Abstract
Aim:
To evaluate and compare the effect of curcumin (CUR) and Nano-curcumin (N-CUR) on human-derived mesenchymal stem cells (MSCs) in a dose-dependent manner.
Materials and Methods:
An experimental study performed with putative MSCs from a total of five systemically healthy subjects with chronic periodontitis. These putative MSCs were isolated by cell culture and were further characterized and identified by colony-forming unit assay and immunocytochemical analysis using cell surface markers CD105, CD146, CD45 and CD73. The identified MSCs were treated with different doses of CUR and N-CUR, and compared with α-minimum essential medium (α-MEM) for its cell viability by performing MTT (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyl tetrazolium bromide) assay for 48 and 72 hr. The statistically analysis was performed using one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test and Bonferroni’s post hoc test.
Results:
Compared to the α-MEM group, both CUR and N-CUR treated cells have shown significantly (P = .029) higher survival rate at lower concentration (0.1 and 0.5 µM/L), at 48 hr incubation. However, there was no statistically significant difference between the CUR and N-CUR groups on cell survival rate at both 48 and 72 hr incubation. When compared between the concentrations of the same group, significantly higher cell viability (P = .001) was observed at lower concentrations (0.1, 0.5 µM/L) in both test groups after incubation for 48 and 72 hr.
Conclusion:
Both CUR and N-CUR have a dose-dependent effect on human derived MSCs survival when incubated for 48 hr, whereas N-CUR shows increased cell survival rate even at 72 hr of incubation. Although, the cautious use of CUR and N-CUR at higher concentrations is recommended.
List of Abbreviations
MSC: Mesenchymal stem cells
CUR: Curcumin
N-CUR: Nano-curcumin
α-MEM: α-Minimum essential medium
MTT: (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyl tetrazolium bromide)
DMSO: Dimethyl sulfoxide
CD: Cell differentiation
BMSCs: Bone marrow derived stem cells
GtMSCs: Granulation tissue derived mesenchymal stem cells
Introduction
Periodontitis is currently considered as one of the main reasons for tooth loss because of its chronic inflammatory nature affecting the supporting tissues of the teeth, resulting in destruction of periodontal ligament and alveolar bone. 1 Various regenerative procedures were reported for the treatment of periodontal defects using bone grafts, membranes, enamel matrix proteins, growth factors and platelet concentrates which were successful in better wound healing and repair of the tissues. But the complete regeneration of lost tooth structures and restoring function are still a debate. 2
With the advances in cellular and molecular biology, application of stem cells for regeneration has been a novel therapeutic strategy which is now the focus of attention for many researchers.3,4 The concept that stem cells-like populations reside in the periodontal tissues which can virtually give rise to all the lost periodontal structures was proposed in the early 1970s and 1980s with the tremendous work carried out by various researchers.5–8
Stem cells are cellular sources for regeneration having the capability to self-renew and maintain their properties for a long period of time and can potentially differentiate into different cell lineages. 9 Although having numerous sources of stem cells, dental stem cells are gaining interest from research point of view because of the ethical and feasibility issues while obtaining embryonic and bone marrow derived mesenchymal stem cells (MSCs). 10 Amongst various dental sources, stem cells from periodontal ligament 11 and dental pulp 12 have been extensively studied for their therapeutic potential in the field of dentistry. The sources of these cells are usually from impacted molars, extracted deciduous teeth or premolars for orthodontic treatment. However, these sources are not always available when needed in individuals in their late 20s and 30s. Because of these reasons, newer sources which can be easily accessible have been sought out. In search of newer sources, researchers found the presence of MSC-like population in the granulation tissue which is considered to be a medical waste during various surgical procedures.13,14 The cells obtained from granulation tissue have shown cementum-like, periodontal-ligament-like structures when transplanted in vivo into nude mice models.14,15
