Abstract
Aim:
The aim of this study was to evaluate the effect of different cooling times on shear bond strength between cobalt–chromium metal frameworks fabricated by different techniques and veneering porcelain.
Materials and Methods:
One hundred twenty cobalt–chromium metal ceramic samples were obtained by three different techniques (casting, milling, and laser sintering). During the porcelain firing, fast and slow cooling protocols were applied. Ten specimens from each group were determined for all groups. The shear bond strength of the metal ceramic samples was measured by a universal testing machine with a constant crosshead speed of 0.5 mm/min. One sample from each group was evaluated by the scanning electron microscopy analysis in terms of surface change and fracture morphology. Variance analysis and Tukey test were used to analyze statistically significant differences between groups.
Results:
It was seen that the difference between the bond strengths of the metal frameworks obtained by different techniques was statistically significant (P < .05). All groups except for difference between the bond strengths of all the frameworks metal groups with G-Ceram porcelain were statistically significant (P < .05). The best bond strengths that were statistically significant were fast cooling G-Ceram (19.65 ± 2.65 MPa) with metal frameworks fabricated by laser sintering, fast cooling with metal framework fabricated by milling Noritake (19.17 ± 2.91 MPa), and the metal framework fabricated by casting was found to be slow cooling Noritake (12.99 ± 2.08 MPa) were seen.
Conclusion:
The porcelain cooling times had significant effect on the shear bond strength of porcelain to casting, milling and laser sintering alloys.
Introduction
Metal ceramic restorations are used for prosthetic treatment for a long time in tooth deficiencies. With increasing use of technology and esthetic demands, metal ceramic restorations are becoming frequently used treatment options for complicated cases. 1
For the clinical success of restorations, it is important that there is a strong connection between the metal substructure and the porcelain. Metal alloys with different metal contents are made with the casting method for many years. One of the metal alloys used cobalt–chromium alloys (Co–Cr) has been used instead of Ni–Cr alloys because of the difficulty of casting, the effect of excessive oxides on the porcelain bond and color, and the allergic or toxic effect of nickel-sensitive patients. 2
In recent years, technology of producing dental metal alloys have improved with the dental technological developments and nowadays the use of Computer-Aided Design/Computer-Aided Manufacturing (CAD/CAM) and laser sinter technology in manufacturing of metal alloys has become popular. 3 Expectations from restorations produced with CAD/CAM in dentistry are obtaining higher and homogeneous quality restorations by using prefabricated blocks and reducing production costs. 3 Shrinkage during casting is eliminated in metal substructures produced in selective laser sintering devices. This is where multimember restorations passively settle on the supporting teeth.3,4
Dental laboratories prefer to use alloys for metal–ceramic restorations due to their higher mechanical properties, high bond strength to porcelain, and corrosion resistance. 5 In studies, it has been stated that the infrastructure is the key factor in mechanically holding and bonding porcelain on the substructure.1,5
Porcelain fractures and breaks usually proceed through the area exposed to the highest tensile stress. Microcracks and pores that progress from the inner surface can increase the internal tension in two-layer restorations and both damage the restoration and negatively affect the porcelain connection. 6 Tuccillo and Nielsen stated that when porcelain is cooled after firing, it can create rupture stresses between the metal substructure and porcelain that negatively affect the bonding strength. 7 They stated that residual stresses on the porcelain metal border may be due to the firing of the porcelain 8 and controlled cooling rates may increase the bonding strength of the porcelain to the metal substructure.7,9 Although there are researches on the effect of cooling rates on the bond strength of veneering porcelain to zirconia material, there are few data on the effect of cooling rates on the bond strength of veneering porcelain to metal alloy.10–12
The compressive forces in the metal–ceramic restoration occur with the properly designed coping and the thermal expansion of the metal coping, which has a slightly higher thermal expansion coefficient than the porcelain coating on it. This small difference in thermal expansion coefficients will cause the porcelain to “pull” toward the metal coping as the restoration cools after firing.5,7 Metal substructure is exposed to very high thermal stress during oxidation and firing. If the metal substructure design is faulty and the wall thickness is not sufficient, heat treatment may cause distortion in the metal substructure and consequently its harmony may be impaired. It may occur in the form of marginal harmonization as a result of residual thermal stress caused by differences in thermal expansion coefficient of metal and ceramic materials during firing and during cooling.8,10 This situation is important for porcelain metal connection.
The aim this study was to evaluate the effect of cooling rates after firing procedures of veneering porcelain on shear bond strength between veneering porcelain and metal alloy frameworks fabricated by different techniques. The null hypothesis was that cooling rates and metal alloy frameworks fabricated by different techniques would not influence on the bond strength of porcelain to metal alloy.
Materials and Methods
This study was approved by the Clinical Research Ethics Committee of Sivas Cumhuriyet University (approval number 2018-01/28).
