Abstract
Aim:
To evaluate the effectiveness of midazolam and zolpidem on postoperative pain perception in patients undergoing implant placement.
Materials and Methods:
In the present in vivo study 60 patients undergoing implant placement were selected based on the inclusion criteria framed and were randomly allocated using sequentially numbered, opaque, and sealed envelope (SNOSE) method into 3 groups with 20 patients each after obtaining informed consent. Group A was the control group, Group B received midazolam 7.5 mg 30 minutes before the procedure. Group C received zolpidem 5 mg 30 minutes before the procedure. The anxiety level of patients was recorded using the Corah scale and postoperative pain was recorded after 2 hours of implant placement using the VAS scale. Statistical analysis was done using analysis of variance (ANOVA), one-way multivariate analysis of variance (one-way MANOVA), and then Tukey’s Honestly Significant Difference (HSD) test for comparison among groups at the 0.05 level of significance.
Results:
Group A had a mean anxiety level of 16 ± 1.451, Group B had a mean anxiety level of 11.2 ± 2.858, and Group C had a mean anxiety level of 13 ± 2.9019 and a statistically significant difference between the groups was observed (P < .05). The mean for the postoperative pain perception for Group A was 6.8 ± 1.1965, for Group B was 3.8 ± 1.3611, and Group C was 5 ± 1.451 and a statistically significant difference between the groups was observed (P < .05).
Conclusion:
This study concluded that both midazolam and zolpidem significantly reduced anxiety levels and postoperative pain in patients undergoing implant placement.
Abbreviations
GABA: Gamma-aminobutyric acid
SNOSE: Sequentially numbered, opaque, and sealed envelope
VAS: Visual analogue scale
Introduction
Anxiety is an emotional response caused by an impalpable and promoting danger, which can in turn activate the autonomic nervous system.1,2,3 Anxiety can be noticed by observing patient behavior and recognizing physiological signs of anxiety such as pupil dilation, pallor, sweating, increased blood pressure, increased heart rate, tremors, dizziness, dry mouth, weakness, and difficulty in breathing.4,5 Anxiety can lead to uncooperative patients during implant surgery that extends the duration of surgery, reduction in effectiveness of local anesthesia and ultimately patient dissatisfaction, increased postoperative pain, delayed wound healing, decreased immune system response, high risk of infection, an increased dose of anesthesia and analgesics.1,6–9 In a Brazilian study, 92.4% of the subjects were anxious during dental treatment, when anxiety was measured using the Dental Anxiety Scale.4,10
A dental implant is a surgical component that interfaces with the bone of the jaw or skull to support a dental prosthesis. 11 Implant placement is known to be mild to moderately painful surgical procedure.12,13 However, the perception of pain varies among individuals because it is a multifaceted emotional and sensory experience that is associated and exaggerated with previous experience or by specific oral surgical procedures in children and adults, stress, clinical situation, and anxiety.14–16 Fear of this pain, represented by anxiety, is reported in 40% of the adult population as dental fear.17,18 Pain experienced during and after implant placement is known to be best predicted by anxiety measured immediately before, immediately after, and 4 weeks after surgery.11,12,17,19–23