Since the last few decades, herbal science has been extensively researched for its therapeutic role in the field of medicine and dentistry. Curcumin (CUR) is one of the mostly studied herbs, because of its medicinal properties such as anti-cancer, anti-inflammatory, anti-microbial, anti-aging and anti-fungal properties. 16
CUR, a natural polyphenolic phyto constituent used as one of the dietary spices in day-to-day life is derived from plant Curcuma longa (turmeric), belongs to Zingiberaceae family. 16 Among all other phytochemicals present in turmeric, CUR accounts for 2%–5% and is responsible for the yellow pigmentation and therapeutic effects. 16 CUR has shown promising results when treated on MSCs for differentiation into mesoderm lineages 17 and also showed synergistic effect when used for the treatment of experimental spinal cord injury. 18 Because of its multiple beneficial properties, CUR has also been largely studied in the treatment of chronic periodontitis for reducing plaque scores, bleeding scores and probing pocket depths (PPDs) when compared to the other delivery agents.19–21
CUR, in its traditional form, comes with certain shortcomings such as poor stability, poor bioavailability and pharmacokinetics, and low penetration and targeting efficiency. 22 With the advances in the field of nanotechnology and nano-preparations, these issues with traditional form of herbs are diminishing.22,23 Various nano-formulations of CUR such as liposomes, nanogels, nano-particles and polymer conjugates have improved the physico-chemical and biological properties of CUR. Because of these enhanced properties, the therapeutic potential of nano-curcumin (N-CUR) has tremendously improved in targeting cancer cells and its use as drug delivery systems. The efficacy of these nano-formulations depends on the size, shape, surface charge and the composition of the nano-particles along with its loading and release properties. However, in spite of the numerous advantageous properties of CUR on MSCs, its dose-dependent effects on survival of human-derived MSCs are less searched.
Among various cell viability assays, MTT is a rapid calorimetric assay used to measure the cellular metabolic activity as an indicator of cell viability, proliferation and cytotoxicity. It is based on the reduction of MTT (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide), a yellow water-soluble tetrazolium dye into water insoluble purple colored formazan crystals by the mitochondrial dehydrogenases present in the living cells. The intracellular insoluble formazan crystals are then dissolved in dimethyl sulfoxide (DMSO) solution and the resultant coloured solution is quantified by spectrophoto- meter. The darker the solution, the higher will be the number of viable and metabolically active cells. 24 The aim of this study is to evaluate and compare the dose-dependent potential role of CUR and N-CUR on survival of stem cells derived from granulation tissue obtained during the routine periodontal flap surgery.
Materials and Methods
Setting and Design
This study was an ex vivo study carried out in Maratha Mandal’s Nathajirao G. Halgekar Institute of Dental Sciences and Research Centre, Belagavi, Karnataka. A small number of subjects were included based on fea- sibility as a part of the preliminary study. A total of five systemically healthy subjects with chronic periodontitis, who reported to the Department of Periodontology, Maratha Mandal’s Nathajirao G. Halgekar Institute of Dental Sciences and Research Centre, Belagavi, Karnataka, were selected with inclusion and exclusion criteria.
Subjects of age ranging from 20–60 years, having moderate to severe chronic periodontitis with the presence of bleeding on probing (BOP), PPD of ≥5 mm and clinical attachment loss of ≥3 mm, and at least one intra bony defect were included. Simultaneously subjects with systemic con- ditions, habit of smoking or alcoholism, periodontal treat- ment within last 6 months, pregnant women and lactating mothers were excluded from the study.