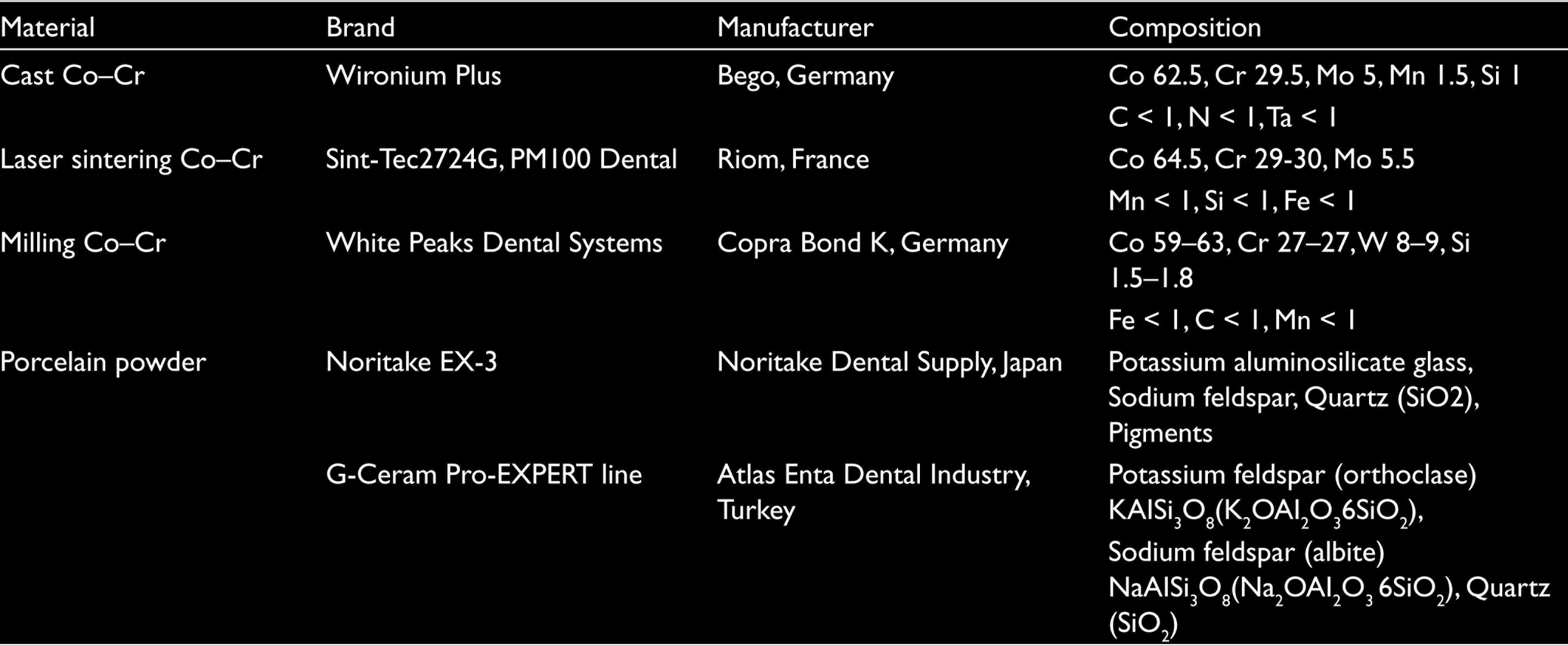
In this study, one castable base metal alloy (Wironium Plus, Bego, Germany), one millable presintered Co–Cr metal alloy (Copra Bond K, White Peaks Dental Systems GmbH & Co. KG, Wesel, Germany), and one laser-sintering powder (Sint-Tec2724G, PM100 Dental, Riom, France) were selected. Two porcelain powders used in the study, Noritake EX-3 (Noritake Dental Supply, Tokyo, Japan), and G-Ceram Pro-EXPERT line (Atlas Enta Dental Industry, İzmir, Turkey), are listed Table 1.
Chemical Compositions of Used Material
Co–Cr samples with a diameter of 7 mm and a thickness of 3 mm were digitally designed on the computer (Exocad GmbH, Darmstadt, Germany). Ten specimens from each group, a total of 120 metal specimens, were prepared (Table 2). The wax molds obtained with the help of the design were invested to create the casting specimens. Co–Cr alloy (Wironia Plus, Bego, Bremen, Germany) was placed to the induction furnace (Mikrotek Dental, Ankara, Turkey). Casting was carried out at 1440°C. Sandblasting was performed using 110-μm aluminum oxide particles to remove investment residues. Then, the specimen surfaces were cleaned by spraying pressurized steam for 5 min according to the dental laboratory protocols and manufacturer recommendations.
