Abstract
Aim:
Different procedures have been attempted to regenerate intrabony defects in periodontitis but complete regeneration is still a herculean feat. Nanoparticles could prove to be advantageous because of their small size and increased surface area. The aim was to evaluate the effectiveness of a chitosan nanohydrogel as a periodontal regenerative material in the management of intrabony defects.
Materials and Methods:
Twenty patients within the age group of 30 to 60 years with intrabony defects >3 mm and clinical attachment levels >5 mm were selected. They were divided randomly into two groups: group-1, which received only open flap debridement with bone graft and acted as control, and group-2, which received open flap debridement, bone graft mixed with a chitosan nanohydrogel, and acted as the test site. Clinical and radiographic measurements were recorded at baseline, three months, and six months. Intragroup comparison was done using a paired t-test and intergroup comparison was done using an unpaired t-test with the statistical value set at P < .05.
Results:
Chitosan hydrogel incorporated bone grafts showed improved bone regenerative potential. The mean reduction in clinical attachment level in group-2 (8.7 ± 0.6 to 1.6 ± 0.8) was significantly higher than that of group-1 (8.6 ± 0.5 to 2.4 ± 0.7). The mean reduction in probing depths in group-2 (test group) was significantly higher (8.3 ± 0.9 to 1.6 ± 0.8) when compared to group-1 (8 ± 0.8 to 2.3 ± 0.7). Significant amount of defect fill was obtained in both the groups from baseline to six-month period.
Conclusion:
Organic materials such as chitosan could have potential bone regenerative capacity.
Introduction
Periodontitis is a multifactorial disease that could be modified by a variety of systemic factors such as diabetes mellitus, cardiovascular diseases, and chronic obstructive pulmonary disorder. 1 Many studies have focused on studying the expression of a variety of molecules, notably endothelin-1, a vasoconstrictor implicated in many systemic diseases, tumor necrosis factor, and interleukin-21.2–5 Studies have shown that in addition to gram-negative bacteria, viruses such as cytomegalovirus and Epstein–Barr virus may also play a role in the etiopathogenesis of the disease. 6 Failure to control the progression of periodontal disease at the initial stages leads to worsening of the disease, and thereby causing severe bone loss and osseous defects which affect the long-term prognosis of the tooth. Periodontitis remains to be the leading cause for the loss of function and aesthetics.7,8 Many clinicians face the need to prosthetically rehabilitate their patients using dental implants over the recent years. 9
The ultimate goal of periodontal therapy is to try and regenerate the attachment apparatus. Traditionally some authors claim that a strict maintenance regimen with mouth rinses could reduce the progression of periodontal disease.10,11 But that may not always work for the cases with severe tissue destruction and disease. Recent developments in the field of dentistry have revolutionized the approach toward the management of periodontal disease. 12 To achieve this, numerous procedures, including guided tissue regeneration, bone grafts, and the use of growth factors, have been developed, but all have their own limitations.13,14
Periodontal regeneration involves a complex series of events aimed to stimulate progenitor cells to reoccupy the defects, which makes growth factors the ideal candidates as regenerative materials.15,16 In addition to growth factors, stem cells have also been tried for regenerative therapy. 17 Nevertheless, success is still elusive. Autografts are the ideal solution to regenerate lost structures. However, limitations such as donor site morbidity and difficulty in procuring adequate amounts of graft have led us to the usage of alternative materials such as alloplasts and xenografts.
Despite the availability of numerous materials, complete regeneration still remains a herculean feat. These materials have numerous limitations as well. Thus, special attention has been driven toward using the materials which are derived from nature to overcome the drawbacks of synthetic ones. Most notably, natural biopolymers have been shown to yield faster healing and are more biocompatible. 18 In particular, chitin and chitosan (poly-N-acetyl glucosaminoglycan), a carbohydrate biopolymer extracted from chitin, have attracted most attention.19,20 Chitin is the second most abundant natural biopolymer next to cellulose. It is a primary structural component of the exoskeleton of arthropods, e.g. crustaceans, the cell wall of fungi, and the cuticle of insects. The derivative of chitin, i.e., chitosan is a very stable polysaccharide and is a linear polymer of N-acetyl-D-glucosamine monomers. Several desirable properties including high osteoconductivity, biocompatibility, ease of application have made it a desirable material. An important category of chitosan-based materials useful for the application in bone tissue engineering is that of chitosan-based hydrogels, which are hydrophilic in nature and thereby play a major role in adhesion and proliferation of cells. 21
Studies have shown that chitosan had the potential to activate osteoblasts, combat the formation of biofilms, enhance neovascularization, and also had a potent antimicrobial property that makes it a good candidate for bone regeneration.22–24 Thus, this study was undertaken to analyze the effectiveness of a chitosan hydrogel as an adjunct to bone grafts to treat bony defects in periodontitis patients.
Materials and Methods
Study Design
This study was a prospective, randomized, and controlled clinical trial. According to a parallel group design, patients were randomly divided into the control group, where defects were filled with bovine particulate graft (osseograft), and the test group, where the defects were filled with bovine particulate grafts mixed with a chitosan hydrogel. The patients were blinded and did not know which material was used for their surgery.
Sample Size Calculation
A sample size of 20 (10 in each group) was calculated using G Power with the power 80 and α error 0.05 based on the study done by Babrawala et al. 25
Inclusion Criteria
Patients with chronic or aggressive periodontitis according to the American Academy of Periodontology’s 1999 Classification were considered for the study. Patients within the age group of 25 to 50 years with probing depths ≥4 mm after Phase I therapy and at least one angular defect of depth ≥2 mm when measured with intraoral periapical radiograph or with a furcation defect were recruited. If multiple sites were present in the same patient, only one site per sextant was included in this study.
