Abstract
Background
Due to its ease of use and quick turnaround times, dental bleaching has grown to be one of the most popular aesthetic procedures. But regrettably, it has the bothersome consequence of dental hypersensitivity. The purpose of this clinical study was to assess the effectiveness of a desensitizing agent used during in-office bleaching with 35% hydrogen peroxide.
Methods
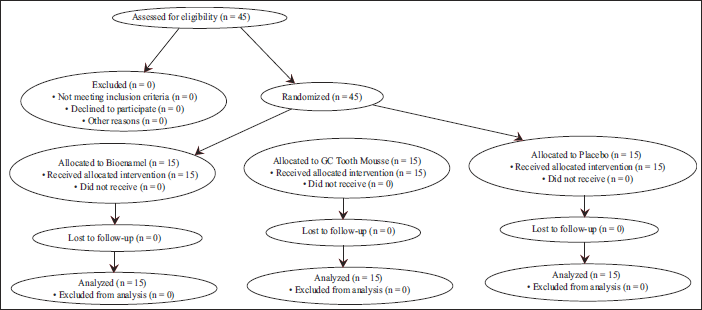
This randomized, double-blind, parallel-group clinical trial (registered at CTRI/2019/05/019123 on 22nd May 2019) included 45 participants randomly allocated into three equal groups (n = 15) using a computer-generated sequence and opaque, sealed envelopes. Group I received Bioenamel (Prevest Denpro, India), Group II received GC Tooth Mousse (GC Corporation, Japan), and Group III received a placebo (Glycerine Liquid, Scortis Health Care). All interventions were applied post-bleaching. The primary outcome was hypersensitivity measured using a Visual Analogue Scale (VAS) immediately, daily for 7 days, and at 1 month. Data were analyzed using the Kruskal–Wallis test.
Results
All participants reported no baseline sensitivity prior to bleaching. Immediately after bleaching, all groups exhibited a significant increase in hypersensitivity (p < .001). Among them, the Bioenamel group showed the lowest median VAS scores, followed by the GC Tooth Mousse group, while the placebo group demonstrated the highest sensitivity levels. A statistically significant difference in hypersensitivity was observed between groups at all evaluation intervals (immediate, 24 hours, and 1 week; p < .05), with Bioenamel consistently providing superior relief. By the end of one week and at one-month follow-up, all groups reported no sensitivity.
Conclusion
Bioenamel (Prevest Denpro) was the most effective at reducing post-bleaching hypersensitivity as compared to GC Tooth Mousse and placebo.
Trial Registration
CTRI/2019/05/019123
Introduction
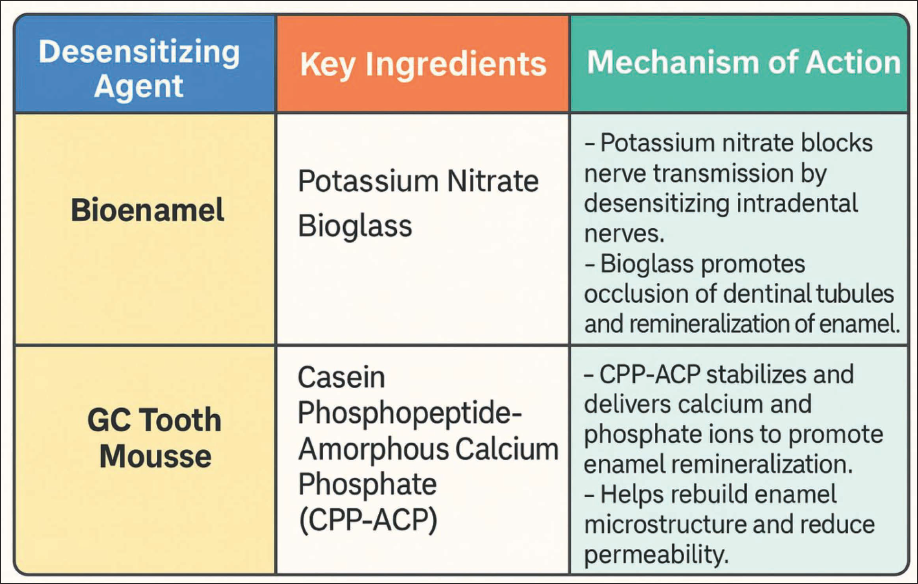
Dental bleaching has become one of the most popular aesthetic treatments worldwide due to its ease of application and rapid results. The use of bleaching agents, especially hydrogen peroxide-based formulations, effectively lightens teeth and enhances the appearance of the smile, making it a favored choice for patients seeking cosmetic dental procedures.1, 2 Despite its popularity, post-bleaching hypersensitivity remains a common side effect, which may hinder the overall experience and outcome of the procedure. This transient sensitivity can manifest as discomfort or pain in response to stimuli such as temperature, touch, or pressure, and it is particularly prominent after in-office bleaching treatments using concentrated hydrogen peroxide. 3 Hypersensitivity associated with dental bleaching is thought to result from the demineralization of tooth enamel, changes in the structure of dentin, or the release of free radicals during the bleaching process, which affect the pulp and nerves. 4 The severity of this condition varies among individuals, with some experiencing no discomfort, while others report significant pain. In-office bleaching with 35% hydrogen peroxide is known to cause more intense sensitivity compared to at-home treatments, making it crucial to explore effective ways to mitigate this side effect. 