Abstract
Aim:
To investigate the effect of sodium ascorbate (SA), an antioxidant and calcium hydroxide (Ca(OH)2), a buffering agent mixture, on the shear bond strength of a luting resin to dentin, and pH changes of the extraradicular medium solution after intracoronal bleaching with hydrogen peroxide.
Materials and Methods:
In this in vitro study, 60 human maxillary central incisors were randomly assigned into six groups (n: 10): Group 1: restored, no bleach; group 2: bleached, bonded immediately; group 3: bleached, treated with 10% SA applications before bonding; group 4: bleached, treated with calcium hydroxide applications before bonding; group 5: bleached, treated with CaOH2 and SA mixture applications before bonding; group 6: delay bonding by seven days. For bleach treatment in groups 2 to 6, intracoronal bleaching was performed with 35% hydrogen peroxide for four days. SA or CaOH2 or mixture of SA and CaOH2 were left in the access cavity for four days. Hybrid blocks, 4 mm × 4 mm wide × 2–mm thick, were luted with resin cements on dentin surface and then the specimens were subjected to shear bond strength testing. Analysis of variance and Tukey post hoc multiple comparison tests were applied (P < .05).
Results:
G2 and G4 showed significantly low bond strength values and they were lower than the other groups (P < .05). There were no significant differences between G1, G3, and G6 (P > .05). The pH values had risen markedly in the groups containing calcium hydroxide (G4 and G5) after four days (P < .05).
Conclusion:
The application of 10% SA or 10% SA and CaOH2 mixture were effective at reversing bleaching effects on bond strength. CaOH2 or CaOH2 and 10% SA mixture increased the external medium pH and were effective for alkalinization after intracoronal bleaching.
Introduction
Discoloration of endodontically treated teeth may occur because of intrapulpal hemorrhage, pulp necrosis, root canal sealer, gutta-percha remaining, and/or iatrogenic negligence at various stages of endodontic treatment. Discoloration caused by root canal treatment should be removed before ceramic laminate veneers treatment. Because veneers are generally fabricated with varying thicknesses ranging from 0.3 mm to 1 mm and reflect the underlying tooth color. Hydrogen peroxide (H2O2) and sodium perborate are used in intracoronal bleaching. Using 30% to 35% H2O2 in nonvital, discolored teeth has shown successful intracoronal bleaching in a short time. 1
Most of the results of bleaching studies for endodontically treated teeth were found sufficiently effectual, but a few studies stated some side effects of the bleaching therapy. The cervical resorption was common side effect for intracoronal bleaching therapy.2,3 It is believed that intracoronal bleaching treatments decrease the pH and generate an inflammatory process around the teeth and implicate the cervical root resorption process. 4 Therefore, a calcium hydroxide (Ca(OH)2) dressing with a high pH (the pH value of the calcium hydroxide and glycerin mixture is close to 12) 5 has been suggested in many studies to neutralize H2O2 and increase the pH during intracoronal bleaching procedures thus preventing cervical resorption. 6
Another common side effect is the decreased bond strength.7,8 The presence of residual oxygen and peroxide from the bleaching agent may provoke the decrement of the bonding strength in bleached teeth according to some bleaching studies.
This residual oxygen is thought to cause inhibition of resin polymerization.9,10 The peroxide radicals affect peritubular and intertubular dentin, dissolving the organic part, increasing the permeability, and decreasing the hardness and elastic modulus. 11 In order to minimize this problem, it has been suggested to apply an antioxidant agent such as sodium ascorbate (SA)12,13 between bleaching and restorative treatment, or delay the restorative process from one week to two weeks.14,15
It is not known how mixing the agents used for the two most common complications after intracoronal bleaching will affect the expected benefit of the agents.
The aim of this study is to investigate the effect of SA, an antioxidant and calcium hydroxide, a buffering agent mixture, on the shear bond strength (SBS) of a luting resin to dentin, and pH changes of the extraradicular medium solution after intracoronal bleaching with H2O2. The null hypothesis of the study is that mixing SA and calcium hydroxide does not affect the expected benefits of the material.
