Abstract
Aim:
To evaluate the therapeutic antioxidant effect of Hypericum perforatum L. (HPL) extract on oxidized dentin as well as to address the following research question: Is experimental HPL-derived antioxidant treatment protocol as effective as sodium ascorbate in terms of reversing the compromised resin bonding to bleached dentin?
Materials and Methods:
Eighty bovine permanent mandibular incisors were included in the shear bond strength (SBS) test and randomly assigned into four groups (n = 20) as follows: negative control (NC), positive control (PC), sodium ascorbate (SA), and Hypericum perforatum L. (HPL). Group NC did not receive bleaching treatment. All specimens except the NC group were oxidized by bleaching with 35% hydrogen peroxide. Experimental antioxidant solutions were prepared from SA powder and HPL extract at 10% concentrations. The bleached specimens in the SA and HPL groups were treated with respective antioxidant solutions. The SBS test was performed at a crosshead speed of 1 mm/min. The SBS data were analyzed with one-way analysis of variance using the post-hoc Bonferroni correction. The total phenolic content and the antioxidant activity of HPL extract were assessed by using the Folin–Ciocalteu assay and the 2,2-diphenyl-1-picrylhydrazyl free-radical scavenging activity test, respectively.
Results:
Considering the SBS test results, statistically significant the highest and the lowest mean values were observed in NC and PC, respectively (P <.05). SA and HPL were not significantly different (P > .05). The total phenolic content (104 ± 1.740 mg gallic acid equivalents (GAE)/gram (GAE/g) dry HPL extract) and the antioxidant activity of the HPL extract were demonstrated.
Conclusions:
The natural HPL plant extract was as effective as SA in reversing the compromised resin bonding to bleached dentin. Antioxidant treatment derived from the HPL extract may enable immediate resin restoration of oxidized dentin.
Introduction
Tooth bleaching is a widely preferred treatment modality for discolored nonvital teeth due to its conservative and cost-friendly characteristics compared to other invasive treatment options such as full crowns. 1 Hydrogen peroxide, the main active ingredient in bleaching agents, can easily penetrate dentin, and then tooth bleaching occurs as a result of the interaction between hydrogen peroxide and organic stain molecules.1,2 Research has shown that the oxidizing action of hydrogen peroxide not only affects stain molecules but also interacts with the organic and inorganic matrix of dentin.2,3 Those interactions may result in some postbleaching clinical adverse effects, such as compromised bonding to oxidized dentin4-7 or decreased fracture resistance. 3
Throughout the bleaching process, the breakdown of hydrogen peroxide results in the release of several reactive oxygen species, namely free radicals such as nascent oxygen, hydroxyl, hydroperoxyl, and superoxide radicals. Those free radicals are mainly held responsible for the compromised resin bonding to bleached tooth structures. 2 Since residual free radicals remained within tooth structures are spontaneously released with time, postponing the final restoration has been suggested. 1 Nevertheless, a time delay between temporary and final restoration would inevitably increase the risk for coronal microleakage 8 and fracture. 3
Postbleaching use of antioxidants has been investigated with the aims of reversing the compromised bonding to resin-based restorative materials, decreasing the risk for fracture, and enabling the immediate resin restoration of the access cavity by eliminating the time delay after bleaching.3,4,9 The sodium salt of ascorbic acid (L-ascorbic acid), sodium ascorbate (SA) (sodium-L-ascorbate), has been the most widely tested antioxidant on oxidized tooth structures.10,11 However, a drawback of using ascorbic acid or its derivatives (i.e., SA) is that they are very vulnerable to environmental conditions such as light, air, temperature, pH, or humidity, which result in a rapid loss in their stability and the antioxidant potency.11,12 In the pursuit of seeking more stable antioxidants, researchers have focused their attention on natural plant-based antioxidant sources that possess additional therapeutic benefits with minimal side effects.
Hypericum perforatum L. (HPL), also known as St. John’s wort, is one of the mostly used medicinal plants in the pharmaceutical industry worldwide. Several researchers13,14 suggested that HPL could exert therapeutic effects by acting as a natural free-radical scavenger/antioxidant and might play a role in improving oxidative stability in biological systems. While extensive research has been performed, the possible antioxidant therapeutic efficacy of HPL on oxidized dental structures has not been investigated to date.