5 To address this issue, various desensitizing agents such as potassium nitrate, sodium fluoride, calcium phosphate compounds (e.g., CPP-ACP), oxalates, glutaraldehyde, and arginine-based formulations have been investigated for their potential to reduce post-bleaching hypersensitivity.6–11 Bioenamel (Prevest Denpro, Jammu) is a product that contains potassium nitrate and bioglass, whereas GC Tooth Mousse contains casein phosphopeptide-amorphous calcium phosphate (CPP-ACP), which is believed to remineralize the enamel and reduce sensitivity.12, 13 Both products have been suggested to offer protective benefits, but limited evidence exists comparing their efficacy directly in the context of post-bleaching sensitivity. This study aims to fill this gap in the literature by comparing the effectiveness of Bioenamel and GC Tooth Mousse in reducing hypersensitivity following in-office bleaching with 35% hydrogen peroxide. The findings of this study could provide valuable insights into the most effective desensitizing strategy, ultimately improving the comfort and satisfaction of patients undergoing cosmetic bleaching procedures (Figure 1).
Composition and Mechanism of Action of Desensitizing Agents.
Materials and Methods
This clinical trial was registered with the Clinical Trials Registry, India (CTRI) under the registration number CTRI/2019/05/019123 on May 22, 2019. This study was a randomized, double-blind, parallel-group clinical trial with an equal allocation ratio of 1:1:1. A computer-generated randomization sequence was used to allocate participants into three groups (n = 15 each). Allocation concealment was ensured using opaque, sealed envelopes prepared by a third party not involved in the clinical procedures or outcome assessment. Both participants and outcome assessors were blinded to group assignments to minimize bias. This randomized, double-blind clinical trial followed the Consolidated Standards of Reporting Trials (CONSORT) 2025 guidelines. The study was conducted at the Department of Conservative Dentistry, Rural Dental College, Loni, Maharashtra, India, between 2019 and 2020. Recruitment Dates: May–July 2019; Follow-up completed by August 2020.
Dissemination Policy
The results of this study were disseminated through peer-reviewed publications and conference presentations. Additionally, participants were informed of the outcomes upon request. The findings were intended to inform clinicians and researchers about effective strategies for managing post-bleaching hypersensitivity.
A total of 45 adult patients (20 males and 25 females), aged between 18 and 40 years, were recruited from individuals seeking in-office teeth whitening. The sample size of 45 (15 per group) was calculated based on previous clinical studies evaluating post-bleaching hypersensitivity, aiming to detect a clinically significant difference in VAS scores with 80% power and a 5% significance level. Participants were included after meeting specific eligibility criteria: presence of six intact maxillary anterior teeth, absence of caries or restorations, good general and dental health, and no reports of spontaneous dental pain.
Exclusion criteria included: Carious lesions or restorations on upper anterior labial surfaces History of bruxism Presence of gingival recession or cracked teeth Use of analgesics Pregnancy and lactation
All participants received a detailed explanation of the study, followed by signed informed consent, which included contact information for follow-up. On scheduled days, participants were reminded by phone calls.
A single calibrated operator carried out the in-office bleaching procedure to eliminate inter-operator variability. Prior to the procedure, oral prophylaxis was done using a low-speed handpiece and DPI Propol prophylactic paste. Baseline hypersensitivity was recorded using a Visual Analogue Scale (VAS) from 0 (no pain) to 10 (severe pain).
During the bleaching appointment, the lips were lubricated with Vaseline, a cheek retractor was placed, and isolation was achieved using cotton rolls and Gingiva Shield VLC (Prevest Denpro, India), a light-cured barrier. The bleaching procedure was performed using Florence™ 35% hydrogen peroxide gel (Prevest Denpro, India). The gel was applied 1–2 mm thick on the labial surface of the anterior teeth and left undisturbed for three cycles of 15 minutes each. After each cycle, the gel was removed with a surgical suction tip or gauze.