Oral midazolam is the most commonly used preanesthetic medication.24,25 The pharmacological actions of midazolam are identical to those of other benzodiazepines, including sleep induction, sedation, anxiolysis, and amnesia.26,27 Midazolam is rapidly absorbed in the gastrointestinal tract and produces its peak effect in 30 minutes, and has a short half-life of 1.5 hours. Midazolam is extensively metabolized with only 0.011%–0.028% of the administered dose being excreted unchanged in the urine during the first 8 hours after dosing. 28 The mechanism of action of midazolam is indirect and is related to GABA accumulation (as it binds to the GABA receptor but does not displace it) and its affinity to the benzodiazepine receptors, combination of these receptor form a common chloride channel, which in turn increases the frequency of chloride channel opening. Hence, both these receptors help in causing membrane hyperpolarization and neuronal inhibition. 29 Contraindication for midazolam includes acute glaucoma, hypotension, shock, cases of kidney and liver diseases, alcohol- and drug-dependent individuals, pregnant individuals, children, and individuals with comorbid psychiatric conditions, critically ill individuals as the accumulation of active metabolite can occur.30–32
In 1922, zolpidem was introduced into clinical practice in the United States, it is now the most commonly prescribed hypnotic as it is clinically efficient, safe, and is well tolerated by patients.33,34 Zolpidem’s time of onset of action on oral administration is about 45 minutes as it is rapidly absorbed from the GI tract and its peak effect seen in 1.5 hours. It is a GABAa receptor chloride channel modulator/agonist that increases GABA inhibitory effects leading to sedation. It is metabolized in the liver, with half-life of 2.5 hours. Zolpidem is one of a few central nervous system (CNS) depressants that is recommended for administration during pregnancy.35,36
Many studies have been done to see the association between preoperative anxiety and postoperative pain, but there are very few studies in which antianxiety drugs are given before surgery and its effect on postoperative pain perception of patients is evaluated. Hence, this study is aimed to evaluate the effectiveness of midazolam and zolpidem on postoperative pain perception in patients undergoing implant placement. The null hypothesis was that there will be no effect of midazolam and zolpidem on postoperative pain perception in patients undergoing implant placement.
Materials and Methods
Setting and Design
The present study is an in vivo, interventional prospective trial involving human subjects. The study was done from April 2019 to December 2019.