For this purpose, in this study, an experimental antioxidant protocol derived from the freshly prepared HPL plant extract was created. Before investigating the antioxidant efficacy of HPL extract on bleached dentin, the antioxidant capacity of the tested substance was investigated by the 2,2-diphenyl-1-picrylhydrazyl free radical (DPPH) assay, and the total phenolic content was analyzed by the Folin–Ciocalteu method.15,16 To the best of our knowledge, this is the first study that comparatively evaluated the free-radical scavenging/antioxidant effect of HPL with SA on oxidized dentin. The null hypothesis tested in the study was that there would be no difference between experimental antioxidant treatment protocols derived from the HPL extract or SA in terms of reversing the compromised bonding to bleached dentin.
Materials and Methods
Setting and Design
This interdisciplinary laboratory research was conducted in three different centers, and all the experimental phases were completed within six months. HPL plant was obtained from the Department of Chemistry, Aydin Adnan Menderes University, Turkey. The extraction of the plant and preparation of the experimental antioxidant solutions have been performed in the same institute. Botanical (taxonomical) identification of the plant was performed in the Department of Biology, Aydin Adnan Menderes University, Turkey.
Sound bovine mandibular incisors obtained from a slaughter house were used as the bonding substrate for the SBS test, which was performed in the central laboratory at the Faculty of Dentistry, Aydin Adnan Menderes University, Turkey. As no permission of any sort is required to use extracted teeth from slaughtered animals, ethics approval was not required for the study. The exclusion criteria consisted of the teeth with cracks, hypoplastic defects, or surface irregularities.
G-Power v.3.1.9.4 software (Heinrich Heine, University of Düsseldorf, Düsseldorf, Germany) was used to determine the required minimum sample size, according to the data of a previous research. 17 Based on the parameters of an alpha-type error of 0.05, a beta power of 0.95, and an effect size of 0.50, the minimal estimated sample size per group was found to be 19. Considering the possible loss in the experiment, 20 specimens for each group (80 specimens in total) were included in the study. The simple random sampling method was used for sample selection by using a free Internet-based program.
Plant Material
Fresh plant samples were washed to remove any dust, and then dried at room temperature keeping away from direct sunlight on a large surface for 24 hours. Flowers and leaves were separated using scissors. Leaves were removed away, and the flowers were dried at room temperature until they were ready for grinding. A sample of the whole plant was dried according to the botanical procedure and used for identification. It was recorded as HPL and stored in the herbarium with the specimen number AYDIN-2133.
Preparation of Experimental Antioxidant Solutions
Methanol, gallic acid, Trolox, SA, DPPH, Folin–Ciocalteu reagent (FCR), and Na2CO3 were obtained from Sigma-Aldrich (Steinheim, Germany).
A 100 g of the dried flower was transferred into a beaker, and 250 mL distilled water was added. The mixture was shaken at 120 rpm with a benchtop shaker (Promax 2020, Heidolph, Kelheim, Germany) in dark at room temperature for 2 hours. The solution was filtered through a black-ribbon filter paper. The residue was treated with 250 mL distilled water and shaken for 2 hours again. The filtration procedure was repeated once more, and the residue was left to shake for 24 hours with 250 mL distilled water. The filtrate was collected, and the last extraction step was applied for 2 hours. The filtrates were pooled and kept at +4 °C until the solvent removal process. The solution obtained after extraction with water was frozen at –20 °C and dried by lyophilization using a lyophilizer (Freezone 6, Model 77520, Labconco Co. Kansas City, MO, USA) at –50 °C, and 0.04 mbar for 72 hours. The dried extract was collected and stored at –12 °C until used.
Based on the findings of a previous pilot study (unpublished), the HPL-derived antioxidant solution at a concentration of 10% was decided to be prepared. To prepare the 10% HPL extract solution, 10 g of HPL extract was dissolved in 100 mL distilled water by stirring at +40 °C for 15 minutes. Then, the solution was kept in an ultrasonic bath for 15 minutes to be completely dissolved. To prepare the 10% SA antioxidant solution, 10 g of SA powder was dissolved in 100 mL distilled water.
