Thus, each material reacted differently upon immersion in the different solutions.
In this study, the pre- and post-immersion microhardness values for Omnichroma were found to be higher than those for Tetric® N-Ceram. This can be attributed to the higher filler loading of Omnichroma (68 vol%) compared to Tetric® N-Ceram (56 vol%), as well as to differences in filler morphology across both composites. Omnichroma is based on supra-nano spherical fillers of silicon dioxide (SiO2) and zirconium dioxide (ZrO2), while Tetric® N-Ceram contains nano-hybrid fillers of barium glass, ytterbium trifluoride, mixed oxide, and silicon dioxide which are a mixture of different sizes and are larger than the supra-nano filled fillers in Omnichroma. Generally, the smoother and more uniform (particularly spherical) filler surfaces showed improved hardness. 36
Although nano-hybrids exhibit a greater surface-area-to-volume ratio and thus higher water sorption potential that accelerates the materials plasticizing effect from aging solutions, 37 the nano-hybrid resin composite tested in this study was less affected by the immersion media compared to the supra-nano-filled composite. Omnichroma exhibited a higher overall percentage reduction in microhardness compared to the nano-hybrid composite Tetric® N-Ceram. It is possible that the supra-nano spherical fillers of Omnichroma exhibited a higher water sorption potential than the nano-hybrid fillers in Tetric N-Ceram, and thus showed a greater overall vulnerability to the immersion media. However, this explanation warrants further investigation. The different reactions of each composite to the different mouthwashes suggest that other factors may have also played a role, such as the composition of the resin matrix and fillers properties, as well as the interactions between specific compositions of each mouthwash and the different resin composites.
Both tested composites incorporate Urethane Dimethacrylate (UDMA) as a base monomer, in addition to other resin monomers in various concentrations (e.g., BisGMA, TEGDMA). The base monomer UDMA (MW = 470.56) is less viscous than BisGMA (MW = 512.59) and exhibits a higher degree of conversion and overall better mechanical properties of the polymer network. 38 However, it still requires the addition of reactive diluent monomers to minimize its viscosity and improve the filler loading. 39 The presence of diluent monomers such as TEGDMA add flexibility to the resin network but may increase the matrix water sorption.38, 40, 41 Additionally, the presence of hydrophilic urethane linkages and ester groups in UDMA resin has been linked to its water sorption potential. 35 It is important to highlight that UDMA represents a wide family of monomers, 42 and thus the particular type used in each composite may affect the overall resin composite properties differently.
Interestingly, samples from both tested resin composites reacted differently to immersion in the distilled water (control). While Omnichroma showed a significant reduction in microhardness, Tetric® N-Ceram showed a significant increase in microhardness post-immersion. The reduction in Omnichroma microhardness after immersion in distilled water corroborate the finding from several studies,32, 36, 43, 44 where a reduction in microhardness values were observed after storage in distilled water. This can be attributed to the plasticizing effect of water on the resin polymer network of various composites. TEGDMA resin, which is the main diluent monomer in Omnichroma, was reported to exhibit higher solubility in water compared to other methacrylate monomers. 40 Water sorption is known to have a weakening effect on the resin matrix and the matrix/filler interface, which can lead to hydrolytic degradation and filler debonding. Such factors can, ultimately, reduce the surface hardness and other mechanical properties of resin composites. 38 The rate of softening and resultant polymer plasticization of resin composite is highly dependent on the uptake rate of immersion solution by the material, which initially impacts the surface properties such as hardness before involving other bulk properties of the material. 35
On the other hand, other studies have shown an increase in surface microhardness after immersion in distilled water,29, 45 as was observed in this study with Tetric® N-Ceram samples. This effect can be explained by the post-irradiation hardness development, where the cross-linking reaction continues, and chemical bonds are progressively formed, resulting in increased surface microhardness.46, 47
In the current study, Colgate® Optic White—which is marketed as a whitening mouthwash—was the only mouthwash that contained hydrogen peroxide in 2% concentration as an active ingredient. Hydrogen peroxide (H2O2) is an oxidizing agent that was used originally to assist oral wound debridement by producing a less favorable environment for anaerobic bacteria. 48
Frequent use of mouthwashes with hydrogen peroxide has, however, been linked to several intra-oral symptoms, including mouth irritation, dryness, loss of taste, elongation of filiform papillae, and diffuse mucosal whitening.48, 49 It is currently the main active ingredient in most OTC bleaching systems, including mouthwashes, which typically incorporate low concentrations (1%–3%) of hydrogen peroxide. 49 Upon interaction with dental materials, it decomposes into free radicals, reactive oxygen molecules, and releases hydrogen peroxide anions. 32
The resin matrix and the organic fillers are both affected by the oxidizing action of hydrogen peroxide, which may result in a softening of the matrix and a reduction in the surface integrity of the resin composite through weakening of the polymer bonds and the creation of surface porosities. 50 This may, in turn, increase the water sorption and ultimately leading to complete or partial debonding of the fillers.50–52 In this study, this effect was observed in both types of resin composites tested, with significant reductions in surface microhardness values post-immersion, although the effect was more pronounced on Omnichroma. This is in line with the findings of another study 14 where a significant reduction in microhardness was reported after immersion in 1.5% hydrogen peroxide mouthwash.
