Abstract
Aim:
To evaluate the effectiveness of photodynamic therapy (PDT), sodium hypochlorite (NaOCl) and chlorhexidine gluconate (CHO) in the endodontic re-treatment in the removing of microorganisms from previously filled infected root canals.
Materials and Methods:
The study samples consist of 54 root canal treated anterior teeth with apical periodontitis were randomly divided into three groups according to the disinfection protocol. Microbiological samples from the root canals were collected after removing the gutta-percha (S1) and following the endodontic re-treatment (S2) with either 5% NaOCl, 2% CHO or PDT procedure for 1 minute. Microbiological samples were cultivated on selective plates to assess the presence or absence of microbial growth and determine the average reduction of viable microorganisms.
Results:
Significant reduction in the total bacterial count in all the groups at post treatment (P < .001). PDT provided statistically higher (99.74%) elimination of Enterococcus faecalis in comparison to NaOCl (95%) and CHO (88%).
Conclusions:
The use of PDT in endodontic re-treatment led to the significant reduction of the remaining bacterial species. So, PDT can be used for routine endodontic disinfection in failed root canal cases.
List of Abbreviations
CHO: Chlorhexidine gluconate
MB: Methylene blue
NaOCl: Sodium hypochlorite
PDT: Photodynamic therapy
PS: Photosensitizer
S1: First microbiological sample
S2: Second microbiological sample
Introduction
The residual root canal infection is the major reason for failure of endodontic treatment.1–3 Disinfection with sodium hypochlorite (NaOCl) and chlorohexidine gluconate (CHO) has a favourable treatment outcome. However, these root canal irrigants do not completely remove the bacteria present in the infected root canals.
Chlorhexidine gluconate solutions of different concentrations have been recommended as endodontic irrigants4,5 and dressings.6-8 NaOCl and Chlorhexidine both have distinct antibacterial activity. In a study irrigation done either with 1% NaOCl or 2% CHO both effectively reduce the total number of viable bacteria (streptococci and E. faecalis) in root canal treated teeth with apical periodontitis. However, there was no significant difference in both the groups. 9
Results of previous research have been inconclusive when comparing the antimicrobial effectiveness of 2% NaOCl and 2% chlorhexidine solutions.10-12 The main drawbacks of conventional root canal treatment are that it’s a time-taking procedure and teeth became weak due to enlargement of the root canal. 13 Still, the complete disinfection of root canal is a challenge. Because of bacterial drug resistance, there is a need of both for new irritants and new techniques to disinfect root canal space.
Photodynamic therapy (PDT) was used to activate a nontoxic photosensitizer, to release nascent oxygen which causes damage to the membrane and DNA of microbes. 14 So, PDT is recommended as a substitute or an additive to currently used disinfection procedures.15,16 In an in vivo study it was used successfully for the removing of multi-drug resistant microorganisms. 17 Bacterial culture methods are commonly used and are easily available to evaluate the antibacterial effectiveness of the different irrigations.
The objective of this in vivo study was to compare the effectiveness of PDT, NaOCl and CHO in disinfecting infected teeth that had previously undergone root canal treatment. The null hypothesis was that there would be no difference in elimination of microbes between the PDT, NaOCl and CHO.
Materials and Methods
Selection of the Patients
This trial was a triple blind randomized clinical trial of three groups. This was performed from September 2016 and July 2017 at King George’s Medical University, Lucknow. This clinical study was approved from the University Ethical Committee (Ref. no. 77th ECMII-B IMR faculty/p15). Standards of the Helsinki Declaration were followed. 18
With reference to the findings of the study by Asnaashari et al., 19 sample size estimation was done using OpenEpi software (Atlanta, GA, USA) considering power of the study = 80%, confidence interval = 95%. The sample size was 18 in each group.
In a total of 54 patients (25 men and 29 women, age between 20 and 70 years, mean age 38 years), each tooth was endodontically treated with apical periodontitis. Patients were chosen from a pool of patients who were referred for root canal re-treatment.
Inclusion and Exclusion Criteria
Patients with previously root canal treated teeth with apical periodontitis were taken for the study. Teeth with a periapical index of 3 or more were incorporated in the study.