Exclusion Criteria
Patients with a known systemic disease, those on medications such as immunosuppressants or corticosteroids, or under antiresorptive medications were not included in the study. Patients having any adverse oral habits, those who have undergone any periodontal therapy in the past six months, pregnant or lactating mothers, those with poor oral hygiene, those with a known allergy to any component of the graft or the chitosan gels were also excluded from the study.
Randomization and Allocation Concealment
Randomization was done by computer-generated random numbers and allotted to either of the groups. The allocation concealment was done by a sealed envelope to avoid bias. A single periodontal surgeon performed all the surgeries to avoid potential bias.
Preparation of Chitosan
The raw material for the production of chitosan is chitin which is derived from the shells of crustaceans, mainly crabs and shrimps. The conversion of this chitin to chitosan can be achieved either by enzymatic or chemical deacetylation. However, the chemical deacetylation is most favored because it is feasible and economical. The crustacean shells are subjected to a variety of treatments such as demineralization with hydrochloric acid, deproteinization with sodium hydroxide, decolorization with potassium nitrate, and lastly deacetylation with sodium hydroxide to form chitosan.
Preparation of Chitosan Nanoparticles
The nanoparticles were prepared as per the procedure described by Calvo et al. 26 A 2.5 mg/mL chitosan solution was prepared by dissolving low molecular weight chitosan in a 0.05% acetic acid solution (HiMedia, Mumbai), and leaving it under stirring for 24 hours. The pH was adjusted using sodium hydroxide (HiMedia, Mumbai). Tripolyphosphate pentasodium, procured from HiMedia Laboratories Pvt. Ltd, was dissolved in deionized water. The suspension was then centrifuged, and sediments obtained were lyophilized and dried.
Preparation of Chitosan Hydrogel
One gram of the chitosan nanopowder was dissolved in 0.05 M acetic acid (100 mL) procured from HiMedia Laboratories Pvt. Ltd. The pH was adjusted to 8.5 to 9.0 by adding sodium hydroxide (HiMedia, Mumbai). The resultant hydrogel (15% chitosan) was decanted several times, washed, and dialyzed using distilled water. After dialysis, the hydrogel was centrifuged and suspended with a blender to render a homogenous gel.
Initial Patient Preparation
At the initial visit, each patient underwent a full-mouth subgingival scaling and root planing and received proper oral hygiene maintenance instructions. The clinical parameters (probing depth and clinical attachment level [CAL]) were recorded using pressure-sensitive probes, and radiovisiographs (RVG) were taken using RVG positioners to standardize the radiographs (Tables 1 and 2; Figure 4A). The defect depths were measured on the computer from a coronal reference point, the level of the cemento enamel junction (CEJ) arbitrarily by looking at the adjacent teeth until the base of the bone defect. Three weeks later, an evaluation was done and further periodontal management by periodontal flap surgery wherever required was decided.
Clinical and Radiographic Parameters in Group-1
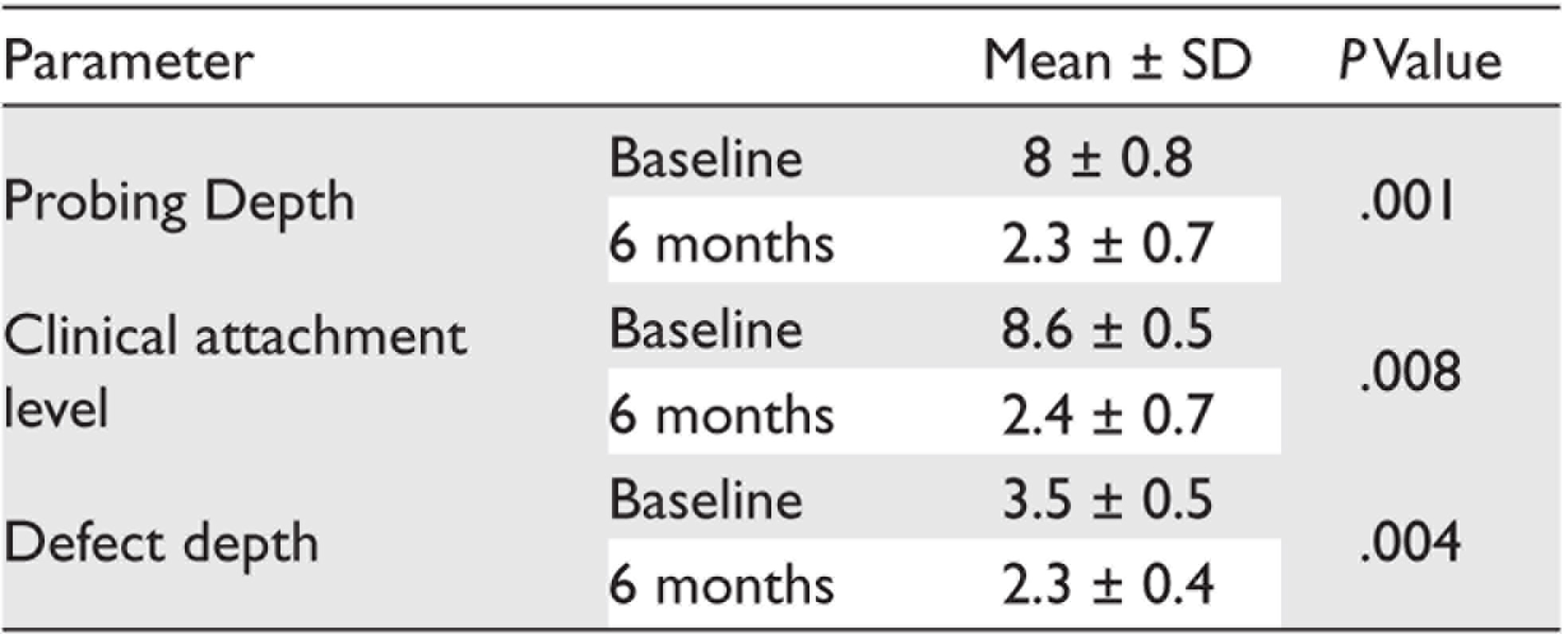
Abbreviation: SD, standard deviation.