The sample size was estimated to be 20 patients in each group using G power with inputs fed from a pilot study done with five samples in each group with Type I error of 0.05, test power of 90%, and effect size of 0.8. A total of 140 patients who underwent implant placement in the Department of Implantology, Saveetha Dental College were assessed.
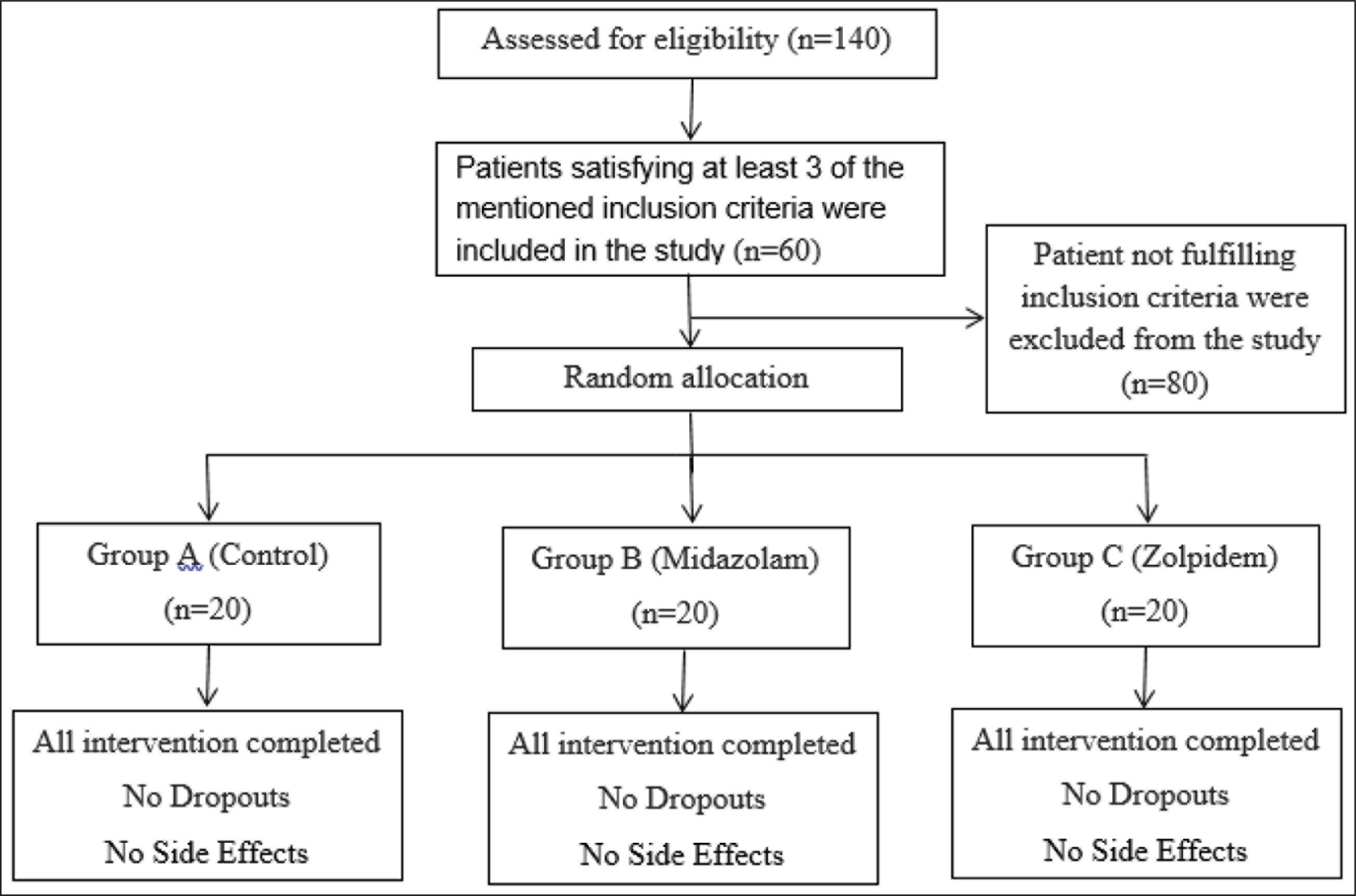
Inclusion criteria were patients indicated for single endo-osseous implants, age within 25–50 years, both male and female, nonsmokers and nonalcoholics, patients taking no medications, patients having no history of allergy to any kind of drugs, with the willingness to participate in the study. Exclusion criteria were patients with any systemic disease, multiple implant placement, immediate implant placement, patients who had previous implant placement done, patients having a history of surgeries done, pterygoid and zygomatic implants. In total, 60 patients satisfying at least 3 of the mentioned inclusion criteria were selected for the study (Figure 1).
Random Allocation
The selected subjects were randomly allocated into three groups A, B, and C, respectively. Allocation concealment was done for the participants using sequentially numbered, opaque, and sealed envelope (SNOSE) method. Tablets of all three drugs were packed in the same type of envelope.
Group A (control) received a placebo 30 minutes before the surgery.
Group B received midazolam (tab. mezolam 7.5 mg, Neon Laboratories Ltd) 30 minutes before the surgery.
Group C received zolpidem (tab. zolfresh 5 mg, ABBO-ABBOTT India Limited H) 30 minutes before the surgery.
Consort Flow Chart
Outcome Measures
Single blinding of the operator collecting the information was ensured by packing the drugs and the placebo in the same type of cover.
Anxiety was recorded using Corah’s scale:
Before giving antianxiety drugs. Thirty minutes after giving antianxiety drugs.
Corah’s scale is a dental anxiety scale; it consists of four questions, which are based on a 5-point Likert scale. Every question is graded from 1 to 5. The total score is a sum of five answers, which ranges from 5 to 20 (Figure 2).
Anxiety rating:
9–12 = moderate anxiety (specific stressors—should be discussed and can be treated) 13–14 = high anxiety 15–20 = severe anxiety (or phobia)
VAS scale was used to record postoperative pain (2 hours after implant placement) for all three groups. VAS scale was used ranging from 0 to 10, in which 0 denoting no pain and 10 denoting maximum pain.
Statistical Analysis
All the collected data was then tabulated and analyzed and using SPSS Statistics software for Windows, version 20.0. Statistical analysis was done using one-way analysis of variance (one-way ANOVA), one-way multivariate analysis of variance (one-way MANOVA), and then Tukey’s Honestly Significant Difference (HSD) test for comparison among groups at the 0.05 level of significance.
Results
The patients enrolled in the study and allocated into the groups had an equal distribution of male and female patients. All the patients completed the interventions with the antianxiety drugs and completed the implant placement. There were no side effects observed with the drugs. There was no loss of patients during follow-up reviews also.

