Abstract
Aim:
To evaluate the effect on vertical bone augmentation of the screw tent-pole technique using particulate allograft in combination with injectable platelet-rich fibrin, and to compare this with autogenous block bone graft.
Materials and Methods:
This split-mouth randomized controlled study involved patients with bilateral partial edentulism and vertical bone loss in the posterior mandible. In each patient, the control side was treated with autogenous block bone graft harvested from the mandibular ramus and, on the test side, the screw tent-pole technique was employed, using particulate allograft in combination with injectable platelet-rich fibrin. All augmented sites were covered by leukocyte and platelet-rich fibrin membrane. The primary outcome variable of this study was the radiographic changes to bone height 6 months after augmentation. The secondary outcome variables were the percentage of newly formed bone and the implant survival rate. The data were analyzed with a significance level of α = 0.05.
Results:
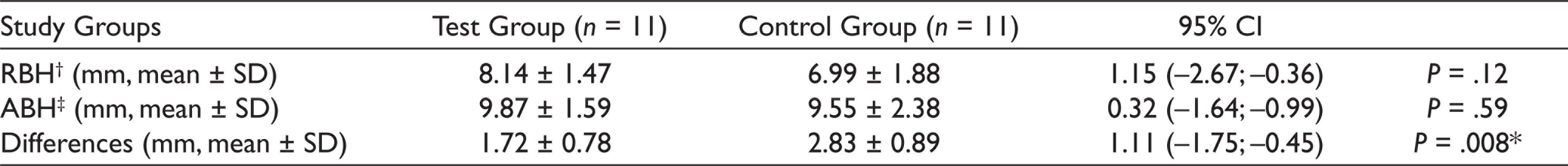
Of the 13 patients included, a total of 11 patients (7 females, 4 males; mean age 50.92) completed the study. The mean values of vertical bone gain were 1.72 ± 0.78 mm for the test group and 2.83 ± 0.89 mm for the control group, which constitutes a significant difference (
Conclusions:
Based on the study results, screw tent-pole can be considered a feasible technique for bone augmentation. Further randomized studies will be necessary to fully evaluate vertical bone augmentation using the screw tent-pole technique, with different graft materials and with larger samples.
Keywords
Abbreviations
AOI: Area of interest
CBCT: Cone beam computed tomography
i-PRF: Injectable platelet-rich fibrin
ICC: Intraclass correlation coefficient
L-PRF: Leukocyte-platelet-rich fibrin
max: Maximum
mg: Milligram
mL: Milliliter
mm: Millimeter
min: Minute
%: Percentage
PRP: Platelet-poor plasma
PRP: Platelet-rich plasma
RBC: Red blood cells
rpm: Revolution per minute
SD: Standard deviation
Introduction
Bone augmentation techniques are required in order to improve esthetic outcomes and long-term prognoses for dental implant treatment.1,2 These techniques involve the use of bone graft materials for the rehabilitation of deficient regions.1,3,4 Bone graft materials originating from human, animal, or synthetic sources are placed to induce osteoblastic activity of the bone and periosteal tissue, or to provide an osteoconductive scaffold that indirectly accelerates bone regeneration.1,5 These materials are manufactured in block or particulate forms to allow them to adapt to different bone defects.3,4 While the block forms have good mechanical properties, the particulate forms can have some disadvantages in reconstructing alveolar bone deficiencies, namely soft tissue migration, lack of structural integrity, which affects stability and also the collapse of the bone graft materials.4,6,7
Previous comparative clinical studies examining vertical ridge augmentation have found block bone grafts to be better than particulate grafts. The latter require the alveolar bone deficiencies to be filled and do not allow adequate three-dimensional bone reconstruction.3–5 In the last decade, “tent-pole” techniques were introduced to prevent the contraction of soft tissue around the bone graft material and to reduce micromovement or physiological resorption of the grafts. 8 These techniques create a space between the bone and periosteum for particulate grafts and provide ample bone volume.8–11 The screw tent-pole method is generally regarded as an effective technique to achieve initial reconstruction of alveolar bone deficiencies. 11 Studies have shown that this technique is recommended in order to minimize bone graft resorption and to obtain predictable, functional, and esthetical outcomes without the use of block bone grafts.6,12,13
In cases of deficient mandibular alveolar ridge, bone augmentation techniques have been focused on biomaterial integration due to its contribution to the long-term outcomes of dental implant treatment. 14 Moreover, a necessary condition for successful outcome of bone augmentation techniques is the biological factors, which affect the ability to heal during bone repair and lead to resorption of grafted bone or even to bone graft loss. 9 Various studies have been conducted into the use of platelet concentrates with different bone graft substitutes, due to their cytokine and growth factor content, which act as signaling molecules in the healing process.15,16 However, these concentrates, such as platelet-rich plasma (PRP) and leukocyte and platelet-rich fibrin (L-PRF) cause heterogeneous modality within the particulate bone graft materials.16–20
Recently, the liquid form of these concentrates has been proposed as a simple and effective method of generating tissue repair and is given the name “injectable platelet-rich fibrin (i-PRF).”21–23 i-PRF is more suitable for mixing with bone graft materials and the use of i-PRF-enriched particulate bone grafts produces predictable results with regard to ease of manipulation. It also appears to be beneficial in terms of bone regeneration.7,16
The purpose of this study is to evaluate the effect on vertical bone augmentation of the screw tent-pole technique using particulate allograft in combination with injectable i-PRF, and to compare this with autogenous block bone graft. The hypothesis of the study was that the screw tent-pole technique using i-PRF-enriched particulate bone graft would demonstrate similar results to autogenous block bone graft for vertical bone reconstruction.