Number of Specimens by Group

The production phase with the laser sintering method was done using the 3D Smart ProX-200 system (3D Systems Corporation, Rock Hill, SC, USA). Specimens were created by melting Co–Cr metal alloy powders (Sint-Tec 2724G, PM100 Dental, Riom, France) with laser beams. In the milling process examples, Co–Cr alloy block (Copra Bond M, White Peaks Dental Systems GmbH & Co. KG) using the five-axis in the milling device (YENADENT, D12, Istanbul, Turkey) were generated using milling. The specimens obtained by laser sintering and the milling method were sandblasted with 110-μm Al2O3 particles under 2 atm pressure and 10 cm from the surface for 15 s. After laser sintering, the specimens were kept in an oven at 950°C for 5 min and oxidation process was performed. After sandblasting, all specimens were cleaned by spraying pressurized steam for 5 min according to the dental laboratory protocols and manufacturer recommendations.
The porcelain-bonding agent (3C-Bond, Alphadent, NV Antwerpen, Antwerpen, Belgium) and opaque [VITA VMK Master (Zahnfabrik, Bad Sackingen, Germany)] applied according to the manufacturers to all metal surfaces. For the porcelain application, a metal mold of diameter and height was prepared to imitate the metal substructure and veneer ceramic. A first plate with a diameter of 7 mm and a height of 3 mm was produced for the framework material, and a second plate with slots of 5 mm diameter and 3 mm height for veneer porcelain after it was placed on the first plate (Figure 1). The application of the porcelain was applied the recommendations of the manufacturers. G-Ceram Pro-EXPERT line porcelain powder was applied to half of the specimens and Noritake EX-3 porcelain powder was applied to the remaining half. After the porcelain firing recommendations of the manufacturers (Table 3), the specimens were subjected to two different cooling protocols: slow and fast. For slow cooling, the furnace tray is lowered a little and left at this level for 4 min ın the first stage, then lowered completely and left to cool at room temperature. For fast cooling, the furnace tray is completely lowered without any waiting time and is left to cool at room temperature. After porcelain firing, the specimens were subjected to thermocycling (5000 cycles between 5°C and 55°C; Gokceler Mechanical, Sivas, Turkey) before the shear bond strength test.
The Image of the Mold Used in Porcelain Application

Firing Schedules of Porcelains

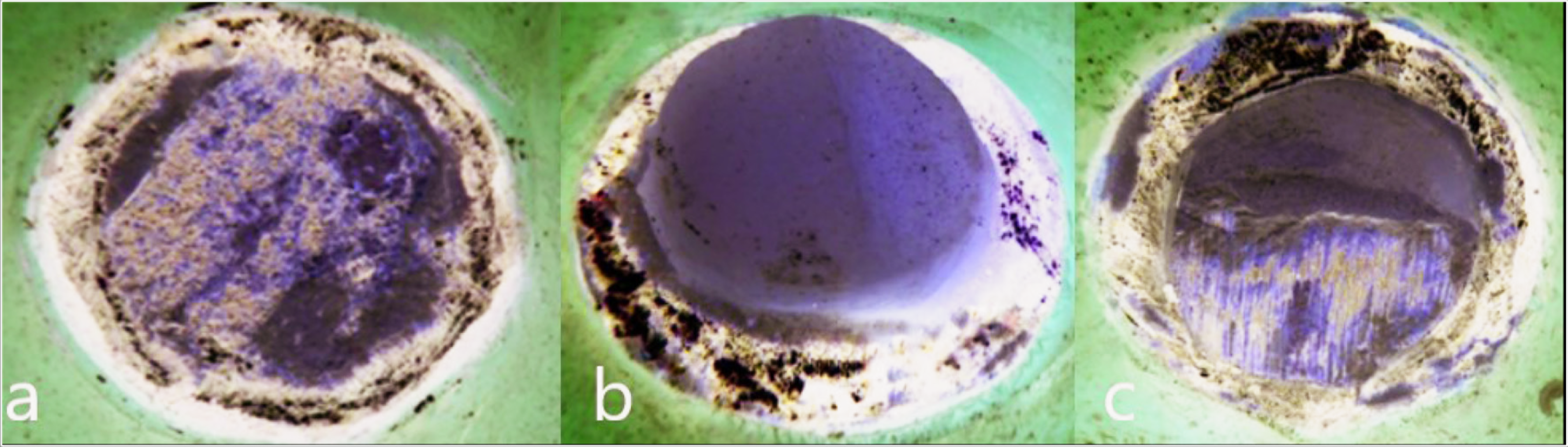
The specimens were subjected to the custom jig of a universal testing machine (Lloyd LF Plus; Ametek Inc., Lloyd Instruments Ltd, Bognor Regis, UK) with a constant crosshead speed of 0.5 mm/min (Figure 2). The resulting bond strength was obtained in megapascals (MPa). To determine the type of failure in all specimens, their interfaces were examined with a stereomicroscope at 8× magnification (Carl Zeiss Microlmaging, Göttingen, Germany). With failure types occurring, it is classified as an adhesive when the metal is completely separated from the substructure, cohesive when the ceramic is completely broken within itself, and combined or both mixed when both types of breakage are observed (Figure 3). The changes on the surface of the porcelain specimens were examined by scanning electron microscopy (SEM; Tescan Mıra3 Xmu, Brno-Kohoutovice, Czech Republic) after rapid and slow cooling.
