Abstract
Aim:
To assess the before- and after-treatment levels of glutathione (GSH) in the serum of patients with chronic apical periodontitis.
Materials and Methods:
The study involved 20 systemically healthy patients with radiographic evidence of chronic apical periodontitis in at least one tooth, of which 11 were finally assessed. 10 mL of the patient’s venous blood was obtained before commencing the endodontic treatment to assess the pre-operative serum GSH levels (T0). The canals cleaned and shaped, and irrigated with 2.5% sodium hypochlorite, and filled with a calcium hydroxide based intracanal medicament. At the subsequent appointment after one week, teeth were obturated. The patients were recalled 30 days after the completion of endodontic therapy, and blood samples were assessed again at time frame T1 for serum GSH. The difference in pre- and post-treatment values for T0 and T1 was statistically analyzed.
Results:
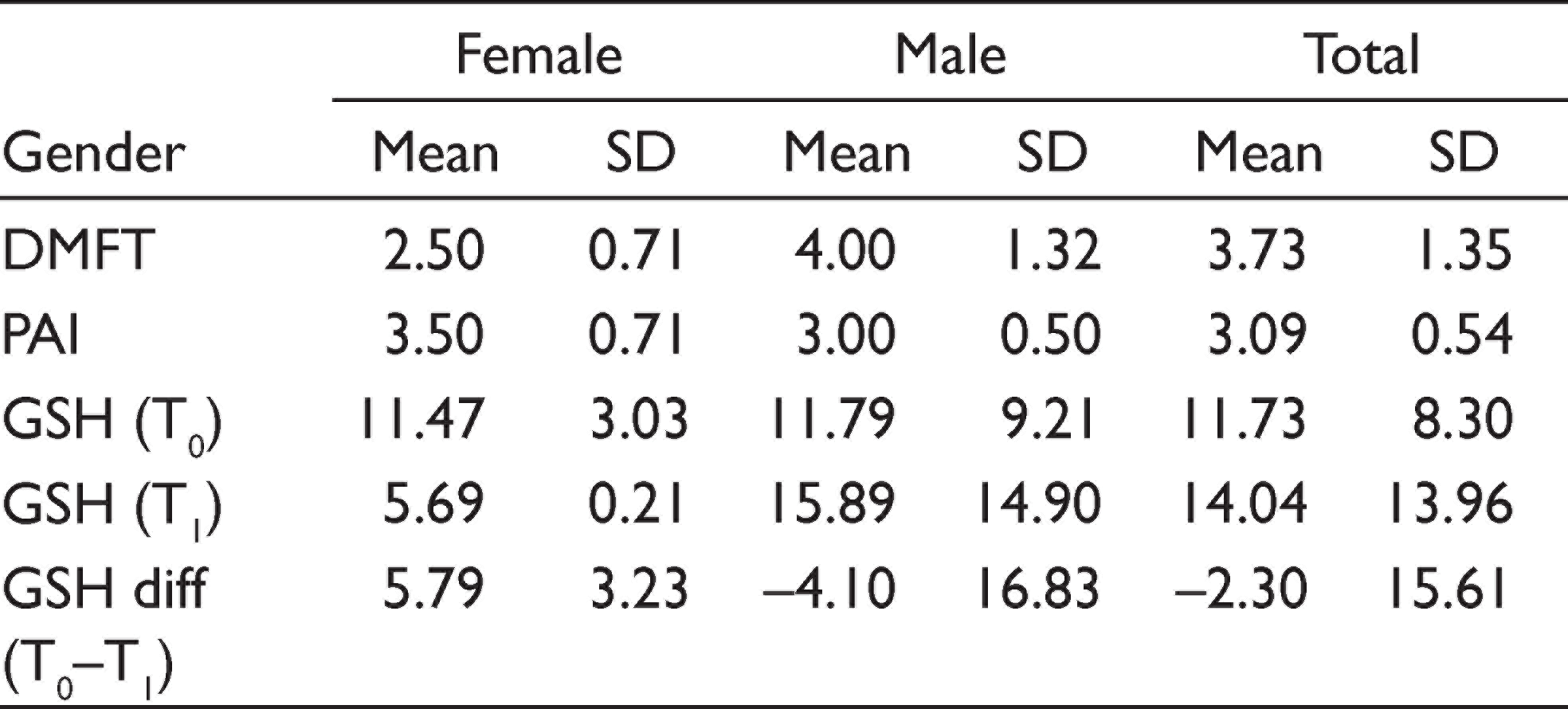
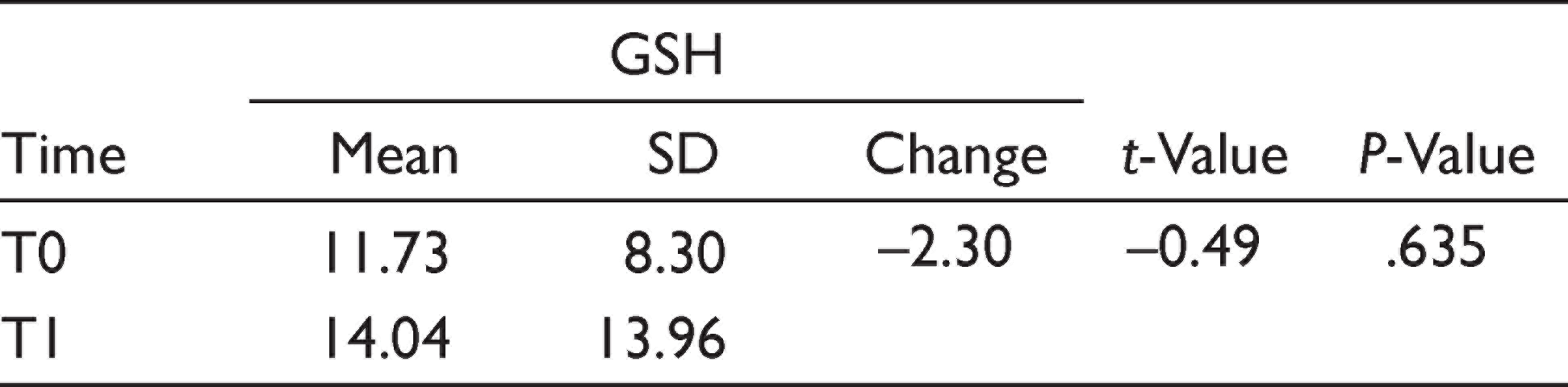
The mean age of the subjects (N = 11) was 21.09±3.24 years, with 19.50±0.71 years in female group and 21.44±3.50 years in the male group. The mean GSH at T0 was 11.73±8.30 units, which was increased to 14.04±13.96 units at T1. However, no significant change was observed in the GSH level from T0 to T1 (change = –2.30, t-value = –0.49, P = .635).
Conclusion:
The results of this study suggest that endodontic therapy may play a role in restoring the systemic oxidative balance, as seen by the increase in post-treatment serum GSH levels.