Materials and Methods
Setting and Design
In order to perform the present in vitro study, 60 freshly extracted, erupted human maxillary central incisors were collected and stored in a solution of 0.5% Chloramine-T for three months at 4°C. Teeth with carious lesion and restoration, cracks caused during extraction, and developmental disorders that affect enamel and/or dentin were excluded from the study. Ethics Committee of Cukurova University Faculty of Medicine, dated November 13, 2020, numbered 45. Informed consent forms were signed by all patients. The procedures used in this study adhere to the tenets of the Declaration of Helsinki.
All teeth were sectioned on 3 mm apical to the cement–enamel junction with a diamond disc using a sectioning machine (Accutom 10, Struers, Rødovre, Denmark) under water. Standard endodontic access cavities were created by measuring with a bone caliper remaining 3 mm of dental tissue from the buccal middle triple with a 12 ≠ round medium grit diamond bur (Diatech, Coltene Whaledent, Altststten, Switzerland) with a high-speed hand piece under water cooling. Radiography confirmed that approximate 2-mm-dentin and 1-mm-enamel thickness remained in each tooth. The coronal pulp tissue was removed with a spoon excavator and medium grit diamond cavities were irrigated with 5 mL of 2.5% sodium hypochloride to dissolve organic tissues. A final flush with 2-mL saline solution was performed. The apical seal produced by 2-mm-thick layer of glass ionomer cement (Fuji IX, GC, Tokyo, Japan). The specimens were randomly divided into six groups (n = 10);
pH Testing
Four cemental–dentinal defects were prepared just below the cemental–enamel junction (CEJ) on each root. The hemispherical defect (diameter 1.0 mm, depth 0.5 mm) was created using a round diamond bur (diameter 1.0 mm) in a high-speed handpiece. Each tooth was suspended in a plastic vial containing 20 mL of distilled water or saliva. The pH measurements were performed immediately (day 0) after immersion of the teeth. The pH was measured four days after the intracoronal bleaching procedure. During this period, the teeth were stored in the same medium without change of distilled water/saliva or bleaching material. Distilled water was replaced and the pH was measured four days after filling with calcium hydroxide or/and SA. The pH of the surrounding distilled water or saliva was evaluated on day 0, 4, and 8.
The pH meter ( PCE-PH 16, PCE Instruments, Germany) was calibrated with standard pH solutions before each experiment. For each sample, the average of two readings was utilized.
Shear Bond Strength Test
The buccal aspect of teeth were ground with 400 grit wet silicon carbide paper on a polishing machine (Exakt 400 cs Apparatebau, Norderstedt, Germany ), to obtain 4 mm × 4 mm flat, midcoronal dentin surface. A total of 60 hybrid blocks (Cerasmart, GC, Tokyo, Japan), measuring 4 mm × 4 mm wide × 2-mm-thick cubes were prepared with a diamond disk using a sectioning machine under water.
All prepareted hybrid block pieces were etched with 9.5% buffered hydrofluoric acid gel (Porcelain Etchant, Bisco, Schaumburg, IL, USA) for 90 s, rinsed with water, and carefully air-dried. Afterward, a primer (G-Multi Primer, GC, Tokyo, Japan) was applied for salinization of the etched restoration surfaces, and thinned with dry air spray.
A dual-polymerized resin cement (G-CEM Link Force. GC, Tokyo, Japan) was used for luting the restoration materials for all specimens. The resin cement was applied to the restoration surface. Soon after, the restoration material was pressed firmly on preparation surface of the tooth. Slightly hardened excess cement was carefully cleaned with microbrushes, and then 20-s light-cured.