In all the cases, root canal treatment was done more than 3 years earlier. There was no overextended obturation in all cases. On clinical examination, all teeth had intact coronal restoration. chosen teeth had no gingival recession and no periodontal pocket more than 4 mm. Before the treatment was initiated, these patients had not taken any antibiotics for a period of at least one month. Patients with any condition that were immunocompromising in nature along with conditions such as diabetes mellitus, chronic infections, rheumatoid arthritis were excluded from the study. Patients on immunosuppressive therapy, pregnant and nursing women were also excluded from the study. The study included the teeth that could not be easily isolated.
Study protocol was explained to all the patients and written informed consent were taken. Root canal treated teeth with chronic apical periodontitis either on maxillary or mandibular anterior teeth was the diagnosis in all of the cases. These patients were randomly divided into three groups based on the intervention used. We have used block randomization method for this purpose. The same practitioner performed the whole clinical protocol. Microbiological samples were collected by another practitioner to avoid any bias.
Endodontic Procedures
Pre-Sampling Procedure for Al Groups
Rubber dam was used to isolate the teeth from the oral cavity. All coronal restorations, carious defects and supra-gingival plaque was removed. Throughout the endodontic treatment and sample collection care was taken to maintain asepsis. To constrain the microbial assessment to a single ecological area, a single root canal was sampled in each case. After accessing the canal with Gates-Glidden burs (Dentsply Maillefer, Ballaigues, Switzerland), up to coronal 3rd of the canal. Further, Rotary ProTaper retreatment files were used to remove the gutta-percha and sealer (ProTaper Universal Retreatment, Dentsply Maillefer, Ballaigues, Switzerland). No chemical solvent of any kind was used to remove the gutta-percha. 20 After removing the gutta-percha, sterile saline solution was used to fill the canal and pulp chamber. Saline was not allowed to overflow from the cavity. Entire saline solution was absorbed from the root canal and pulp chamber by using paper points. Paper points are highly absorbent and they absorb all the fluid present in the root canal. They are easy to take microbial samples from the canals, while the endodontic files and Gates-Glidden drills are used only when we require the dentin chips for sample.
Paper points were transferred to the Eppendrof tube (Eppendorf, Hamburg, Germany) comprising anaerobe transport media (thioglycolate broth, Anaero GRO, Hardy Diagnostics, USA). Paper points were inserted into the canal to the full working length (which was assessed using a preoperative radiograph) and kept for 1 minute. This sample was the first microbiological sample (S1) sent for microbial isolation. Now at this stage, patients were randomly distributed to different groups according to disinfection protocol.
Group 1: Disinfection with 5% Sodium Hypochlorite
After the first sample (S1), root canal was irrigated with a copious amount of 5% NaOCl for 5 minutes using a 5-mL syringe and 30-G needle, which was placed 2 mm short of the working length. Root canal preparation was completed with hand files. After each treatment, the canals were thoroughly irrigated with 2 mL 5% sodium thiosulfate followed by 2 mL normal saline solution, to neutralize the effect of NaOCl solution.
Then, according to the working length, which was assessed with a preoperative radiograph, a sterile paper point was inserted into the canal to take the microbial sample. The paper point was kept in the canal for 60 seconds to completely absorb NaOCl. Subsequently, the paper points were transferred to 1 mL 0.85% sterile saline in the Eppendorf tube and vortexed vigorously for 1 minute. This was the second microbiological sample (S2) of this group.
Group 2: Disinfection with 2% Chlorehexidine
Using a 5-mL syringe and 30-G needle, which was positioned 2 mm short of the WL, the root canal was irrigated with 5 mL 2% chlorhexidine for 60 seconds. After each treatment with chlorhexidine, the root canals were irrigated with 2 mL 5% sodium thiosulfate to neutralize the effect of chlorhexidine solution followed by 2 mL normal saline prior to take microbial sample.
For microbial sampling, a sterile paper point was inserted into the canal up to the working length and kept in the canal for 60 seconds. Subsequently, the paper points were transferred to 1 mL 0.85% sterile saline in the Eppendorf tube and vortexed vigorously for 1 minute. This is the second microbiological sample (S2) of this group.
Group 3: Disinfection with PDT
A FotoSan Device (FotoSan; CMS Dental, Copenhagen, Denmark) at 628 nm was used throughout the experiment. For PDT on teeth, a long-tapered tip (endodontic tip) was used. The apical size of the tip was 500 µm and a 0.03 taper in the apical part; it could be inserted into the canals up to 3 mm short of the working length and guide the light to the apical parts.
