Abstract
Aims and objectives:
The aim of this in vitro study was to investigate the antibacterial and cytotoxic effect of cements: zinc polycarboxylate, glass ionomer, self-adhesive resin cement, eugenol-containing and eugenol-free temporary cements.
Materials and methods:
The agar-diffusion test was carried out on 4 types of bacteria: Streptococcus mutans (S. mutans; ATCC 35668), Streptococcus salivarius (S. salivarius; ATCC 13419), Streptococcus sangius (S. sangius; ATCC 10556), and Lactobacillus casei (L. casei; ATCC 27139). Freshly prepared cement samples were placed on a brain heart infusion medium and left at 37°C for 24 hours in a CO2 incubator. Ampicillin disks of 10 mg were used as positive controls. Antimicrobial effects were determined using the zone of inhibition measurement in millimeters at 24 and 48 hours. Cytotoxicity was assessed through a 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay (SERVA Electrophoresis GmbH, Heidelberg, Germany). To evaluate cell viability, an optical density microplate reader (Tecan™ Sunrise, Männedorf, Switzerland) was used at 24 and 48 hours at 570 and 630 nm, respectively.
Results:
Except the positive control group, the Adhesor luting cement showed a higher inhibitory effect on S. sangius and L. casei. RelyX Temp E temporary cement showed an inhibitory effect on S. sangius and L. casei, whereas the Cavex temporary cement showed an inhibitory effect on only S. sangius at 24 and 48 hours. At 100% concentration, Cavex temporary cement showed the least toxicity (23.6% pulp cell and 9.9% gingival cell) and highest cell viability (76.4% pulp cell and 90.1% gingival cell) at 24 hours. However, Meron, Adhesor, Panavia SA luting, and RelyX Temp E temporary cements showed the highest toxicity (above 94% gingival cell and 95% pulp cell) at 100% concentration.
Conclusions:
Meron, Adhesor, Panavia SA luting, and RelyX Temp E temporary cements may have a cytotoxic potential. None of the cements showed an inhibitory effect on S. mutan and S. salivarius, whereas Adhesor luting cement has a higher inhibitory effect than that of S. sangius and L. casei.
Introduction
Physiologically, different bacterial species live in the eco-logical system in the mouth. However, ecological changes in the oral environment through elements such as caries may cause the number of pathogenic microorganisms to increase. 1 Pathogenic microorganisms should be removed to protect oral health while preserving function, phonation, and esthetics with prosthetic tooth treatment. 2
One of the important factors for the success of all permanent prosthetic restorations is the cementation phase. 3 Nowadays, there are various types of cements that can be used in the cementation process with various dental materials.4,5 The properties of the selected cement for the cementation of the restoration affect the success of the prosthetic restoration. It is known that pathogenic micro-organisms especially in the area between the prosthetic restoration and the tooth are colonized after microleakage plays an important role in the development of undesirable effects such as secondary caries and pulpal reactions.6,7 The presence of antimicrobial and cytotoxic properties of the cement to be used in the cementation process reduces the effectiveness of pathogenic microorganisms that are the main causes of pulpal and periapical tissue diseases.7,8
In this study, it is aimed to investigate the cytotoxic properties of different luting and temporary cements used in the cementation of prosthetic restorations on pulpal and periodontal tissues and to evaluate their antimicrobial activity by using the agar-diffusion test. The first hypothesis is that 5 commercially different luting and temporary dental cements have no antimicrobial activity against 4 different microorganisms. The second hypothesis is that the 5 commercially different luting and temporary dental cements induce cytotoxic effects on gingiva and pulp living cells.
Materials and Methods
Before the preparation of cement, especially prepared Teflon™ (Wilmington, Del.; polytetrafuoroethylene) molds (2 mm height and 6 mm diameter) were sterilized in autoclave (Figure 1). For antimicrobial activity, 20 samples from each cement group were prepared according to the manufacturer’s instructions on powder and liquid propor- tions under disinfected conditions (Table 1). After the polymerization, the cement specimens were stored 37°C for 24 hours. Then, using ethylene oxide gas, all specimens were sterilized in sealed packages.