Determination of the DPPH Free-Radical Scavenging Activity
Radical scavenging activity of HPL and standard antioxidants (i.e., gallic acid, Trolox, and SA) toward DPPH free radical was evaluated by the method proposed by Brand-Williams et al.12,16,18,19 Three milliliters of extract solutions with increasing concentrations (5–500 μg/mL) were mixed with 500 μL of the 0.5 mM methanolic DPPH solution. Reaction mixtures were vortexed and kept at room temperature in the dark for 30 minutes. Absorbances were measured at 517 nm with a UV-Vis spectrophotometer (UV–Vis 1601, Shimadzu, Kyoto, Japan). The blank sample was prepared by using extract solutions and methanol for each concentration to prevent the contribution into absorbance from the sample itself. Negative control was also prepared in the absence of the extract solution. The percentage of DPPH radical scavenging activity was calculated by using the following formula:
DPPH radical scavenging activity = [(A0–A1)/A0]×100,
where A0 was the absorbance of the control and A1 was the absorbance of the sample. The results were expressed as the IC50 (μg/mL) values that represent the antioxidant concentration able to scavenge 50% of DPPH radical present.
Determination of Total Phenolic Content
The total amount of phenolic compounds in HPL extracts was determined according to the method reported by Singleton et al.15,16 The extract solution (1.0 mg/mL) was prepared in distilled water, 500 μL of the solution was transferred to a 100 mL Erlenmeyer flask, and 22.5 mL of distilled water was added. FCR (0.5 mL) was pipetted, and the solution was stirred for 3 minutes before the 1.5 mL 2.0% (w/v) Na2CO3 solution was added. The resulting solution was shaken with a benchtop shaker at 120 rpm in dark and at room temperature for 120 minutes. The absorbance of the solution was measured at 760 nm using distilled water as blank. The same procedure was applied for gallic acid, which was used as the representative of phenolic compounds, and the total phenolic content of the extract was expressed as the mg GAE/g extract using gallic acid concentration versus the absorbance standard plot.
Preparation of Tooth Specimens
Freshly extracted bovine incisors were cleaned away from residues and stored within a 0.1% thymol solution in a refrigerator at 4 °C for one week. Afterward, all teeth were examined under a stereomicroscope at ×40 magnification to exclude any tooth exhibiting cracks, hypoplastic defects, or surface irregularities. Eighty sound bovine mandibular incisors, incorporated in the study, were stored in daily refreshed distilled water throughout the experiment and used within four weeks. Coronal tooth segments were obtained by horizontally sectioning 3 mm below the cementoenamel junction with a water-cooled diamond saw in a precision-cutting machine (Precision Cutter, Mod Dental, Ankara, Turkey), and the roots were removed. Vestibular enamel surfaces facing upward all coronal tooth segments were horizontally embedded in auto-polymerizing acrylic resin placed within standard sized polyvinyl chloride (PVC) molds. An individual number was assigned to each specimen and recorded on the relevant PVC mold. To expose superficial dentin, vestibular enamel of each specimen was removed with a diamond bur attached to a high-speed handpiece under copious water. Thereafter, exposed dentin surfaces were wet grounded with a 600 grit SiC paper for 15 seconds to obtain a standard smear layer. Using an Internet-based random sampling program, all specimens were randomly assigned to 4 groups of 20 each (n = 20). The groups were as follows: negative control (NC)/no bleaching, Positive control (PC)/bleached-only, sodium ascorbate (SA), and HPL.
Bleaching Treatment and Antioxidant Application
Negative control specimens in NC were not bleached. Specimens in PC, SA, and HPL were bleached with 35% hydrogen peroxide gel using the walking bleach technique according to the manufacturer’s instructions. Gently dried dentin surfaces were completely covered with 1–2-mm-thick bleaching gel (Opalescence Endo, Ultradent Products Inc., South Jordan, UT, USA), and a cotton pellet was placed onto the gel-applied surface. Afterward, specimen surfaces were thoroughly covered with temporary cement (Cavit, Espe, Seefeld, Germany) to the level of surrounding acrylic area, and the specimens were kept into an incubator at 37 °C, 100% relative humidity for 3 days (72 hours). At the end of this process, temporary cement, cotton pellet, and the bleaching gel were removed from the surfaces, and the specimens were rinsed with distilled water for 60 seconds.
Experimental antioxidant solutions, 10% SA and 10% HPL, were applied onto the bleached dentin surface of each specimen in SA and HPL, respectively, by agitating with a microbrush in an active manner for 10 minutes. The solutions were kept refreshing once in 2 minutes. Then the specimens were rinsed with distilled water for 60 seconds. Positive control specimens in PC did not receive antioxidant treatment.
