Abstract
Background and Objective:
Mesenchymal stem cells (MSCs) from many different dental tissues have enormous potential in tissue regeneration and immunotherapy. MSCs isolated and cultured in many different ways are known for their heterogenecity expressed through multiple phenotypes which in turn may reflect their distinct functional properties; it is important to analyze and compare the characteristics of each cell lines cultured. The objective of this study is to isolate MSCs from 3 different sources and analyze and compare their morphological and phenotypic characteristics.
Materials and Methods:
The samples were collected and mononuclear cells were isolated using enzymatic digestion method and the adherent cell cultures were maintained till passage 3 and the harvested cells were analyzed for different phenotypic markers such as CD34, CD45, CD29, CD105, CD73, CD90, SOX2, OCT3/4, and neuroectodermal stem cell marker (NESTIN). The comparative statistical analysis of characterized cells was done by standard deviation methods using SPSS software (Canada).
Results:
Our study was successful in isolation, culture, and characterization of stem cells from all the mentioned 3 sources. All established cultures were more than 95% positive for mesenchymal markers and less than 5% positive for hematopoietic markers. The cells also expressed pluripotency markers and neural markers. Further, the comparison of mean positive expressions among the groups using analysis of variance showed the different significance levels of expression of different markers in groups.
Conclusion:
MSCs from different sources show promising expression of different markers; therefore, it can be concluded that cells isolated from dental sources can be used for treating various neurodegenerative diseases in the future.
Keywords
Introduction
Stem cells can be defined as a group of cells that have the ability to self-renew and have the multilineage differentiation capacity. 1 In general, stem cells are composed of pluripotent stem cells like embryonic stem cells and induced pluripotent stem cells, and multipotent stem cells, such as adult or somatic stem cells. Pluripotent cells have the capacity to differentiate into various types of cells but with the risk of tumorigenesis. 2 On the other hand adult stem cells like mesenchymal stem cells (MSCs) have been identified in almost all postnatal tissues including adult bone marrow, adipose tissue, etc. and have the ability to regenerate into desired tissues and are easily accessible without a risk of tumorigenesis. These cells have potential use in treating many degenerative diseases. 3 MSCs are multipotent progenitor cells derived from many tissues of the body; these cells can be useful in tissue regeneration and immunotherapy because of their self-renewal capacity, multilineage differentiation potential, and immunosuppressive properties. 4 MSCs from the dental tissue are among many other stem cells residing in specialized tissues that have been isolated and characterized successfully. MSCs from orofacial region can be isolated from 11 different sources such as dental pulp stem cells (DPSCs), stem cells isolated from human exfoliated deciduous teeth (SHEDs), stem cells derived from human natal dental pulp, periodontal ligament stem cells, stem cells isolated from the apical papilla (SCAPs), stem cells isolated from dental follicle precursor cells, bone marrow-derived MSCs from orofacial bone (BMSCs), oral epithelial progenitor/stem cells, gingival-derived MSCs, periosteum-derived stem cells, and salivary gland-derived stem cells. Many previous studies have demon-strated that a particular subgroup of MSCs exists in human dental tissues, including DPSCs, SHEDs, and SCAPs. 5 These cells have the potential to be used in tissue regeneration and immunotherapy.6,7 However, it is important to note that although it is known that MSCs from different types of dental tissue share a lot of common characteristics, they also present significant heterogeneity, expressed through multiple phenotypic differences which in turn may reflect their distinct functional properties. The heterogenecity may be significantly enhanced as a function of MSCs’ tissue microenvironment.8,9 This issue has become more complicated as different researchers have used many different methods to isolate and culture dental MSCs, and to evaluate their differentiation potential.
Therefore, the aim of this study is to isolate MSCs from three different sources, namely exfoliated deciduous teeth, impacted third molars, and apical papilla of impacted third molars, and analyze and compare their morphological and phenotypic characteristics.
Materials and Methods
A presented comparative cross-sectional study was evaluated on extracted human third molars obtained from the Department of Oral-Maxillofacial Surgery, YMT Dental College and Research Institute, Kharghar, Maharashtra, India. Exfoliating deciduous teeth were obtained from the Department of Pediatric and Preventive Dentistry. The teeth specimens obtained were further carried and processed at ReeLabs Pvt. Ltd, Mumbai. 10 deciduous teeth and 10 impacted third molars with open apices were selected for the culture study. The study was carried out for the period of 20 months from 2018 to 2019. Necessary ethical approvals were obtained. The random sampling method was used to collect samples for this study.
The study included participants from both the sexes between the age group of 7 and 12 years for exfoliated deciduous teeth collection and 16 and 22 years for impacted third molars. Carious teeth, teeth with developmental anomalies, and third molars with closed apices were excluded, and only noncarious exfoliated deciduous teeth and impacted third molars with open apices were collected for the study.