Clinical and Radiographic Parameters in Group-2
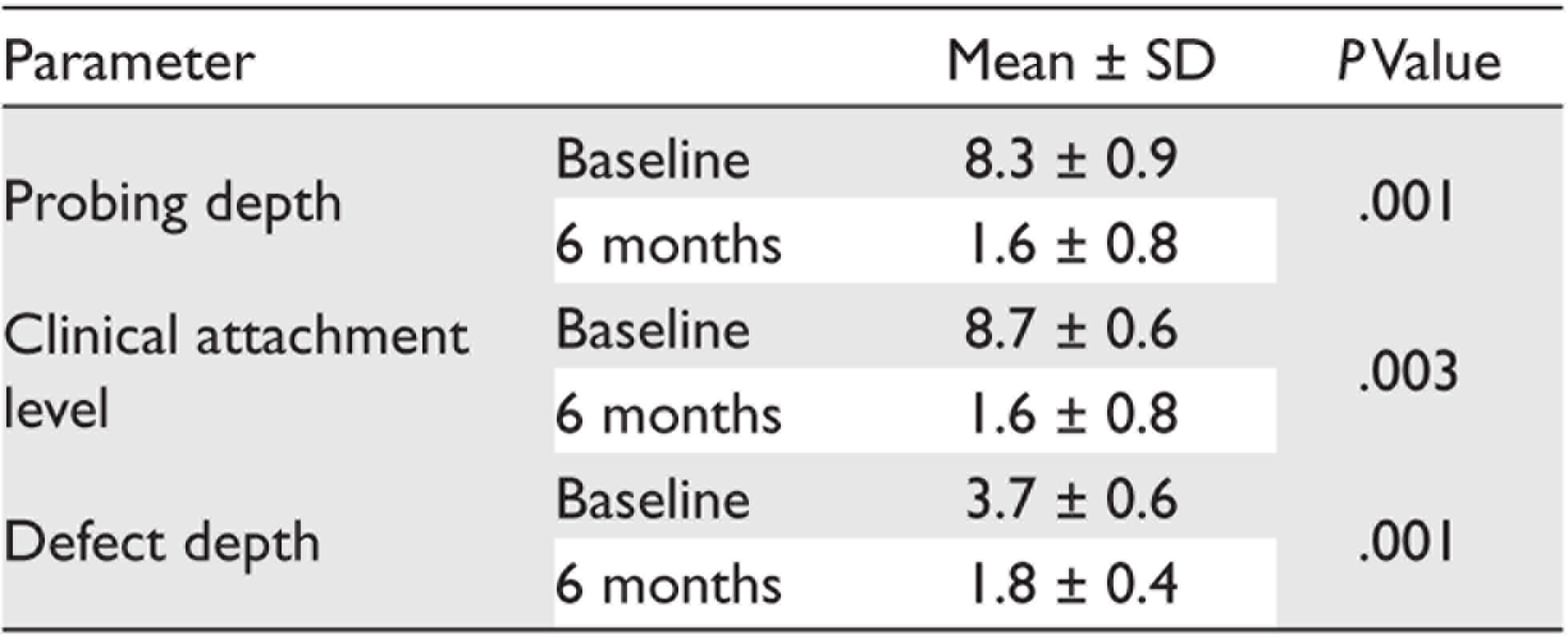
Abbreviation: SD, standard deviation.
Clinical and Radiological Evaluation
Clinical parameters such as probing pocket depth, CAL, and defect depth were recorded at baseline, three months, and at six months. A radiographic evaluation was done at six months and the defect depth was measured to estimate the amount of bone fill (Figures 4B and C).