Test Specimen Placed in the Test Machine

Types of Failure on the Metal Surface of Porcelain With a Stereo Microscope (Magnification: 8X). (a) Adhesive Failure. (b) Cohesive Failure. (c) Combined Failure
Statistical Analysis
The data obtained from our study were evaluated using the SPSS 22.0 (SPSS Inc., Chicago, IL, USA) package program. The Kolmogorov–Smirnov test was evaluated in the province whether the data were suitable for normal distribution. When parametric test assumptions were fulfilled in paired groups, significance test between two means, analysis of variance when comparing more than two groups, and Tukey test were used to find the group or groups that differed when the significance decision was made. The level of error (P) was taken as .05.
Results
Mean and standard deviation values of the bond strength of different metal substructures in different cooling periods are shown in Table 4 and Figure 4. In the study, the difference between metal substructure production techniques was statistically significant as a result of Tukey test, one of the parametric tests, since the distribution of bond strength values provided a normal distribution condition (P < .05). The difference between the bond strength of different brands of ceramics with metal specimens was statistically significant (P < .05). The difference between the bond strength of different cooling protocols on different metal substructures and porcelains was statistically significant (P < .05). When Noritake EX3 porcelain is applied to the frameworks obtained by casting, laser sintering, and milling methods with slow and fast cooling protocols, the difference between the bond strength was significant in the casting and milling groups (P = .003, P = .001, and P < .05, respectively), and laser sintering group was not significant (P > .05). The group with the highest bond strength statistically significant is the MNF group (19.17 ± 2.91 MPa).
Comparing Bond Strengths Within Each Group (MPa)
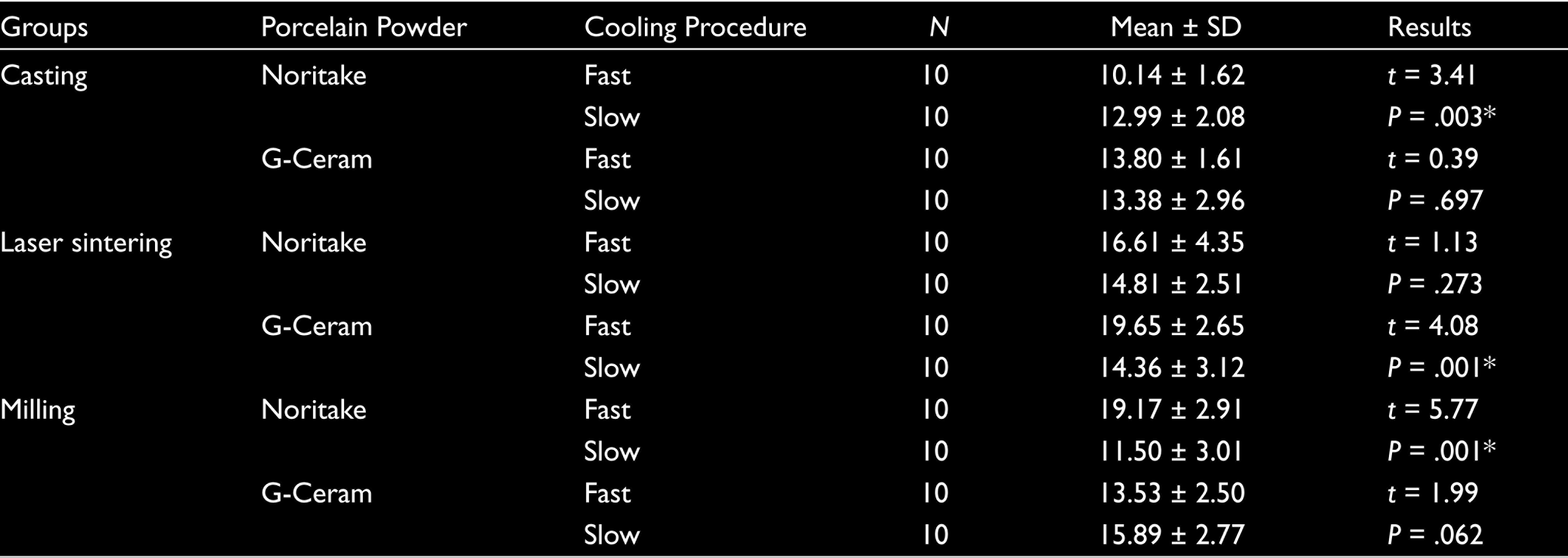
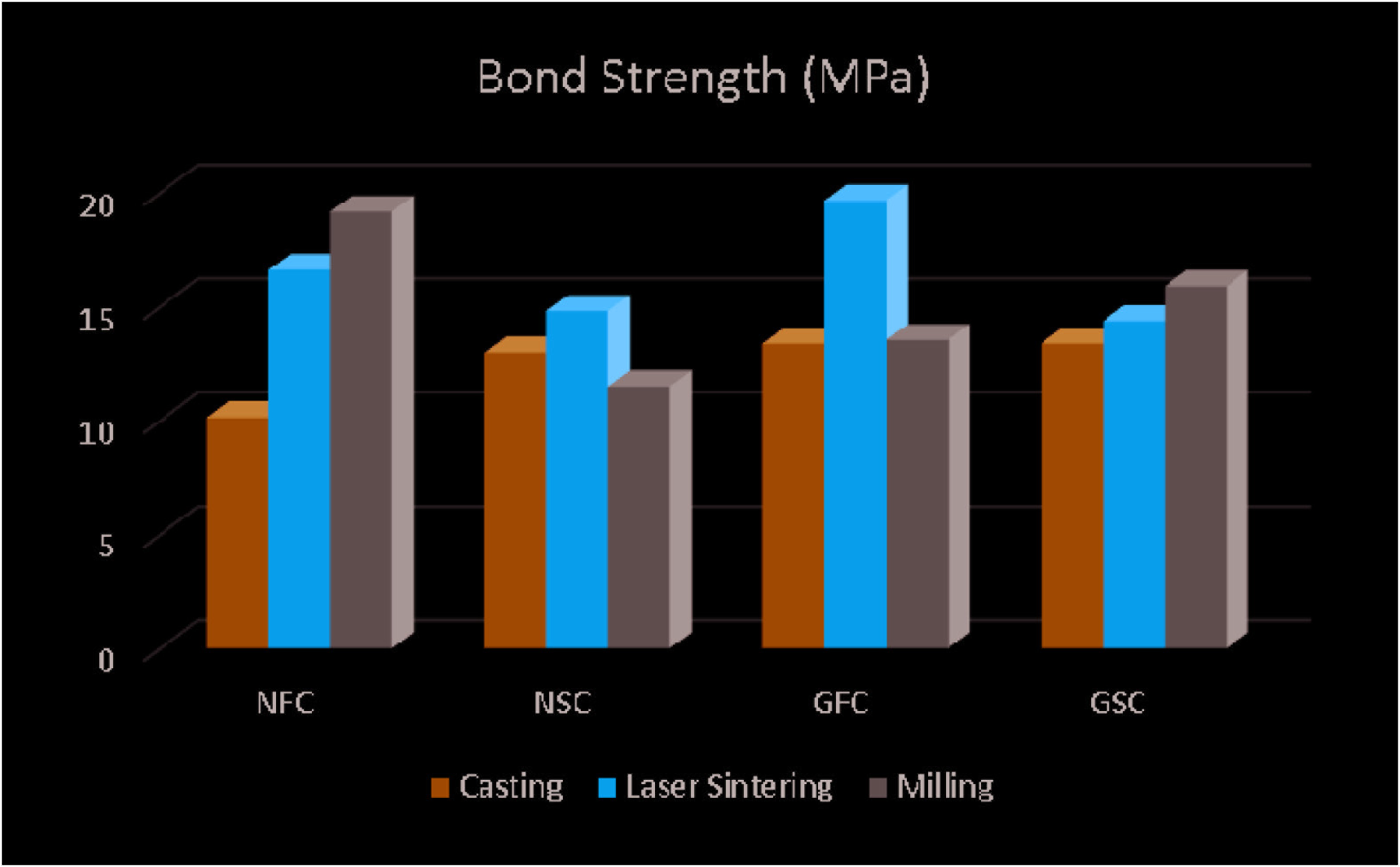
When G-Ceram porcelain was applied to frameworks obtained by casting, laser sintering, and milling methods with slow and fast cooling protocols, the difference between bond strength was significant in the laser sinterization group (P = .001, P < .05), whereas it was not significant in the casting and milling groups (P > .05). The group with the highest bond strength statistically significant is the LGF group (19.65 ± 2.65 MPa).
When examined in terms of bonding between groups, a statistically significant difference was found between casting, laser sintering, and milling methods and in all processes except G-Ceram slow cooling process (P < .05).
