Abstract
Aim:
Polymethyl methacrylate is the common material used as a denture base. Ease of application, stability in the oral environment are its advantages; however, its mechanical properties should be enhanced. This study aimed to evaluate the effect of different ratios of polypropylene fiber (PPF) in addition to denture base materials on impact strength, water sorption, and cytotoxicity.
Materials and Methods:
Heat-cure acrylic resin specimens were prepared according to the manufacturer’s instruction by adding PPFs of 6 mm length in different ratios (1, 3, 5, 10, and 20 wt%). In order to determine the impact strength, specimens were subjected to a Charpy impact test machine after being kept in distilled water at 37°C for 48 h. A span of 40 mm was adjusted and a 0.5 J pendulum was used. The fractured surface of specimens was also analyzed using a scanning electron microscope. In addition, mouse fibroblast cells and agar diffusion tests were used for cytotoxicity determination. The results were analyzed using the Kruskal–Wallis and the Mann–Whitney
Results:
5 wt% PPF group exhibited the highest water sorption and impact strength values, and the difference was statistically significant (
Conclusion:
Addition of PPFs in acrylic resin increased the impact strength and decreased water sorption without any cytotoxic effects.
Introduction
Polymethyl methacrylate (PMMA) denture base resin material has been widely used in prosthetic dentistry. It is the main material for the fabrication of dentures because of its simple processing technique, relatively low cost of fabrication process, and high aesthetic properties.1,2 However, despite these satisfying properties, poor mechanical properties including low fatigue resistance and low impact strength should be improved to obtain long-term clinical success.3,4 Within the 3 years of using prosthesis, the fracture rate of acrylic resin was determined at 68%, especially from the midline of the prosthesis under masticatory forces. 5
Different techniques have focused on incorporation of various reinforcing substances into acrylic resin matrix by adding copolymerization and cross-linking agents of monomers 6 and by adding filling materials including metal wires, plates, and fibers.4-10 Difficult manipulation, 8 unaesthetic property,6,7,10 minor positive influence on flexural fatigue resistance, 11 and poor adhesion to acrylic resin7,10 inhibit using metal wire reinforcement. In addition to the drawback of metal wires, metal plates require additional devices for the casting procedure. 8 Similarly, no significant improvement in strength properties was achieved by copolymerization and cross-linking monomers. 7 Therefore, the best way to improve the strength of the denture base acrylic resin seems to be addition of fibers. On the other hand, there are various types of fibers, such as carbon, glass, aramid, whisker, and polyethylene fibers,4,10 whose poor adhesion to acrylic resin, polishing difficulties, unacceptable aesthetic appearance,6,8,9 and mucosal irritation 12 made them unfavorable for clinical use. 9
Because of these disadvantages, researchers are focused on alternative reinforcing materials. One of them is polypropylene fiber (PPF) which consists of long-chain synthetic polymer composed of at least 85% by weight of propylene. 4 Lightweight fibers exhibit abrasion and chemical deterioration resistance. PPF is widely used in surgery because of low density, high strength, and high biocompatible. 13 In addition, they enable the natural looking and biocompatible strengthening properties of PMMA. 4 In the literature, limited studies focused on strengthening of PMMA with PPF.13,14 The influence of PPF addition into the PMMA on the physical properties of denture base resin is still exactly unknown. Therefore, the purpose of this study was to evaluate the effect of addition of different ratios of PPF into denture base materials on impact strength, water sorption, and cytotoxicity of denture base resin. The null hypotheses tested were: (a) impact strength of denture base resin is increased by addition of PPF, (b) water absorption of PMMA is independent of the addition of different ratios of PPF, and (c) PPF groups show higher cytotoxic effect than control group.
Materials and Methods
This study was approved by the Clinical Research Ethics Committee of Cumhuriyet University, Sivas (approval number 2020-03/16). A total of 186 PMMA specimens were used in the present study. Of these samples, 90 samples were prepared for impact resistance, 90 samples for water absorption and 6 samples for cytotoxicity. Samples of different sizes and shapes were prepared for the tests. The number of specimens to be studied was estimated on the basis of a power analysis that assumed a power of 90% and a significance level of 0.05. The number of samples was determined as 15 in each group.
