Abstract
Aim and Background:
35% carbamide peroxide can cause adverse effects on the structure and composition of teeth. However, the addition of calcium and fluoride in bleaching agents may reduce enamel demineralization. The aim of the study is to evaluate the chemical changes in a tooth submitted to 35% of carbamide peroxide containing chitosan and carbopol as a carrier system using scanning electron microscopy/energy dispersive x-ray analysis (SEM-EDX).
Materials and Methods:
30 freshly extracted anterior teeth were given root canal treatment. The teeth were randomly divided into 3 groups. Group 1 was Opalescence PF (35% carbamide peroxide), group 2 was chitosan with 35% carbamide peroxide, and group 3 was a control group (no treatment). All canals were filled with 0.06 tapered gutta-percha and AH-plus sealer. Then, intracoronal bleaching was applied. SEM-EDX analysis was performed to determine whether bleaching procedures changed the mineral content of the tooth. While SEM-EDX analysis, the tooth was analyzed based on elemental content and elemental distribution from standardized points under 300× magnification. Statistical analysis was performed with one way analysis of variance.
Results:
There was no significant difference in C, O, Na, P, Mg, K, and S values and the Ca/P ratio obtained by SEM-EDX analysis among groups (P > .05), whereas the Ca level was significantly lower in Opalescence PF group than the control group (P < .05).
Conclusions:
The results of this study show that chitosan does not cause mineral loss and dissolution in the inorganic content of a tooth. Thus, chitosan can be used as an alternative carrier system in the bleaching agent.
Introduction
Nowadays, bleaching systems can be done in different concentrations and applications. Thanks to the practical availability and efficacy of the agents offered, this treatment has become a method that everyone can use and thus has become increasingly popular in society. The contents of the bleaching agents are oxidizing agents, thickening agents, urea, carriers, preservatives, and sweeteners. The most effective and most commonly used bleach solutions are hydrogen peroxide and carbamide peroxide with different concentrations depending on the method opted for. 1
The carbomer polyacrylic acid derivative, which is frequently used in bleaching agents, is a high molecular weight, synthetic anionic polymer and has mucoadhesive properties. The formulations are used for ophthalmic, rectal, and tropical purpose in creams, gels, lotions, and ointments. In tablet formulations, carbomers are used as controlled release agents and/or as binders. 2
Among the natural polymers is chitosan, which is obtained from the shells of the crustaceans. It is a bioadhesive, biodegradable, and biocompatible cationic polymer that exerts also various bioactive properties such as tissue regeneration and has antimicrobial effects.3-6
Recently, chitosan has been found to be as effective as the carbopol in terms of its delivery system for intracoronal bleaching treatment. 7 There is an absence of scanning electron microscopy/energy dispersive x-ray analysis (SEM-EDX) reports of dental hard tissue alteration as a result of chitosan with carbamide peroxide application.
A number of studies indicate that the bleaching leads to changes in the structure of the teeth, which in turn changes their biomechanical properties.8-10 This leads to a significant difference in the ratio of the organic/inorganic structure and dentin permeability, which negatively affects the connection and sealing properties of dental materials to the dental tissue. 11 Bleaching agents can cause adverse effects on mineral contents of root dentin, so it is important to know what effects each agent will have on root dentin before their clinical use. In this study, we aimed at developing a polymeric system for local delivery of carbamide peroxide for the treatment of bleaching. For this purpose, carbamide peroxide–incorporated chitosan polymer was used.
This study was designed to examine whether different carrier systems within bleaching agents can change the amount of mineral in the dentin. Therefore, the null hypothesis was that the 35% carbamide peroxide bleaching agent containing either chitosan or carbopol would not change the surface mineral content of root canal dentin.