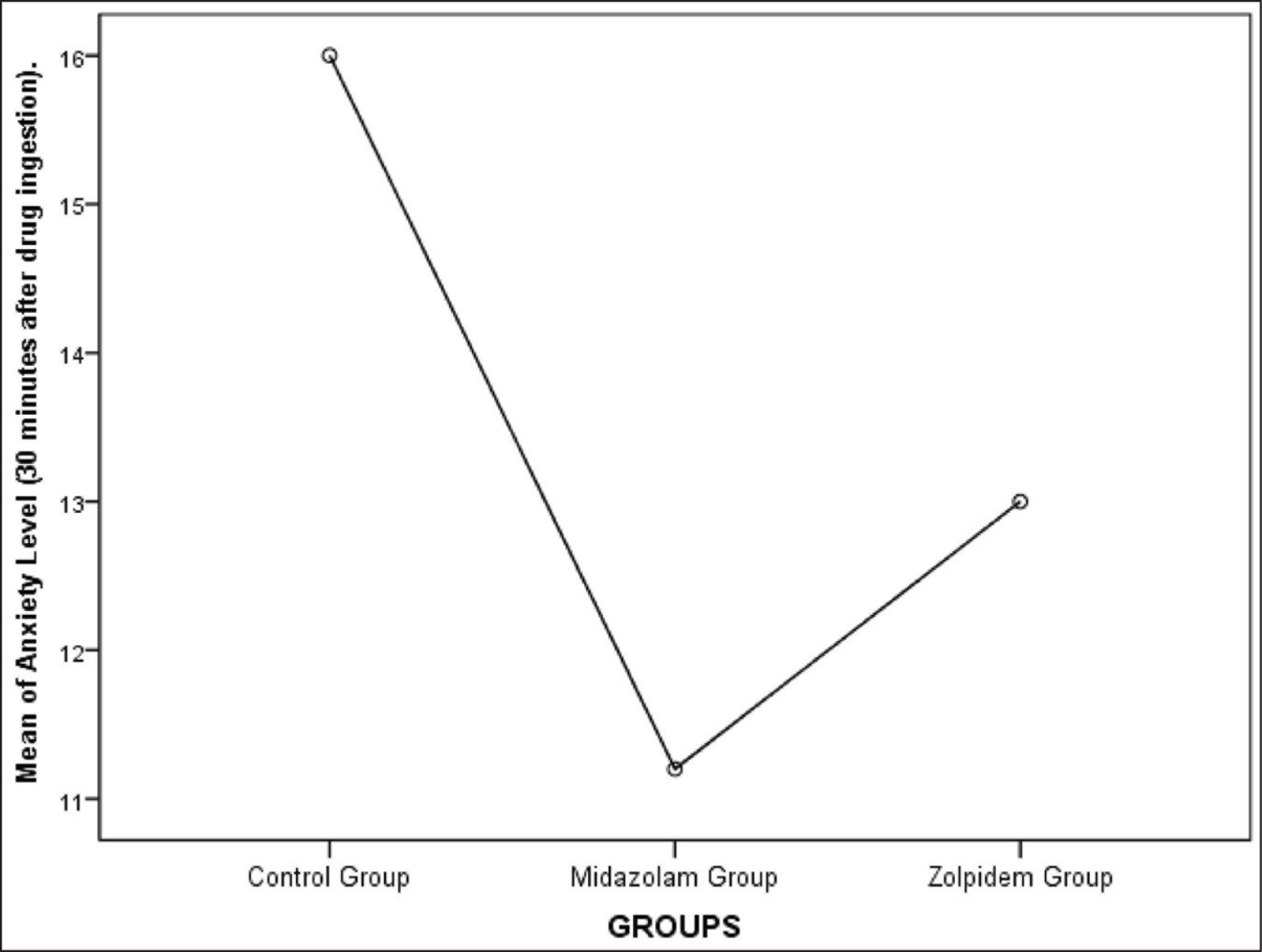
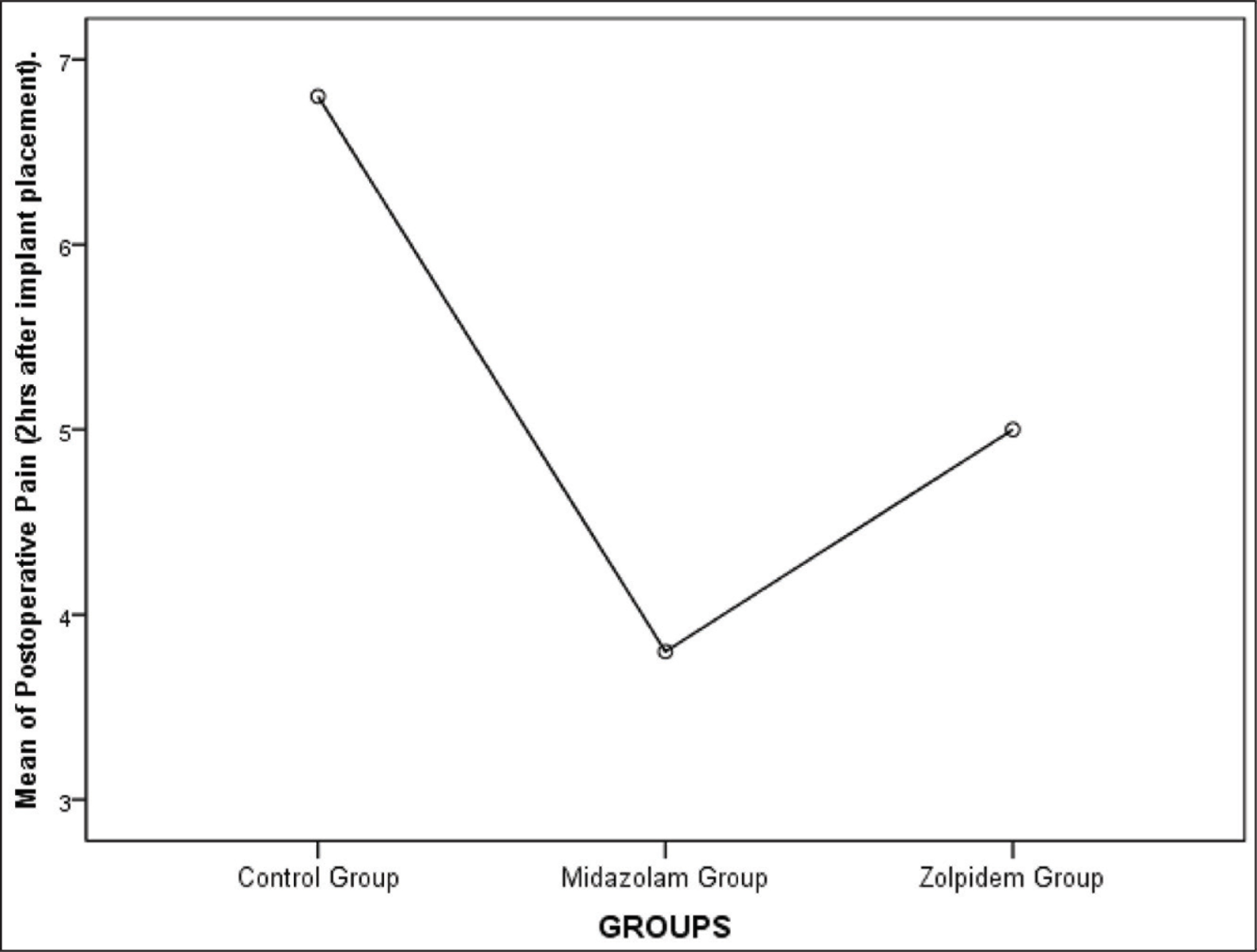
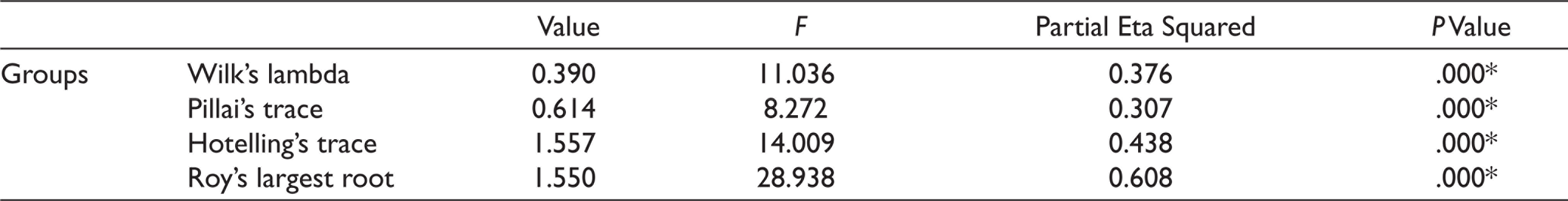
Group A had a mean anxiety level of 16 ± 1.451, Group B had a mean anxiety level of 11.2 ± 2.858, and Group C had a mean anxiety level of 13 ± 2.9019 when measured using Norman Corah’s dental anxiety scale. The values were statistically significant (P < .01). Anxiety level was least for the Midazolam group. The mean for the postoperative pain perception for Group A was 6.8 ± 1.1965, Group B was 3.8 ± 1.3611, and Group C was 5 ± 1.451. The values were statistically significant with the least postoperative pain seen for the midazolam group (P < .01) (Table 1, Figures 3 and 4). One-way multivariate ANOVA (one-way