Heat-cure acrylic resin (Meliodent; Bayer Dental, Newbury, UK) and no-surface treated PPF at different ratios (Dost Kimya, Istanbul, Turkey) with a length of 6 mm were used in this study.
Impact Strength Test
To obtain impact test specimens, wax patterns were prepared with a dimension of 60 × 7.5 × 4mm according to the ADA No:12 standards by using a stainless steel mold (Figure 1A). Different ratios of PPFs including 1%, 3%, 5%, 10%, and 20% by weight were added in to the PMMA denture base resin after they were soaked in a monomer for 10 min to ensure better bonding of fibers with the PMMA resin. Conventional polymerization procedures which were suggested by the manufacturer were performed for control and fiber groups and a total of 90 PMMA test specimens were fabricated (n:15). The size of the specimens was verified by measurements of 3 different sections with a digital caliper (Altas 905; Gedore-Altas, Istanbul, Turkey). Following to keeping specimens in distilled water at 37°C for 24 h, they were subjected to an impact test machine (Zwick HIT50P, Zwick Roell Group, Ulm, Germany). Impact strength test (Charpy) was carried out using a 0.5 J pendulum at a distance between 2 supporting points of 40 mm. The impact strengths of the specimens were calculated using the following formula:
where
After the specimens were broken, the fractured surfaces of specimens were examined by scanning electron microscopy (LEO 440; Zeiss, Oberkochen, Germany).
Water Sorption Test
The water sorption (Wsp) test specimens were prepared in accordance with ISO1567:1999 standards with a dimension of 10 × 10 × 2 mm (Figure 1B). Fabrication procedures were the same as impact strength test specimens. 90 PMMA test specimens were stored in a desiccator containing silica gel at 37 ± 2°C for 24 h; then, to obtain a constant mass (W1), the specimens were kept at room temperature for an hour. Prior to retrieving water storage, they were weighed using a precision scale (XB 220A, Precisa Gravimetrics AG, Dietikon, Switzerland). Immersion procedure was carried out in a water bath machine (BM302, Nuve, Ankara, Turkey) with distilled water at 37ºC for the following time intervals: 1 day, 1 week, and 1, 3, and 6 months.
After completing each storage duration, the specimens were removed from the water, dried with a clean towel to remove excess of visible surface liquid, and weighed again (W2). Wsp values of all samples were calculated using the following formula:
Test specimens. (A) Impact strength, (B) water sorption, and (C) cytotoxicity


The data were statistically analyzed by SPSS for Windows (Version14.0, SPSS Inc., Chicago, USA). Between groups, Kruskal–Wallis and Tukey’s range tests, in addition to Friedman and Wilcoxon tests, were utilized to identify the significant differences in each group (
Cytotoxicity Test
Wax models with standardized dimension (5 mm diameter and 1 mm thickness) in accordance with ISO 10993-5:1992 were used to fabricate heat-cure acrylic resin specimens according to the conventional denture manufacturing protocol (Figure 1C).
L929 fibroblast cells (mouse connective tissue, ATCC) were incubated in Dulbecco’s Modified Eagle Medium (Sigma, St. Louis, USA) supplemented with 10% fetal calf serum (Sigma, St. Louis, MO) and 2 mM/ml L-glutamine without adding antibiotics. The cells were incubated at 37°C in a 5% CO2 atmosphere for 7 days before performing the agar diffusion tests. Briefly, the cell cultures were harvested using 0.25% trypsin solution (Gibco, Germany) and were seeded in petri dishes (Nunc, Wiesbaden, Germany) at a density of 1 × l06. After the formation of a confluent cell layer, the medium was removed and replaced with complete medium containing 1.5% agarose (FMC BioProducts, Rockland, ME). After solidifying agarose, the cells were stained with a vital dye (neutral red; Sigma). In all phases of experiments, cells were protected from light to avoid cell damage caused by photoactivation of the stain. Test specimens were placed on the agar surface so that the bottom surface of each specimen was in contact with agar. A phenol-impregnated blotting paper was used as positive control and a Dulbecco’s Modified Eagle Medium-impregnated blotting paper as negative control. After an exposition period of 24 h at 37°C, the cell responses were evaluated by observing them under an inverted microscope.