List of Abbreviations
AO = Antioxidant
CAP = Chronic apical periodontitis
GSH = Glutathione
ROS = Reactive oxygen species
Introduction
Recent evidence views chronic low-grade infections of the oral cavity in a new light where these are seen as a contributing factor to the total systemic inflammatory burden.1,2 Furthermore, long-lasting chronic inflammatory conditions are known to be related to the production of certain reactive oxygen species (ROS), which is defined as partially reduced metabolites of oxygen that possess robust oxidizing potentials. 3 Oxidation is the “chemical reaction involving the transfer of electrons or hydrogen from a substrate to an oxidizing agent.” While oxidation is vital for the existence of any organism, it is also responsible for the production of certain ROS. 4 These ROS initiate chain reactions, which may further lead to cell damage or death. Common examples of ROS are the superoxide radical (O2−), hydroxyl radical (OH) and nitric oxide (NO) radical (NO−), hydrogen peroxide (H2O2), and hypochlorous acid (HOCl).5, 6 To counterbalance these reactions, certain molecules known as antioxidants (AO), present at low concentrations, stop the damage initiated by ROS, by inhibiting the oxidation of the substance. Oxidative stress (OS) leads to molecular damage due to a disparity between oxidants and antioxidants. OS is linked to the pathogenesis of various chronic inflammatory disorders. These include noninsulin-dependent diabetes mellitus, cardiovascular disease, and acute respiratory distress syndrome. 7 Glutathione (GSH) is a low-molecular-weight, non-protein cellular thiol, present universally in all the cells of the body. Sometimes referred to as “the master antioxidant,” it is a powerful AO that prevents the impairment to crucial cellular components caused by ROS, and functions as a cofactor for enzymes in the destruction of ROS. 8
Chronic Apical Periodontitis (CAP) is an oral inflammatory pathological condition resulting from the infectious incursion of the periradicular tissues from a diseased radicular pulpal tissue, leading to an inflammatory response at the root end and consequent clastic-breakdown of the periradicular bone. 9 CAP is a common condition in adults, as reported in several epidemiological studies conducted, which show a prevalence ranging from 27% to 70% . 10 Over the last few years, researchers have conducted studies to assess if CAP is correlated to oxidative stress and systemic inflammation. 11 Few cross-sectional clinical researches have shown that there is an upsurge in inflammatory mediators, such as the interleukins (ILs), matrix metalloproteinases (MMPs), and tumor necrosis factor (TNF-α) in patients with CAP in contrast to the healthy patients. 12
Treatment of CAP involves chemomechanical cleaning of the infected root canal to remove and reduce the total microbial load of the infected tooth and subsequently find a resolution to the inflammation.
Although a few studies have been conducted to see the association between apical periodontitis and oxidative stress, no studies have evaluated GSH values. Also, no studies have longitudinally followed up the patients to see the effect of endodontic treatment on serum GSH levels. Thus, the present pilot study aims to estimate the change in serum concentration of GSH before and after root canal treatment in patients with CAP.
Materials and Methods
Setting and Design
This pilot study was a short duration longitudinal clinical trial. It was a pre-post study, where each patient acted as his/her control. It was a secondary analysis of the data, which was primarily collected to evaluate the influence of endodontic therapy on serum CRP levels of patients with CAP.
The study population comprised of otherwise systemically healthy patients who presented with radiographic signs of periapical periodontitis visiting the Department of Conservative Dentistry and Endodontics, King George’s Medical University, Lucknow, between November 2016 and January 2018. Patients on any medication, antimicrobials, or any anti-inflammatory analgesics during the last three months, with systemic conditions (pregnancy, cardiovascular diseases, thyroid disorders, diabetes mellitus, obesity, allergies), less than 25 teeth in the oral cavity, or poor oral hygiene were removed from the study. Finally, 20 patients were recruited and signed informed consent before initiating the procedure.
Study Method
Patients meeting the inclusion criteria were further investigated for clinical and radiographic baseline parameters (periapical disease condition according to the periapical index [PAI] described by Ostravik et al.) 13 The decayed, missing, filled teeth (DMFT) score of the patients was also recorded. Before starting the endodontic treatment, a 10 ml sample of patient’s venous blood was collected. Serum was separated from the blood sample and stored at –80° C for further processing and estimation of GSH at time frame T0 (pre-treatment).
A single operator carried out all the steps of endodontic treatment to reduce interobserver bias. Root canal therapy was performed according to standard protocols of isolation and sterilization. Pulpal space were prepared K files (Dentsply, Maillefer, Switzerland) and copiously irrigated with 2.5% NaOCl. After placing an intracanal medicament (Metapex, META Biomed Co. Ltd., Korea) for a week, the canals were obturated. Clinical and radiographic parameters after the completion of therapy were noted.
No antibiotics or analgesics were prescribed, and patients were refrained from taking any medication without prior consultation with their dentist. They were recalled after 30 days of root canal treatment, and blood samples were obtained at time frame T1 to assess postoperative serum GSH levels. The change in pretreatment and posttreatment values for T0 and T1 was statistically calculated to measure the influence of endodontic treatment on serum GSH levels. Further, the difference in the levels of GSH was correlated to the clinical healing and the PAI score. Patients who reported with acute pain or swelling during or after completion of the treatment were dropped from the study.