Once extracted, teeth were placed into sterile containers with 20 ml of sterile phosphate buffered saline on ice pack and transported to the lab for processing within 24 hours. Before digestion of the dental tissue, teeth were disinfected by a protocol adopted and modified from cornea banking. 7 The pulp tissue was gently removed from the crown and root. 8 Root apical papilla was gently separated from the surface of the root with sharp excavator, then minced into small parts and finally placed into a culture dish.
The explants were digested using type I collagenase (0.3 mg/ml; Sigma Aldrich, USA) and 0.1% dispase II (Sigma Aldrich) for 1 hour at 37°C with regular shaking on mechanical shaker (REMI, India). After tissue digestion single-cell suspensions (5 × 103 per well) of dental pulp were seeded into a T-25 flask (Corning, USA) at a density of 1 tooth digest per flask with Dulbecco’s modified Eagle medium (Gibco, USA) containing 15% fetal bovine serum (Gibco) with 100 units/ml antibiotic solution (Himedia, India), and was placed into a high humidity 37°C 5% CO2 incubator (Memmert). No selection of stem cells was performed directly, and instead selective culture was used to maximize stem cell recovery. Culture flasks were monitored daily, and any contaminated flasks were removed immediately and recorded. Noncontaminated flasks were monitored for cell growth, with culture medium changes taking place every third day.
After 14 to 15 days of growth, adherent cells were dissociated from flasks using 0.25% trypsin containing 1 mM ethylene diamine tetraacetic acid (Himedia) and a cell count of the total cells per flask obtained via standard trypan blue dye using hemocytometer counting methods. The flasks containing adherent cells from all the 3 sources were subcultured and maintained till passage 3. At passage 3, cells were detached from the flasks, resuspended in complete medium, and analyzed to whether they meet the minimal criteria to be called as MSCs by flow cytometry based on the International Society for Cell Therapy (ISCT) position paper. 9 Antibodies to human CD73/APC, CD90/FITC, CD105/PE, CD29/PE, CD34/PE, CD45/FITC, SOX2/ALEXA FLUOR, and OCT3/4/PERCP, NESTIN/APC, were obtained from BD Biosciences, USA. Cells were stained with the above antibodies and appropriate isotype controls as per the manufacturer’s instructions were analyzed using a fluorescence-activated cell sorting Calibur instrument with Cell Quest software (BD Biosciences), after which a comparative analysis was performed with analysis of variance to study the characterization of DPSCs, SHEDs, and SCAPs.
Statistical Analysis
All the statistical analyses were carried out using SPSS (version 23) software by agency in India. Results were tabulated and graphs were drawn according to the obtained results. The mean percentage expression of each marker and standard deviation of the particular marker in each group (group 1 = DPSCs, group 2 = SHEDs, and group 3 = SCAPs) were calculated. The percentages of mean positive expression between the groups were compared with the analysis of variance technique. Wherever significant difference/rejection in null hypothesis was found (P value < .05) between the groups, Turkey’s multiple comparison test was performed to find out the actual difference in significance levels.
Results
The samples were successfully collected and were transported to a research laboratory in sterile phosphate buffer saline on ice pack. The mononuclear cells were isolated from the samples, and cultures were established from dental pulp (DPSCs), human exfoliated deciduous teeth (SHEDs), and apical papilla (SCAPs). Total 10 cell cultures were established from each of the source, namely DPSCs, SHED, and SCAP. We obtained about 10 to 15 million DPSCs and SHEDs, and 4 to 6 million cells from apical papillae per sample. Each passaging was done after reaching 70% confluence. By the third to fourth day, colonies of high density were observed in SHED and SCAP cultures, whereas in DPSC cultures, small number of colonies could be detected only after the sixth to eighth day. SCAP and SHED cell cultures reached confluency by the fifth to sixth day. Most DPSC cultures, on the other hand, needed 12 to 15 days to produce subconfluent monolayers.
Characterization With Flow Cytometry
All samples were evaluated by flow cytometer for MSC markers (CD29, CD90, CD105, and CD73); hematopoietic stem cell markers (CD34 and CD45); pluripotency markers (SOX2 and OCT4), and neural marker NESTIN. The results were tabulated (Table 1); all cultures were more than 95% positive for CD90, CD29, CD105, and CD73, and less than 5% positive for the antigens CD34 and CD45. SOX2 was expressed as 1.63%, 1.44%, and 1.36% in DPSCs, SHED, and SCAP. While OCT4 was expressed at 4.64% of DPSCs, 4.19% of SHED, and 5.3% of SCAP, DPSCs, SHEDs, and SCAPs were 70.82%, 64.9%, and 82.24% positive for NESTIN, respectively (Figures 1–3).