Shear Bond Strength Test
A composite resin build-up was restored onto the dentin surface of each specimen using an etchant gel (K-etchant, Kuraray Co., Osaka, Japan), a two-step etch and rinse adhesive system (AdperTM Single Bond 2, 3M ESPE, St. Paul, MN, USA), and a nanohybrid composite (Filtek Z 250, 3M ESPE, St. Paul, MN, USA) according to the manufacturer’s instructions. Transparent plastic tubes with 3 mm internal diameter and 4 mm height were used as templates, and the restorations were completed in two subsequent increments. After photopolymerization, all specimens were incubated at 37 °C, 100% relative humidity for 24 hours. The contents of the materials are demonstrated in Table 1.
The Contents of the Materials Used in the Study

SBS was measured using a shear test device (Micro Tensile/Shear Device, Standard Model, Mod Dental, Ankara, Turkey) equipped with software. Before starting the test, relevant data, such as the bonding surface area (7.065 mm 2 ), crosshead speed of the knife edge (0.5 mm/min), and the maximum debonding force to be applied (500 N), were entered into the computer. The software package automatically calculated the SBS values in both Newton (N) and megapascal (MPa=N/mm²), by dividing the maximum applied force at the moment of fracture by the surface area.
Statistical Analysis
SBS values were analyzed by one-way analysis of variance (ANOVA) with Bonferroni post-hoc comparisons at a significance level of .05, in SPSS Statistics V25. Fractured areas of specimens (resin–dentin interfaces) were evaluated under a stereomicroscope at ×40 magnification, and failure modes were classified as cohesive, adhesive, or mixed.
Results
DPPH Radical Scavenging Activity and Total Phenolic Content
DPPH radical scavenging activities were calculated as inhibition percentages. IC50 values were also presented to express the potential of the samples to scavenge 50% of DPPH present in the reaction medium. Smaller IC50 values mean high antioxidant activity. Although IC50 of the HPL extract was higher than gallic acid, Trolox, and SA, which was an expected result since the extract was a complex medium with respect to the standard antioxidant compounds. Figure 1 depicts the DPPH radical scavenging activities of HPL and standard antioxidants. Maximum DPPH scavenging activities and IC50 values of HPL and gallic acid, Trolox, and SA can be seen in Table 2.
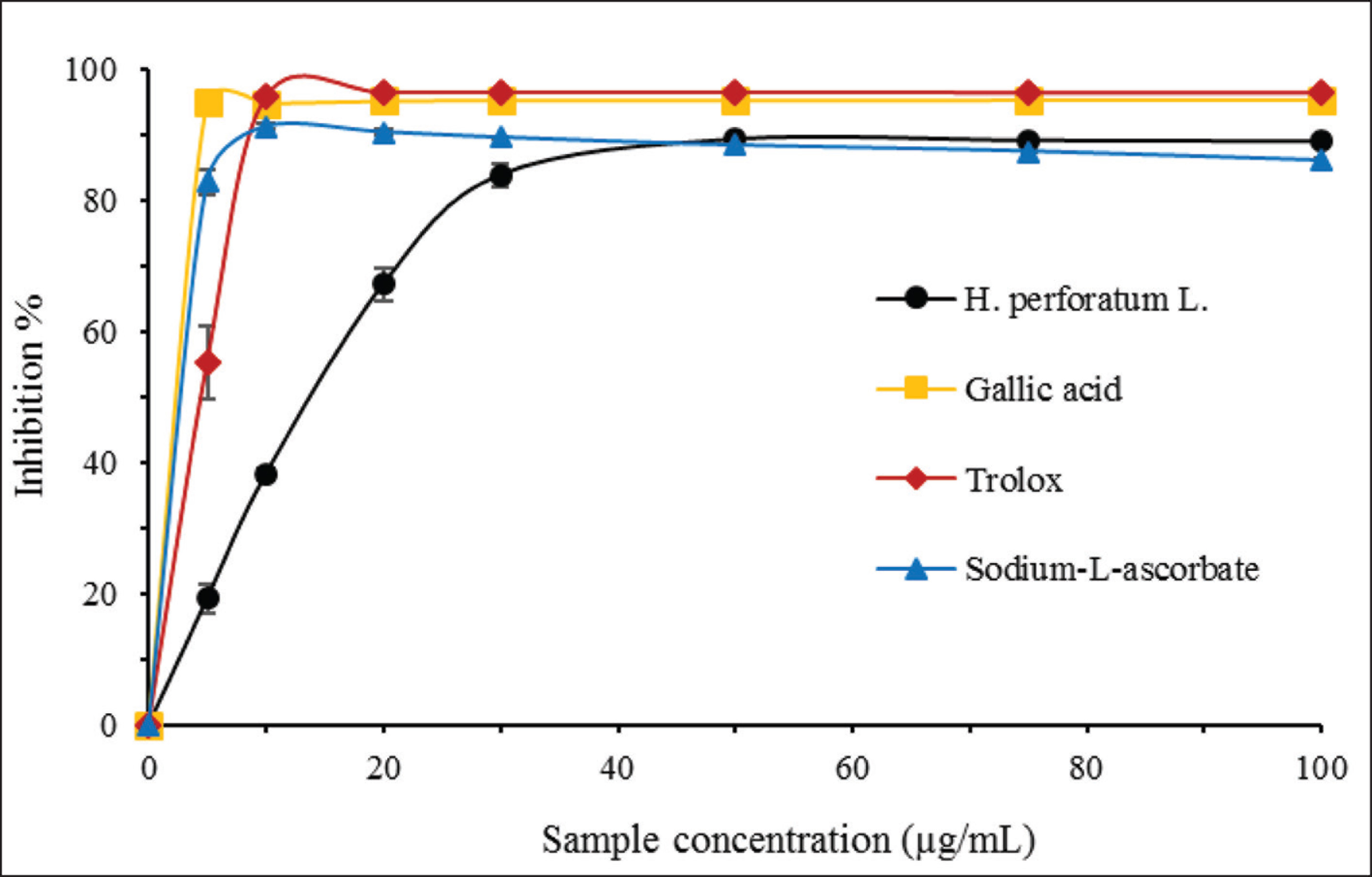