Collection of the Sample
All the subjects underwent through scaling and root planning with ultrasonic tips and curettes one week prior to the surgery. After administering local anaesthesia (XICAINE) of 1:80,000 2% adrenaline, a full thickness mucoperiosteal flap was raised. The unattached necrotic granulation tissue was removed and the attached granula- tion tissue from the base of the pocket was scraped with the help of curettes for investigations. The tissue was then transferred into α-minimum
Cell Culture
The samples collected were sent to the Central Research Laboratory at Maratha Mandal’s Nathajirao G. Halgekar Institute of Dental Sciences and Research Centre, Belagavi, Karnataka. The collected samples were processed by first treating with Betadine (WIN-MEDICARE Pvt. Ltd, Uttarakhand, India) for 1 min followed by washing with phosphate buffer saline (HiMedia Laboratories Pvt. Ltd, Mumbai, India) for three times. The tissues were then min- ced with scalpel in a culture plate with basal media; α-MEM (Gibco, USA) with 10% fetal bovine serum (HiMedia Laboratories Pvt. Ltd, Mumbai, India), 0.29 µg/ml glutamine, 100U/ml penicillin and 100 mg/ml streptomycin (HiMedia Laboratories Pvt. Ltd, Mumbai, India). The minced tissues were then transferred to a 6 well culture plate with basal media and incubated at 37 ºC with 5% O2. The media was then refreshed every 3 days until sub-confluence was seen. The passages of the subculture were used for further analysis. 25
Colony-Forming Unit (CFU) Fibroblast Assay
The cells of passages less than P5 were placed in 10 cm diameter culture dishes with basal medium at a density of 1×103 /well and were cultured for 2 weeks. The cells were then fixed with 4% paraformaldehyde and stained with 1% toluidine blue (HiMedia Laboratories Pvt. Ltd, Mumbai, India). A group 50 or more cells were obtained and viewed under the microscope and scored as CFU. 25
Immunocytochemical Analysis
The cells of passages less than P5 were sub-cultured into chamber slides and incubated for a week and then fixed with 4% paraformaldehyde (HiMedia Laboratories Pvt. Ltd, Mumbai, India) for 30 min. The fixed tissues were then blocked with blocking solution and the chamber slides were then kept in hot water bath. The slides were then subjected to primary antibodies CD146, CD105, CD45 and CD73 (PathnSitu Biotechnologies Pvt. Ltd, Telangana, India) based on the manufacturer’s instructions. The stained cells were then observed under inverted light microscope.
Preparation of Nano-curcumin
Initially, chitosan solution (0.2 % w/v) was prepared by dissolving in dilute acetic acid (2 % v/v) and the pH was adjusted to 5.0 using 4M NaOH. This solution was stirred at 600 rpm on a magnetic stirrer and ethanolic solution of pure CUR (purity >95% total curcuminoids; Kancor Ingredients Ltd, Kerala, India) was added drop-wise. Afterwards, sodium tripolyphosphate (HiMedia Laboratories Pvt. Ltd, Mumbai, India) (TPP, 0.1 % w/v) solution was added to the above mixture to achieve 3:1 mass ratios of chitosan to TPP. The suspension obtained was stirred further for 30 min at room temperature and centrifuged at 12857 × g at 20 °C for 20 min to pelletize the prepared nanoparticles. The prepared pellet was subsequently redispersed in deionised water and stored at 4 ºC. 26 The pure CUR (1 mM) was prepared by dissolving it in dimethyl sulphoxide DMSO (Sigma-Aldrich Pvt, Ltd, Bangalore, India) and diluted subsequently to obtain five different concentrations of 0.1, 0.5, 1, 5 and 10 µM/L.
MTT Assay
The cells of passages less than P5 were treated with different concentrations of pure CUR and N-CUR along with culture media and compared to α-MEM. These cells were incubated in a 96 well culture plate for 48 and 72 hr. After 48 and 72 hr of incubation, 20µL of 5 mg/ml MTT reagent was added to each well and incubated for 4 hr. The media was then replaced with 150 µL of DMSO and subjected to spectrophotometer for differences in absorbance at 48 and 72 hr for all the treated cells.
Statistical Analysis
The statistical analysis was performed by using IBM SPSS Statistics for windows, version 2.0 (IBM Corp., Armonk, NY, USA). One-way analysis of variance followed by Tukey’s post hoc test analysis was used for inter-group comparison. Additionally, a pair-wise Bonferroni’s post hoc test was performed for intra-group comparison. The P value of <.05 was considered statistically significant (the degree of freedom 2 with confidence interval of 95% was used).