In this study, cell lysis was scored as follows: 0, no cell lysis detectable; 1, less than 20% cell lysis; 2, 20% to 40% cell lysis; 3, 40% to 60% cell lysis; 4, 60% to 80% cell lysis; and 5, more than 80% cell lysis. For each sample, one score was given and the median score value for all parallels from each sample was calculated for the lysis zone. Cytotoxicity was then classified as follows: 0 to 0.5 = noncytotoxic; 0.6 to 1.9, mildly cytotoxic; 2.0 to 3.9, moderately cytotoxic; and 4.0 to 5.0, markedly cytotoxic. The median (instead of the mean) was calculated to describe the central tendency of the scores because the results were expressed as an index in a ranking scale.
Results
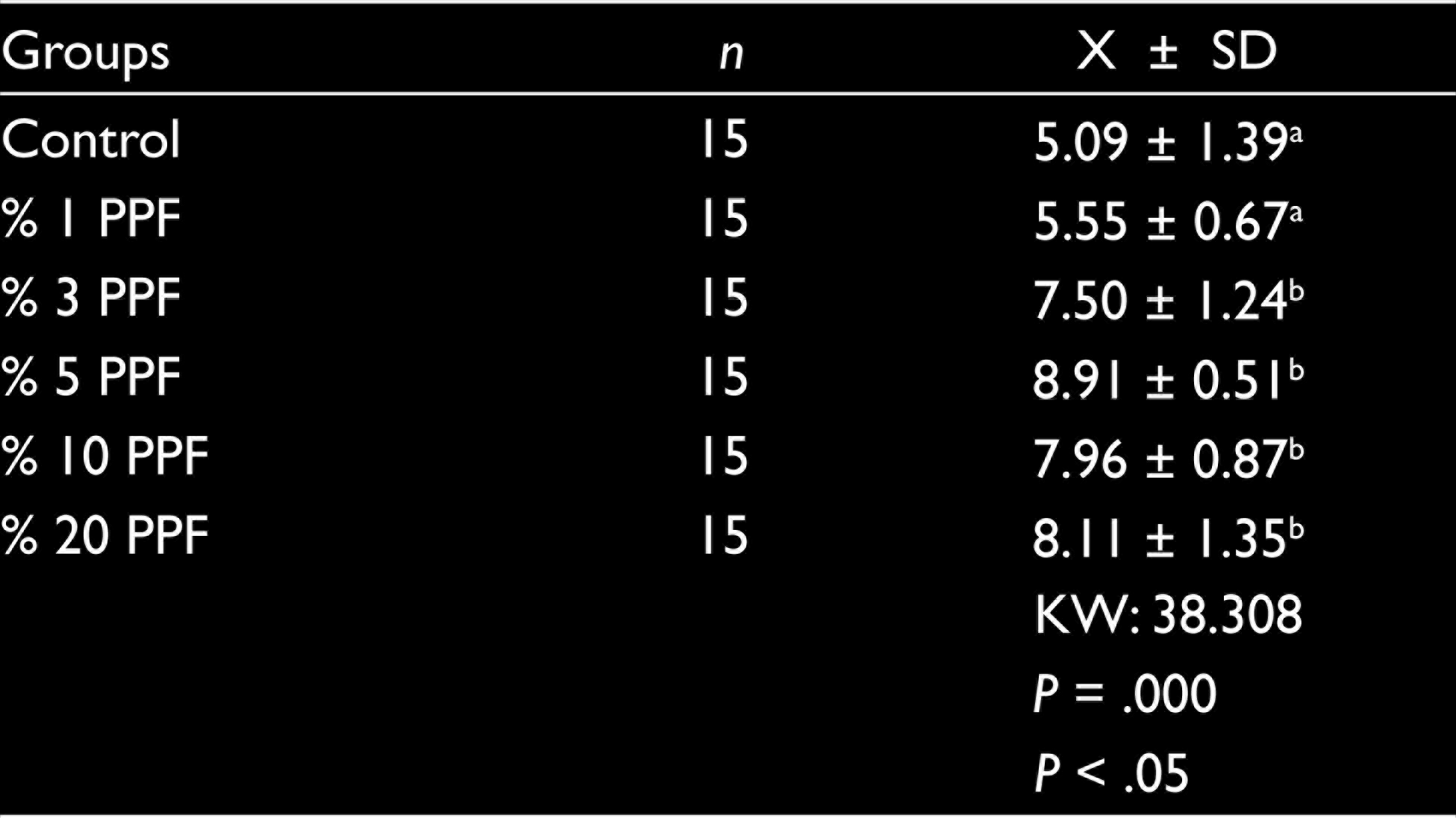
Impact strength values are depicted in Table 1. The 5% PPFs group exhibited the highest impact strength values. Control and 1% PPFs group of specimens presented same impact strength values (
Impact Strength (kJ/m2) of Denture Base Materials (Mean ± SD)
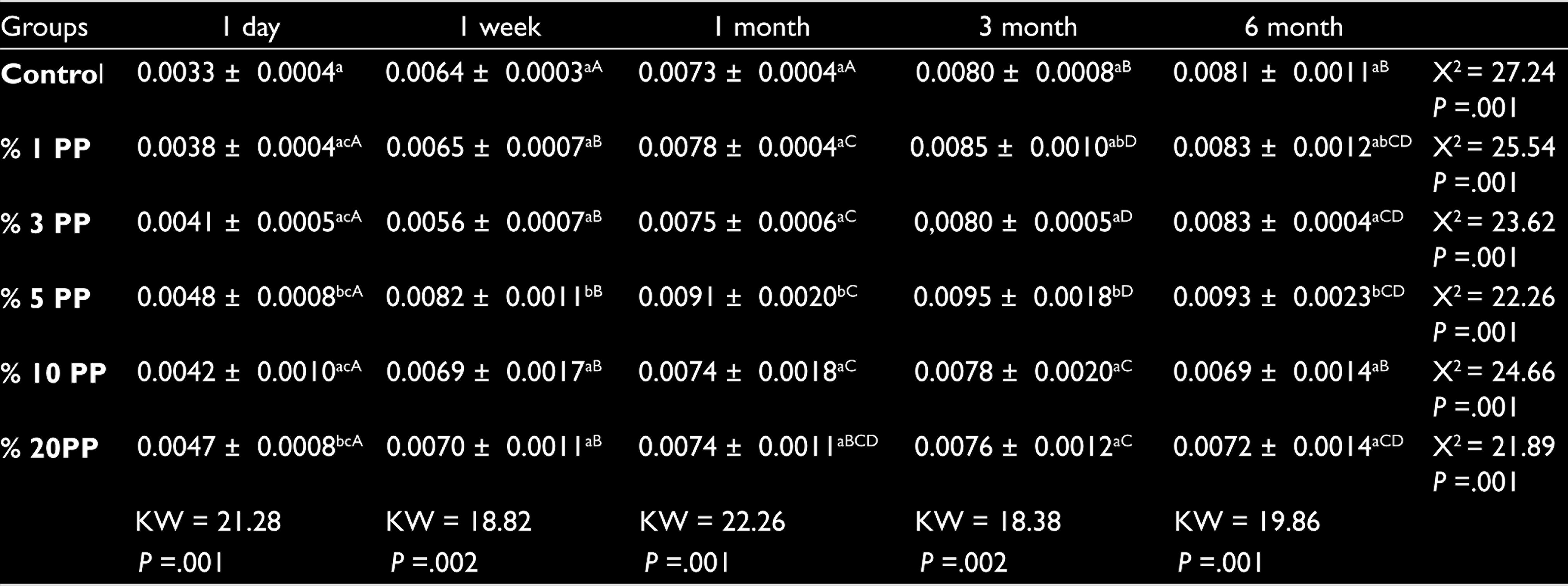
In addition, Wsp test results including the mean values and standard deviations for all tested specimens are summarized in Table 2. The highest Wsp values were detected in 5% PPFs group for all time intervals. After 6 months, the lowest Wsp values were seen in 10% PPFs group, followed by 20% PPFs group. The analysis of data revealed that there is significant difference in Wsp values between 5% PPFs group and all other groups (
Mean and Standard Deviation for Water Sorption (g/cm3) of Denture Base Materials
According to the cytotoxicity tests, polypropylene exhibited no cytotoxic effects for the cells in the concentration of 1%, 3%, 5%, 10%, and 20%. There was no decolorization zone around the samples. Although the cells were directly in contact with the extract in the culture media, they did not show any signs of injury and kept their morphological characteristics and wholeness like those in the controls. The lysis index score was 5 (markedly cytotoxic) for the positive control group, while for the negative control group it was 0 (noncytotoxic).