Materials and Methods
30 human premolar teeth extracted for periodontal reasons were used in this in vitro study. All samples were radiographically confirmed as they have single straight canals. Teeth with any abnormality such as immature apices, cracks, calcification, or resorption were not included. The teeth were stored in normal saline at room temperature until required. The endodontic access cavity was opened under water cooling using 12 diamond burs (Diatech, Coltène Whaledent, Altstätten, Switzerland). Then, the canals of the samples were prepared with 0.06 tapered, size 25 One-Shape files (Micro-Mega, Besançon, France), attached to an endodontic motor (VDW Reciproc, Munich, Germany) using settings for One-Shape recomm-ended by the manufacturer (400 rpm; 2.5 N cm). All canals were filled with 0.06 tapered gutta-percha and AH-plus sealer (AH Plus, Dentsply, Konstanz, Germany). All samples were kept in a moist environment at 37°C for 1 week to provide complete setting of the sealer. Then, gutta-percha was cut 2 mm below the enamel-cement boundary. The canal orifices were filled with glass ionomer cement (Vitrebond, 3M Dental Products, St. Paul, Minnesota, USA) to be 2 mm thick. Teeth were randomly divided into 3 groups with 10 teeth per group. Table 1 shows the contents of the groups. Subsequently, bleaching procedures were carried out according to the Opalescence PF user guide. After the bleaching treatment was completed, the samples were analyzed with SEM-EDX (Zeiss Supra 