Results
Isolation and Identification of Granulation Tissue MSCs (GtMSCs)
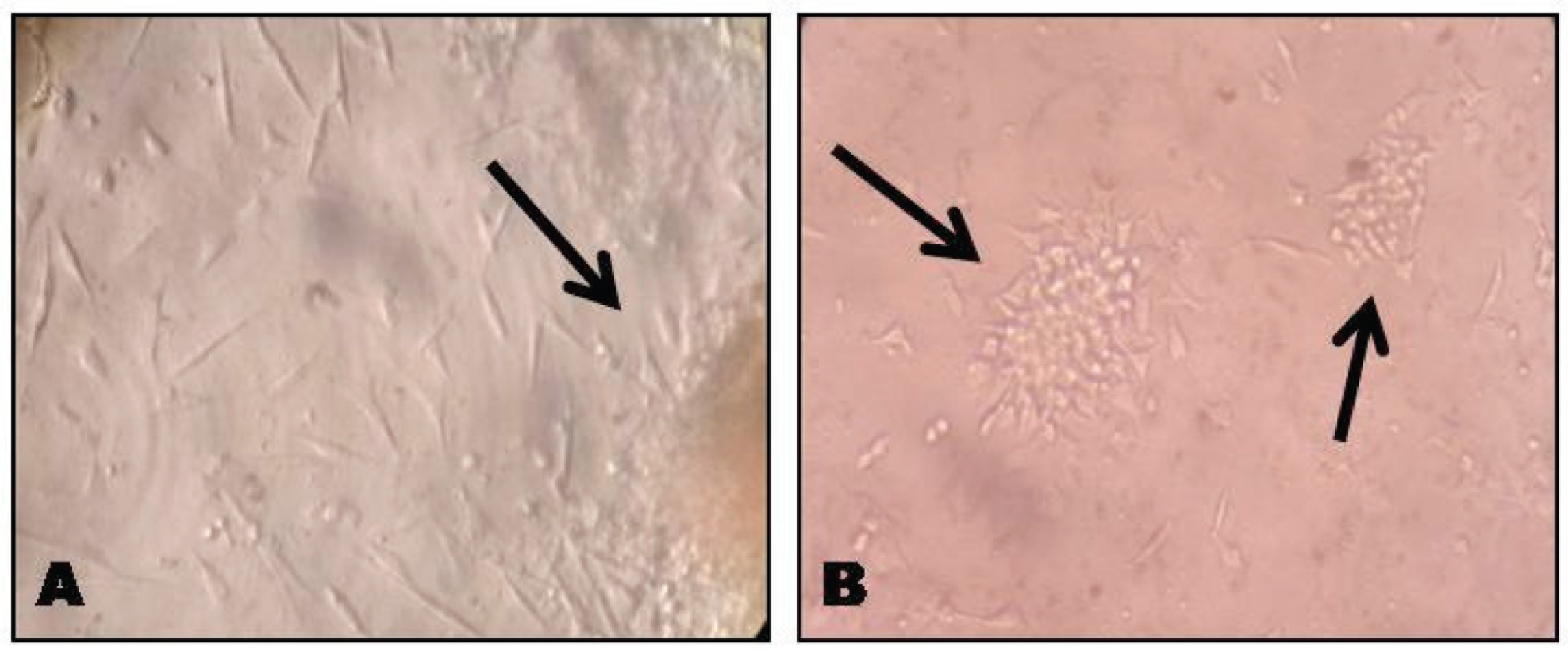
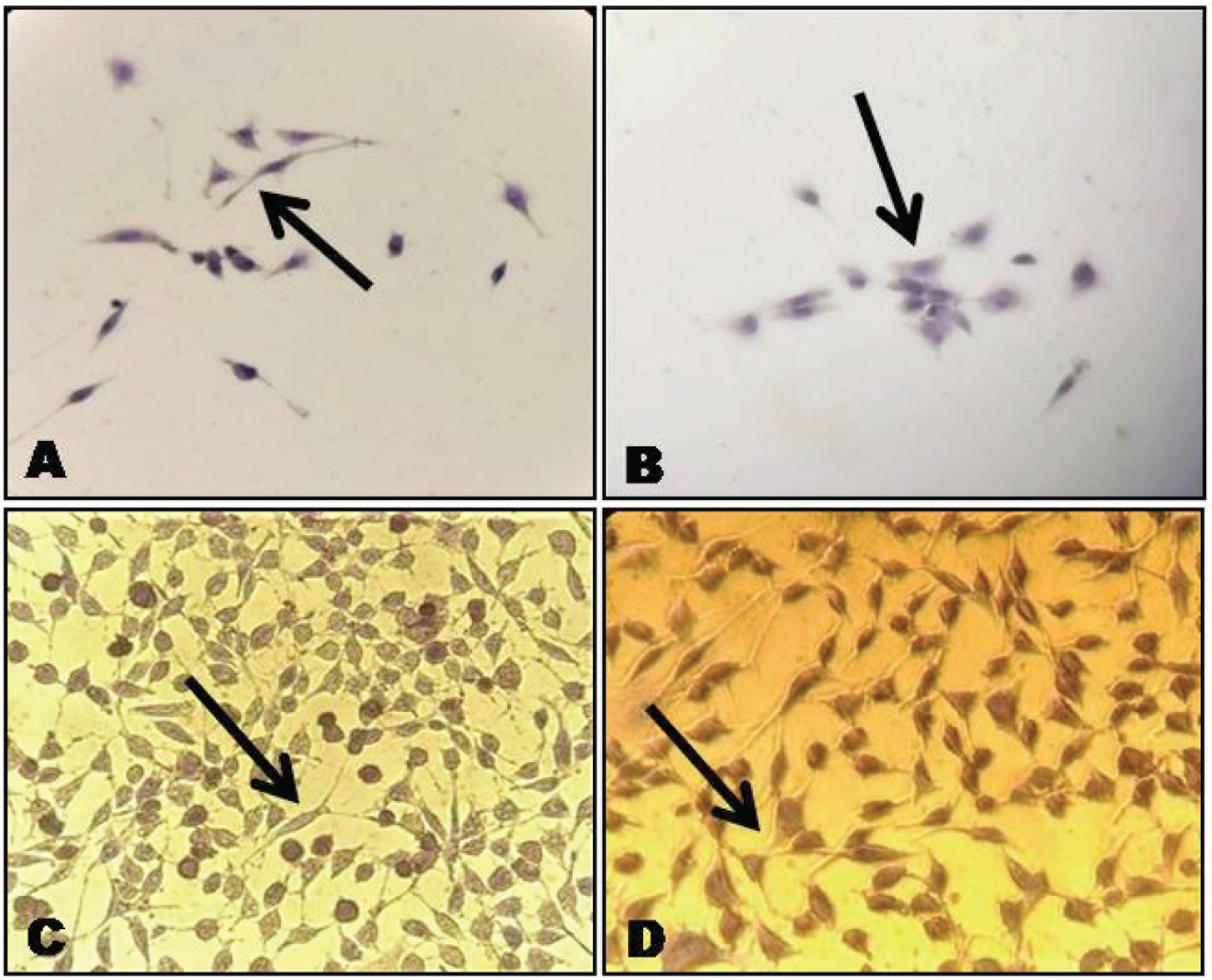
The primary cell culture after 7–10 days showed pronounced heterogenous population of cells with a predominance of elongated spindle-shaped cells adherent to culture plate, which organized them into small colonises after 14 days as shown in Figures 1(A) and (B). The obtained cells expressed MSC markers positive for CD105, CD146 and CD73, and negative for CD45 by immunocytochemical []analysis as shown in Figure 2 (A), (B), (C) and (D).
Microscopic Images of Cell Culture in Granulation Tissue, Under 20× Magnification (A) Colonies after one Week of Cell Culture (B) Colonies Formed after 14 Days
By Immunocytochemical Analysis: (A) CD146 Positive Cells Showing Brown Pigmentation of Cytoplasm (B) CD105 Positive Cells Showing Brown Pigmentation (C) CD73 Positive Cells Showing Brown Pigmentation (D) CD45 Negative Cells Showing Basophilic Stain
Preparation of N-CUR
The N-CUR was successfully prepared by ionic gelation method using chitosan polymer and the TPP as cross linker. The prepared N-CUR has a particle size of 167.3 ± 1.9, and zeta potential +18.1 mV and entrapment efficiency of 88.4 %. The drug release was 41.5 ± 1.1% at 24 hr time according to the published work of Nair et al.. 26
Effect of CUR and N-CUR on Survival of GtMSCs at 48 Hours
The results obtained from MTT assay showed that CUR and N-CUR at different concentrations enhanced the cell viability in a dose-dependent manner when compared to α-MEM when incubated for 48 hr. Both CUR and N-CUR at 0.1 and 0.5 µM/L concentrations showed increased cell survival of GtMSCs, whereas N-CUR has shown a significantly higher cell survival rate at 0.1 µM/L (P = .029) concentration when compared to the α-MEM treated cells as shown in Table 1. Further, there was also a decrease in viability of the cells in both CUR and N-CUR treated cells when compared to α-MEM treated cells at higher concentrations (Table 1). No significant difference was observed between CUR and N-CUR treated cells on the cell survival rate after 48 and 72 hr incubation. Subsequently, when compared between the concentrations of the same group, 0.1 µM/L in CUR (P = .003) and both 0.1 and 0.5 µM/L in N-CUR (P = .001) group showed significantly higher cell viability compared to the higher concentrations 5.0 and 10 µM/L.