Mean Positive Percentage Expression and Standard Deviation of Each Marker in 3 Groups
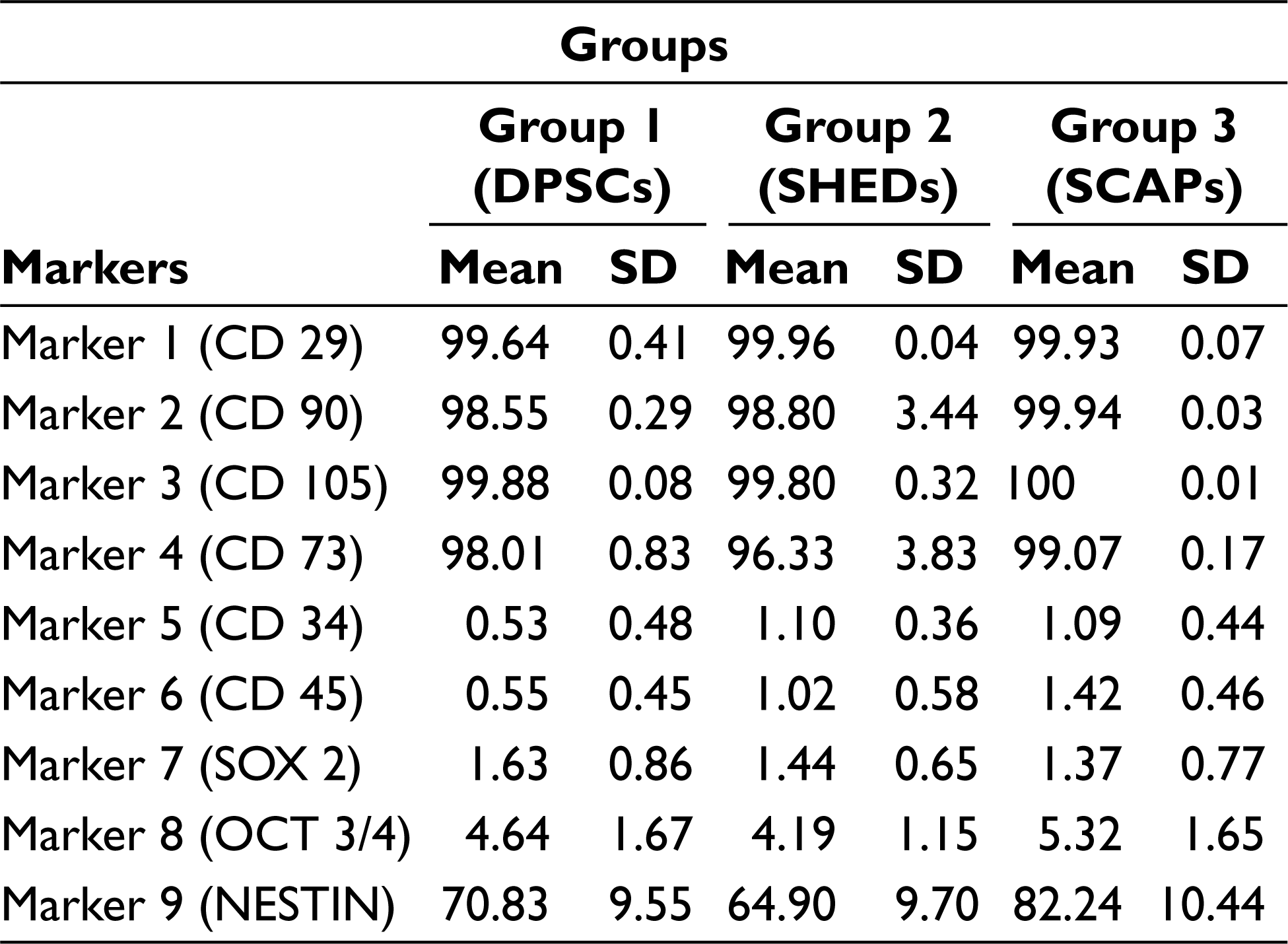
Comparative Expression of DPSCs, SHEDs, and SCAPs for Mesenchymal Stem Cell Markers (CD29, CD90, CD105, and CD73)
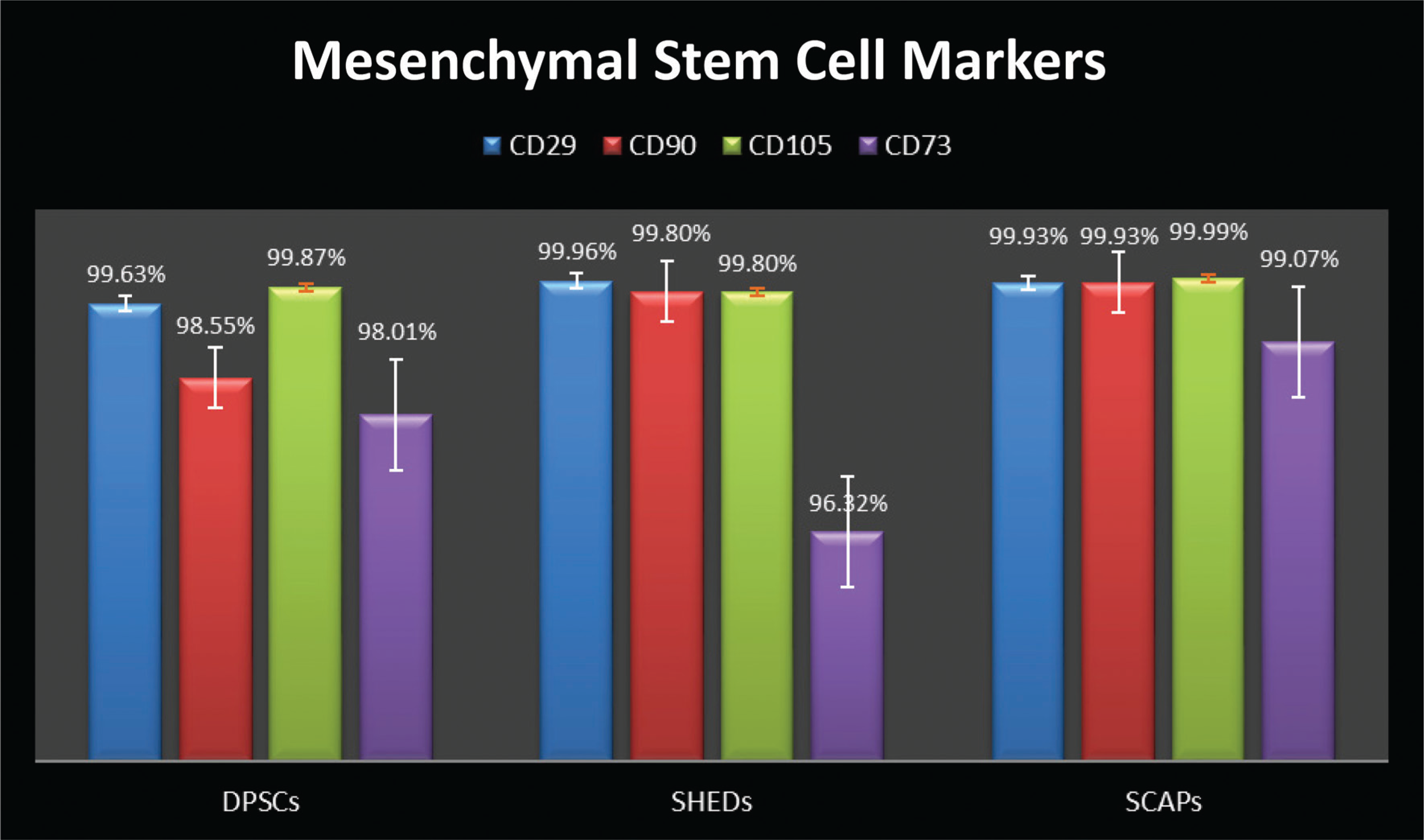
Comparative Expression of DPSCs, SHEDs, and SCAPs for Hematopoietic Stem Cell Markers (CD34 and CD45)
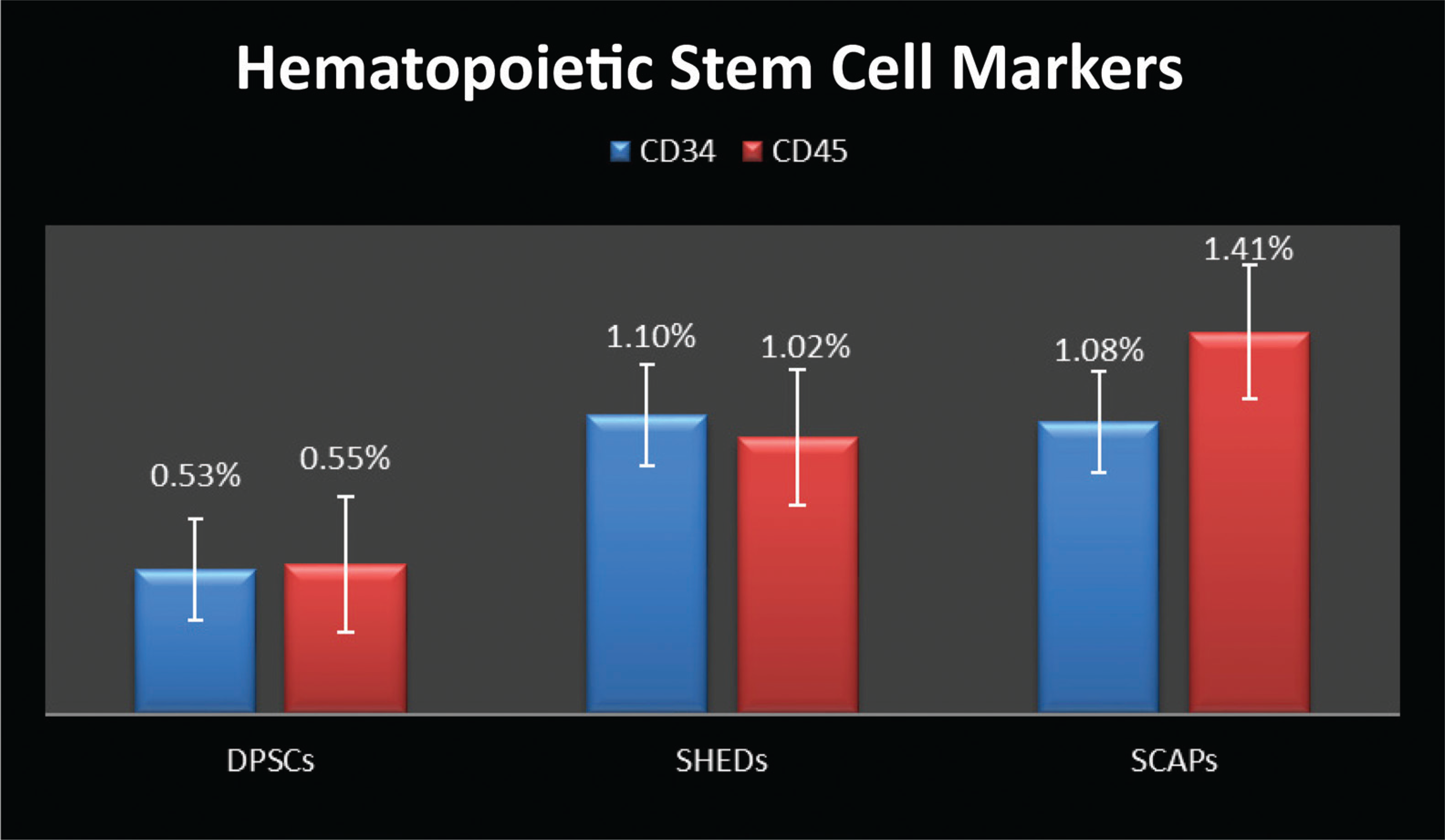
Comparative Expression of DPSCs, SHEDs, and SCAPs for Pluripotency Markers (SOX2, OCT3/4)
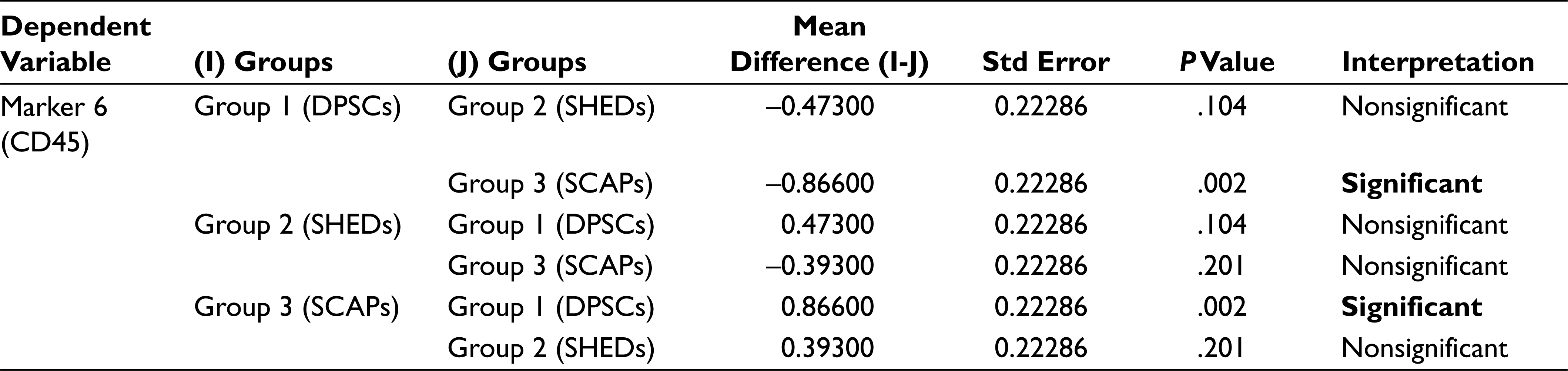
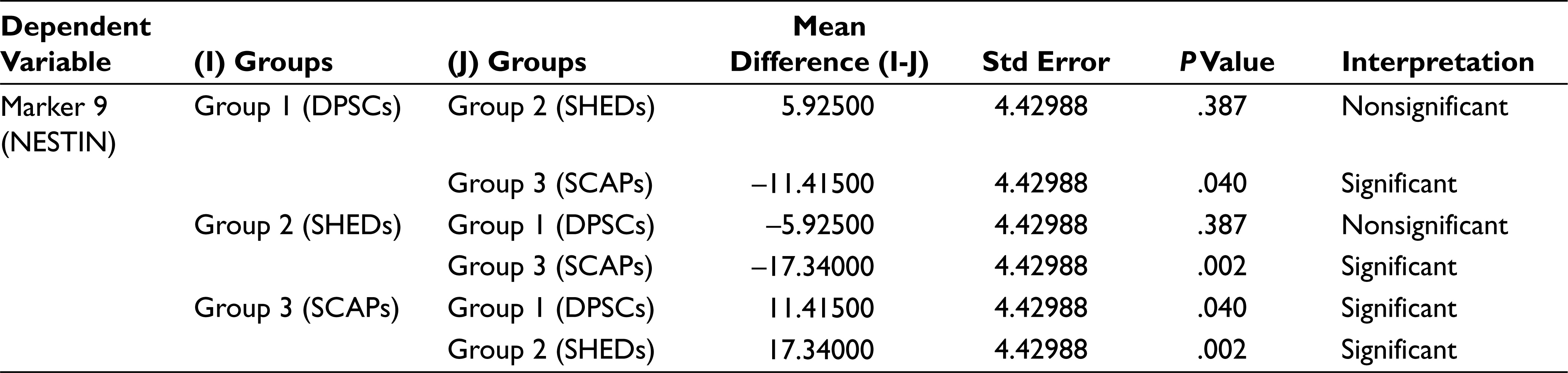
In analysis of variance (Table 2) the P value for the between groups for markers CD90, CD105, SOX 2, and OCT3/4 is greater than that of .05 (alpha value), which thus indicates that there is no significant statistical