A watery solution with concentration of 100 µg/mL of toluidine blue O (TBO; Sigma-Aldrich, St. Louis, MO), a thiazine dye of the quinone-imine family, was prepared and injected into the canal with a sterile single-use endodontic syringe (VMK-Endo needle; Vedefar, Dilbeek, Belgium). A sterile endodontic K-file moved gently up-down in the canal to eliminate air bubbles (ISO 15, stainless steel; Dentsply Maillefer). After this, PDT procedure was carried out. Sixty seconds after inoculation, irradiation treatment was performed with LED lamp for 60 seconds. After irradiation, the root canals were thoroughly irrigated with 2 mL 5% sodium thiosulfate followed by 2 mL normal saline to neutralize the solution remaining in the root canal prior to take the microbial sample. Then the microbiological sample was taken with sterile paper points. Subsequently, the paper points were transferred to 1 mL 0.85% sterile saline in the Eppendorf tube and vortexed vigorously for 1 minute. This was the second microbiological sample (S2) of this group.
Apical preparation was completed with nickel-titanium hand files according to the root canal anatomy and initial diameter of the canal. Each canal was dried using sterile paper points, 5% sodium thiosulfate solutions (Sigma-Aldrich) was used to neutralize any residual NaOCl or CHO and photosensitizer. 21
Lateral compaction technique with AH plus sealer (Dentsply, De Trey, Konstanz, Germany) was used to obturate the root canals in a one visit root canal re-treatment. Surplus gutta-percha and root canal sealer was eliminated from the pulp chambers. The access cavities were sealed with composite resin (Gaenial, GC, Tokyo, Japan) after a base of glass ionomer cement (Fuji IX GP Extra, GC, Tokyo, Japan) was placed.
Microbiological Procedures
Within 1 hour of the sampling, the microbiological samples were forwarded to the Department of Microbiology, in an anaerobic jar with Gas Pack (BBL, Becton Dickinson, Heidelberg, Germany). 22
A temperature of 37±1 °C for 48 hours was used for the incubation of the plates. Light microscopy at 400× was used to examine microbial growth after incubation. Colony-forming units (CFU) was used for microbial counting, and the average percentage reduction of viable microorganisms was determined.
Statistical Analysis
Kruskal–Wallis H and Mann–Whitney U test as a post hoc test (P = .05) were used to statistically analyse the data. Wilcoxon test was used to measure variations between paired groups’ median values for the number of bacteria between (initial versus after PDT; after chemo-mechanical preparation versus after PDT; initial versus after chemo-mechanical preparation).
Results
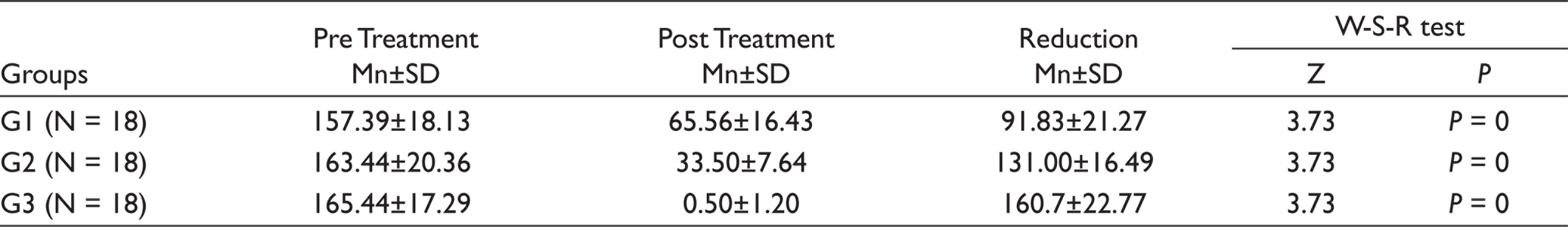
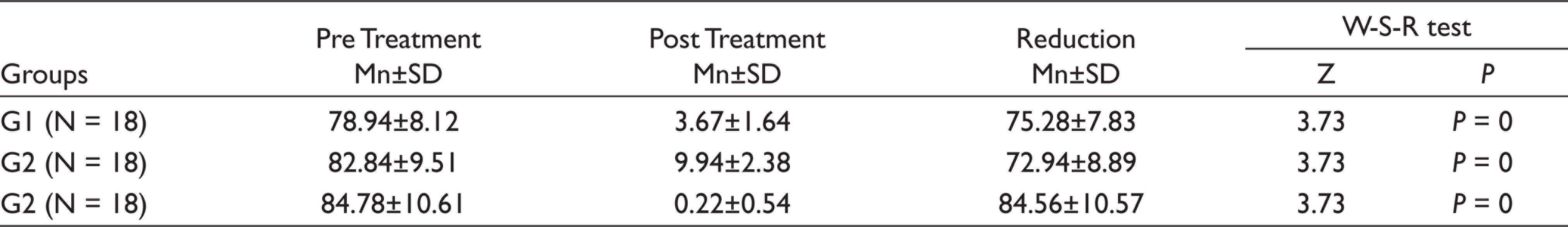
In all 54 cases, the initial sample revealed bacterial growth, with a mean value of 4.57 bacteria species per canal. For each of the groups, the mean percentage reduction of viable microorganisms in CFU/mL is shown in Table 1. For all groups, there was a statistically significant (P < .001) reduction in the number of CFUs from S1 to S2. The null hypothesis was rejected. The significant reduction of E. faecalis displayed that it was more immune to destruction when instrumentation using 2% CHO was employed as compared to 5% NaOCl. However, after the PDT procedure, maximum reduction was seen (99.74% reduction, Table 2), while the other two groups have a significant reduction of E. faecalis.