Inter- and Intra-group Comparison of Mean MTT Assay Values for CUR, N-CUR and α-MEM at 48 Hours by Using One-Way ANOVA Followed by Tukey’s Post Hoc Test and Bonferroni’s Pair-Wise Analysis
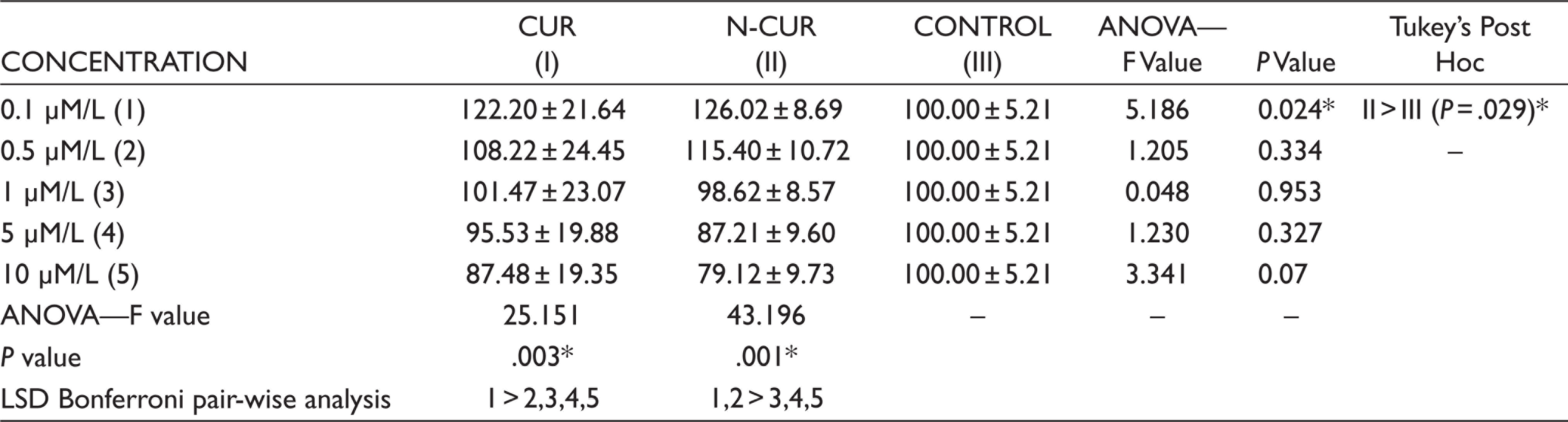
*P <.05 statistically significant, control—α-MEM.
> Statistically significant in relation to the variable.
= Statistically non-significant.
Effect of CUR and N-CUR on Survival of GtMSCs at 72 Hours
As shown in Table 2, the results of MTT assay reveals that N-CUR has increased the cell survival of GtMSCs at lower concentrations 0.1, 0.5 and 1.0 µM/L when compared to CUR and α-MEM treated cells after 72 hr of incubation, whereas the cell survival rate has considerably decreased at higher concentrations 5.0 and 10 µM/L for both CUR and N-CUR treated cells when compared to α-MEM treated cells. However, there was no statistically signif- icant difference between the groups. Additionally, when compared amongst the concentrations of the same group, 0.1 and 0.5 µM/L in CUR (P = .001) and 0.1 µM/L in N-CUR (P = .001) group have shown statistically significant increase in cell survival compared to higher concentrations as observed in Table 2.