MANOVA) results was statistically significant (Table 2). This showed that there is a positive relation between preoperative anxiety level and postoperative pain. Tukey’s Honestly Significant Difference (HSD) post hoc tests showed there was a statistically significant difference (P < .01) between Group B and C (midazolam and zolpidem) with respect to postoperative pain (Table 3). There was a significant decrease in postoperative pain perception after giving midazolam prior to implant placement. However, there was no statistically significant difference (P > .05) between Group B and C (midazolam and zolpidem) with respect to anxiety levels (Table 4).
One-way ANOVA Results for Anxiety Level and Postoperative Pain After Administration of Antianxiety Drugs

Means Plot Showing Mean of Anxiety Level (30 minutes After Drug Ingestion) for All Three Groups. The Mean Anxiety Level for Group A, B, and C Was 16 ± 1.451, 11.2 ± 2.858, and 13 ± 2.9019, respectively
Means Plot Showing Mean of Postoperative Pain Level (2 hours After Implant Placement) for All Three Groups. The Mean for the Postoperative Pain Perception for Group A, B, and C was 6.8 ± 1.1965, 3.8 ± 1.3611, and 5 ± 1.451, respectively
One-way Multivariate Analysis of Variance (MANOVA) Results for Anxiety Level and Postoperative Pain After Administration of Antianxiety Drugs
Post Hoc Test Results for Postoperative Pain
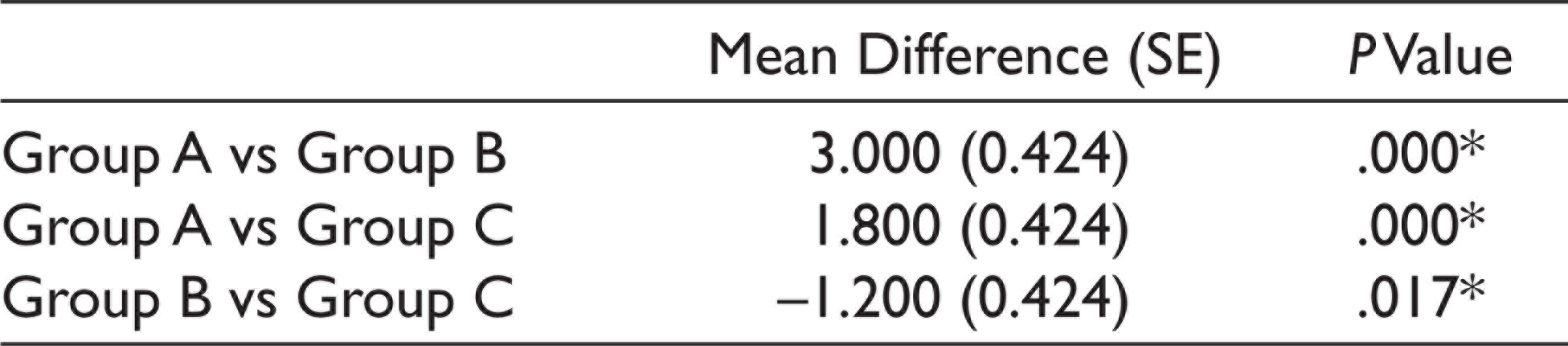
*The mean difference is significant at the 0.05 level.