Materials and Methods
Setting and Design
This split-mouth randomized controlled study was conducted at the Department of Oral Surgery of the School of Dentistry. Before the participants were selected, sample size was calculated using PASS 2000 software.
24
A power analysis was performed, based on a significance level of 0.05 and a power of 95%, to estimate sample size by dependent group
Selection of the participants was determined by clinical and radiological examination in line with the inclusion criteria. The inclusion criteria are as follows: (a) bilateral partial edentulism with vertical bone loss in the posterior mandible 25 ; (b) an indication for a fixed implant-retained prosthesis; (c) insufficient bone height for insertion of short dental implants 26 ; (d) absence of/previously treated periodontal disease; (e) ability to understand and accept the requirements of the study. The exclusion criteria are as follows: (a) medical conditions that compromised tissue healing (osteoporosis, hemophilia, anemia, etc.); (b) treatment with an interfering medication (bisphosphonate or steroid therapy); (c) a total platelet count lower than 150,000/mm3; (d) radiation therapy within the last 2 years; (e) pregnancy or lactating; (f) a smoking habit; (g) <18 years old.
One examiner informed all patients about the treatment protocol of the study and the risks and benefits associated with participation. Each patient provided written informed consent prior to enrollment.
Randomization
Randomization and allocation concealment were performed by an examiner (GI). The allocation sequence was carried out using a computer-generated method
27
and concealed by means of sequentially numbered sealed envelopes. Before augmentation surgery, the envelopes were opened by the same examiner and the bilateral edentulous posterior mandible in the case of each patient was randomly assigned to one of the two study groups:
The test group (screw tent-pole technique + particulate allograft with i-PRF + L-PRF membrane). The control group (autogenous block bone graft + L-PRF membrane).
Preparation of Platelet Concentrates
Blood samples were collected by a nurse at the beginning of the surgery and the materials were prepared step by step by a single examiner (GI).
The L-PRF preparation was performed as follows: 17 the venous blood samples were collected in 9 mL × 4 glass tubes without anticoagulant and immediately centrifuged at 2,700 rpm (equivalent ~700× g RCF-max) for 12 min. This procedure was carried out using an EBA 20 centrifuge (Hettich® Instruments, LP) with a radius of 86 mm and a fixed angle rotor. After the centrifugation of the samples, the red blood cells (RBC) and platelet-poor plasma (PPP) were separated from the buffy coat described as L-PRF. The L-PRF was then placed on a special press kit in order to obtain the required membrane form.
The i-PRF preparation was performed as follows 18 : the blood sample collected in an 8 mL, noncoated plastic tube without anticoagulant was immediately centrifuged at a protocol of 2,700 rpm (equivalent ~700× g RCF-max) for 2 min (EBA 20, Hettich® Instruments, LP). The liquid autologous fibrin material, comprising the 1 mL upper layer of the tube was then collected, using a disposable syringe. Finally, the resulting i-PRF was mixed with allograft (0.5–1 mm particle size) material to obtain “sticky bone.”
Surgical Protocol
All surgeries were performed under local anesthesia by the same oral surgeon (TG). Before surgery, patients were asked to rinse their mouths with 0.2% clorhexidine gluconate as an antiseptic mouthwash for 60 s.
A midcrestal incision was made on either side to expose the edentulous alveolar ridge. Then, for the control side, a posterior releasing incision was made over the external oblique ridge to provide access to the donor site.
The ramus donor site was prepared using a similar method to that described by Misch. 28 To avoid inferior alveolar nerve injury, extreme care was taken. Vertical and horizontal osteotomies were performed using a 6-mm diameter separation disc (Meisinger Saw Basic Kit®, Düsseldorf, Germany), under copious irrigation with saline. Horizontal osteotomy was carried out as appropriate for the size of the recipient site. The outlined graft was mobilized from its bed through careful application of a thin chisel. This was placed parallel to the posterior border of the horizontal osteotomy and the block graft was subsequently disengaged by the application of equal strength and pressure from posterior to anterior, using a hammer. Sharp edges around the block graft were rounded off under saline irrigation using a 4-mm round bur. The screw holes were made carefully on both the recipient site and the block graft using a 1.3 mm drill. The graft was firmly fixed with 1.3 mm screws and a compatible driver (Meisinger BTX100 Screw System®, Düsseldorf, Germany). The periphery of the graft and the underlying gap between the screws and the recipient site were covered with a mixture of particulate allograft (demineralized freeze-dried bone allograft, ETB-BISLIFE: Multi tissue Center, Tissuelab® S.p.A, Milan, Italy) and sterile saline solution in order to minimize resorption and to improve the adaption of the graft.
Surgery on the opposite side (the test side) was performed in the same session. The position and number of screws were assigned according to planned prosthetic rehabilitation. The screw holes were made on the recipient site using a 1.3 mm drill. Screws of 7-mm length were fixed on the recipient site and stabilized by a compatible driver. i-PRF was collected using a 0.5 mL disposable syringe and mixed with allograft material in a sterile metal box. Polymerization was completed in 3–5 min, then this material was applied to the underlying gap between the screws and the recipient site.
Decortication was carried out using a 2-mm diameter round carbide bur and six to eight cortical perforations were formed on the recipient beds for both study groups. All augmented sites were covered by L-PRF membrane and flap closure was affected with a 4/0 silk suture (Figure 1).