The teeth were embedded horizontally into the acrylic resin making the buccal surface facing upwards. Shear load was applied in a direction parallel to the bonded interface via a universal testing machine (MOD Dental MIC-101, Esetron Smart Robototechnologies, Ankara, Turkey). The load speed was set at 0.5 mm/min and the loading was performed until failure occurred. The recorded shear bond force in newton was converted to megapascal (MPa) to symbolize the bond strength.
Statistical Analysis
The statistical analyses were performed using SPSS (IBM SPSS, Inc., Chicago, IL) 23.0 software. The data were normally distributed based on the results of the Shapiro–Wilk test (P > .05); thus, one-way analysis of variance was used for the SBS data analysis. The analysis of variance for repeated measurements was used for pH changes. Tukey post hoc test was used for the pairwise comparisons (P < .05). In order to see the pH changes in detail, the differences of pH values were taken for the measurements of day 0 and day 4, day 4 and day 8, and also on day 0 and day 8. These differences were analyzed by using one-way analysis of variance for group differences. The failure mode ratios were evaluated using the chi-squared test (P < .05).
Results
Bond Strength
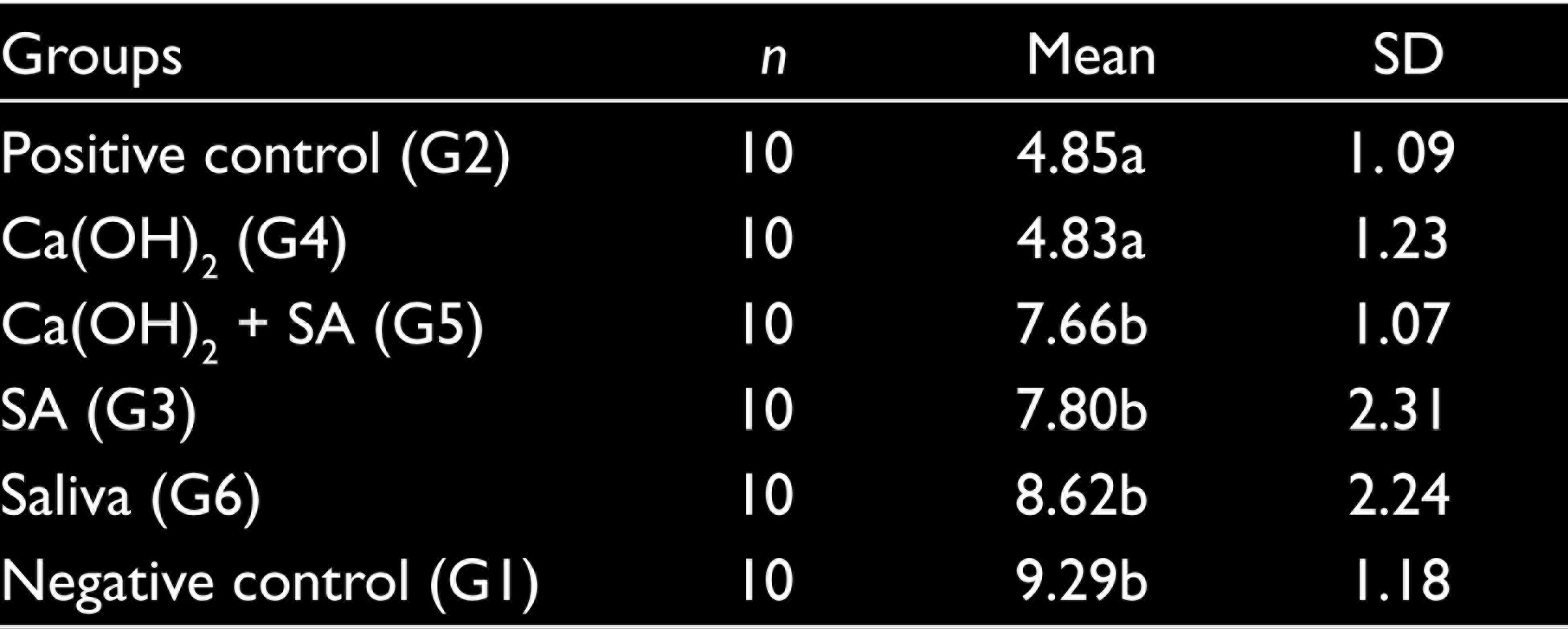
Mean SBS values with standard deviation and differences between the groups are listed in Table 1. The analysis of variance indicated a statistically significant difference among the groups as a result of differences in sample treatment procedures (P = .000). Therefore, data were further analyzed by using the Tukey test for pairwise comparisons. There were no significant differences between the saliva (G6), negative control (G1), and ascorbic acid applied (G3, G6) groups (P = .072). G2 (bonded with composite resin immediately after bleaching), and G4 (calcium hydroxide applied) groups showed significantly low bond strength values and they were lower than the other groups (P = .001).