40 Vp FESEM, Carl Zeiss, Germany) in terms of elemental content and elemental distribution under 300× magnification. Data on oxygen (O), carbon (C), calcium (Ca), phosphorus (P), magnesium (Mg), sodium (Na), sulfur (S), and potassium (K) and the Ca/P ratio were collected from the SEM-EDX analysis.
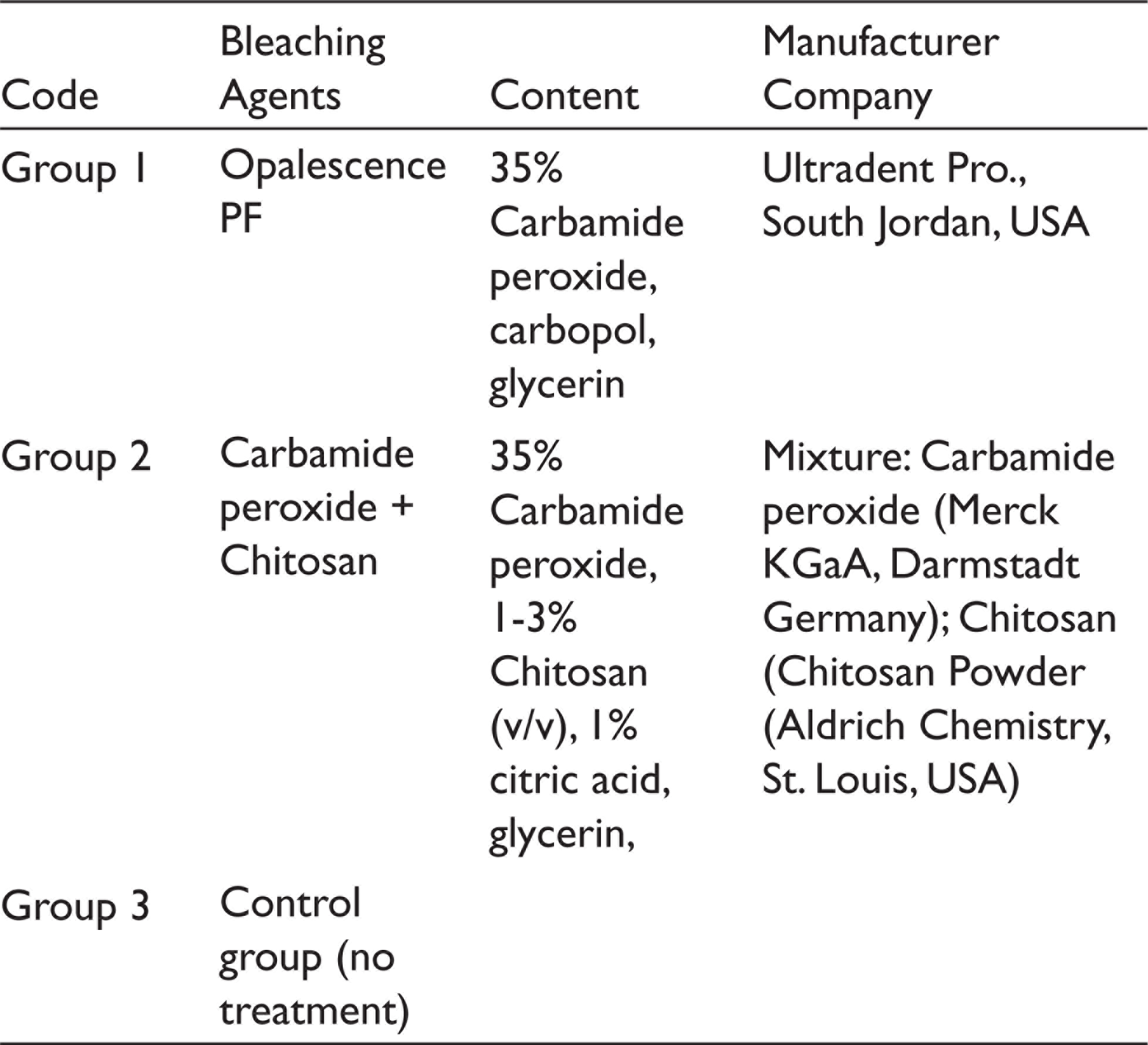
Content of the Bleaching Agents Used in the Study
Statistical Analysis
All data were performed by using SPSS 22.0 (SPSS Inc., Chicago, IL) program using one-way analysis of variance. All variables were presented as the mean ±SD. Probabilities of P < .05 were considered statistically significant.
Results
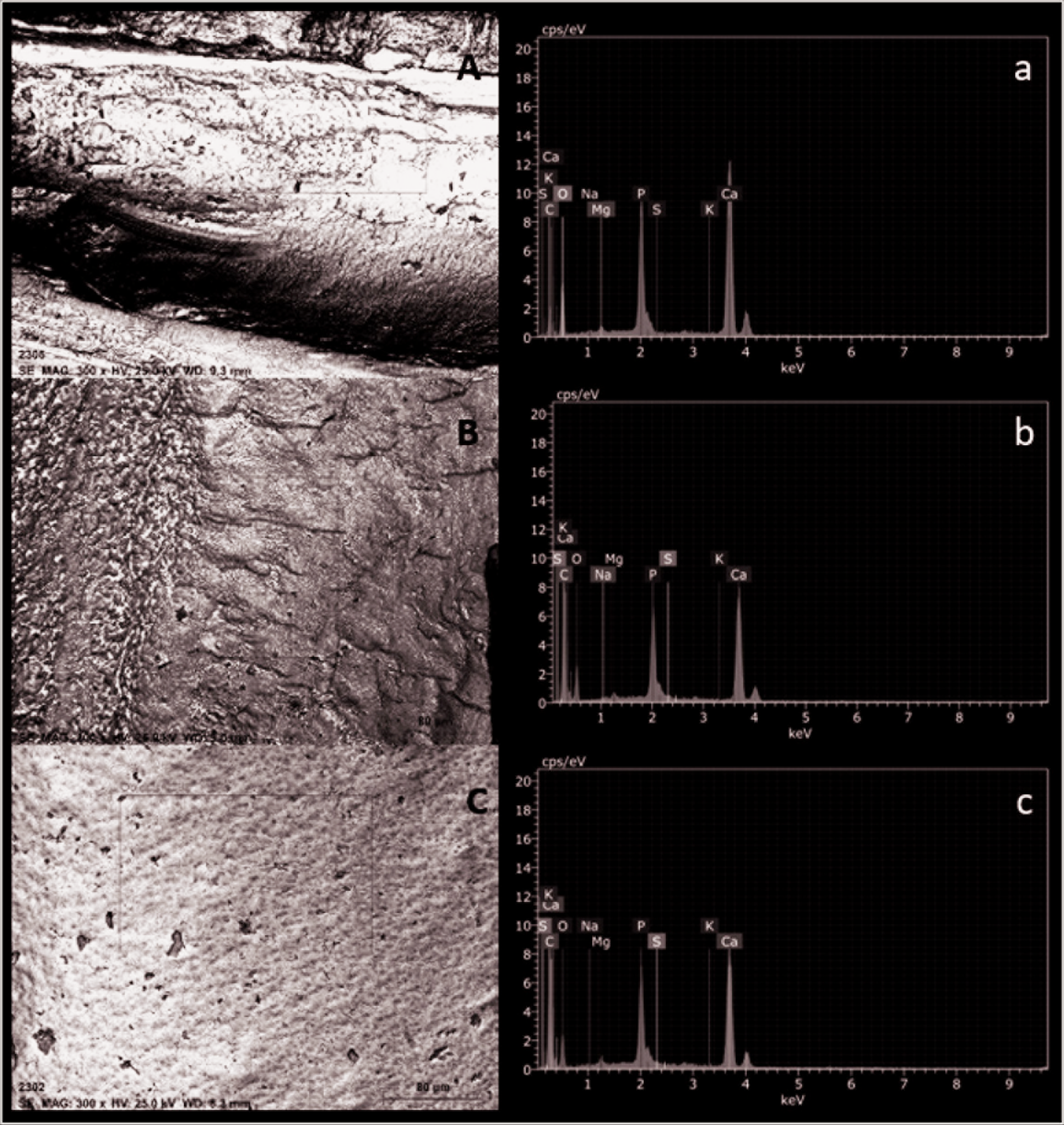
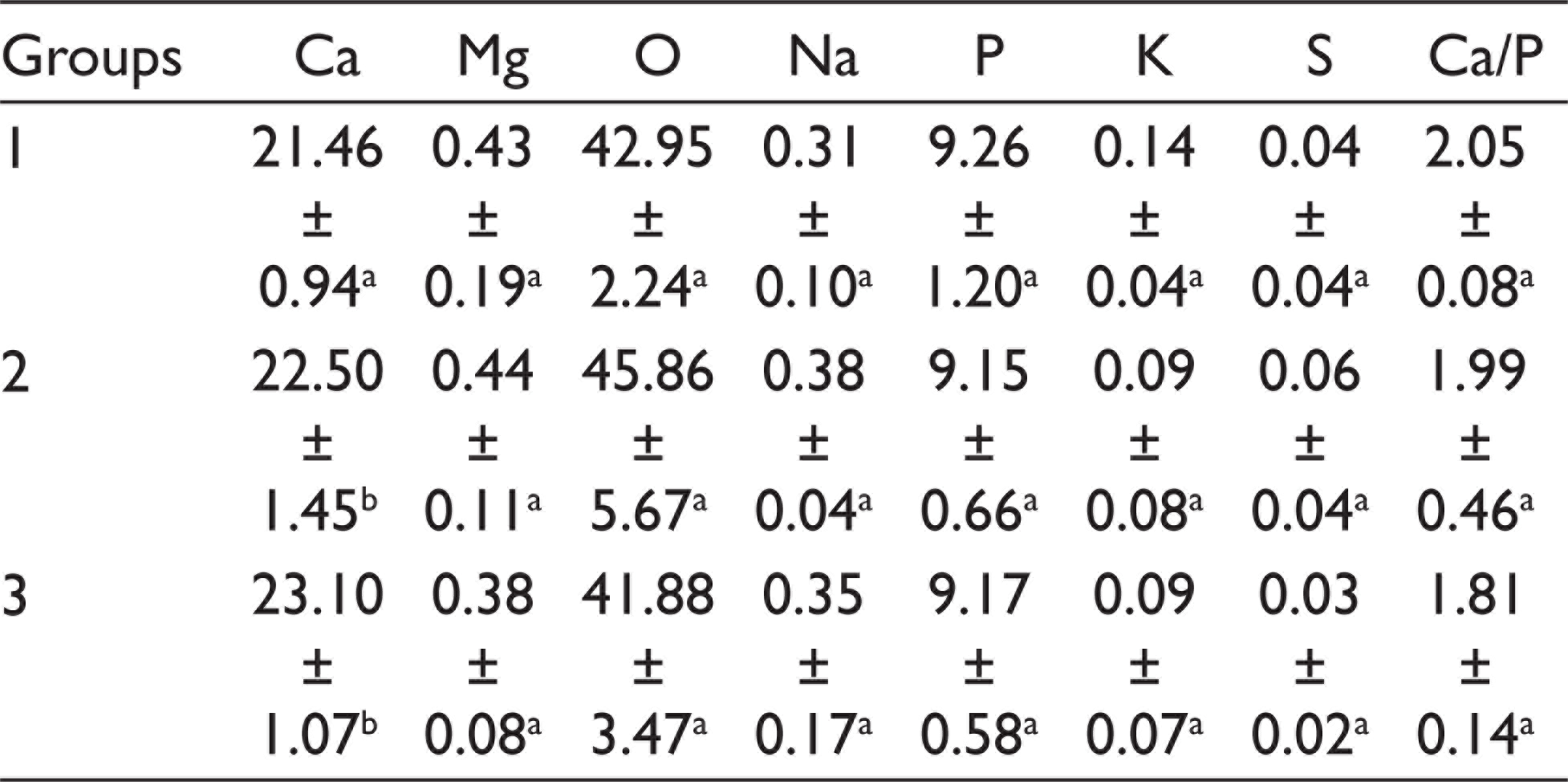
The mean percentage weights of the elements in the dentin after bleaching treatments are shown in Table 2. There was no significant difference in C, O, Na, P, Mg, K, and S values and the Ca/P ratio obtained by SEM-EDX analysis among the groups (P > .05), whereas the Ca level was significantly lower in group 1 than group 3 (P < .05). Figure 1 shows the representative results of the chemical composition and the main elements of the control and test groups according to EDX microanalysis. EDX profiles on control and test groups’ surfaces revealed high peaks for Ca, O, and P, and traces of Na, Mg, K, and S.
Representative Scanning Electron Microscopy (SEM) Image of Surface of Specimens (Original Magnification 300×) and Energy Dispersive X-Ray Analysis (EDX) Spectra With Peaks of Ca, C, O, Mg, K, S, Na, and P on Different Treatment Dentin Surface: (A) Group 1, (B) Group 2, and (C) Group 3