GSH estimation in serum samples was done in the biochemistry lab according to the instructions in the GSH assay kit (Cayman Chemicals, MI, USA).
Statistical Analysis
The results were analyzed using descriptive statistics and making comparisons among various groups. A paired t-test compared the T0 and T1 values of GSH. Pearson’s correlation was applied to check if there is any correlation between GSH changes with other study parameters. Statistical analyses were performed using SPSS version 19.0 (SPSS Inc., Chicago, IL, USA). A value of P < .05 was considered statistically significant.
Results
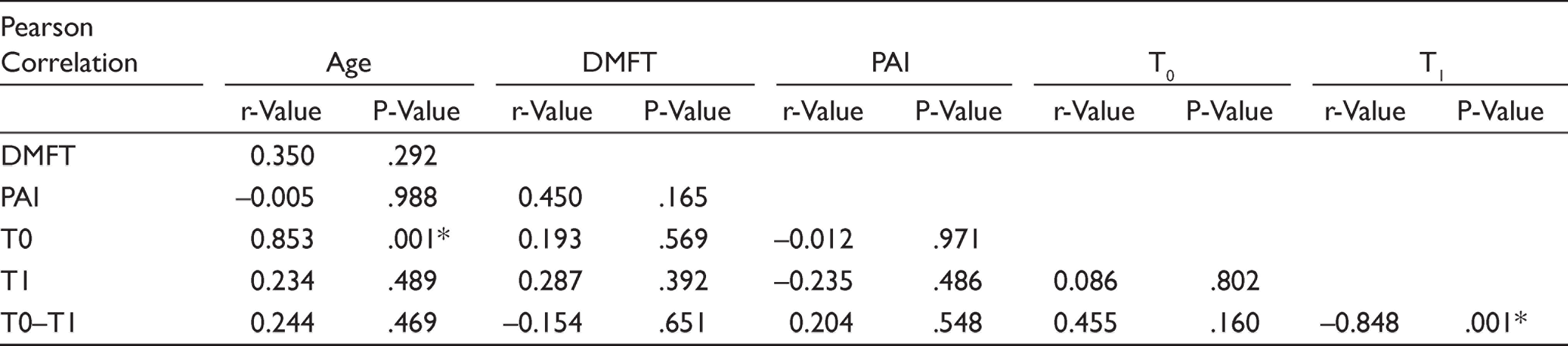
Out of a total of 20 patients enrolled for the study, 15 fulfilled the eligibility criteria. Of these, two patients did not follow-up and two reported acute pain during treatment; therefore, they were omitted from the trial. Ultimately, only 11 patients were evaluated. In the study, the mean age of the subjects (N = 11) was 21.09±3.24 years, with 19.50±0.71 years in the female group and 21.44±3.50 years in the male group. A descriptive summary of patients is illustrated in Table 1. The mean GSH at T0 was 11.73±8.30 units, which was increased to 14.04±13.96 units at T1. However, no significant change was observed in the GSH level from T0 to T1 (change = –2.30, t-value = –0.49, P = .635) (Table 2). The Pearson correlation statistics showed that there was no significant correlation between PAI or DMFT to the change in GSH values; however, a significant positive correlation was observed between age and GSH values of time T0, while significant negative correlation was observed between GSH change from T0 to T1 and TSH value at T1 (Table 3).