Means and Standard Deviations of the Shear Bond Strength Values (MPa)
Fracture Modes
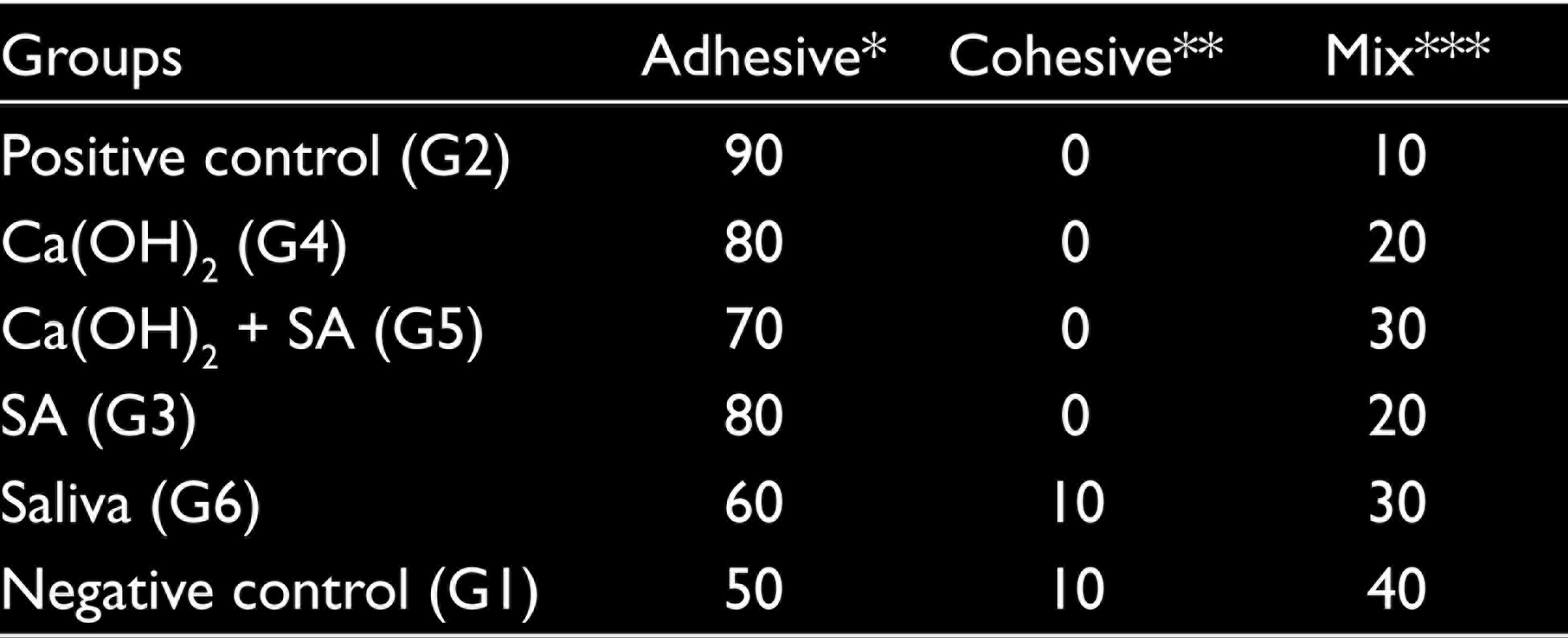
The fracture modes of groups are shown in Table 2. In each group, mainly adhesive fractures were observed. The number of adhesive fracture modes was high in the immediately bonded after bleaching group (positive control). The number of cohesive type of fracture mode was high in the saliva and negative control group. There were no significant differences among the study groups in terms of fracture modes (P = .657).