Maximum DPPH Scavenging Activities and IC50 Values of Hypericum perforatum L. and Standard Antioxidants (n = 3)
Phenolic compounds of HPL were extracted into distilled water. The quantification of phenolics was utilized by the Folin–Ciocalteu method.15,16 Results showed that 1 g extract contained 104.0 ± 1.740 mg GAE phenolic compounds.
Shear Bond Strength Results
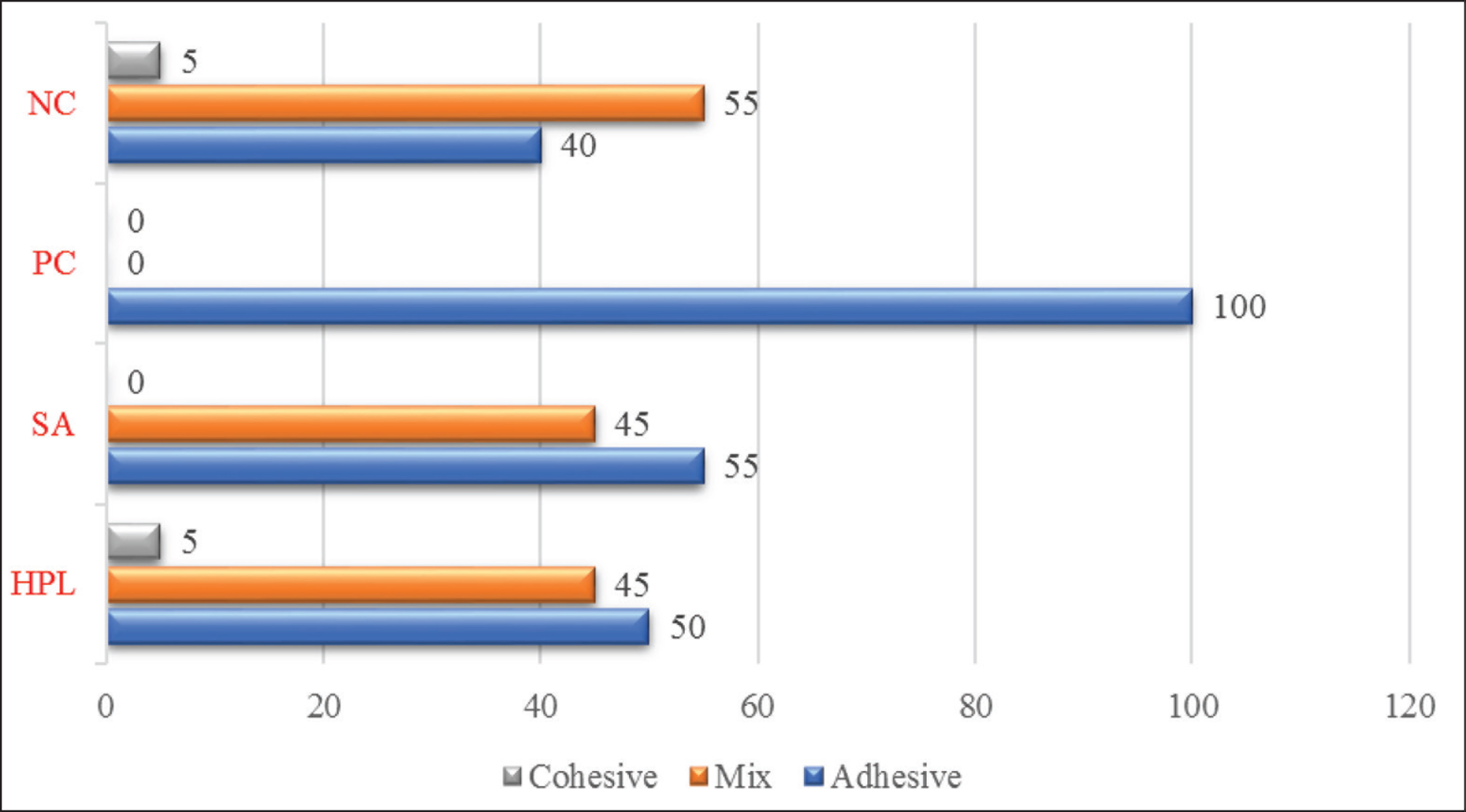
Table 3 shows the minimum, maximum, mean SBS values, and standard deviations in MPa. ANOVA revealed that statistically significant difference existed between at least two of the groups. The highest and the lowest statistically significant difference values mean SBS values were observed in NC and PC, respectively (P < .05). Pairwise comparisons revealed that the difference between SA and HPL was not statistically significant (P > .05). Figure 2 depicts the percentages of failure modes observed in the groups.
Minimum, Maximum, Mean Shear Bond Strength Values and Standard Deviations in MPa
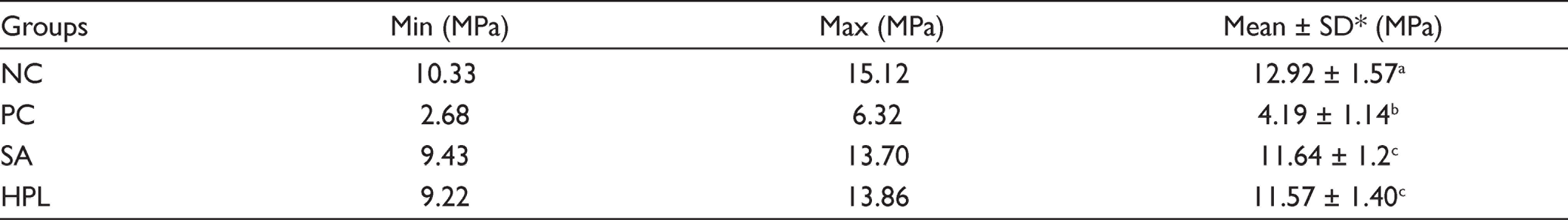
*Different superscript lowercase letters in the same column indicate statistically significant differences (P < .05).
Abbreviations: NC = negative control; PC = positive control; SA = sodium-L-ascorbate; HPL = Hypericum perforatum L.