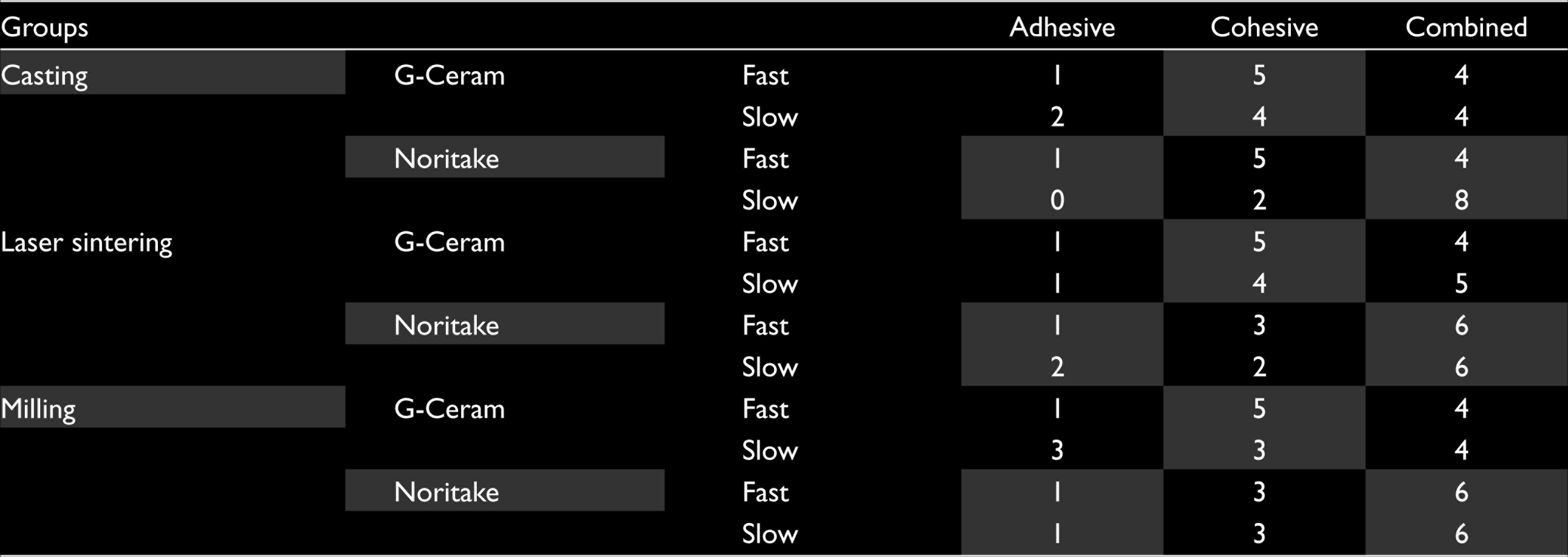
When the specimens are evaluated according to their failure type, 10% adhesive, 40% cohesive, and 50% combined failure in the casting group, 12.5% adhesive, 35% cohesive, and 52.5% combined failure in laser sintering group, and 15% had adhesive, 35% cohesive, and 50% combined in milling group were seen (Table 5).
Failures Types in the Groups