Teflon™ Molds
Material, Composition, Manufacturer, and Lot Number

The assessment of the antimicrobial activity of cements was conducted through the agar-diffusion test using the bacteria of Streptococcus mutans (S. mutans; ATCC 35668), Streptococcus salivarius (S. salivarius; ATCC 13419), Streptococcus sangius (S. sangius; ATCC 10556) and Lactobacillus casei (L. casei; ATCC 27139). Bacterial strains were propagated using the brain heart infusion medium and then extensively seeded by adding 0.1 mL of 0.5 McFarland suspensions prepared in the phosphate-buffered saline (PBS) solution to blood agar petri dishes.
The cement disks were placed at intervals on the surface of the petri dish with bacterial inoculation (Figure 2) and incubated at 37°C for 24 hours in a CO2 incubator. The bacterial growth inhibition zone diameters formed in petri dishes where 10 μg ampicillin disks were used as positive controls were measured in millimeters (mm) at 24 and 48 hours, respectively.
Five Different Cements and Control in 4 Types of Bacteria
For the cytotoxicity test, 6 specimens were prepared from each cemented sample in sterile Teflon™ molds and placed in sterile plastic tubes. After weighing each cemented sample with a precision scale, 10% fetal calf serum (FCS), 100 U/mL penicillin, 100 μg/mL streptomycin and 5 μg/mL fun-gizone were added to the tubes at 0.2 g/mL according to ISO 10993-12 guidelines into the alpha-Minimum Essential Medium (alpha-MEM, Lonza) solution, as the extraction solution (3 mL). The tubes were incubated at 37°C for 24 and 48 hours and 5% CO2 incubator. The extracts of specimens were prepared according to ISO 100993-12 (sample surface area/mL cell culture medium).
All participants provided their written informed consent to participates in this study. Healthy human third molars (extracted for orthodontic reasons) and gingival tissue (because of gingivectomy) from systemically patients were removed aseptically.
Human gingiva and pulp fibroblast cells (passage 4) were replicated in T75 culture dishes using 10% FCS, 100 U/mL penicillin, 100 μg/mL streptomycin, and 5 μg/mL fungizone-added cell culture producing alpha-MEM. After removing 80 to 90% of the confluent cell layer using trypsin/ethylenediaminetetraacetic acid, the cell suspension was prepared at 1 10 5 /mL in the culture medium and distributed as 100 μL/well in a 96-well cell culture dish. The culture dishes were incubated for 24 hours at 37°C and 5% CO2. After that, the replication medium was removed, and the wells were added at 100 μL/well, and 50%, 25%, and 12.5% dilutions were prepared from the cement extraction solution (100%) that was obtained 24 and 48 hours later and prepared with a replication medium. Culture dishes that had only the replication medium were added as a control group instead of the extraction solution and were incubated for 48 hours at 37°C and 5% CO2. At the end of the incubation period, the culture dish contents were drained, and the wells were washed 2 times by adding alpha-MEM without FCS. Then, the wells were added in the form of 100 μL/well of 1% FCS, 100 U/mL penicillin, 100 μg/mL streptomycin, and 5 μg/mL fungizone-supplemented cell culture replication medium alpha- MEM. After waiting 30 minutes at 37°C and 5% CO2 incubator, 10 μL of the 3-(4,5-dimethyl-2-thiazolyl)-2, 5-diphenyltetrazolium bromide (SERVA Electrophoresis GmbH, Heidelberg, Germany) solution was added to each of the wells at 5 mg/mL. After leaving for 3 hours in the incubator, the culture dish contents were drained, and dimethyl sulfoxide (Merck, Darmstadt, Germany) was added to the wells at 100 μL/well. The resulting color was measured by the optical density (OD) microplate reader (Tecan™ Sunrise, Männedorf, Switzerland) at 24 and 48 hours at 570 and 630 nm, respectively.
Statistical Analysis
The data of the positive control were not used in statistical analyses. The cement extraction liquid cytotoxicity was calculated using the [100 (ODTest/ODcontrol) ×100] formula. Differences between the groups were statistically analyzed by the 3-way analysis of variance (ANOVA) and Tukey honestly significant difference tests with SPSS software, SPSS, version 16.0 (SPSS, Chicago, IL, USA). The significance level was set at α = .05.