Comparison of Total Bacterial Counts in Three Groups after Irrigation Procedure
Reduction of E. faecalis in Three Groups after Irrigation Procedure
Discussion
Persistent microbes in the root canal are the main reason for endodontic treatment failure. Some microbes become resistant to the disinfectants and root canal medicaments. They remain viable even after obturation. 23
The culture methodology in this research revealed an average of four bacteria species in every obturated root canal which was associated with chronic apical periodontitis. The prevalence of gram-positive bacteria was 55% in these samples while that of gram-negative bacteria was 45%. In a study done by Siqueira et al, 20 in a polymerase chain reaction-based analysis, they observed that gram-positive bacteria species was most commonly found in root canal treated teeth associated with peri radicular lesion. Gram-positive bacteria are more resistant to biomechanical treatment due to presence of peptidoglycan layer (15–80 nm) in the cell membrane. Hence, they are more prevalent in failed root canal cases. 24
Laser is a novel methodology to disinfect the root canal. In vitro research employing a low-level diode laser (Fotosan) and a photosensitizer to disinfect the root canals, mainly against E. faecalis, have yielded conflicting results.25,26
Many in vitro27,28 and in vivo17,29 research has been shown that PDT is highly effective in the reduction of bacterial load in infected root canals. The findings of this study revealed that a significant antimicrobial effect, especially against E. faecalis, was achieved when PDT was used immediately after the removal of root canal filling material. This hence demonstrates that PDT has higher antimicrobial efficacy as compared to NaOCl (Table 3).
Ng et al. 30 concluded that when chemo-mechanical preparation and PDT were combined, 86.5% of root canals were microbe-free compared to 49% root canals that were microbe-free after only chemo-mechanical preparation. In this study, PDT resulted in 99.74% reduction of bacteria while chemo-mechanical preparation with 5% NaOCl resulted in 95.30% reduction of bacteria and CHO killed 88.00% bacteria (Table 3). In contrast to our findings, Souza et al. 31 reported no substantial synergistic effect of the PDT using either methylene blue (MB) or toluidine blue dye with a 660 nm diode laser (40 mW).
Intra Group Comparison of E. faecalis in All Three Groups
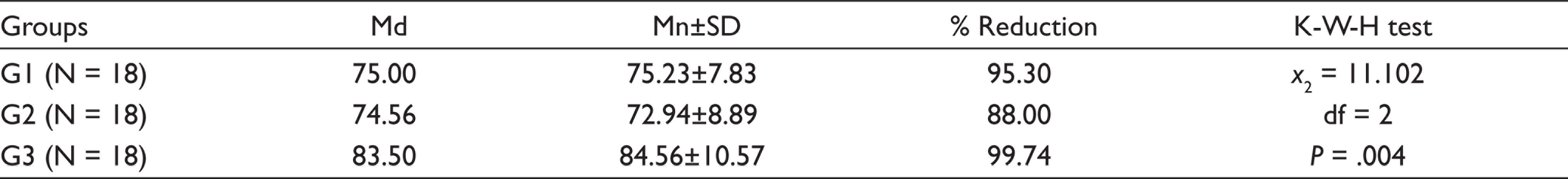
The PDT regimen with toluidine blue and 628 nm diode laser was compared with NaOCl and CHO for endodontic re-treatment in this clinical trial. In comparison to NaOCl and CHO, PDT proved to be more successful in the elimination of both gram positive and gram-negative bacteria species.