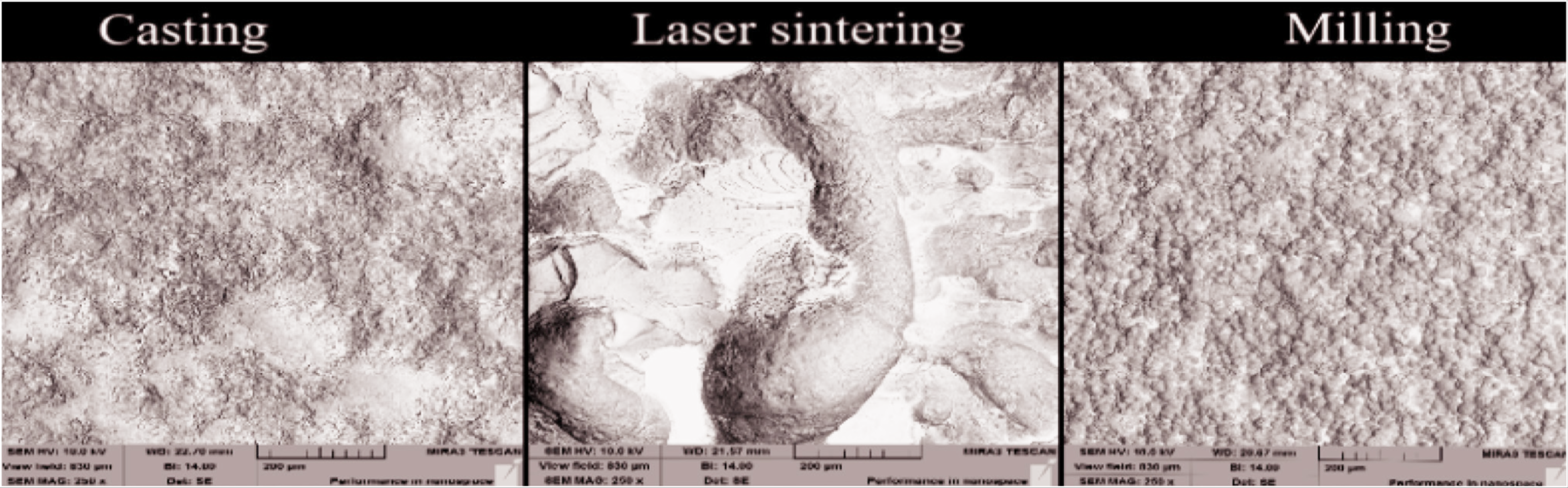
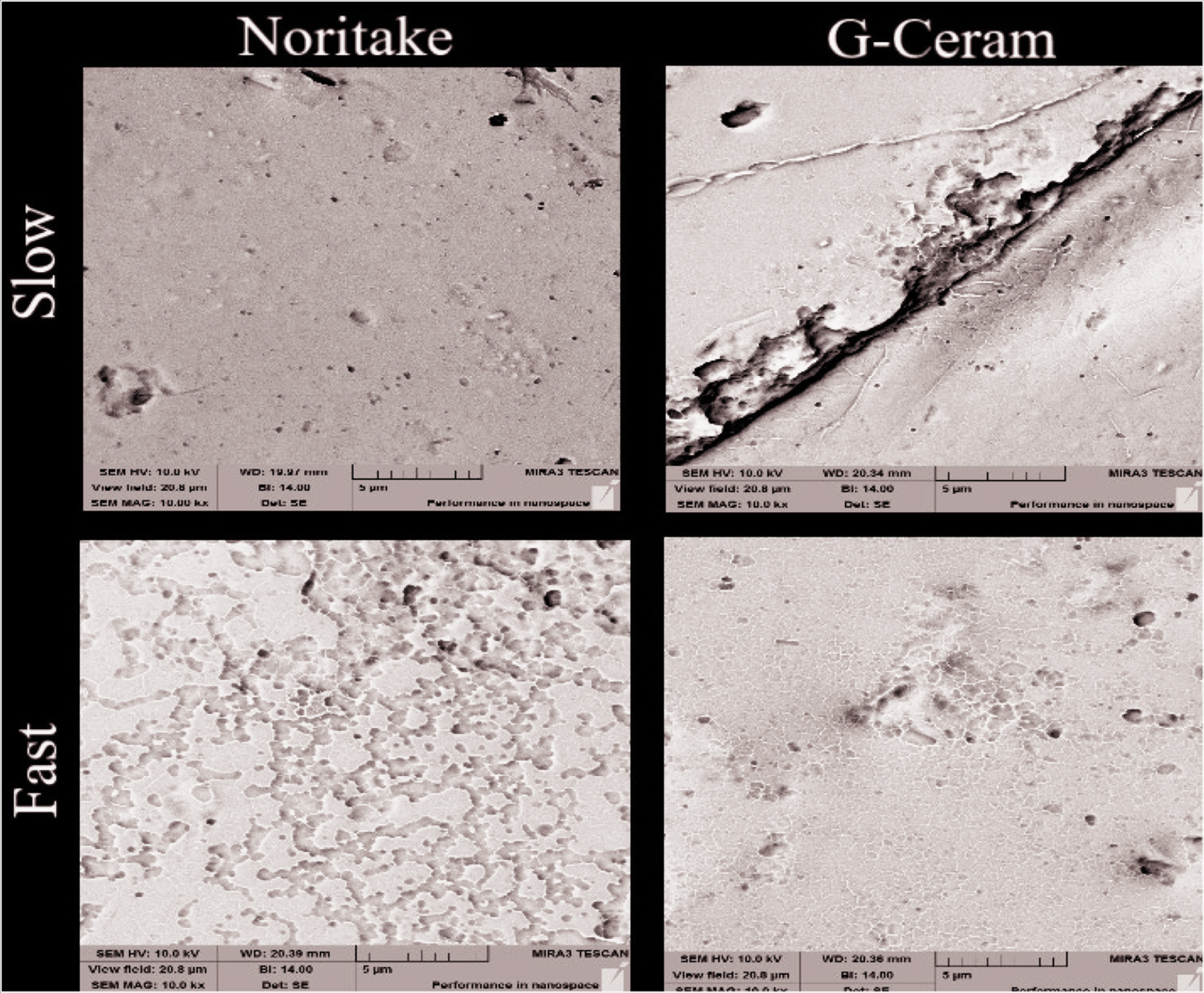
In the SEM analysis, it was seen that the specimen surfaces obtained by laser sintering were rougher than other metal frameworks (Figure 5). In addition, it was observed that the cooling processes created cracks of different sizes in different types of porcelain (Figure 6).