Failure Mode (%) of the Groups
pH Measurements
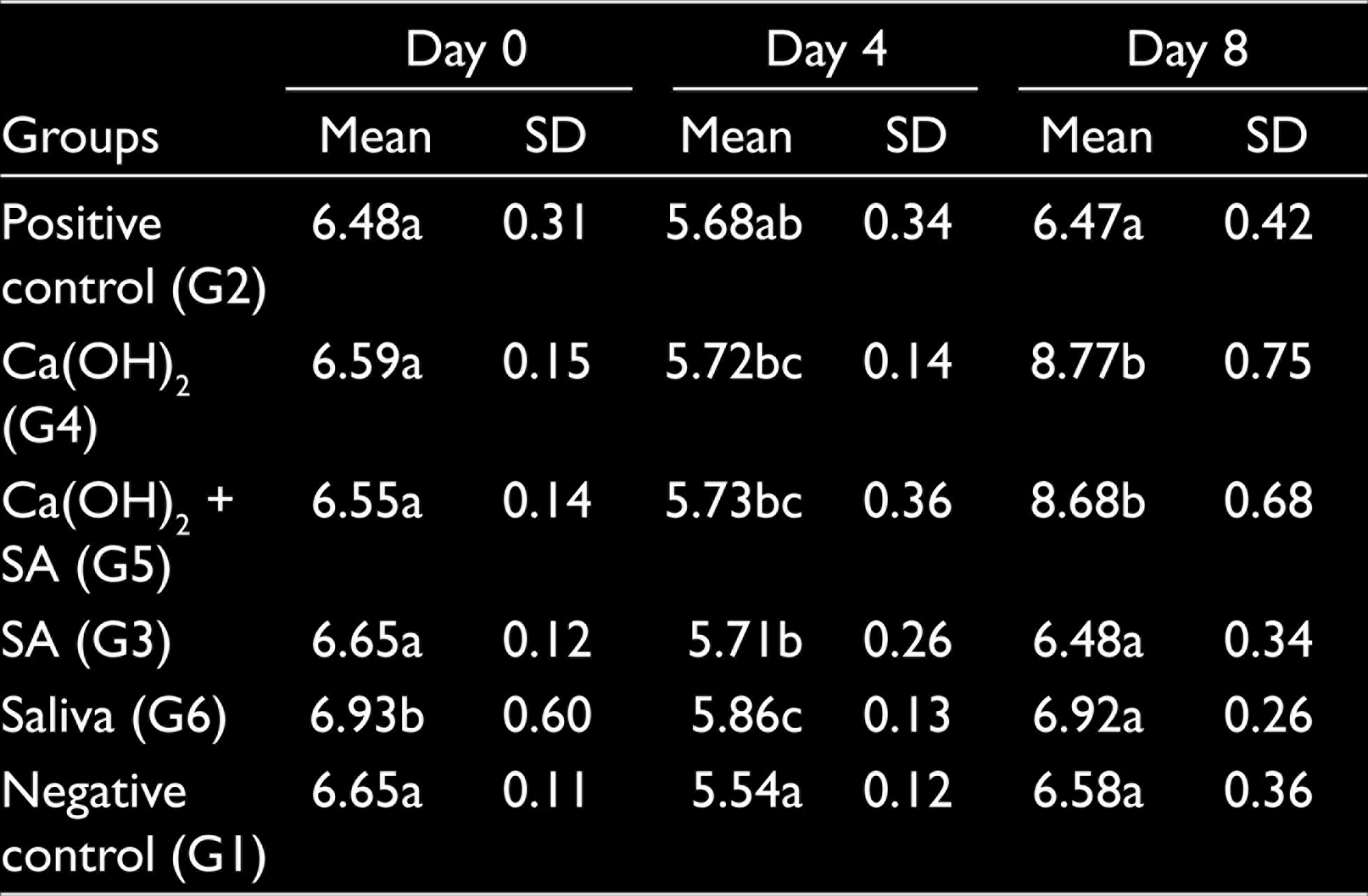
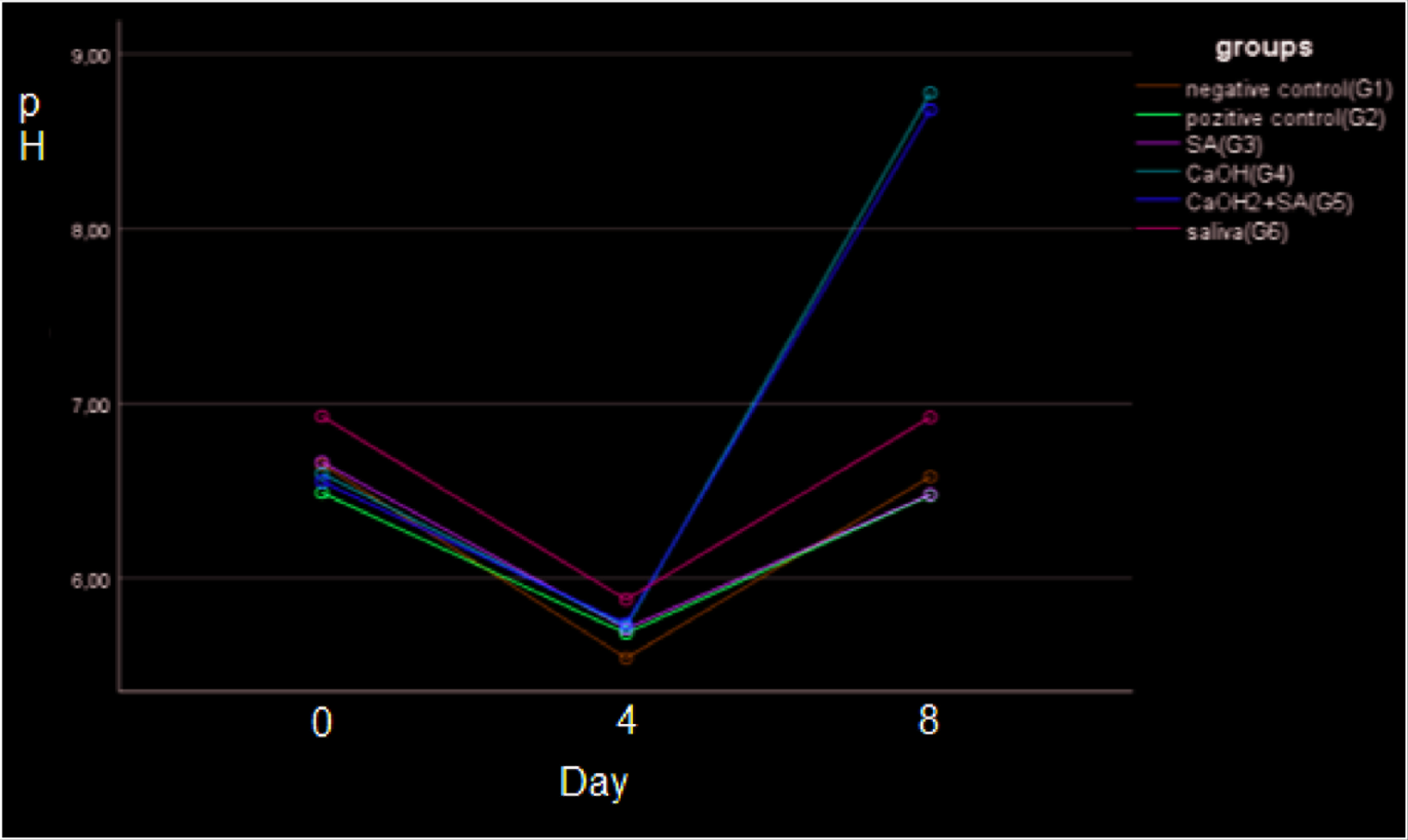
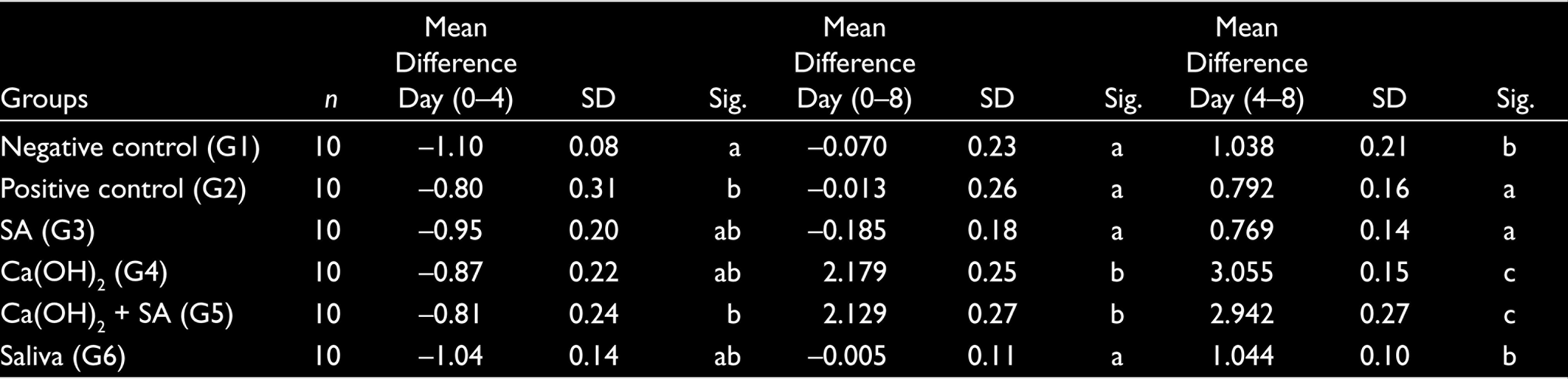
The pH changes of the extraradicular medium solution are summarized in Table 3. The base line pH values of the teeth in the groups are compatible with the pH of distilled water and saliva. Four days after the placement of bleaching agent, mean pH was recorded within a narrow band between pH 5.54 ± 0.12 and pH 5.86 ± 0.13 (Figure 1). Four days after placement of the calcium hydroxide paste, the pH values had risen markedly in the groups containing calcium hydroxide and were statistically different from the other groups (P < .001). At day 8, the mean differences of pH changes in groups using Ca(OH)2 are statistically significant compared to other groups. (P < .001; Table 4)
Summary of pH of Extraradicular Medium Solution
The Mean Differences of pH Changes From Day 0 to Day 4, Day 0 to Day 8, and Day 4 to Day 8
The Mean Differences of pH Changes From Day 0 to Day 4, Day 0 to Day 8, and Day 4 to Day 8, Also Significances of Groups
Discussion
One of the most undesirable effects observed in internal bleaching is cervical resorption and the other is compromised bond strength. There is a possibility of these two adverse effects to be seen together in intracoronal bleaching treatment. The current study assessed the effect of antioxidant and buffering agent mixture on dentin shear bond strengths of laminate veneer, and the pH change of external medium after intracoronal bleaching. Null hypothesis is not rejected. Mixing of 10% SA and calcium hydroxide did not adversely affect their expected benefits.