Surgical Procedure and Postoperative Care
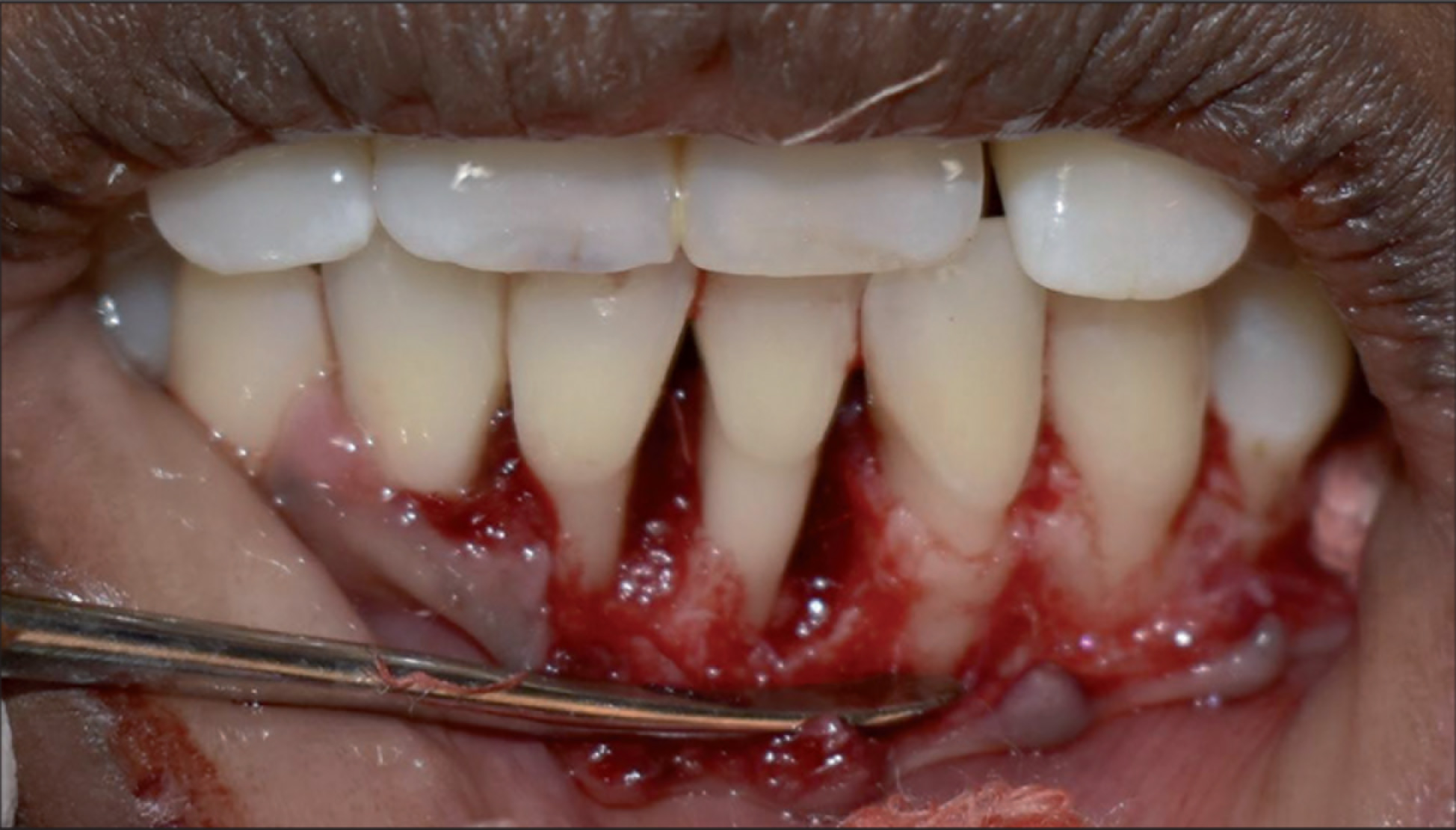
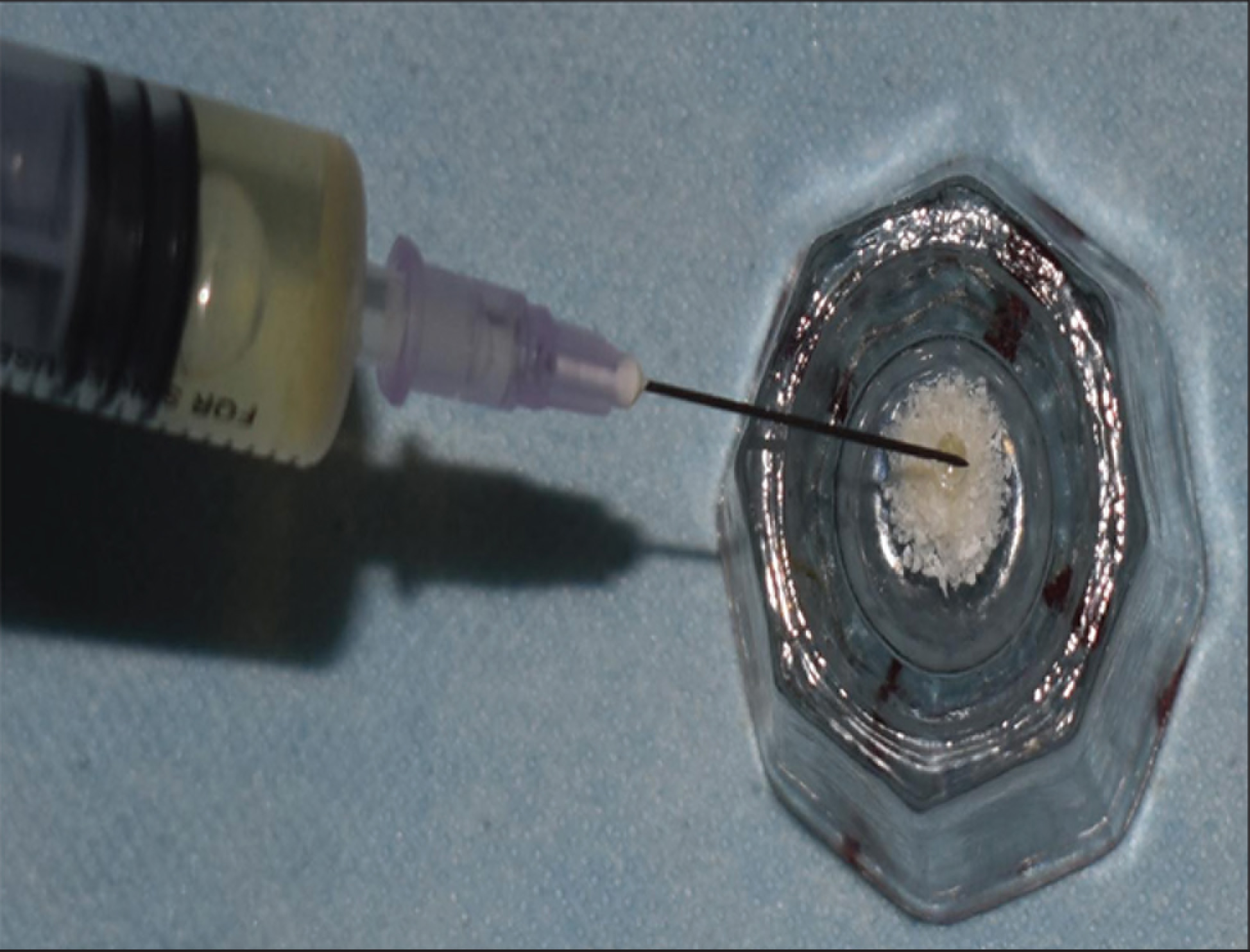
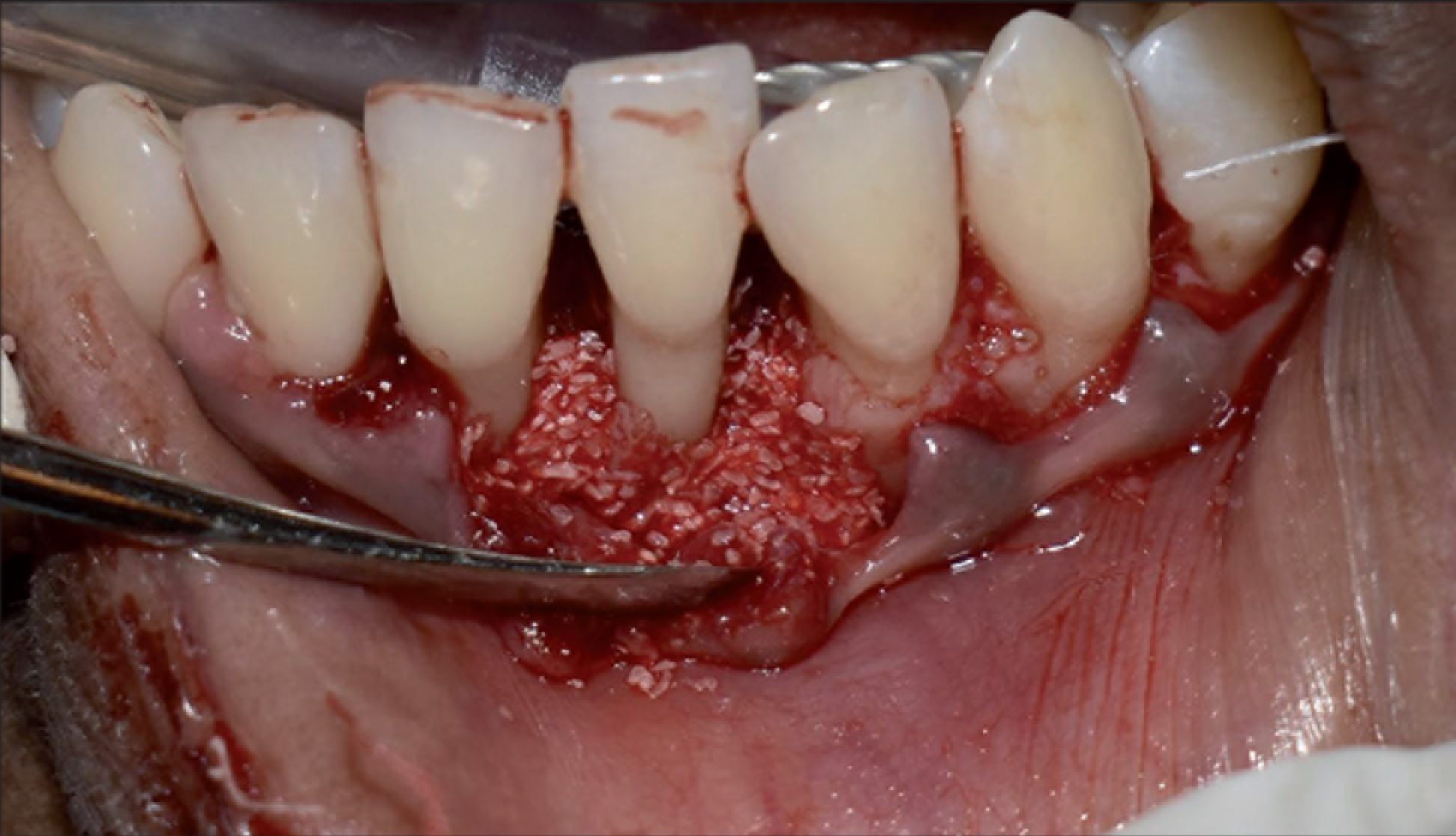
Patients were asked to rinse using 0.12% chlorhexidine digluconate prior to the commencement of surgery. The surgical sites were treated under local anesthesia (2% lignocaine with 1:200,000 epinephrine). Intrasulcular incisions were made on the buccal and lingual/palatal aspects and full-thickness mucoperiosteal flaps were elevated. Thorough root planing and debridement of granulation tissue were done (Figure 1). No osseous recontouring was done. Bone grafts were placed in defects wherever required in the control group. Bone grafts that were mixed with a chitosan hydrogel were placed in the bone defects in the test group (Figures 2 and 3). The flap was secured using interrupted 3-0 nonabsorbable silk sutures (Ethicon, Johnson and Johnson, Somerville, NJ, USA). Antibiotics and analgesics (amoxicillin 500 mg with lactobacillus 60 million spores thrice per day; metronidazole 400 mg thrice per day for five days; and diclofenac sodium twice a day for three days, along with a gastroprotective agent Pantop 20 mg twice a day for three days) were prescribed and postoperative instructions were given.