PDT with paste-based azulene lowered E. faecalis population in root canals by 99.2%, while 0.5% NaOCl reduced E. faecalis by 93.25%. These results contrast with those of Silbert et al. 32 who concluded that in contaminated root canals 40% destruction of E. faecalis could be achieved using MB and a 670 nm diode laser. The conditions involved in the effectiveness of this therapy may explain the conflicting report. Disparate bacterial species for example are susceptible to PDT in different ways because of which reactive oxygen species are not equally toxic to all microorganisms. 33 The wavelength of light, photosensitizer absorbance, light energy, light intensity, and exposure time are important factors which can play a role in the elimination of microbes. Light source effectively photosensitizes the photosensitizer for better results. 34 Another important factor is optic fibre which should be reached to the maximum length of canal for more and uniform illumination in all the inaccessible areas of root canal. 35
Results of our study showed that PDT is more effective than 5% NaOCl to reduce the bacterial load from the root canals in a given time of exposure (1minute). Seal et al. 13 observed that use of 3% NaOCl for 10 min, was more efficient than PDT but, in this study, the time was same (1 minute) for PDT, NaOCl and CHO. Results of this study showed that PDT was still more effective than NaOCl and CHO. Furthermore, Siqueira et al. 36 observed significant inhibition zones against E. faecalis at varying concentrations of NaOCl solution. It is accepted that because of antimicrobial property of NaOCl, it should be used in higher concentration in root canal disinfection. So, the aim of this study was to suggest the routine use of PDT in the treatment and re-treatment of root canals as well as an alternative of NaOCl.
It is widely accepted that PDT is more effected in lowering the bacterial load from the infected root canal. Results of this study showed that there is significant reduction of bacteria and E. faecalis from the infected earlier root canal treated teeth. So, it can be advised that in failed root canal treated cases, PDT alone or with NaOCl can be used as routine disinfection protocol. Thus, null hypothesis was rejected as the results of this study show the significant difference in between the groups.
The outcome results indicate promising usage in in vivo conditions despite the fact that PDT showed even better reduction in microbial load. Despite comparing our results to other studies, further investigations of PDT should be carried out for clinical use. Results of this study are promising, however further research of a large number of samples of infected root canal are required to detect the exact outcome of PDT.
MB may cause tooth discoloration when it is used as a photosensitizer. As MB is viscous, it adheres to dentine surface forming a smear layer that obliterates dentinal tubule, which decreases the bond strength of root canal filling material to root canal dentine.
PDT appears to be effective against antibiotic-sensitive and antibiotic-resistant microorganisms. In addition, there is no evidence of resistance development in the target bacteria after PDT even after repeated applications treatment.37,38
Conclusion
Many studies concluded that E. faecalis was commonly present bacteria in failed root canal treated cases. NaOCI in its various concentrations and 2% CHO are used to treat the E. faecalis infection in root canals and have promising results. Application of PDT as an alternative to traditional endodontic treatment might lead to a rapid reduction of pathogens without causing any damage to the healthy soft tissue. This study also suggests that PDT can achieve better infection control in the apical region of canal. Clinical and radiological outcome in all three groups is almost similar except PDT is more effective than NaOCI and CHO.
Footnotes
Acknowledgements
The study was supported by Research cell as an Intramural research project at King George’s Medical University, Lucknow. It was presented at the Annual World Dental Congress (FDI), 4–8 September, 2019, at the Moscone Convention Center, 747 Howard Street, San Francisco, CA 94103, USA.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Ethical committee of King George’s Medical University UP Lucknow approved the study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Research Cell King George’s Medical University, Lucknow, Uttar Pradesh.
Patient Declaration of Consent (If In-vivo Study/Case reports)
Consent of patients was taken before commencement of study.