Study Groups. In Control Group: (a) Autogenous Bone Graft Fixed Recipient Bed; (b) L-PRF Membranes Covered the Augmentation Site; (c) İntraoral View of Control Group at Postoperative 6 Months; (d) Osseointegration of Block Bone Graft with Recipient Site. In test Group: (e) Screws Used for Tent-Pole; (f) i-PRF Enriched Allograft Material Covering the Augmentation Site; (g) İntraoral View of Test Group at Postoperative 6 Months; (h) Osseointegration of Graft Material with Recipient Site.
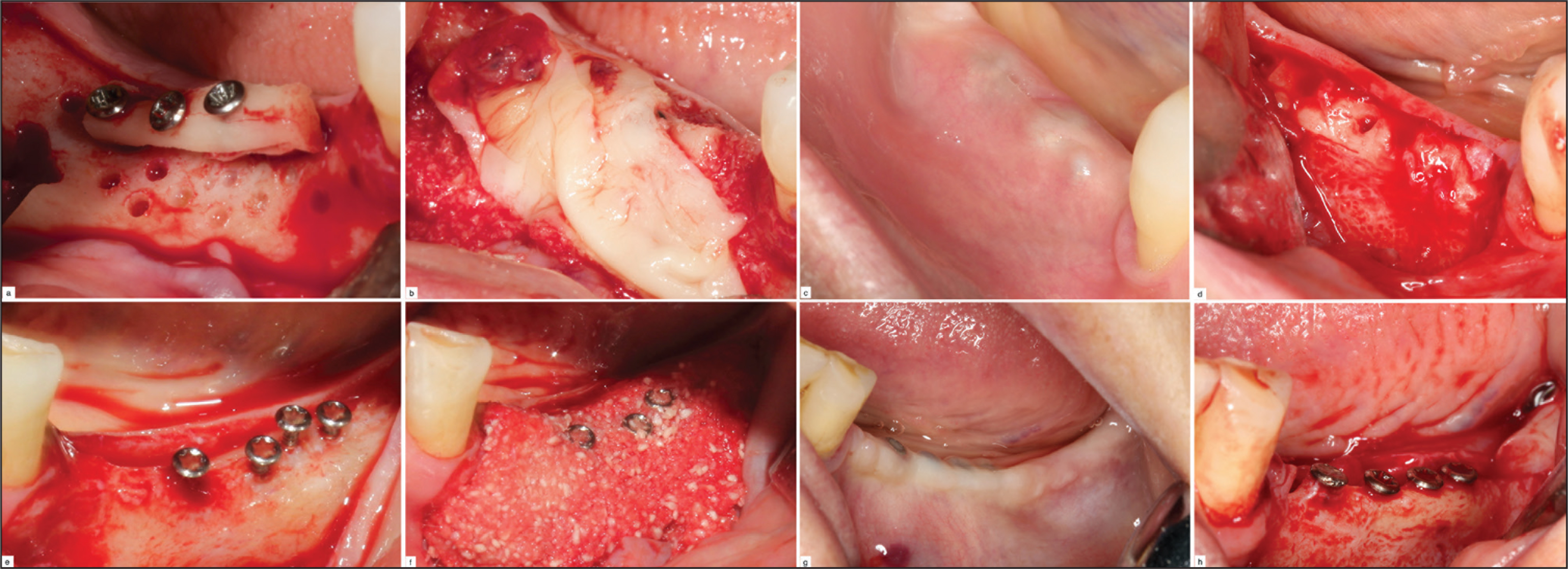
After surgery, amoxicillin/clavulanic acid (875/125 mg) and naproxen sodium (275 mg) were prescribed by the surgeon, to be taken every 12 hours for 14 days, in order to control postoperative pain and surgical side infection. Also, the patients were required to take an antiseptic mouthwash of 0.2% clorhexidine gluconate for 7 days. Sutures were removed after 14 days. Six months postoperatively, all patients underwent the second stage of the surgery. Implant site preparation was performed as recommended by the manufacturer’s guidelines (Bio3 Implants®, Pforzheim, Germany). Each group received a type of tapered implant, with triangular thread and a hydrophilic microporous surface. These tapered implants were preferred to minimize any potential trauma during the procedure and to prevent graft separation or fracture at the augmented sites.
Study Outcomes
The predictor variable of the study was the treatment technique in the groups. The primary outcome variable of this study was the radiographic changes to bone height 6 months after augmentation. The secondary outcome variables were the percentage of newly formed bone and the implant survival rate 12 months after implant placement.
Radiological Assessment
To evaluate the height of the alveolar bone, radiological analysis was conducted, using the same cone-beam computed tomography device throughout (Kodak Dental System, Caresteam Health, Roschester, NY, USA), by a blinded independent examiner. This device was calibrated with the same irradiation time, voxel size, and section thickness to eliminate interchange on the radiographic image. Also, the optimal relation of the Frankfurt horizontal plane and centric occlusion were evaluated.
Analysis of alveolar bone height was carried out in two anatomical planes: the alveolar bone crest and the mandibular canal in the atrophic mandible. The first and last of the measurements for each of the study groups were taken on the coronal sections. Vertical bone gain was measured by cone beam computed tomography (CBCT) at two different times—before the augmentation surgery and at the time of dental implant placement. All radiographs were taken with a surgical template compatible with the mandibular arch.