Participants were randomly assigned to three groups (n = 15 per group): Group I: Bioenamel (Prevest Denpro Limited, India) applied post-bleaching Group II: GC Tooth Mousse (GC Corporation, Tokyo, Japan) applied post-bleaching Group III: Placebo (Glycerine Liquid, Scortis Health Care) applied post-bleaching
Participants were instructed to avoid desensitizing toothpaste and pigmented beverages (e.g., tea, coffee, cola) for seven days. Hypersensitivity was assessed using the VAS at four-time intervals: immediately after bleaching, at 24 hours, 1–7 days, and 1 month. Each participant maintained a pain diary using a VAS daily and brought it during follow-up evaluations. The VAS score was determined by measuring the distance (in cm) from the zero-anchor point to the patient’s mark. Assessment was carried out by a blinded evaluator using a standardized printed chart. While both the participants and the evaluator were blinded, the operator was not, due to differences in material consistency despite similar appearance. Data were analyzed using IBM SPSS Statistics for Windows, Version 23.0 (Armonk, NY: IBM Corp.). Non-parametric data were expressed as median and interquartile range (IQR). Intergroup comparisons were conducted using the Kruskal–Walli’s test, while categorical data were presented as frequencies and percentages. A p ≤ .05 was considered statistically significant (Figures 2 and 3).
Application of Florence Teeth Whitening Gel.
CONSORT 2025 Flow Diagram of Participant Enrollment, Allocation, Follow-up, and Analysis.
Results
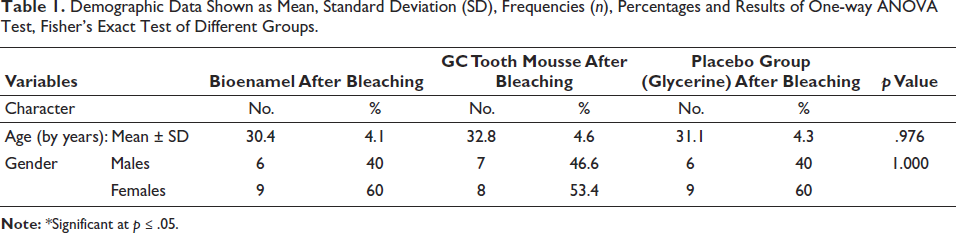
Forty-five adult patients (20 men and 25 women) participated in this study. The mean age of the patients, who ranged in age from 18 to 40, was 30.4 ± 4.1, 32.8 ± 4.6, and 31.1 ± 4.3 for the Bio enamel group, GC Tooth Mousse and the placebo group, respectively. There was no statistically significant difference in the mean age values between any of the groups (p = .965) (Table 1). In terms of patient gender, there were six males and nine females in the Bio enamel group, seven men and eight females in the GC Tooth mouse group, and six males and nine female’s placebo control group. There was no discernible difference in the distribution of genders (p = 1.000). All of the study group cases had no pain before bleaching (p = 1.000, effect size = 0.000). Immediately following bleaching, a significant change was seen in all groups (p < .001, effect size = 3.149). Concerning the group’s pairwise comparison, Group 1 Bioenamel (Prevest Denpro Limited) showed the lowest median pain score respectively followed by Group II (GC Tooth Mousse). Highest median pain score after bleaching was observed in Group III placebo (Table 2). A substantial difference between the groups was found after one day (p < .001). A statistically significant difference was seen on the second day following bleaching (p = .092, Effect size = 1.876. All three groups’ instances revealed no pain on days 4, 5, 6, and 7 and one month later.
Demographic Data Shown as Mean, Standard Deviation (SD), Frequencies (n), Percentages and Results of One-way ANOVA Test, Fisher’s Exact Test of Different Groups.
Results of the Kruskal–Wallis Test for Sensitivity Score Comparison Between the Groups.