According to the scanning electron microscopy analysis of the fractured surface of fiber-reinforced acrylic resin, effective impregnation which allows the fibers to contact with the polymer matrix of the resin could be seen (refer to Figure 2).
Connection Between Polymer Matrix of the Resin and the Different Ratios of Fibers: (A) 1%, (B) 5%, and (C) 20%

Discussion
Based on the present study results, 1% PPFs addition did not improve the impact strength of the resin material. However, 3%, 5%, 10%, and 20% PPF addition were found to be effective for increasing the impact strength value. Therefore, the first hypothesis was partially rejected.
Flexural and impact loading is an important parameter for the dental prosthetic materials as it imitates situations they undergo. Under loading, the fibers apply a force opposing the crack propagation, and thus, the strength of the prepared composite increases. 15
In dental literature, there is limited study which is about reinforcement of denture base resin by using PPF. In accordance with the present study, Movade et al. 4 found that 2% PPF group of specimens exhibited improved flexural strength values and they also presented that PPF groups of specimens had higher toughness values than glass fiber and polyethylene fiber groups of specimens. Similarly, Nagakura et al. 16 suggested that glass fiber and PPF reinforcements have beneficial effect on the physical properties of denture base resin.
Mathew et al. 14 suggested that among the PPF reinforced test group (3, 6, and 12 mm length of the fiber and 2.5, 5, and 10 wt%), a 6 mm long fiber reinforced in 2.5 wt% resulted in the best flexural strength value. In addition, Mathew et al. 13 suggested that fiber weight percentage, fiber length, and adhesion between fiber and matrix play an important role in flexural characteristics of the hydrogen plasma‑treated PPF‑reinforced PMMA polymer. In the present study, 5 wt% added PPF supports the study conducted by Mathew et al. that the addition of PPF 5 and 10 wt% is required to get better mechanical properties coupled with an easy and cost‑effective reinforcing procedure.
Another clinically relevant factor that may affect the strength of the relined denture is the effect of water storage. Over time, mechanical properties such as hardness, impact strength, and fatigue limit are negatively affected by water penetration. 17 A study by Jagini et al. 18 showed that water storage for 15, 30, 60, and 120 days decreased the flexural strength of a denture base acrylic resin. According to ISO 1567, the value of Wsp for denture base materials should be less than or equal to 32 µg/mm3 for 1 week of storage. Both 5% and 20% PPF groups were not adhered to the ISO standardization. Furthermore, based on the present study results, the second hypothesis was rejected because the highest Wsp values were detected in 5% PPF groups of specimens, and it was found to be statistically significant for all time intervals. In accordance with the present study, Ladizesky 19 emphasized that the decrease of sorption in a fiber-reinforced polymer was related with the fiber concentration and that adding polyethylene fibers in acrylic reduced the Wsp by 25%.
Jagger mentioned that the increase in the amount of residual monomer increases the amount of water absorption. 20 The major part of the monomer conversion of PMMA forms at the early stage of water sorption and is drastically decreased after the third month. Thereby, the cytotoxic effect of PMMA may be related to storage time. 21 The decrease in the amount of residual monomer in PMMA results in a less cytotoxic effect. It was exhibited that the use of glass-fiber reinforcement in PMMA increased the release of a residual monomer. 22 However, there is no data in dental literature about the cytotoxicity of PPF-reinforced acrylic resin material. To ensure the safety of this material, in vitro cytotoxicity tests have been developed as a preliminary screening test to evaluate material biocompatibility. 23 Consistent with the present study results, Meric 24 found glass-fiber-reinforced PMMA denture base resin to be noncytotoxic according to cell viability assessed by the MTT assay.
In the present study, the change of the mechanical and physical properties of the fiber, which was added to acrylic in certain proportions, was evaluated. In addition, the effects of long-term use can be evaluated with the reflection of the oral environment. For this reason, the results can be compared by applying with the short- and long-term aging processes. In addition, more studies are needed to add the fiber used in different lengths and to compare with different acrylics.
Conclusion
Under the conditions of the present study, the following conclusions were drawn:
Reinforcement with PPF increased the impact strength of PMMA denture base resin. Wsp values of PPF added heat-cure acrylic resin did not change after the third month. PPF can be used as an alternative for reinforcing denture base materials.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