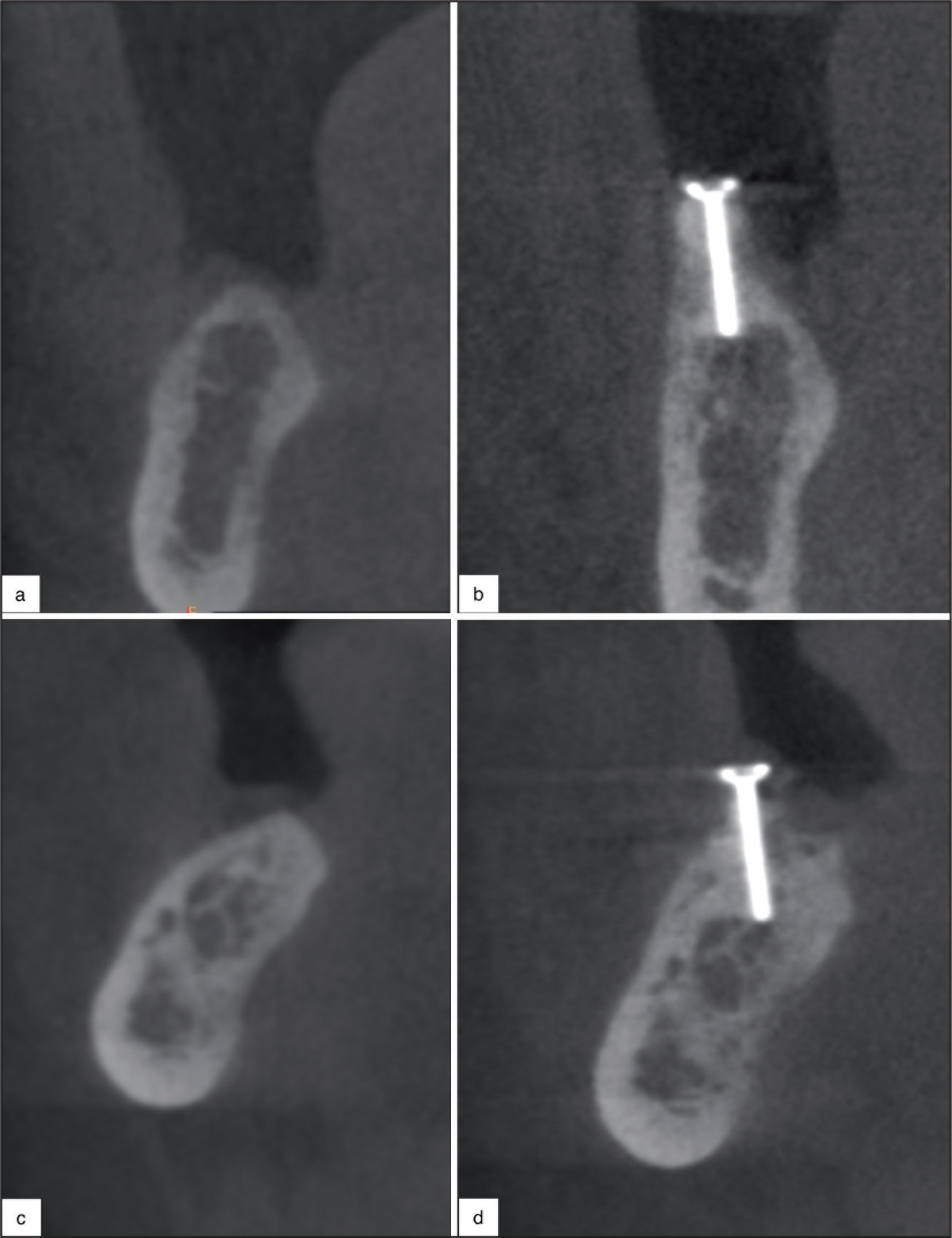
The residual bone height was set at 2 mm from the mandibular canal for both study groups before the augmentation surgery. This measurement was correlated on the second CBCT with a surgical template and the augmented bone height was measured postoperatively after 6 months. The extent of bone augmentation was evaluated on the basis of the difference between the residual and augmented bone measurements (Figure 2). After 3 months, the CBCT measurement process was repeated by the same examiner, in order to provide reliability and prevent bias (
CBCT View. (a) and (c) Height of Residual Bone Before Augmentation Surgery; (b) and (d) Vertical Bone Gain at Postoperative 6 Months
Histological Assessment
Bone core biopsies were harvested from the implant placement sites at postoperative six months. Two different trephine burs, of the same size, were chosen for each study group to prevent interfusion of the biopsies. The biopsies were retrieved in parallel to the augmented region by using a trephine bur (3 mm diameter and 10 mm long; Meisinger Trephine Basic Kit®, Düsseldorf, Germany) from both surgical sites for each patient.
The biopsies (3 mm diameter and 6 mm long) were fixed in 10% buffered formalin solution for seven days and decalcification was carried out in 15% EDTA over a period of four to six weeks. The decalcified biopsies were dehydrated using 50%, 60%, and 70% alcohol, respectively, and embedded in paraffin. The paraffin blocks were produced in 20-µm-thick sections and stained with hematoxylin–eosin alone for histological and histomorphometrical assessment.
The specimens were routinely processed for measurement using a light microscope (Olympus® BX51 System Microscope, Tokyo, Japan) with a digital camera (Olympus® DP 72, Tokyo, Japan) connected to a computer. Analysis was carried out by means of the Image J programme (NIH, Bethesda, MD, USA) for both groups, by a blinded examiner (YU). As an area of interest (AOI), histomorphometric analysis of the augmented region formed by the alveolar ridge augmentation techniques was represented on the biopsy area by a rectangular AOI in 2 mm2 (10× magnification) of grafted bone. The relative quantities of newly formed bone within both groups were planimetrically calculated as a percentage.
Clinical Assessment
Implant survival was assessed postoperatively twice in the first year at 6-month intervals. This assessment was based on the following criteria, as previously described 29 : absence of pain; erythema or any other signs of infection; absence of exposed implant surface leading to implant mobility; and absence of peri-implant radiolucency. Each implant was monitored by a blinded examiner (MÖY).