In this study, the least reduction in surface microhardness was observed when the samples from both resin composites were immersed in Biorepair® mouthwash. This could be attributable to the distinct viscosity of Biorepair® mouthwash (it is marketed as “high density” by the manufacturer), which may have contributed to the adsorption of a protective layer with zinc-HA micro-clusters for a longer time on the surface of the tested resin composites. This longer adsorption period could have delayed the loss of resin composite contents and minimized the disintegration of the surface layer. However, this assumption requires further investigation.
The efficacy of Biorepair® mouthwash against acid-induced enamel demineralization has been demonstrated in situ. 7 Transmission electron microscopic analysis revealed the accumulation of HA microclusters on the outer layer of the examined pellicle after the application of Biorepair® mouthwash, which may act as a reservoir for subsequent remineralization. 7 Occlusion of dentinal tubules and reduction of fluid flow has also been demonstrated in vitro after exposure to Biorepair® mouthwash. 53 Similarly, the zinc-HA that Biorepair® incorporates in its toothpaste formulation has been shown to have a protective effect on enamel against erosive challenges in vitro, 54 with significant enamel remineralization potential. 55
Fluoridated mouthwashes are the most frequently used types of oral care products, with fluoride in its various formulations, fulfilling an established role as an effective preventive and antibacterial agent, particularly against streptococcus mutans.7, 56, 57
Various studies have investigated the effect(s) of fluoride mouthwashes on resin composites.13, 14, 58 Some studies have shown, for instance, that fluoride particles can have potentially undesirable effects on composite resin matrices, particularly the monomer contents. 59 On the other hand, other studies have not found any significant difference in the microhardness of a nano-filled resin composite (Filtek supreme XT) upon intermittent immersion (1 minute daily-60 days) in different fluoridated mouthwashes. 60
In this study, Listerine® in its alcohol-free formulation was used which incorporates fluoride (in addition to essential oils) as an active ingredient. This mouthwash has a long history of use in oral health care since the 19th century. 61 In vitro studies have consistently shown that immersion of different resin composites in Listerine® (in its various formulations) results in a considerable reduction of surface microhardness.29, 30, 62
Our findings corroborate the findings from previous research, with the surface microhardness of both resin composites being significantly reduced after immersion in Listerine® mouthwash. Although Listerine® mouthwash is based on sodium fluoride, it is important to note that the observed reductions cannot be attributed solely to the mouthwash’s fluoride content as other ingredients (e.g., essential oils) may have also contributed to this effect.
Apart from the effects induced by their active ingredients, mouthwashes also contain other constituents such as preservatives, stabilizers, solvents, salts, and artificial flavors that can also affect the surface properties of resin composites.1, 13 For example, some mouthwashes may incorporate essential oils, which is the case with both Listerine® (e.g., Thymol, Eucalyptol, and Menthol) and Biorepair® (e.g., communis seed oil, mentha arvensis leaf oil). The daily exposure to such ingredients is expected to affect the surface integrity of resin composites, especially if they exhibited relatively low pH levels or have abrasive potential. 37 The low pH levels have deleterious effects on the resin matrix (e.g., hydrolysis of ester groups in dimethacrylate monomers), as well as on fillers (e.g., surface erosion and potential debonding). 37
The findings from the present study provide some insights into the possible effects of daily use of commercially available alcohol-free mouthwashes on surface microhardness of resin composites, particularly the recently introduced single-shade universal resin composite. Nevertheless, it is important to acknowledge the limitations of the current in vitro study. The current findings are based on simulated and controlled exposure of resin composites to some mouthwashes in vitro, which only reflect some aspects of the clinical condition. The applied in vitro protocol did not fully simulate the multi-factorial clinical environment, where the interplay of diverse factors (e.g., saliva, oral biofilm, eating habits, and use of oral care products) may modulate the effect of mouthwashes on the resin composites. Particularly the possible dilution effect of saliva on various ingredients and their potential interaction with salivary enzymes.
Another limitation is that the pH level of the tested mouthwashes was not directly measured in our study, it was only obtained from the product information provided by the manufacturers. Due to its importance and effect on resin matrix degradation, pH level should be measured in future studies.
Well-designed clinical trials are recommended to evaluate the in vivo effects of mouthwashes on various resin-based restorative materials. This would facilitate an informed selection of mouthwash types appropriate for each oral health condition.
Conclusions
Within the limitation of this in vitro study, the following can be concluded:
The tested alcohol-free mouthwashes resulted in a significant reduction in surface microhardness of both resin composites (nano-hybrid and supra-nano filled). The observed effects were dependent on the compositions of the resin composite and the mouthwash contents. Omnichroma exhibited significantly higher microhardness values than Tetric® N-Ceram both at baseline and after immersion. However, a higher overall percentage reduction in microhardness was observed for Omnichroma compared to Tetric® N-Ceram. Biorepair® mouthwash with zinc-HA resulted in the least reduction in surface microhardness for both resin composites.
Footnotes
Authors’ Contribution
Loulwa MA: Research idea, experimental design; monitored the experiment, wrote the manuscript, performed statistical analysis, discussed the results, proofread the manuscript, and commented on the manuscript at all stages. Lina MA: Contributed to the experiment as part of her internship project: prepared the samples, performed the microhardness test, contributed to introduction writing, and proofread the manuscript. Dhayah SA: Contributed to the experiment as part of her internship project: prepared the samples, performed the microhardness test, contributed to introduction writing, and proofread the manuscript.
Data Availability Statement
All data sets used in the current study are available within the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval Institutional Statement
This study was registered at the college of dentistry research center/ CDRC (IR 0410).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article from any funding agency in the public, commercial, or not-for-profit sectors.