Results
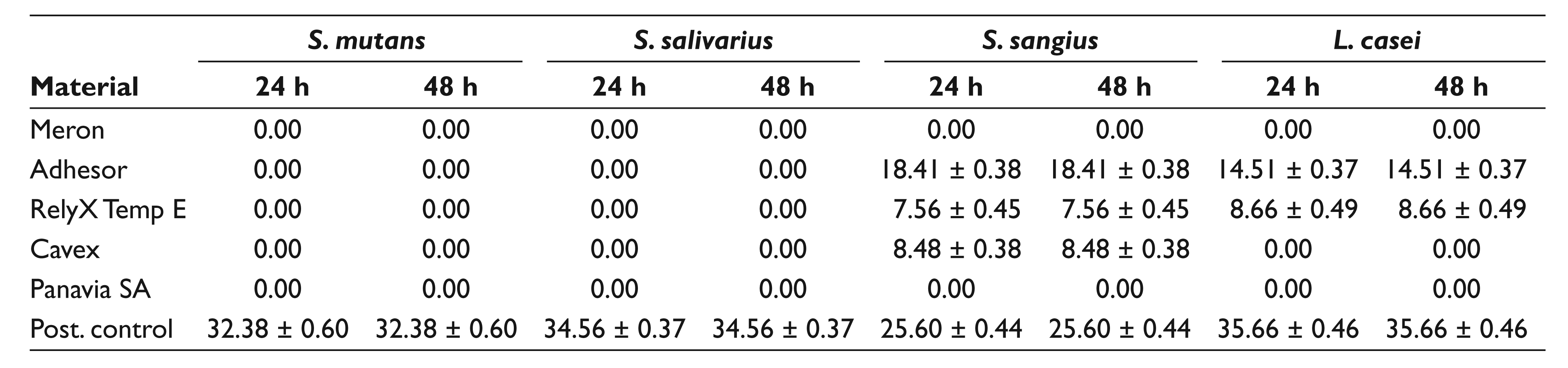
Measurements of the inhibition zone in mm at 24 and 48 hours are shown in Table 2 and Figure 3. The positive control showed the highest zone of inhibition in all the bacteria species. There was no inhibitory effect of Meron and Panavia SA luting cements against S. sangius, S. mutans, S. salivarius, and