Data Collection and Management
Outcome data were recorded on a data collection form and transferred using a computer programme as a data management system for analysis. To avoid bias, data were collected and entered only by the blinded examiners.
Statistical Analysis
Statistical analysis was carried out using a software programme (IBM SPSS Statistics for Windows, Version 22.0. Armonk, NY: IBM Corp.). Data were summarized using means, standard deviations, frequencies, and percentages. The normality assumption of the numerical data was evaluated by use of the Shapiro–Wilk test and a paired samples
Results
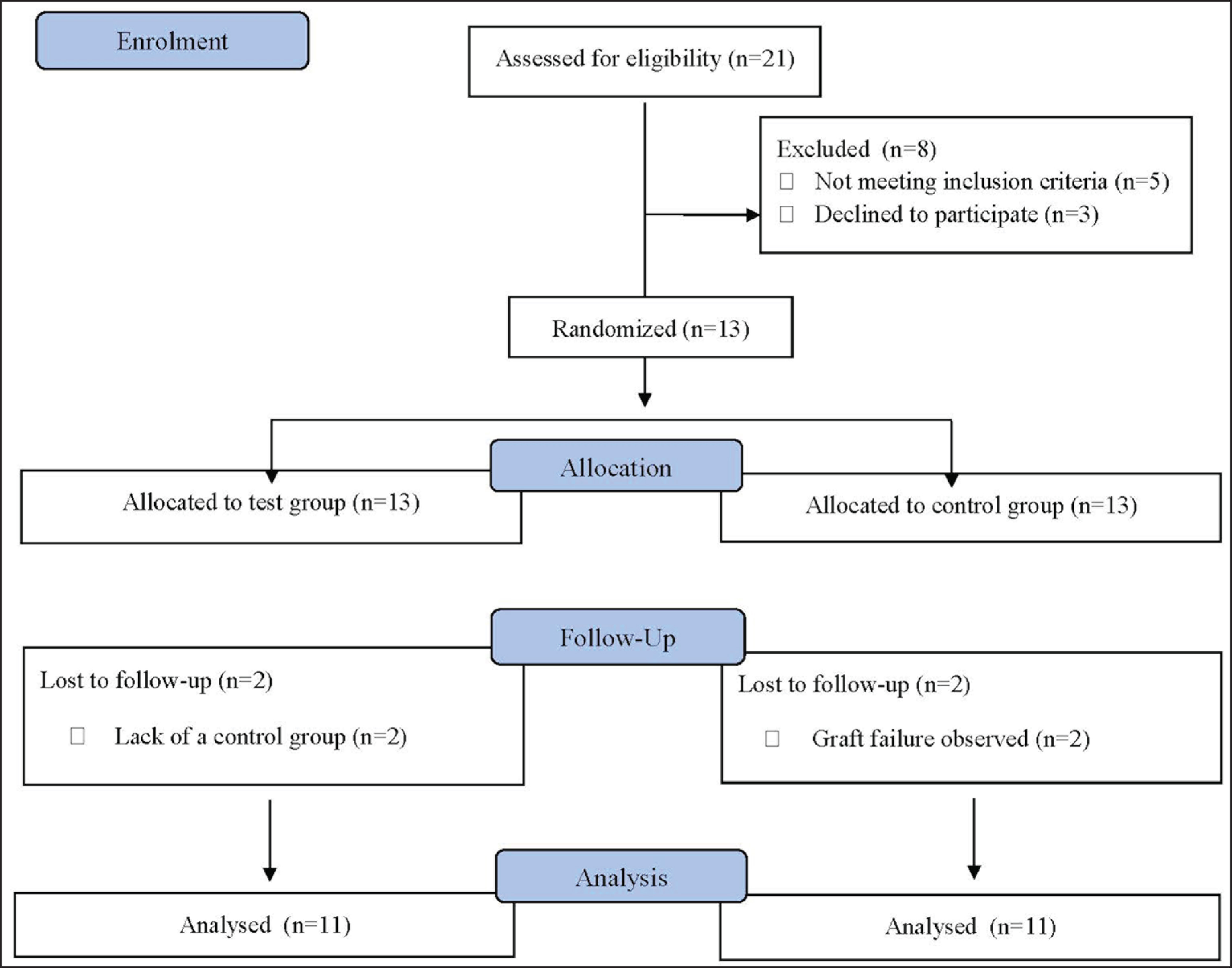
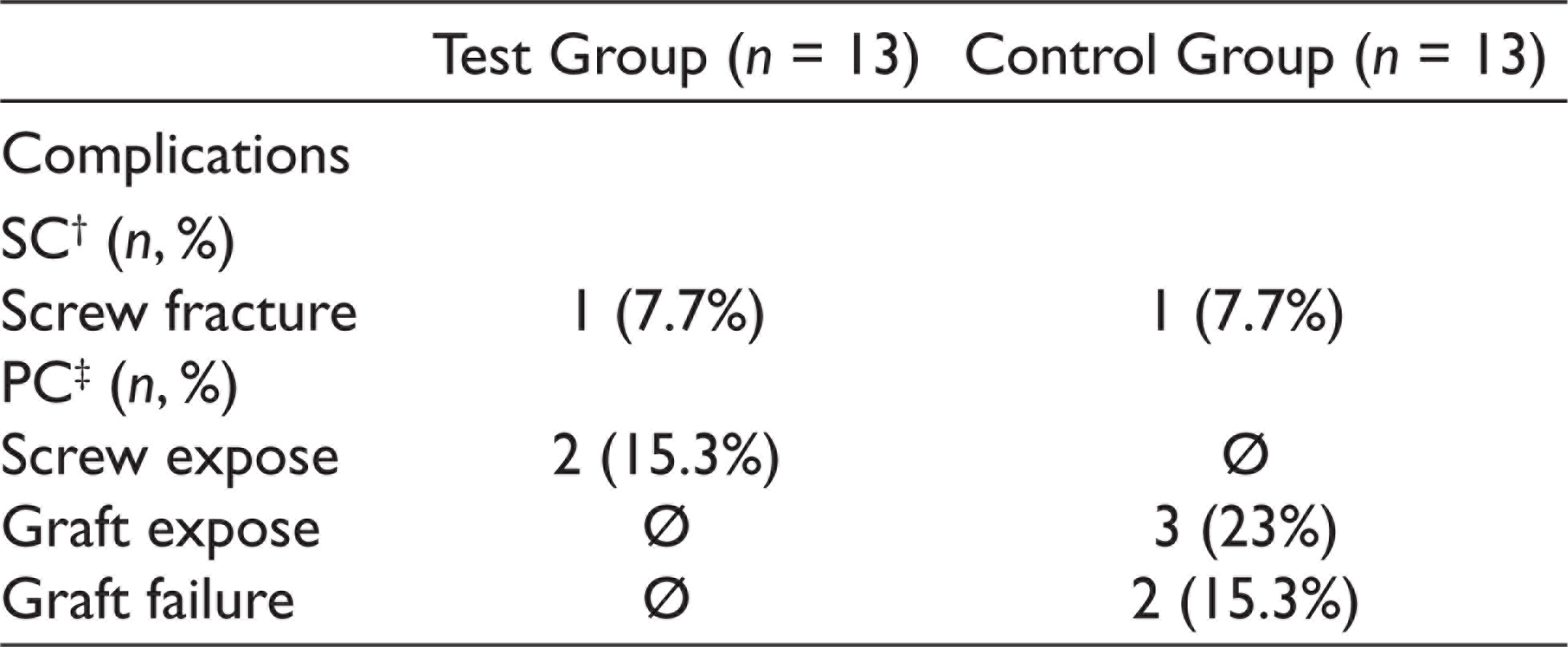
Thirteen patients underwent both screw tent-pole and autogenous block bone graft treatment from December 2016 to July 2018. Graft failures were observed in two surgical sides. This required the patients concerned to be excluded as it was not possible to match the control and test groups for comparison (Figure 3). Thus, 11 patients (4 males and 7 females; aged between 20 and 69; mean age 50.92 years ± 13.01) were evaluated in this study. Details of the complications are summarized in Table 1.
Detail of Study Participants and Exclusions
Description of Surgical and Postoperative Complications
Radiological Findings
Analysis of the augmentation measurements revealed the presence of bone reconstruction during follow-up. The mean of residual bone height was 8.14 ± 1.47 for the test group and 6.99 ± 1.88 for the control group. There was no significant difference between the groups (
Comparison of Vertical Bone Gain Between Test and Control Groups
Histological and Histomorphometric Findings
Analysis of the bone core samples retrieved from the test group revealed that graft material was observed in various portions, with empty osteocyte lacunae. Newly formed bone was analyzed throughout for graft material, and the interface between those portions of biopsy was clearly identified by the presence of increased osteoblastic activity. On the other hand, analysis of bone core samples retrieved from the control group revealed that graft material was not in direct contact with the newly formed bone in some portions. Bone remodeling was evident from the presence of perivascular mesenchymal stem cells, which differentiated into osteoblasts. There was no evidence of infiltration of acute or chronic inflammatory cells, or of foreign body reactions, in either of the groups (Figure 4).