Post Hoc Test Results for Anxiety Level (30 minutes After Drug Ingestion)
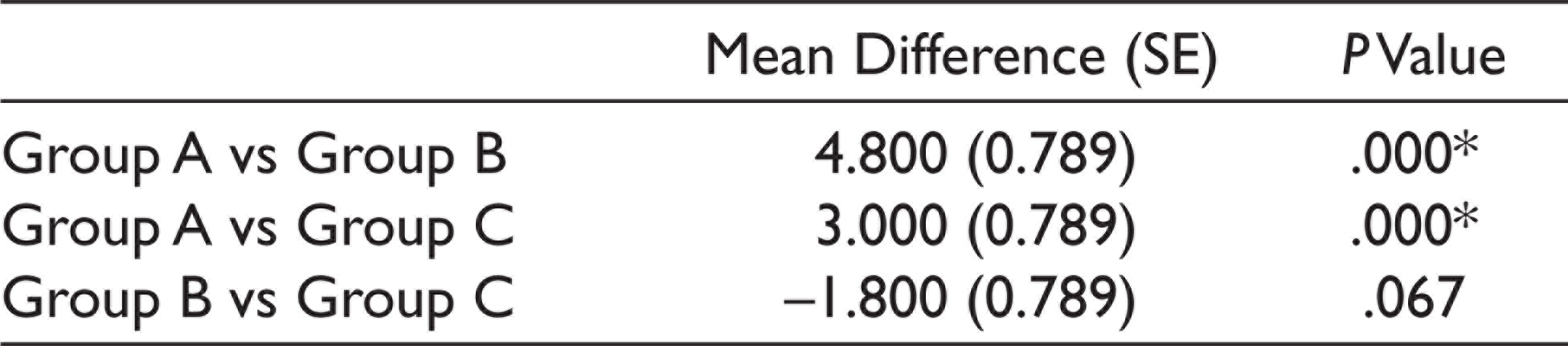
*The mean difference is significant at the 0.05 level.
Discussion
Anxiety is most commonly seen during oral surgical procedures. Anxiety can lead to uncooperative patients during implant surgery, which will extend the duration of surgery, reduce the effectiveness of local anesthesia, and ultimately patient dissatisfaction. 1 Sedation is an effective method to reduce anxiety, and midazolam is widely used for this purpose because it provides profound anterograde amnesia and satisfactory sedation with minimal changes in the cardiovascular and respiratory systems. Midazolam has become the standard drug because it is an ideal sedative for this type of surgical procedure. 31 Its short action is consistent with the average duration of a dental procedure. 4
In our study, 20% of participants reported severe anxiety, 46.6% had moderate anxiety, and 33.3% had mild anxiety. A similar study was done by Khorshidi et al., 1 10% of the participants reported symptoms of severe anxiety, 5% had moderate levels of anxiety and 85% had mild levels of anxiety before the procedure. These results were different from results obtained from a study done by González-Lemonnier et al., 38 which showed that low preoperative anxiety was reported in 27.8% of patients, moderate in 50%, and high in 22.2%. Many clinical studies indicate that like other dental treatments, dental implant surgeries also cause a different range of anxiety. 1 Commonly women easily display their feelings of fear during dental treatment when compared with men. Authors believed gender could influence the capability of managing fear. 1 A study done by Liliane-Poconé Dantas et al. 4 showed that women had higher levels of anxiety than men. However, our study and study done by Campos et al. 39 did not find a significant association between anxiety and gender but did corroborate that age did not influence anxiety levels.