Intraoperative Image of the Bone Defect
Bone Graft Mixed With the Prepared Chitosan Gel
Intraoperative Image Depicting the Bone Graft Mixed With Chitosan Placed in the Defect
Postsurgical Measurements
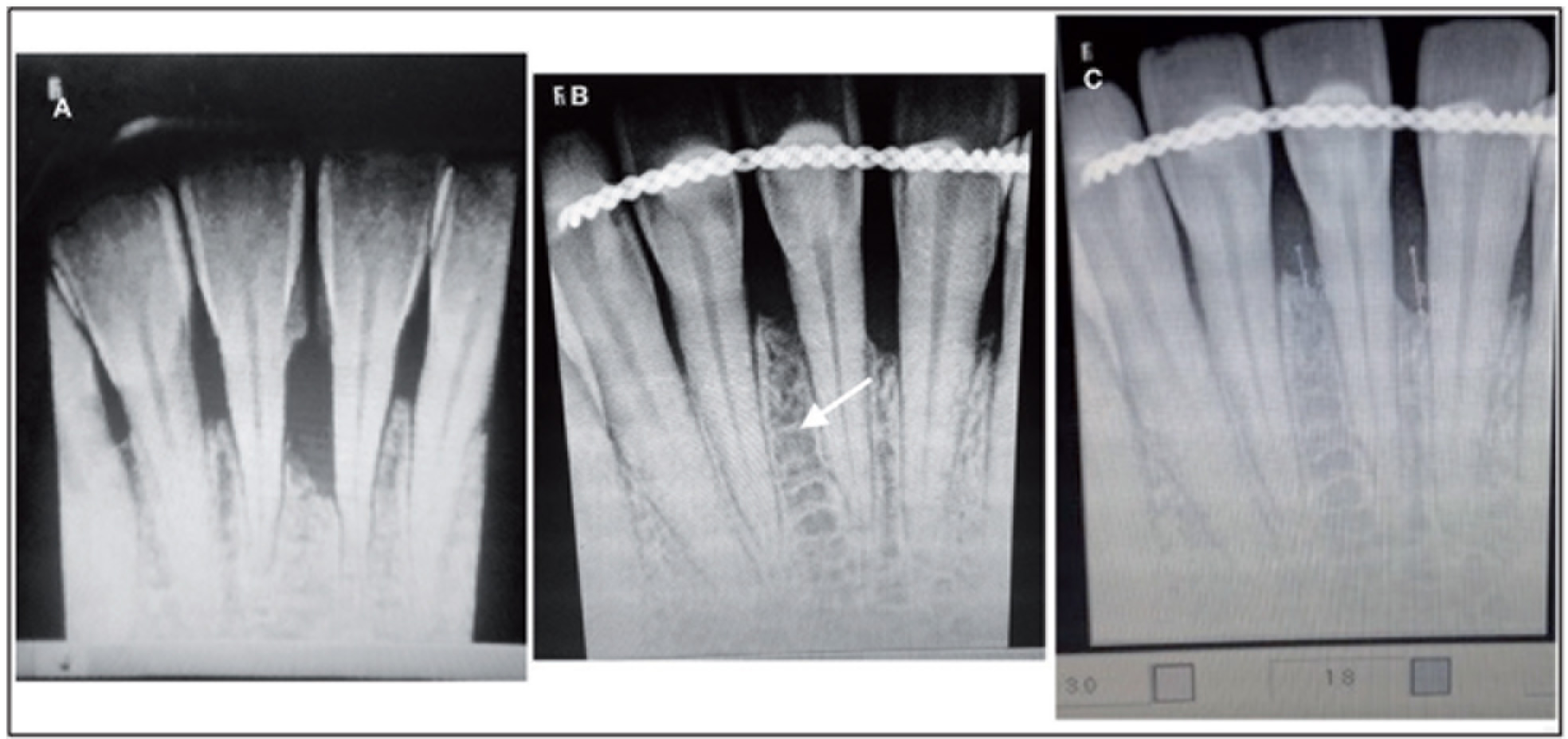
A clinical and radiographic evaluation was performed six months after surgery. Clinical parameters such as probing depth and CAL measurements were repeated with the previously used acrylic stents. For a radiographic intrabony defect depth measurement, RVG was taken using standardized positioners, and a similar measurement that was followed at baseline was also done (Figure 4).
(A) Preoperative Radiograph, (B) Postoperative Radiograph, and (C) Measurement of Defect Depth in RVG
Statistical Analysis
Intragroup comparison was done using a paired t-test and intergroup comparison was done using an unpaired t-test. The significance level was set as P value <.05.
Results
A total of 20 defects in 10 patients in both the test and the control groups were evaluated. The treated sites were evaluated for clinical parameters at baseline, three months, and six months postoperatively. No patient dropped out during the study and uneventful healing was observed. Good oral hygiene was maintained by the patients during the study period.