difference between the mean percentage expression of different groups for mentioned markers, Similarly, the P values for between the groups for markers CD29, CD73, CD34, CD45, and NESTIN were less than .05 (alpha value), which indicates there is significant statistical difference between the mean percentage expression of different groups, thus concluding the rejection of the null hypothesis. To find to exact significance of difference, the Tukey’s multiple comparison tests for the mentioned markers was done, from the data. For CD29 Marker there is statistically significant difference in the mean positive percentage expression of CD29 between DPSCs compared with SHEDs and SCAPs. There is no significant difference statistically in mean positive percentage expression of CD29 between SHEDs and SCAPs. For CD73, there is statistically significant difference in the mean positive percentage expression of CD73 between SHEDs and SCAPs, and no significant statistical difference between DPSCs and SHEDs. For CD34, there is statistically significant difference in the mean positive percentage expression of CD34 between DPSCs with SEHDs and SCAPs. No significant statistical difference was observed between SHEDs and SCAPs; similarly, for CD45 there is statistically significant difference in the mean positive percentage expression of marker CD45 between DPSCs and SCAPs. There is no statistically significant difference between SHEDs and SCAPs; for NESTIN there is statistically significant difference in the mean positive percentage expression of neural marker NESTIN between all 3 groups except when DPSCs and SHEDs are compared (Tables 3–7).