SEM Topography Images of Metal Alloys Fabricated by Different Techniques. (a) Casting, (b) Laser Sintering. (c) Milling (Magnification: 250x)
SEM Images of Porcelains After Cooling (Magnification: 10.0 kx)
Discussion
The present study evaluated whether rate of cooling and metal alloy frameworks fabricated by different techniques would influence the bond strength of porcelain to metal alloy. Based on the findings in the present study, different metal frameworks and rate of cooling have changed the bond strength. Therefore, the null hypothesis was rejected.
Co–Cr alloy was preferred in this study, considering the need for more work on Co–Cr alloys that are frequently used today. The success of a metal–ceramic restoration depends on the bond strength between porcelain and metal framework. 13 Mechanical tests such as tensile, microtensile, shear, and three-point bending bond strength tests are used to evaluate the metal–ceramic bond. 14 In the studies, they stated that since the force transmitted in the shear test can be applied directly to the metal–ceramic connection surface, it will not be affected by the elasticity coefficient of the metal and this test is suitable for use in bond strength studies.14,15 For this reason, shear bond strength test was preferred in this study.
The main bond strength between metal framework and porcelain occurs chemically and includes ionic, covalent, and metallic bonds thanks to the oxide structure between the two layers.14,16 Also, metal surface is important in bond strength. Different metal substructure production techniques affect metal surface properties.17,18
Wu et al. 19 compared the mechanical properties of Co–Cr alloys produced by laser sintering and casting technique and their connection with porcelain. İt is stated that the specimens with substructure produced by laser sintering have superior mechanical and physical properties. While they found the bond strength of specimens prepared by laser sintering 55.78 MPa, they found the bond strength of specimens prepared by casting method 54.17 MPa. Our study was in parallel with the work done, and the superiority of laser sinterization in two different types of porcelain was observed in the bond strength compared to casting.
Stawarczyk et al. 20 evaluated the bond strength of Co–Cr–Mo framework produced by milling, laser sintering, and casting techniques with three different porcelain. They stated that those obtained by milling and laser sintering showed higher bond strength than casting. Our study was in parallel with the work done, however, we can say that the difference in bonding values is due to the differences in the content of metal alloy and the different porcelain brands we use.
Bae et al. 21 stated that there was no significant difference in the bond strength between laser sinterization and casting, but laser sinterization was more effective in bonding. In our study, a significant difference was observed in the rapid cooling of Noritake and G-Ceram porcelains on laser and casting infrastructures. The reason for this may be the alloy, ceramic brand, and firing procedures used.
Hangwei et al. 22 found the selective laser melting technique as a good alternative metal production technique as in our study. As in the study of Wang et al., 16 the bond strength between cast and laser specimens showed a significant difference in our study. Akova et al. 23 found no significant difference between the bond strength of cast Co–Cr specimens and laser sintered Co–Cr specimens. In our study, a significant difference was found between cast Co–Cr specimens and laser sinter Co–Cr specimens, and cast Co–Cr specimens indicated higher bond strength.
The type of rupture between the metal substructure and porcelain is determined after the bond test. The type of failure is generally classified as adhesive, cohesive, and mix. 24 Combined failure was the most common in this study. In study, they stated that slow cooling increases the content of leucite crystals in vita porcelains and significantly increases flexural strength. Leucite is attributed to the structure that creates resistance to crack propagation. 15 In our study, the most common cohesive failure was observed after mix rupture. We can say that the excessive cohesive failure may be caused by the cooling protocols.
In the study carried out by Akova et al., all specimens with Co–Cr cast showed combined failure, whereas half of the laser specimens stated that there was an adhesive and half of them in the combined failure. Since this type of bonding was seen in the laser sinter group and it did not disrupt the metal–ceramic interface, they offered the laser sinter technique as an alternative to cast alloys as a good alternative. 23 Similarly, in our study, the laser sinter technique showed a significantly higher bond strength value than the casting.
Conclusion
Based on a study on shear bond strength of two dental porcelains, one used to veneer metal casting frameworks, and the other for metal laser sintering frameworks and milling frameworks, the following conclusions can be made.
Regardless of the cooling protocol, the highest bond strength was seen in laser sintering.
A significant difference was found between the fast and slow cooling protocols in the bond strength of metal substructures with porcelains. The biggest difference between fast and slow cooling times in terms of bond strength was seen on the milling framework.
It was observed that the surfaces of the metal specimens we examined with SEM images are very different from each other, and especially in metals produced by laser sintering, there are more rough and hollow areas. The positive effect of these areas on bond strength is supported by the results.
With the limitations of the study, cooling protocols have an important factor in bonding porcelain to metal frameworks. For this reason, in future studies, more porcelain types can be researched, and the efficiency of bonding can be evaluated and the most effective method can be determined.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors gratefully acknowledge the financial support provided by the Scientific Research Project Fund of Cumhuriyet University Project DIS-213.