Histological View of the Bone Core Biopsies Retrieved from (a–d) Autogenous Bone Graft (Control Group) and (e–h) i-PRF Enriched Allograft (Test Group). (a–d); Residual Graft Material with Empty Osteocyte Lacunae (). Newly Formed Bone Not in Direct Contact with Grafted Bone in Some Areas (). Mesenchymal Stem Cells Around Newly Formed Bone (). (e–h); Residual Graft Material () Surrounded by Newly Formed Bone (). Bone Remodeling İdentified by the Presence of Bone Spicules (), Osteoblast (), and Osteoclast cells (). (a and e): Hematoxylin-Eosin Staining, Magnification 10× and (b–d and f–h): Hematoxylin-Eosin Staining, Magnification 20×
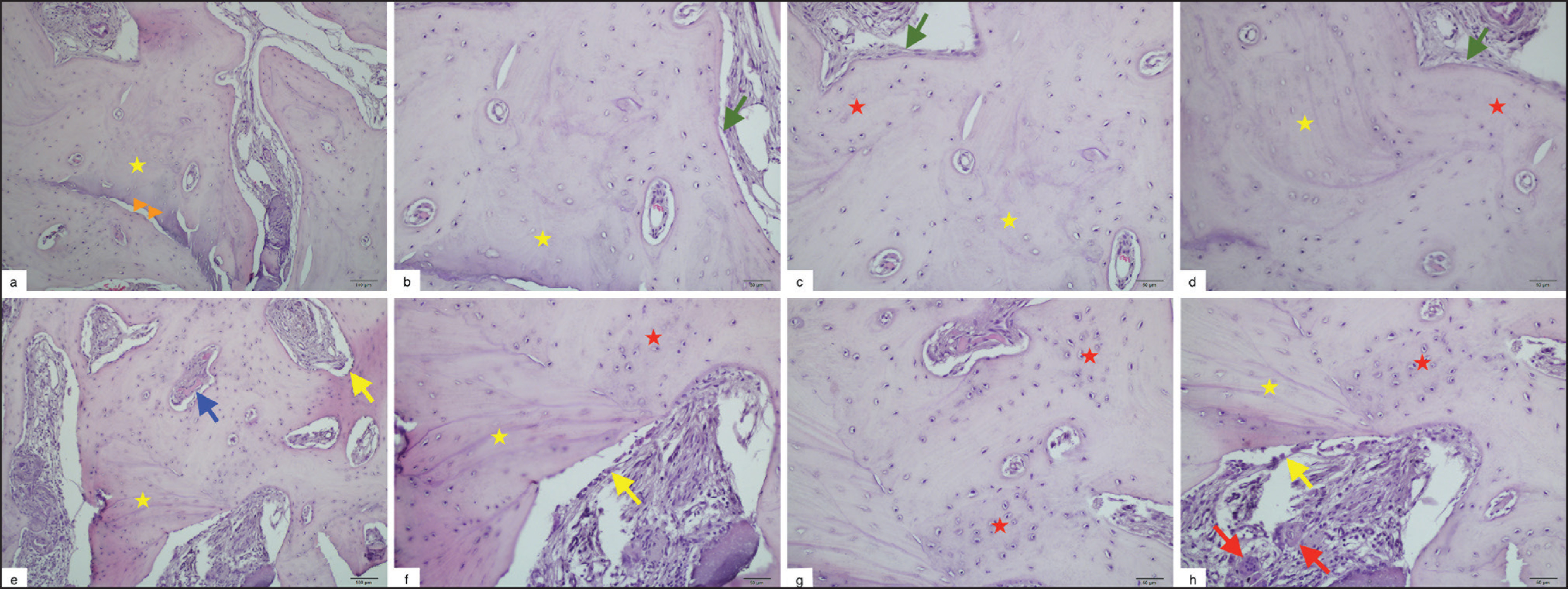
The percentage of newly formed bone was 18.08% ± 2.17% for the test group and 14.26% ± 1.76% for the control group. The difference between the groups was statistically significant (
Comparison of Newly Formed Bone, Graft Material, and Residual Bone Percentages (%) Among Test and Control Groups

Clinical Findings
A total of 48 implants (24 for the test group and 24 for the control group) were placed in accordance with prosthetic planning. Additional grafting was required around six implants in the control group. The osseointegration period was completed without infection, marginal bone loss, or implant failure. Twelve months after implant placement, the survival rates were 100% in both augmented sites.
Discussion
Various different grafting techniques have been proposed in the past with the aim of achieving efficient and reliable reconstruction of alveolar deficiencies.1,4,11,12 One of these techniques, screw tent-pole, has been examined in several studies.11–13 However, there have been few clinical randomized studies into the efficacy of this technique for vertical bone augmentation.3,11 In a recent study, Rocchietta et al. 3 used the screws to create a tent-pole effect and reported that the autogenous block grafts showed statistically higher bone volume in the augmented region compared to the autogenous particulate grafts. Systematic reviews of bone augmentation reported that nonautogenous bone grafts were deemed to be equally effective and beneficial methods when compared with autogenous bone graft.1,2,5,30 Therefore, it was considered that a different particulate bone graft might be used to build on this preliminary result when compared against autogenous block bone graft. Nevertheless, the results of our study demonstrated that vertical bone augmentation in the screw tent-pole group was lower than the autogenous block bone graft group. Thus, our hypothesis was not accepted.