Analysis of Variance Results Between Different Marker Groups
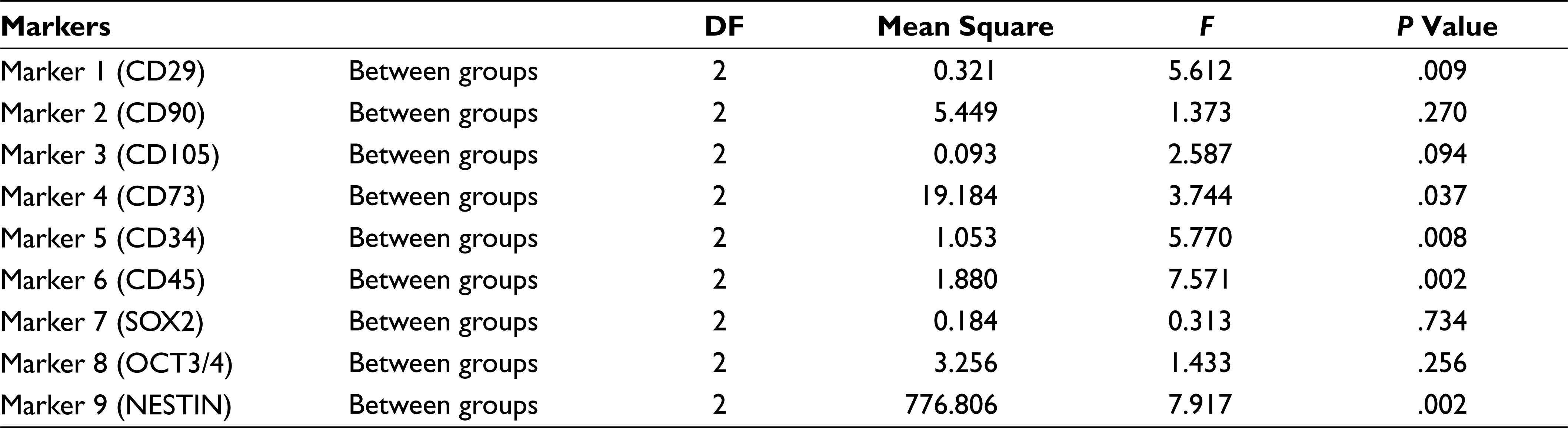
Tukey’s Multiple Comparison Test for Marker 1 (CD29)
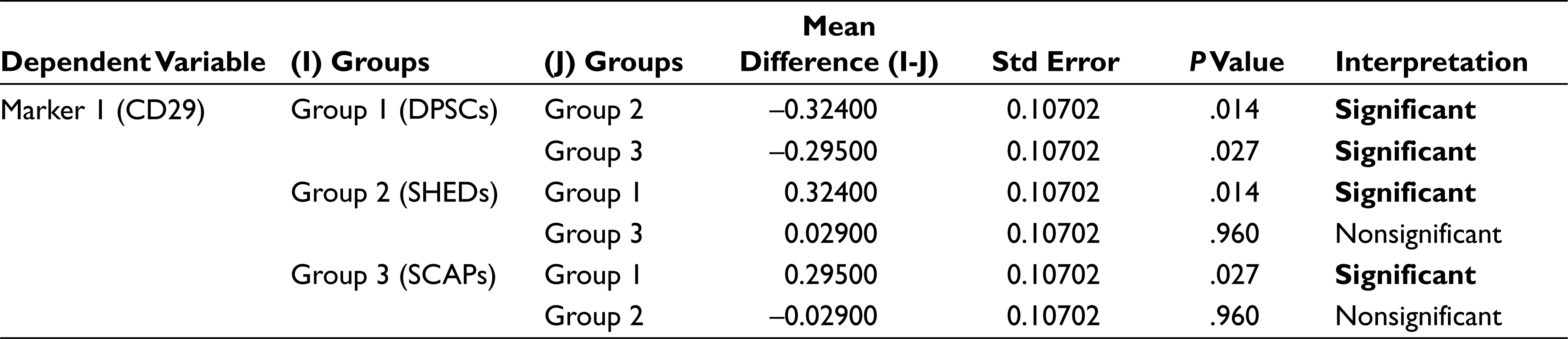
Tukey’s Multiple Comparison Test for Marker 4 (CD73)
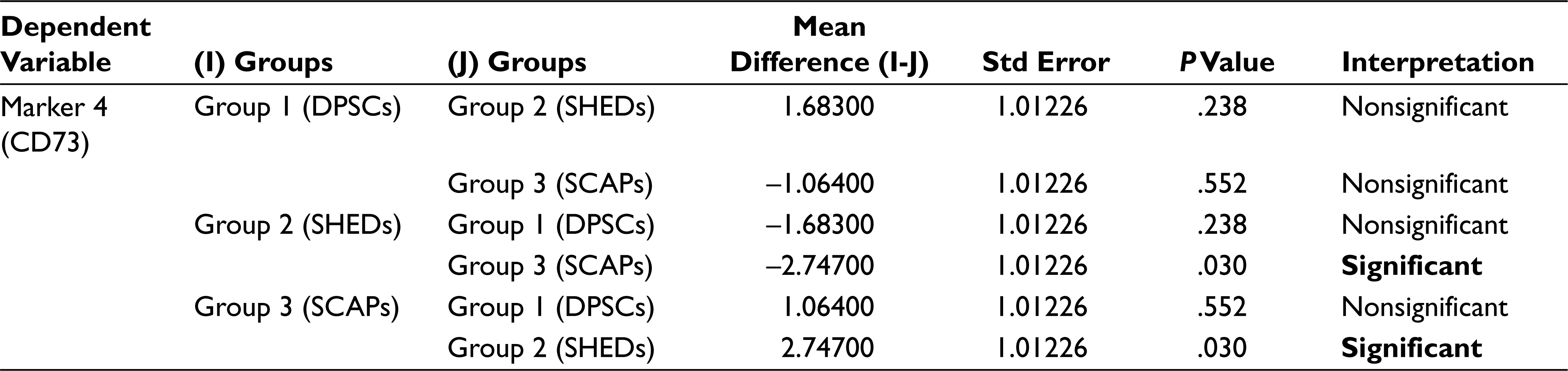
Tukey’s Multiple Comparison Test for Marker 5 (CD34)
Tukey’s Multiple Comparison Test for Marker 6 (CD45)
Tukey’s Multiple Comparison Test for Marker 9 (NESTIN)
Discussion
In recent years MSC-based therapies have been considered to be a promising tool for tissue repair and regeneration, but still the selection of the optimal cell source for the clinical applications remains unclear. Umbilical cord tissue, dental pulp, placenta, bone marrow, and menstrual blood are easily available for MSCs isolation without any ethical issues.10–12 The researchers have isolated dental pulp stem cells from adult human dental pulp, which had the ability to regenerate a dentin-pulp-like complex. DPSCs, SHEDs, and SCAPs have received extensive attention and importance since past few years13–15 and their proposed uses in spinal cord injury and neurodegenerative diseases raised expectations among the general public as well as professionals of possible therapies utilizing dental pulp cells. 16 The results of this study entailed are of significant interest to the fields of adult stem cell research and dental pulp biology. The purpose of this study is to isolate stem cells from dental pulp, from exfoliated deciduous teeth, and from apical papilla, and carry out comparative characterization of these stem cells with the help of flow cytometry.