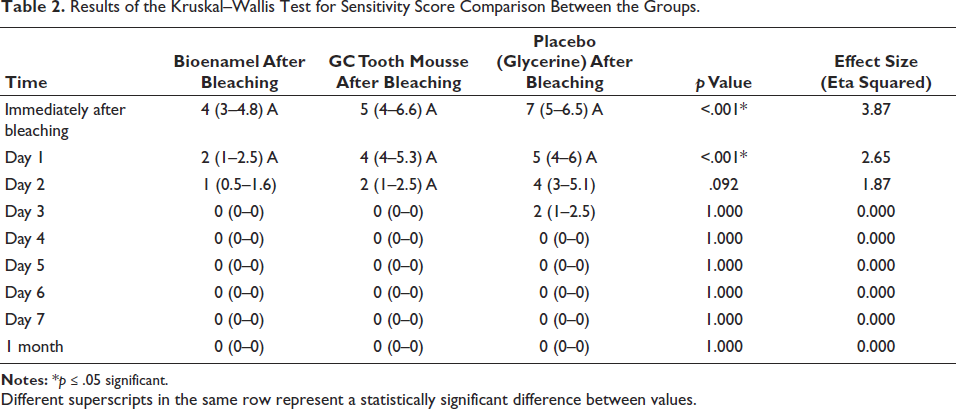
Different superscripts in the same row represent a statistically significant difference between values.
Discussion
Post-bleaching hypersensitivity is a common clinical concern, reported in approximately 55%–75% of patients undergoing in-office tooth whitening with high-concentration hydrogen peroxide agents.14, 15 This transient yet uncomfortable condition typically results from enamel porosity, dentinal tubule exposure, and pulp inflammation induced by peroxide penetration.16, 17 The present study demonstrates that Bioenamel, containing bioglass and potassium nitrate, provides significantly better desensitization outcomes compared to GC Tooth Mousse and placebo. The superior performance of Bioenamel can be attributed to its two key ingredients—bioglass and potassium nitrate—both of which contribute to rapid and effective reduction in hypersensitivity. Bioglass (calcium sodium phosphosilicate) releases calcium and phosphate ions in the presence of saliva, leading to the formation of a hydroxycarbonate apatite (HCA) layer on the tooth surface. This HCA layer effectively occludes open dentinal tubules, thereby reducing fluid movement within the tubules and blocking external stimuli that can trigger pain responses. 18 Additionally, the bioactivity of bioglass initiates a regenerative process, mimicking natural tooth mineral composition and sealing exposed dentin. Potassium nitrate, another active component in Bioenamel, acts by penetrating the dentinal tubules and depolarizing nerve endings within the pulp. This reduces the nerve’s ability to transmit pain signals, leading to a rapid decrease in sensitivity symptoms. 19
The dual mechanism of action—tubule occlusion and neural desensitization—allows Bioenamel to offer both immediate and sustained relief from post-bleaching discomfort. In contrast, GC Tooth Mousse contains casein phosphopeptide-amorphous calcium phosphate (CPP-ACP), a compound derived from milk protein casein. CPP-ACP stabilizes calcium and phosphate ions and delivers them to enamel and dentin surfaces to promote remineralization. 20 This remineralization helps reduce dentinal fluid flow, which is the principal cause of sensitivity. However, unlike Bioenamel, CPP-ACP primarily relies on passive mineral deposition, which may take longer to achieve effective tubule occlusion, thus potentially delaying symptom relief. 21 Our findings are consistent with earlier clinical and in vitro studies reporting faster relief from sensitivity with potassium nitrate-based agents than with calcium-phosphate-based systems.22, 23 Notably, all groups in this study showed a return to baseline (no sensitivity) within one week, highlighting the transient nature of bleaching-induced hypersensitivity, as also observed in previous clinical trials.24, 25
This trial’s strengths include its randomized double-blind design, the use of a placebo control, and standardized application of the bleaching protocol across all participants. Furthermore, VAS scores were collected at multiple time points to capture both immediate and delayed effects. However, this study also has certain limitations, including the relatively short follow-up period of one month, the limited sample size (N = 45), and the inability to blind the operator due to differences in the desensitizing agents’ consistency. Future studies with larger cohorts, crossover designs, and longer follow-ups could offer more robust conclusions. Moreover, the individual variation in pain threshold and the subjective nature of VAS scoring may influence sensitivity outcomes. 26 Nevertheless, this study contributes meaningful clinical insight, suggesting that Bioenamel may be the superior choice for practitioners aiming to minimize discomfort following in-office bleaching procedures.
Conclusion
Within the constraints of this study, it was discovered that the desensitizing agent Bioenamel (Prevest Denpro Limited) was the most successful in lowering post-bleaching hypersensitivity, followed by GC Tooth Mousse. All groups experience a steady decrease in hypersensitivity two days following in-office bleaching.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The institutional ethical committee, in its meeting held on 10/1/2019, has reviewed and discussed your application to conduct the dissertation proposal titled “A randomized clinical trial comparing post-bleaching hypersensitivity using two desensitizing agents” Registration Number: PMT/RDC/IEC/2019/03. Decision of the Committee: Approved, Recommended for a period of 2 years.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Self-funded.
Informed Consent
Written consent was taken before beginning the procedure.