Average and Standard Deviation of Mineral Content of Dentin Among Group
Discussion
This investigation analyzed the behavior of dentin submitted to bleaching with experimental agents containing chitosan and carbopol. The purpose was to verify the efficacy of experimental gel in inhibiting or controlling mineral loss of root canal dentin.
According to the SEM-EDX analysis, bleaching with each of the two bleaching agents imparts changes in weight percent of Ca, C, Na, O, Mg, K, S, and P, and in the Ca/P ratio of dentin. There was no significant difference in the values of C, O, Na, P, Mg, K, and S, and the Ca/P ratio obtained by SEM-EDX analysis among groups (P > .05), whereas the Ca level was significantly lower in group 1 than group 3 (P < .05). There are studies reporting that dental surface properties, mineral content change, and microhardness decreased.12,13 A decrease in the Ca/P ratio in bleached samples was also reported in some previous studies.14,15 On the contrary, there are also studies reporting that the Ca/P ratio has not changed.16-18 In the present study, however, the ratio of Ca/P among groups was not found to have any significantly different change.
Tanaka et al reported that carbamide peroxide causes more surface demineralization and decomposes into its components on the surface of tooth hard tissues. 19 Calcium loss is reported in human enamel exposed to carbamide peroxide. 20 Moreover, it is also known that carbopol, which is frequently used in most bleaching agents, inhibits the growth of hydroxyapatite crystals, and the reason for this is that the calcium-binding capacity of carbopol is very high. 21 In the present study, the Ca level in group 1 (Opalescence PF) was significantly decreased compared to that of group 3 (Control). The decreased mineral content was attributed to this finding. The higher values of Ca loss reported in the present work might be due in part to carbopol delivery system and can also be due to the remineralizing property differences between carbopol and chitosan. Moreover, there are also studies in which chitosan supports remineralization in dental tissue.22,23 Therefore, the results obtained in the present study can be partly attributed to this feature.
Conclusion
The null hypothesis was accepted by the results of the present study. Within the limitations of the present study, it can be concluded that chitosan and carbopol with bleaching treatment did not affect the mineral contents of root dentin. The addition of chitosan to an experimental bleaching agent could control the decrease of Ca values for teeth surfaces. In the present study, bleaching materials with the same concentrations were used. Further studies must be conducted to determine whether different concentrations and time have an effect on the mineral content of root canal dentin.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