Several studies have recommended a time delay after bleaching to achieve optimum bond strength. However, there is no consensus on the exact time in the literature; some studies have been recommended for one week,17,18 and some studies have reported that two weeks19,20 or even three weeks21,22 are required. As most of the studies suggested delaying for one week, in our study, the specimens in delayed group were immersed in artificial saliva solution for one week. Natural saliva which contains vitamin E and ascorbic acid acts as an oxidizer. In this study, it was shown that delay of one week increased the bond strength to the level of the nonbleached group. It is thought that during the storage of the samples in artificial saliva, the oxygen remaining in the dentin tissue is removed and the damaged dentin may be repaired.
Natural, nontoxic, and biocompatible SA activity is one of the most evaluated antioxidant after tooth bleaching. Several researchers reported that 10% SA is sufficient to reverse the reduction in bond strength for dentin and enamel after tooth whitening.12,13,23,24 Therefore, 10% SA was preferred as an antioxidant in our study. In the recent study, the use of SA for equal of bleaching time was found to be sufficient to reverse the compromised bonding in the treated enamel.
The use of SA alone or mixed with calcium hydroxide increased the bond strength. There is no statistical difference between the two groups. The results of this study were well matched with the results of above mentioned studies; the bond strength for negative control and saliva groups were similar to the SA group’s strength.
Calcium hydroxide did not reduce the effect of SA. In this study, calcium hydroxide Ca(OH)2 did not increase the bond strength when used alone. No significant difference was found between the calcium hydroxide Ca(OH)2 and positive control groups.
The pH values of the samples at day 0 were compatible with the extraradicular medium. The pH values of samples stored in distilled water were between 6.65 ± 0.12 and 6.48 ± 0.31. The pH of the sample stored in saliva was 6.93 ± 0.60. There was significant reduction in the pH values after four days of intracoronal bleaching application in all samples. The pH of the 35% H2O2 gel used in the current study is 5. 25 Therefore, the use of H2O2 as an intracoronal bleaching agent decreased extraradicular pH in all groups. This study suggests that there was a pH change of the external medium, in agreement with the results of Rokaya et al., 25 de Sá et al., 26 and Lou et al. 27
Cai et al. 28 and Mustafa et al. 29 found that the pH of the dentin increases after placing calcium hydroxide Ca(OH)2 in the root canal. de Sá al. 26 reported that calcium hydroxide increased the pH of external medium. They hypothesized that alkaline pH may interfere with osteoclastic activity and also stimulate hard tissue formation; therefore, it has a preventive effect on cervical resorption. In this study, calcium hydroxide or/and SA mixture dressing placed in the pulp chamber for buffering the acid pH created statistically significantly higher pH than the other groups. A 10% aqueous SA solution has a pH of 7.3 to 7.6. Therefore, it did not affect the pH of the highly alkaline calcium hydroxide. In the saliva group, the acidic pH reached near the initial value again via the buffering of saliva.
Today, intracoronal bleaching of nonvital discolorations is a common treatment. However, the presence of cement defects can increase the risk of cervical root resorption or causes low bond strength in adhesive restorations. A mixture of calcium hydroxide and SA can be considered as a dressing to avoid these two complications.
Conclusion
Treating the bleached dentin surfaces with 10% SA or a mixture of calcium hydroxide and SA, which reversed the compromised bond strength, were good alternatives to delayed bonding. Calcium hydroxide or a mixture of calcium hydroxide and SA increased the external medium pH and were effective for pH alkalinization after intracoronal bleaching.
Footnotes
Acknowledgements
The author would like to acknowledge Professor Dr Yuksel Bek, Department of Biostatistics and Medical Informatics, Ondokuz Mayis University, Turkey, for his assistance in statistical analysis.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