Mean Values of Various Parameters of the Study Population
Change in Serum GSH Level at T0 (Pre-treatment) and T1 (After 30 Days)
Correlations Between Study Parameters
Discussion
Inflammation is the defense mechanism of our body towards injurious exogenous and endogenous noxious agents. The current concept says that while inflammation can initiate oxidative stress, oxidation also can stimulate inflammation. 14 Depletion in levels of GSH can be seen in a state of inflammation, which in turn inhibits the resolution of the state of inflammation. Research shows that decreased GSH levels are associated with an increase in pro-inflammatory mediators and inflammatory potential. Therefore, it is sensible to consider GSH levels and function when addressing chronic inflammatory disease states. 15 In this pilot project, a slight increase in the mean serum GSH values was seen from time frame T0 (before treatment GSH = 11.73±8.30) to T1 (1 month after treatment GSH = 14.04±13.96), though the rise in levels was not significant (P = .635). GSH has a low redox potential, and an increased concentration in the cells. In the presence of electrophilic elements, such as free radicals and ROS species, GSH easily oxidizes nonenzymatically to GSH disulfide. The thiol group of GSH is responsible for its antioxidant action and itself becomes oxidized when GSH reduces the target substrate molecules. 16 Studies report a significant rise in GSH peroxidase and GSH transferase, and reduced GSH and total GSH levels in gingival tissues and serum of periodontitis patients. 17
Ichingolo et al., 18 conducted a prospective clinical study to evaluate the oxidative stress (by measuring reactive oxidation metabolites: d-ROM) in plasma samples of patients with CAP, and the role of endodontic treatment in normalizing the oxidation imbalance (by assessing biological antioxidant potential: BAP). They concluded that patients with CAP exhibited significantly higher levels of oxidative stress than the healthy control group subjects. The reactive oxidation metabolites were shown to decrease, and the biological antioxidant potential values were seen to rise in patients with CAP following the endodontic treatment. The results of their study suggested that a well-performed endodontic therapy (aka root canal treatment) could restore a disturbed oxidative imbalance.
A clinical study by Vengerfeldt et al. 19 compared the magnitude of oxidative stress in saliva and the root canal contents in patients with various endodontic pathologies and healthy control group. Higher levels of oxidative stress markers were seen in patients with diseased pulp and periapical tissues, while controls exhibited the lowest oxidative stress marker levels in both root canal contents and saliva. Also, substantial positive correlations between oxidative stress levels, PAI scores, and pain were disclosed. Hernández-Ríos et al., 20 in a review article, illustrated the role of oxidative stress in apical periodontitis local and systemic events. The authors opined that oxidative stress is crucial to the pathogenesis of apical periodontitis. Reactive oxidation species are critical as a protective host defense mechanism against endodontic bacterial invasion; yet, oxidant imbalance contributes locally to the formation and development of periapical pathologies. Additionally, some initial evidence is indicative of an underlying connection between systemic oxidative stress and atherosclerosis associated with chronic periapical periodontitis. 21 As the root canal infection progresses into a periapical infection, there is a release of inflammatory mediators, for example, cytokines and matrix metalloproteinases. Also, the binding of the Toll-like receptors on phagocytes’ surface brings about specific humoral and cellular responses which, in turn, prompt the generation of reactive metabolites. 22 Elevated breakdown products of peroxidation, and reduced GSH activity in periapical granulomas are also reported. 23
Limitations
This study was a pilot clinical intervention with a very small sample size. Also, the period of follow-up was not very long. Though care was taken to exclude patients with systemic diseases or on medication, as these affect the serum GSH levels, it is still impossible to rule out all factors, which may have influenced the results.
Conclusion
This study focused on the interrelationship between oxidative stress and chronic inflammatory conditions. Like other chronic, long-standing inflammatory conditions of the body, CAP also seems to affect the systemic oxidative stress, as shown by slight increase in GSH levels after endodontic treatment. More clinical trials, with larger sample sizes, should be conducted to figure out a correlation between the size of periapical lesions, the number of teeth affected by CAP, and the magnitude of change in GSH or other oxidative stress markers.
Future Scope
The results of this pilot study can serve as a basis for well-designed, clinical trials with a larger sample size to study the correlation between the systemic GSH levels and oxidative stress, and chronic apical periodontitis.
Author Contributions
Rhythm Bains: Concept and design of the work, acquisition of data
Wahid Ali: Analysis and interpretation of data
Promila Verma, Rhythm Bains: Drafted the article and revised it critically for important intellectual content
Aseem P. Tikku, Anil Chandra: Approved the version to be published
Data Availability Statement
The data related to the study can be made available on request.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
The institutional ethical committee approved the study protocol (Ref No. 77th ECM B-IMR-faculty/P2).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded in part by the research cell, King George’s Medical University, Lucknow.
Patient Declaration of Consent
A written, informed consent was taken from all patients enrolled in the study.