Our study results may be explained by the different variables affecting the outcomes of the bone augmentation. Clinical complications were the main cause of unsatisfactory treatment outcomes in rehabilitating vertical alveolar bone deficiency. 4 There are many reports in the literature of soft tissue dehiscence or membrane exposure as a consequence of different augmentation techniques.1,2–4 Platelet concentrates as a fibrin matrix promotes the wound healing process through the inclusion of cytokines and growth factors. 17 Therefore, several studies have reported that this postoperative complication can be prevented by the use of platelet concentrates.15,17
The absence of clinical complications, and the beneficial effects on wound healing, may be related to the fact that these concentrates are derived from autologous sources. 31 On the other hand, Ehrenfest et al. 32 reported that the results may be due to the biological characteristics of platelet concentrates as a consequence of clinical preparation methods and protocols. Also, they may differ depending on the ability of fibrin cells to release growth factors. Similarly, the study by Miron et al. 33 revealed significant changes in the macroscopic morphology and size of PRF membrane and asserted that this was related to age, gender, and the time interval between blood collection and the start of centrifugation.
In our study, postoperative complications associated with screw and graft exposure were not eliminated by the use of L-PRF membrane. This could be explained by our observation of a very low rate of augmented bone height in the study groups compared with the previous studies.4,6,8–10 Nevertheless, the value of our findings in this study may be enhanced through further research involving different centrifugation protocols and patient groups with similar platelet values. Moreover, gingival biotype has the potential for soft tissue dehiscence during the healing period. Gingiva with a thin biotype may result in relatively high postoperative complications and affect the predictability of augmentation prognosis. 12 Therefore, further randomized studies, with the benefit of knowledge of the gingival biotype, are needed in order to evaluate the treatment outcomes of augmentation techniques.
In studies of the use of autogenous bone block graft as a tent-pole, Morad et al. 10 reported that clinical outcomes in terms of alveolar bone gain and rate of graft resorption were found to be better in the cortical tenting group compared with the autogenous bone block group. Khojastesh et al. 9 reported that cortical tenting using localized ridge augmentation provided sufficient bone regeneration for dental implant surgery. On the other hand, the authors reported that maxillary bone augmentation with cortical tenting produced better results than mandibular bone augmentation.