Discussion
In the current study, the statistically highest SBS values were observed in the NC group and the lowest SBS values were observed in the PC group (P < .05). All specimens in PC exhibited adhesive failure. These findings indicated that bleaching treatment compromised the resin–dentin bond strength. Deterioration of the immediate resin–dentin bond strength was probably due to free radicals remained within the bleached dentin structure, which inhibits resin polymerization and compromises bonding, as previously reported.4-7
Numerous substrates either synthetic or natural (plant derived) have been tested regarding their free-radical scavenging/antioxidant efficacy against the free radicals exerted within bleached tooth structures. However, there still lacks a clinically adapted antioxidant treatment modality for oxidized tooth structures. SA has been the most widely investigated synthetic antioxidant in vitro. 10 The therapeutic antioxidant effect of 10% SA on bleached dentin has been demonstrated4,20-23; yet, some controversial results7,24 exist as well, depending on the experimental design variations among the studies. In the present study, SA was capable of reversing the compromised resin bonding after bleaching by scavenging free radicals remained within bleached dentin structure, which is in line with the previous research.4,20-23
Whether for vital or nonvital teeth, in-office bleaching techniques are applied chairside, and a clinically feasible treatment duration would be required for subsequent antioxidant application. Several investigators examined relatively prolonged durations for the application of 10% SA on bleached dentin, such as 3 hours 23 or 40 hours, 20 reflecting a time period which is equal to one-third of the bleaching duration, 25 whereas others reported that a duration as short as 2 minutes 4 or 10 minutes 22 would be satisfactory for the application of SA, provided to be applied in an active manner and the antioxidant solution was kept refreshing continuously. This has been also demonstrated in the current study. Cortez et al. 26 examined three different protocols for the application of SA on dentin bleached with 35% hydrogen peroxide and reported that active application modes yielded better results. However, application durations were not notified in the relevant study. Park et al. 9 indicated that in case the passive application strategy was used, the penetration of SA into the bleached tooth tissues would occur slowly and take time due to simple diffusion. In the current study, both experimental antioxidant solutions, SA and HPL, were applied on bleached dentin in an active manner, and the solutions were refreshed during the application. Both antioxidant treatments applied in SA and HPL could manage to improve immediate resin bond strength to bleached dentin (P < .05), and a lower percentage of adhesive failure was detected in those groups than that of the PC group. No significant difference was observed between SA and HPL, which means natural HPL was as effective as synthetic SA in terms of improving the compromised bond strength to bleached dentin (P > .05). Therefore, the null hypothesis tested in the study has been accepted.
Medicinal plants have been investigated extensively in biomedical and pharmaceutical research since they constitute natural sources of antioxidant compounds acting as free-radical scavengers and less toxic than synthetic antioxidants such as hydroxyanisole or butylated hydroxytoluene.14,27
In an attempt to examine the antioxidant effect of some plant-based extracts on bleached teeth, several investigators17,28,29 created experimental antioxidant treatment protocols in their study, using the over-the-counter (OTC) herbal and dietary supplements, such as grape seed extract, Aloe vera, green tea extract, pomegranate peel, or pine bark extract. However, as a drawback, the purity of capsule ingredients in herbal supplements may be highly suspicious. Some OTC herbal and dietary supplements were reported to contain hazardous additives such as pesticides, heavy metals, mycotoxins, and adulterants, 30 which may lead to great uncertainty regarding the purity of the natural plant ingredients in the supplements. Booker et al., 31 after analyzing 47 commercially available St. John’s wort (Hypericum perforatum) supplements, reported that significant compositional variation and adulteration were detected among the products, due to the incorporation of incorrect species or food dyes. The purity of a tested plant in a laboratory study is important to provide consistent evidence, accuracy, and reproducibility of the scientific research. 32 Therefore, the accurate botanical characterization and the purity verification of the plant(s) are necessary in the early stages of an experimental investigation. 32 In the current study, contrary to other investigations, taxonomical identification and the purity verification of HPL were provided prior to the plant extraction procedures.
The antioxidant activity of the Hypericum perforatum extract is mainly attributed to its rich content of phenolic compounds.13,33,34 Quantity and variety of phenolic compounds in plants or plant products are generally related to their antioxidant effectiveness. Phenolic compounds are plant secondary metabolites bearing one or more aromatic rings substituted with a hydroxyl group(s). Phenolic compounds are generated by plant itself in order to facilitate growing under unfavorable conditions as a defense mechanism, and they constitute the largest phytochemical molecules in plants. 27 Research has shown that the plant material may contain various bioactive contents at different amounts according to the localization, different floral development stages, and to the part of the plant examined. 35 Several classes of phenolic compounds such as flavonoids, phenolic acids, naphthodianthrones, and phloroglucinols have been identified in the Hypericum perforatum plant extracts.34,36,37 Among those phenolics, flavonoids (quercetin, kaempferol glycosides, aglycones, and biflavonoids) and phenolic acids (caffeoylquinic acids) have been specifically linked to the antioxidant properties of the plant.13,33,34 In the current study, the amount of phenolic compounds was determined as the 104.0 ± 1.740 mg GAE/g dry HPL extract.