Both groups exhibited a significant reduction in probing pocket depths and CAL at six months. The mean reduction in probing depths in group-2 (test group) was significantly higher (8.3 ± 0.9 to 1.6 ± 0.8) when compared to group-1 (8 ± 0.8 to 2.3 ± 0.7). The mean reduction in CAL in group-2 (8.7 ± 0.6 to 1.6 ± 0.8) was significantly higher than that of group-1 (8.6 ± 0.5 to 2.4 ± 0.7; Figure 5).
(A) Parameters in Control Group, (B) Parameters in Test Group, and (C) Reduction in Defect Depth
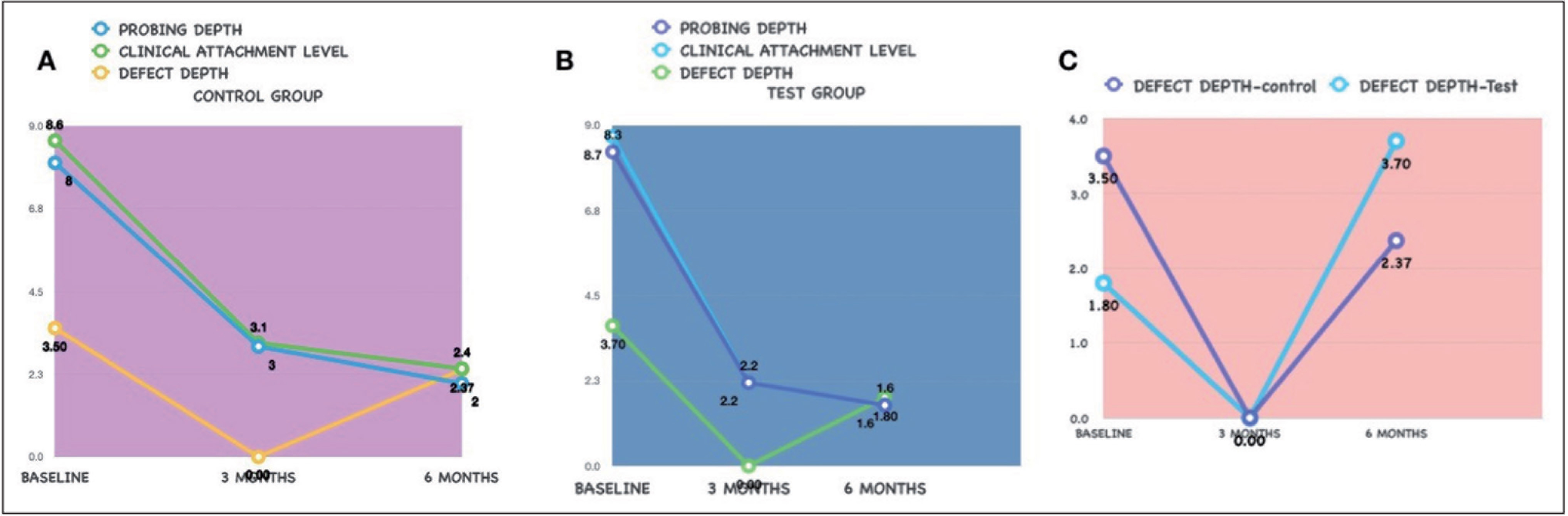
The mean amount of defect fill observed in group-1 (3.5 ± 0.5 to 2.3 ± 0.4) and group-2 (3.7 ± 0.6 to 1.8 ± 0.4) from baseline to six months was recorded. Statistically significant differences in the amount of defect fill were observed between the two groups at six months (Tables 1 and 2).
Intragroup analysis done by a paired t-test demonstrated statistically significant changes from baseline to six months in both the groups. Intergroup analysis done by an unpaired t-test showed statistically significant differences between the groups at the end of six months (P value <.05).
Discussion
Although the healing effects of chitosan on mammalian wounds have been appreciated for centuries, it did not become popular until the 1960s. Reynolds laid the foundation for the wound healing properties of chitosan. 27 Subsequently, a variety of animal studies followed that reported chitosan had the capability to promote wound healing. 28 One notable study was done by Park et al. where the effects of chitosan on the regeneration of periodontal tissues in beagle dogs were studied. They demonstrated, histologically, an increase in new bone formation and cementum regeneration at eight weeks. 29 Chitosan is one of the most used biopolymers in tissue engineering because of its wide availability, biodegradability, and nontoxicity. Its structural similarity to the components of extracellular matrix makes it an ideal material for bone and tissue regeneration. 30
It is a promising scaffold material because of its capacity to potentiate the differentiation of osteoprogenitor cells that facilitate bone regeneration. 31 It is structurally similar to glycosaminoglycan hyaluronic acid found in the extracellular matrix, which plays a crucial role in tissue regeneration.
In this study, the effect of a chitosan hydrogel in combination with bone grafts in the regeneration of bone defects was compared to that of using bone grafts alone. We wanted to assess the bone regenerative potential of chitosan in combination with bone grafts, without the usage of barrier membranes in order to assess if the resultant regeneration achieved was because of inherent potential of chitosan itself. Also, clinically a chitosan gel behaved like a binder and held the graft particles in place. To the best of our knowledge, this is the first study to use a chitosan hydrogel prepared from chitosan nanoparticles combined with bone grafts to regenerate bone defects. Therefore, a direct comparison with similar studies was not possible.