Inter- and Intra-group Comparison of Mean MTT Assay Values for CUR, N-CUR and α-MEM at 72 Hours by Using One-Way ANOVA Followed by Tukey’s Post Hoc Test and Bonferroni’s Pair-Wise Analysis
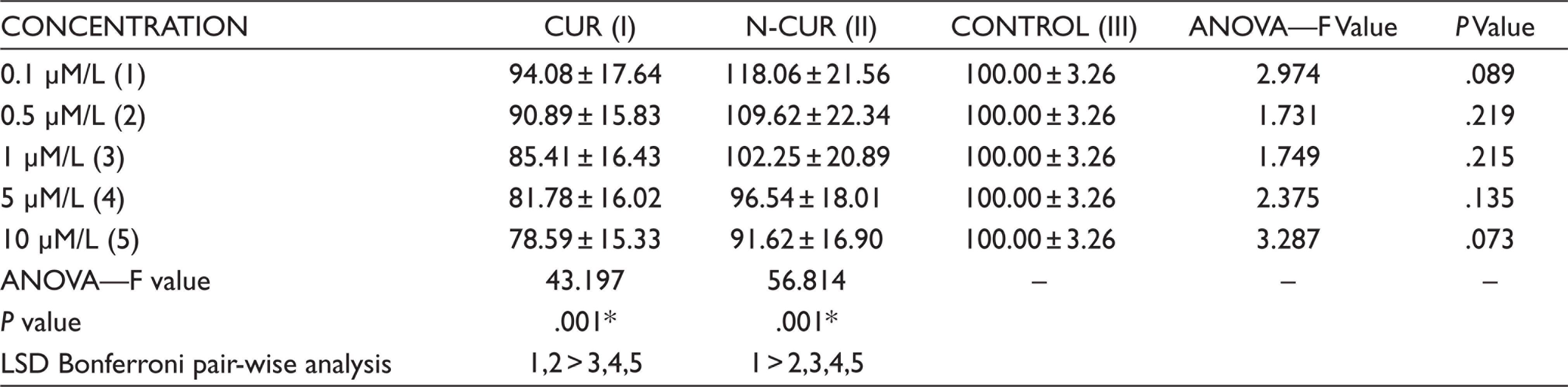
*P <.05 statistically significant, control—α-MEM.
>Statistically significant in relation to the variable.
= Statistically non-significant.
Discussion
Chronic periodontitis is considered to be one of the biggest threats to the oral health, which is more commonly seen in older individuals. 27 While treating these patients surgi- cally, the removal of granulation tissue from the diseased sites is of utmost importance. 28 This granulation tissue is considered to be a medical waste and had shown some amount of MSC-like populations which could be easily accessible and used as auto-transplant in individual’s subsequent surgical procedures. 14 So, this study was completely focused on the isolation of MSCs from the alternative sources, which would be available for all the individuals irrespective of their age, by means of granu- lation tissue collected during routine periodontal flap surgery. There are very few studies evaluating the different doses of CUR on rat BMSCs and neural stem cell for proliferation and cell viability.29–31 These studies concluded that specific dosage of CUR for a specific period of time influences the results, and they also found that CUR behaves as a double edged sword which can be even toxic at higher concentrations. However, there is no literature evidence on evaluating the effect of CUR and its nano preparations on human-derived MSCs. Therefore, we have also aimed to evaluate the effective role of CUR and N-CUR on these stem cells.
The cell culture results of our study showed that granulation tissue obtained possess a significant population of putative MSCs, which have expanded under particular experimental conditions. These cells also demonstrated their stem cell-like properties by adhering to the culture plate, forming colonies with spindle shaped cells and also expressed cell surface markers for MSCs CD146, CD73, CD45 and CD105 by immunocytochemical analysis.
In this study, we have also observed the multiple dose-dependent effects of CUR and N-CUR on isolated GtMSCs pertaining to its viability by MTT analysis. After exposing the different doses of CUR and N-CUR to GtMSCs, we found that lower doses (i.e., 0.1 and 0.5 µM/L) of both CUR and N-CUR were effective in increasing the cell survival compared to the α-MEM treated cells when incubated for 48 and 72 hr. On the other hand, the higher concentration (i.e., 5.0 and 10.0 µM/L) of CUR and N-CUR resulted in decreased cell viability of GtMSCs when compared to α-MEM. The results of our study were in association with the studies conducted by Attari et al., 29 Kim et al. 30 and Ormond et al. 31 who also stated the dose-dependent effect of CUR on proliferation and rate of survival of rat BMSCs and neural stem cells when compared to DMSO at 48 and 72 hr. To the best our knowledge, this was one of its kind studies evaluating and comparing the effect of CUR and N-CUR with α-MEM culture media on human-derived GtMSCs. The discrepancies observed among the studies can be attributed to the fact that our study was performed on human-derived MSCs, and the different doses of CUR were compared with α-MEM media. Futhermore, the granulation tissue obtained from different individuals of different age with different inflammatory conditions which would have impacted the results.