To date, few studies12,19 examined the antioxidant activity of potential antioxidant materials to be applied to tooth structures by using DPPH radical scavenging activity analysis. DPPH is a stable free radical used to evaluate the radical scavenging abilities of antioxidant compounds for more than 50 years. This spectrophotometric assay is based on the DPPH radical quenching reaction that proceeds by the hydrogen atom or electron transfer between the radical and the antioxidant.12,16,18,19 In this study, at concentrations higher than the 50 μg/mL HPL extract’s scavenging percent was very close to the standard antioxidant compounds. Similar to our results, Gioti et al. 38 reported that both shoot and flower extracts of HPL scavenged approximately 100% of the DPPH radical present in the test medium. Researchers 38 also stated that phloroglucinols, naphthodianthrones, phenolic acids, and flavonoids were the main constituents in HPL. Flavonoids are well known for their antioxidant properties. Silva et al. 33 reported that in Hypericum perforatum, quercetin and its glycoside derivatives and hydroxycinnamates represent 57 and 10% of total phenolics, respectively. Since these compounds bear a catechol moiety in the ring, 2,3-double bond in conjunction with a 4-oxo group in the C-ring and hydroxyl moieties at 3 and 5 positions, these structural aspects allow them to maintain high antioxidant activity. Flavonoids in Hypericum perforatum species were detected by several researchers as kaempferol, quercetin, luteolin, catechin, rutin glycosides and quercetin glycosides.33,39
To the best of our knowledge, this is the first study that revealed the therapeutic antioxidant efficacy of an experimental protocol derived from natural HPL on an oxidized tooth structure. Nevertheless, we would like to highlight that the results of this in vitro research do not propose the direct application of the HPL extract-derived antioxidant solution on newly bleached teeth, since it would be unsuitable in the clinical setting due to the chromatic characteristics of the plant extracts. Preliminary researches as such offer a suitable method for investigating the therapeutic potential of the antioxidant compounds within the composition of plant extracts and construct a scientific basis for further laboratory studies. Among several classes of phenolic compounds identified in HPL, the most potent bioactive compound(s) in scavenging free radicals within oxidized dental structures remain(s) unclear yet and need(s) to be clarified.
HPL has a great number of medicinal applications, owing to its broad-spectrum biological activities such as antibacterial, antiviral, antifungal, anti-inflammatory, wound healing, analgesic, and antidepressant.39,40 This research has only focused on the antioxidant efficacy of HPL on a tooth structure; yet, HPL has additional therapeutic benefits that make it a promising candidate for use in the oral environment. In a recent study, Suntar et al. 40 examined the antimicrobial effect of the HPL extract against oral bacteria and biofilm formation and suggested that HPL could be used as a natural antibacterial agent in oral care products. It is an undeniable fact that many medications have been developed from the active ingredients of medicinal plants. 32 In this manner, further research on HPL extract would contribute to the development of a chemically stable product with predictable and reproducible therapeutic results for use in everyday dental practice.
We tested only a single experimental antioxidant protocol and evaluated only the immediate resin bond strength to the dentin bleached with a single bleaching technique (walking bleaching). Hence, further investigation is required to reveal the long-term bonding efficacy of various HPL-derived antioxidant treatment protocols on improving adhesion to enamel/dentin oxidized due to various vital/nonvital dental bleaching treatments, ozone therapy, or sodium hypochlorite irrigation.
Future Scope and Clinical Significance
Considering the limitations of this study, the experimental antioxidant treatment protocol prepared from the HPL extract at 10% concentration was statistically as effective as 10% SA in terms of reversing the compromised resin bond strength to bleached dentin.
The HPL extract seems to be a promising potent antioxidant source and a natural alternative to synthetic SA and might facilitate immediate adhesive procedures on oxidized dental structures. Further research is needed to generate a chemically stable antioxidant product derived from HPL for use in dental practice.
Footnotes
Acknowledgements
The authors would like to thank Dr Ozkan Eren of the Department of Biology at Aydin Adnan Menderes University for the botanical identification of the Hypericum perforatum L. plant and Kursat Ozkan for assistance with statistical analysis.
Author Contributions
Details on study conception: NAY, RY, and AAK
Data collection, data acquisition and analysis: NAY, RY, and AAK
Data interpretation: NAY, RY, and AAK
Manuscript writing: NAY, RY, and AAK
All the authors approved the final version of the manuscript for publication.
Data Availability Statement
The data set used in the current study is available within the article or its supplementary materials.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Policy and Institutional Review Board Statement
Ethics approval was not applicable for this study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Aydin Adnan Menderes University Scientific Research Project under grant ADU-BAP DHF-17004.