In studies where the screw-guided bone regeneration technique was used, Chasioti et al. 12 and Caldwell et al. 13 reported that the screws gave effective support and protection for horizontal bone regeneration against the various mechanical constraints. However, the authors asserted that this technique is not appropriate for vertical augmentation of atrophic ridges or large defects as bone resorption is relatively high. Le et al. 6 reported that adjunct grafting protocol was needed for the completion of prosthetic rehabilitation after the use of screw tent-pole technique in vertical bone augmentation of the posterior mandible. They suggested that this method can be used successfully as an effective correction for localized or single-tooth defects.
Consistent with these studies, our results showed that vertical bone gain in the screw tent-pole group was lower than the autogenous block bone graft group after bone augmentation in the posterior mandible. Thus the choice of patients, including those with total or bilateral edentulism, and also alveolar bone augmentation in the posterior mandible with its related occlusal forces, may be an accelerating factor on the poor clinical outcomes.
Bone regeneration may be successfully achieved due to the biological and mechanical attributes of the grafting materials.3,14 The biological features of bone augmentation materials demonstrate superiority in the proliferation and differentiation of cells for appropriate regeneration. 5 However, when platelet concentrates are used, the regenerative capacity of grafting materials may be accelerated through limited connective tissue penetration and increased osteoblastic activity. 15
Recently, several studies were conducted with i-PRF, using different centrifugation protocols with the aim of improving outcomes.7,16,18,22,23 i-PRF acts to promote the formation of a fibrin network, embedding leukocytes, platelets, and growth factors, which are effective in tissue healing.7,22 Moreover, i-PRF significantly increases the migration of cells and promotes osteogenic differentiation. Thanasrisuebwong et al. 16 reported that i-PRF may contribute improved biological and physical properties for the handling and stability of the grafting material application. They went on to suggest that the mixture with the particulate graft materials might imitate the block bone grafts by preserving their structures. 16
However, there is only limited research in the literature on the augmentation pattern of i-PRF enriched with grafting materials. Scarano et al. 23 reported that liquid platelet concentrate increased the mechanical resistance of particulate graft materials. Similarly, Sohn et al. 18 analyzed the effect of liquid platelet concentrates enriched with particulate graft materials as sticky bone. The authors concluded that they improved clinical outcomes, with stable bone augmentation and minimal soft tissue in-growth.
Our results showed that particulate allograft material with i-PRF was found to be rich in osteoblast cells when compared with autogenous block bone graft. Even though the hypothesis is that i-PRF, which acts as a dynamic gel, preserves its structure for more than 10 days, 22 no evidence was found in this study that vertical bone gain was any greater than that found in the case of particulate graft materials. 3
Several studies on autogenous block bone graft reported high success rates for implant survival.1,4 However, there has been only limited data from previous studies that used the screw tent-pole technique.6,12,13 In these studies, stable bone levels and healthy peri-implant tissues were indicated after implantation,12,13 and there was no evidence of peri-implant bone loss in the clinical and radiographic follow-up. 6 Consistent with these findings, our results showed that no complications were indicated in the osseointegration period and prosthetic rehabilitation was observed to be stable for both augmented sites in the 12 months after implant placement. While short-term outcomes have been encouraging, long-term follow-up is required to determine the potential impact of this treatment technique on implant survival rates.
The most important limitation of the study is the small number of participants. This detracts from the wider validity of the results and the scope for them to be extrapolated more generally. Another limitation is that this study did not evaluate gingival biotype, which may have provided further insight through correlation with the resorption pattern of the graft materials. This limited the scope of our study results on vertical bone augmentation. Moreover, this study presented short-term outcomes on implant survival rates. Given these limitations, it is clear that further randomized studies will be required in future, involving larger sample groups and long-term follow-up, in order to investigate the effect of screw tent-pole technique on vertical bone augmentation.
In conclusion, our study results showed that vertical bone gain in the screw tent-pole group was lower than the autogenous block bone graft group after bone augmentation in the posterior mandible. However, given the rate of new bone formation in the augmented region and the survival rate of implants, screw tent-pole may be considered a feasible technique for bone augmentation. Further randomized studies will be necessary to fully evaluate vertical bone augmentation using the screw tent-pole technique, with different graft materials and with larger samples.
Future Scope
Based on the study results, screw tent-pole may be considered a feasible technique for bone augmentation. However, further studies will be required to fully evaluate the effect of screw tent-pole technique on vertical bone augmentation, with different graft materials and with larger samples.
Footnotes
Acknowledgements
The authors are grateful to Prof. Dr Mehmet Nurullah Orman at the Department of Biostatistics and Medical Informatics, of the University of Medicine, for contributions to statistical analysis and the interpretation of the results.
Author Contributions
GI and TG conceived the ideas and planned the research; TG performed the surgeries; YU and HK contributed the biopsy sample preparation; YU analyzed and collected the histological data; MOY analyzed and collected the clinical data; GI led the writing of the manuscript. All authors discussed the results and contributed to the final manuscript.
Data Availability Statement
The datasets used and/or analyzed during the study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Policy and Institutional Review Board Statement
The research protocol and informed consent forms for all procedures on patients were approved by the local ethics committee of the university (16-5.1/11), in accordance with the Helsinki Declarations of 1975. The study was registered on ClinicalTrials.gov (NCT04133090;
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Scientific Research Committee of the University (16-DIS-026).