We also found that N-CUR has significantly increased the viability of GtMSCs at lower concentration, that is, 0.1 µM/L at 48 hr when compared to α-MEM treated cells, whereas N-CUR has also consistently increased cell viability even after 72 hours when compared to CUR and α-MEM treated cells, although the results were statistically insignificant. These findings explain the effectiveness of both CUR and N-CUR over α-MEM culture media and increased availability of the drug in nano-preparations even at 72 hours. Nevertheless, the properties of N-CUR such as loading and release kinetics of the drug determine the efficacy. Further detailed analytical in vitro studies on N-CUR are essential to understand the results better.
Additionally, when compared among the concentrations, considerably 0.1 and 0.5 µM/L concentrations have shown significant increase in cell viability in both CUR and N-CUR groups at both 48 and 72 hr. This observation was also in relation to the previously mention studies29–31 where CUR at concentration 0.1 and 0.5 µM/L has shown increased proliferation and cell viability of BMSCs and neural stem cells. This suggests the safe use of CUR and its nano-preparations at lower concentrations to increase the viability of MSCs and substantial toxic nature of compounds at higher concentrations.
Different synthetic and semi-biological substances such as recombinant cytokines and growth factors were used to increase the rate of proliferation and differentiation of MSCs. These agents also come with certain drawbacks and adverse effects when treated with the stem cells, which make the use of these agents questionable. Various herbal extract mixtures or individual compounds were extensively studied on stem cells and proven to show increased proliferation and differentiation properties with less toxicity. The increasing research on herbal medicine in stem cell therapy offers the field of tissue engineering with alternative sources for synthetic materials, with less expense, minimal toxicity and reliable results. 32
Research proves that properties of stem cells decrease with aging of an individual, 33 and stem cells require an antioxidant mechanism to survive and repair cells. As periodontitis is usually seen in elderly patients, curcumin with its antioxidant properties become a potential candidate to be applied in stem cell therapy research. Stem cells treated with curcumin and its nanoparticles show better proliferation potential at 48 and 72 hrs of incubation compared to stem cells treated without curcumin.
Conclusion
Within the limits of our study, we found that granulation tissue from periodontal flap surgery could be a relatively accessible source of MSCs. And from the results of our study, it is clear that both CUR and N-CUR play a beneficial role in increasing the cell viability of human MSCs in a dose-dependent manner. As our results also suggest the toxic nature of the compound at higher concentrations, it should be used more cautiously.
Future Scope
A relatively smaller sample size of five subjects with different age groups and with disease activity possess limitations to the study. However, the results of our study furnish an appropriate base for future extensive research with larger sample size and age-matched subjects, and detailed investigation on nano-preparations with clinical trials would lay out a better clinical therapy to treat periodontitis. Further research has to be focused on different biochemical aspects of CUR on granulation tissue-derived MSCs.
Footnotes
Acknowledgements
We acknowledge the support received from Central Research Laboratory Maratha Mandal Dental College team especially from Dr Chetana (BDS), Ms Geetha and the other research officers.
Author Contributions
P Hameeda: Details on study conception, data collection, manuscript writing. Sandeep Katti: Design of the work and details on study conception. Rajkishore Jammalamadugu: Data acquisition and analysis and interpretation. Kishore G Bhat: Made a substantial contribution to the concept or design of the work. Malleswara Rao: Drafted the article and revised it critically for important intellectual content. Vijay Kumbar: Data interpretation.
Data Availability Statement
Available on request from Dr Hameeda (e-mail id:
Ethical Policy and Institutional Review Board Statement
The study protocol was approved by Institutional Ethical Board Committee for relevance and feasibility of the study (1478).
Patient Declaration of Consent
All the subjects selected were explained about the study and a written informed consent was obtained from all the participants.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