Earlier it was demonstrated that all primary human cells have finite proliferative capacity during culture and that they enter cellular senescence with limited number of cell divisions. 17 Further studies have also stated that MSCs may undergo alterations during in vitro expansion, spontaneous transformation, reduction in homing, and also alterations in gene expressions.18–20 However, it has been also reported that there is no marked indication of culture degeneration or spontaneous differentiation in SHEDs during long-term culture till P19. 21 In our study we could not find any marked indications for culture degeneration till passage 10, but we have used cells from passage 3 for the characterization. In another study carried out by Wataru Sonoyama et al, to check whether cells in the apical papilla proliferate at a different rate compared with cells in pulp when the extracted teeth are placed in cultures. They used bromodeoxyuridine (BrdU) to label the cells in the tissues in cultures. Their results showed that the apical papilla contains 2- to 3-fold more BrdU-positive cells than the pulp tissue in the same tooth, 3.5 hours or 18 hours after the BrdU incorporation. Their data suggested that once the tissues are placed in cultures, cells in the apical papilla start to enter the growth cycle more than those in the pulp. 14 Similarly, in our study we found that by the third–fourth day, colonies of high density were observed in SHED and SCAP cultures, whereas in DPSC cultures, small number of colonies could be detected only after sixth–eighth day. SCAP and SHED cell cultures reached confluency by the fifth– sixth day. Most DPSC cultures, on the other hand, needed 12 to 15 days to produce subconfluent monolayers. This shows that SHED and SCAP exhibited higher proliferation rate as compared to that of DPSCs.
In our study using flow cytometry, we characterized our cultured cells for MSC markers (CD29, CD105, CD73, and CD90); hematopoietic stem cell markers (CD34 and CD45); pluripotency markers (SOX2 and OCT3/4), and neural marker (NESTIN). DPSCs showed high positivity for MSC markers and showed less positivity for hematopoietic stem cell markers, suggesting their stromal origin and the absence of hematopoietic precursor contamination. In another similar study, the researchers isolated pluripotent stem cells from human third molar dental pulp and reported a new population of pluripotent stem cells isolated from dental pulp. The cells were isolated by culturing them in media containing leukemia inhibitor factor, epidermal growth factor, and PDGF (platelet-derived growth factor). DPPSCs were identified as cells with the phenotypes CD13+, SSEA4+, OCT3/4+, NANOG+,SOX2+, LIN28+, CD14+, CD29+, CD105+, CD34-,CD45-, CD90-, STRO1- and CD146-. In their study an increase in the percentage of specific markers of DPSC cells (CD13, SSEA-4, OCT3/4, and CD34) was observed with respect to the culture time. CD13, SSEA-4, and OCT3/4 expression levels increased from 19% to 54%, 6% to 30%, and 0.4% to 6% during weeks 1 to 3, respectively. 22 However, in our study we did not isolate “dental pulp pluripotent stem cells” (DPPSCs) but carried out fluorescence-activated cell sorting analysis of DPSCs for pluripotency markers such as SOX2 and OCT3/4 which was 1.6% and 4.64%, respectively. In comparative flow cytometry analysis for pluripotency markers such as SOX2 and OCT3/4, DPSCs showed maximum mean positive expression for SOX2 (1.6%) when compared with that of SHEDs (1.44%) and SCAPs (1.36%), while SCAPs showed maximum mean positive expression for OCT3/4 (5.30%) when compared with that of DPSCs (1.6%) and SHEDs (4.19%). In comparative flow cytometry analysis for neural marker (NESTIN), SCAPs showed maximum mean positive expression for NESTIN (82.24%) when compared with that of DPSCs (70.82%) and SHEDs (64.9%). So it can be stated that SCAP expressed more positivity for pluripotency markers OCT3/4 and neural marker (NESTIN) as compared to SHEDs and DPSCs. The comparative characterization study in expression patterns in cells isolated from different sources was useful to know the exact difference in the expression levels, thus proving their immature status, the cell sources may be further studied to investigate the mechanism involved in the in vitro differentiation capacities of cell, thus aiding in selection of cell sources to be used in different clinical studies. However, this study was to isolate, culture, and analyze the difference in phenotypic expression levels, further studies investigating the other distinct biological characteristics, differentiation potentials of each cell sources, gene expression profiles, and different pathways involved in short-term and long-term culture conditions may be explored.
Conclusion
Unlike all other MSCs from sources such as umbilical cord, menstrual blood, bone marrow, etc. the cells isolated from different dental sources also exhibit self-renewal and multipotency characteristics based on their phenotypic marker expressions, thus exhibiting distinct biological characteristics. Furthermore, the results provide insights into the immature status of the dental stem cells as the isolated MSCs expressed mesenchymal markers, along with pluripotency and neural marker. Ease of availability and utilization from biomedical waste makes dental stem cells to stand out as the best from waste. Therefore, it can be concluded that dental stem cells can be easily isolated and characterized, and can be one of the sources in clinical applications for various neurodegenerative diseases in the future.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