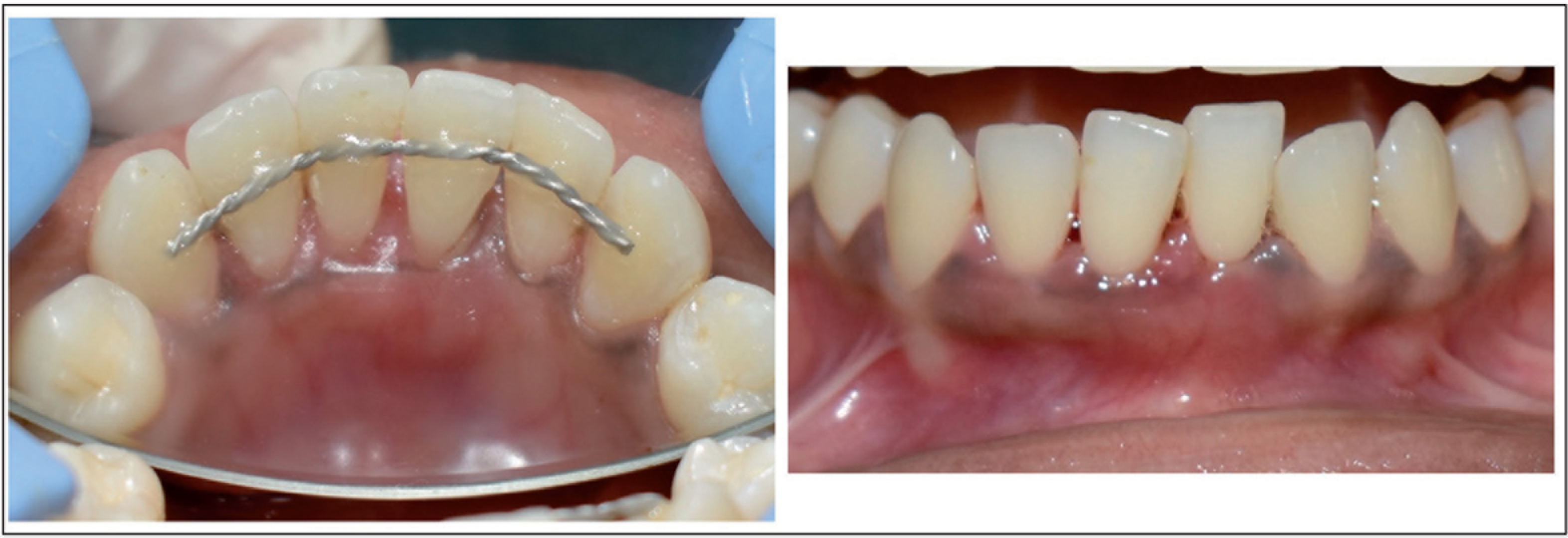
The results of the present study demonstrated that chitosan showed significant improvement in clinical as well as radiological parameters. Defects treated with a chitosan gel mixed with bone grafts exhibited a significant reduction of probing depth, improvement in CAL, and a significant defect resolution at six months. In the follow-up visit at 12 months, there was very good soft tissue healing clinically with no recession evident (Figure 6). In a case series by Babrawala et al., they used a combination of a 15% chitosan gel with bovine porous mineral for three-walled defects and found significant bone regeneration and soft tissue healing, which is in accordance with our study. 25 Boyneugri et al. studied the potential of a 1% chitosan gel as an adjunct to demineralized bone matrix and observed a significant defect resolution at six months compared to open flap debridement alone. 32
Postoperative View at 12 Months
In our study, we have also included patients in whom splinting was done. One could argue that splinting could have had a role in healing, but then in order to achieve regeneration, all the factors playing a role in healing need to be addressed. Several studies have investigated the effects of chitosan on bone healing and stated different hypotheses on its mechanism of actions. Klokkevold et al. proposed that chitosan had an ability to potentiate the differentiation of osteoprogenitor cells that may facilitate bone regeneration. 33 Mukherjee et al. evaluated a paste of chitosan glutamate and hydroxyapatite as a synthetic bone graft material in rats and found favorable outcomes. 34 Chevrier et al. postulated that chitosan was capable of increasing the vascularization of blood vessels and stimulating fibroblasts. 22 Park et al. as well as Lee et al. reported that spongy chitosan could activate osteoblasts and could increase osteogenic potential.35,36 In a study by Zhang et al., chitosan was used along with mannitol and calcium phosphate cement for bone healing. They reported that this new formulation could be used for shaping hydroxyapatite in surgeries and implants. 37
One of the main strengths of our study was that we included one-walled defects as well, where generally regenerative procedures are not performed. Because chitosan behaved like a binder, we used it in one-walled defects as well, and a surprisingly good amount of regeneration was obtained. However, one of the limitations of our study was the lack of histological evaluation, which could show evidence of true regeneration. We did not include teeth with hopeless prognosis and thus did not want to compromise the site of interest with a bone biopsy. Another limitation is that we did not have a long-term follow-up. Further research with histologic analysis could establish the usage of chitosan as a potential regenerative material.
Conclusion
In conclusion, this study demonstrates that a chitosan hydrogel in combination with a bone graft showed superior bone regenerative potential and could prove to be an excellent candidate for bone regeneration.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement:
The protocol was approved by the Institutional Ethical Committee and Review Board (MDS/ SRB//PERIO/18–19/012). All procedures were performed in compliance with the Declaration of Helsinki and followed the Consolidated Standards of Reporting Trials (CONSORT) 2010 statements. All participants received detailed descriptions of the study and a written informed consent was obtained prior to the intervention.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