Several studies have reported somnolence as a common adverse effect of midazolam. In a study by Ritwik et al., 40 66.7% of the subjects showed somnolence after oral administration of midazolam, which was similar to our results and results obtained by Liliane-Poconé Dantas et al. 4 Previous studies done by Ng et al. 41 in the medical field also showed that anxiolytic premedication helps to reduce preoperative anxiety level and reduces the pain level during the time of surgery. But premedication was associated with moderate to severe drowsiness postoperatively. A study done by Gupta et al. 42 showed that the mean preoperative anxiety level was less when midazolam was given preoperatively. In the same study, it was seen that postoperative pain 30 minutes after the surgery was less when preoperative midazolam was given (preoperatively midazolam is given: 3.13 ± 0.86, preoperatively midazolam not given: 5.73 ± 1.2). These results were similar to our study. Some studies showed that Midazolam premedication did not help in reducing anxiety level 39 . Another study done by Kim et al. 40 showed that midazolam premedication did not improve the postoperative quality of recovery, but the midazolam group showed shorter induction time and less consumption of propofol.
Midazolam and zolpidem have helped in the reduction of postoperative pain perception after implant placement. The results were statistically significant with P value <.00001. Hence, these drugs can be used to reduce anxiety, reduce postoperative pain perception, and also help in increasing patient cooperation during the treatment. The preference for midazolam can be explained by the fact that this drug can cause anterograde amnesia, resulting in participants not remembering what happened in surgery and eliminating the possible formation of negative memories of the procedure. Memory interference was the only variable resulting in a statistically significant difference between the protocols; therefore, it may be the reason participants preferred midazolam.4,43
The limitation of our study was that the anxiety levels were not checked in the same patients before and after giving antianxiety drugs, only subjective pain was evaluated and anxiety level in the same patient during implant placement with and without anxiolytic premedication was not evaluated. If future studies are done to overcome the limitations of this study, it can help in future treatment for anxious patients.
Conclusion
Anxiety causes uncooperation during implant surgery and extends the duration of surgery, insufficiency of injected local anesthesia, and ultimately patient dissatisfaction. This study concluded that both midazolam and zolpidem significantly reduced anxiety levels and postoperative pain in patients undergoing implant placement. Anxiolytic premedication is commonly used in the medical field before various surgeries. Hence, antianxiety medication can be given as premedication even in some dental procedures as it helps in the reduction of postoperative pain sensation and patient satisfaction.
Future Scope
Further studies can be done to evaluate differences in anxiety level before medication and after medication; the effect of antianxiety drugs and associated postoperative pain can be checked on the same patient: one side implant placement without any antianxiety premedication and one side implant placement with premedication given. In our study only subjective pain was evaluated, further studies can include subjective and objective pain evaluation. In future studies, the dosage of the antianxiety drugs can be reduced, and the anxiety level and postoperative pain can be evaluated.
Footnotes
Acknowledgements
We would like to acknowledge xxx Dental College and hospital for providing their constant help and support for this research.
Authors’ Contributions
First author (Minal Tulsani) contributed to conception, study design, data collection, analysis, interpretation and wrote the manuscript. Second author (Dhanraj M Ganapathy) helped with study design, analysis, interpretation, and critically revised the manuscript. Third and fourth authors (Divya Rupawat and Sanjana Devi) critically reviewed the manuscript. All the authors have discussed results and revised the manuscript.
Data Availability Statement
The data analyzed during the current study are available from the corresponding author upon request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Policy and Institutional Review board statement
The study was presented before the institutional ethical and scientific review board and permission was obtained (Ethical Clearance No.: Saveetha/PROSTHO/1809/19/151 and IHEC Ref No.: IHEC/Saveetha-PROSTHO-1804/19/147). The study protocol conformed to the ethical guidelines prescribed by the WHO and Helsinki declaration.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
The selected subjects were clearly explained about the study protocol and informed consent was obtained from them for participation.