L. casei at 24 and 48 hours. Except the positive control group, the Adhesor luting cement showed a higher inhibitory effect on S. sangius and L. casei, but no inhibition effect was observed against S. mutans and S. sangius at 24 and 48 hours. Compared with other examined cements, the RelyX Temp E cement showed an inhibitory effect on S. sangius and L. casei, whereas the Cavex cement showed an inhibitory effect only on S. sangius at 24 and 48 hours.
Measurements of the Inhibition Zone (mm)
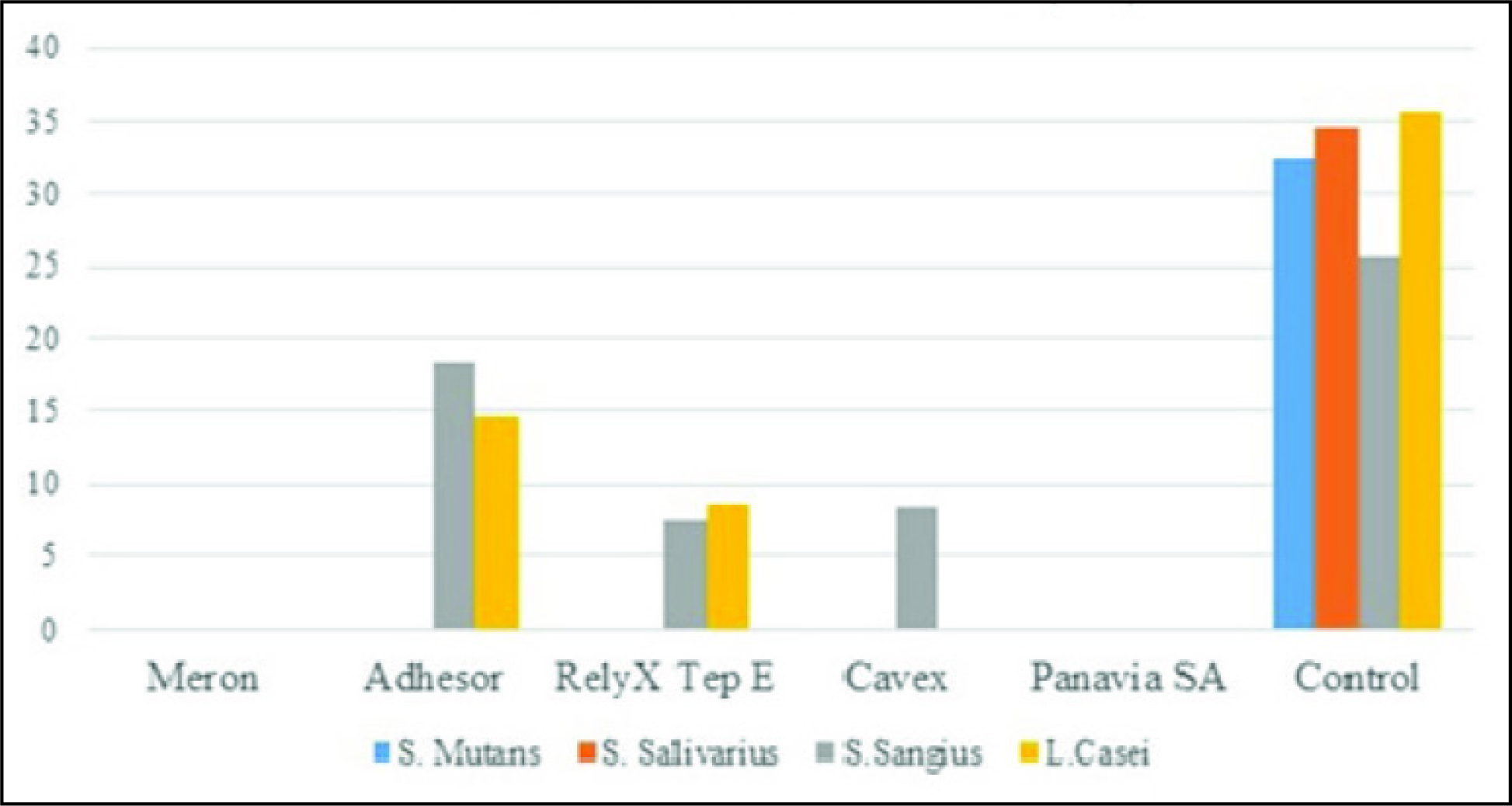
Measurements of Inhibition Zone in Millimeters at 24 and 48 Hours for Cements
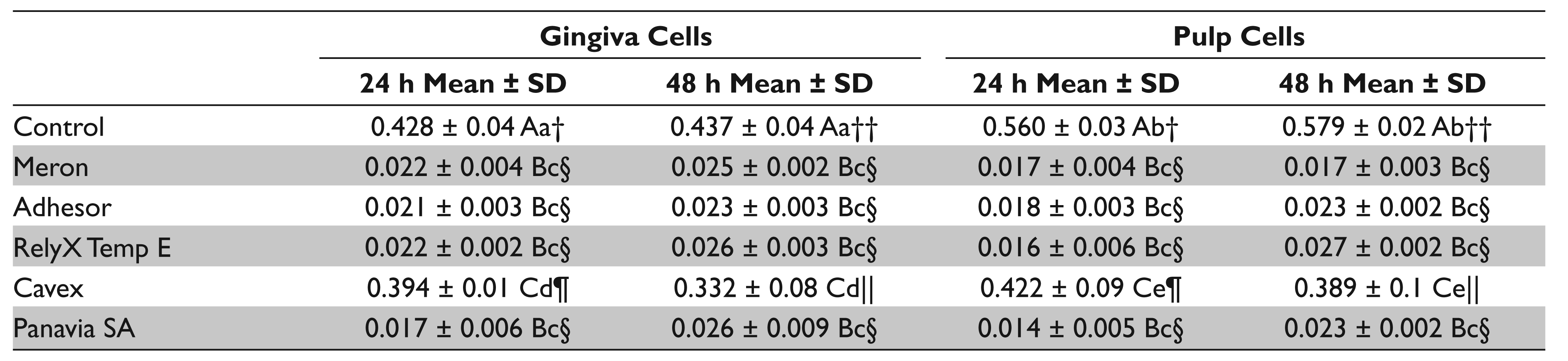
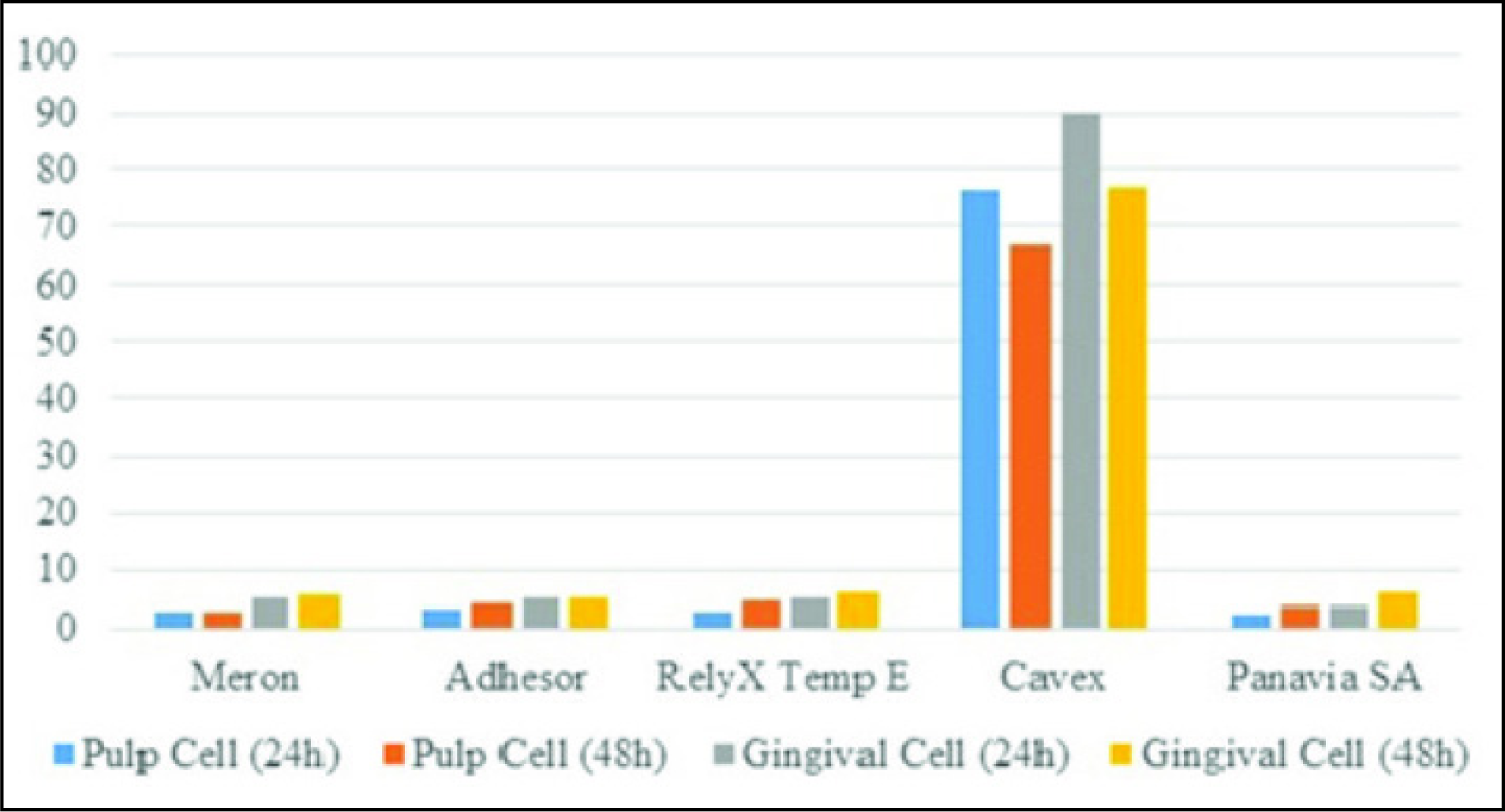
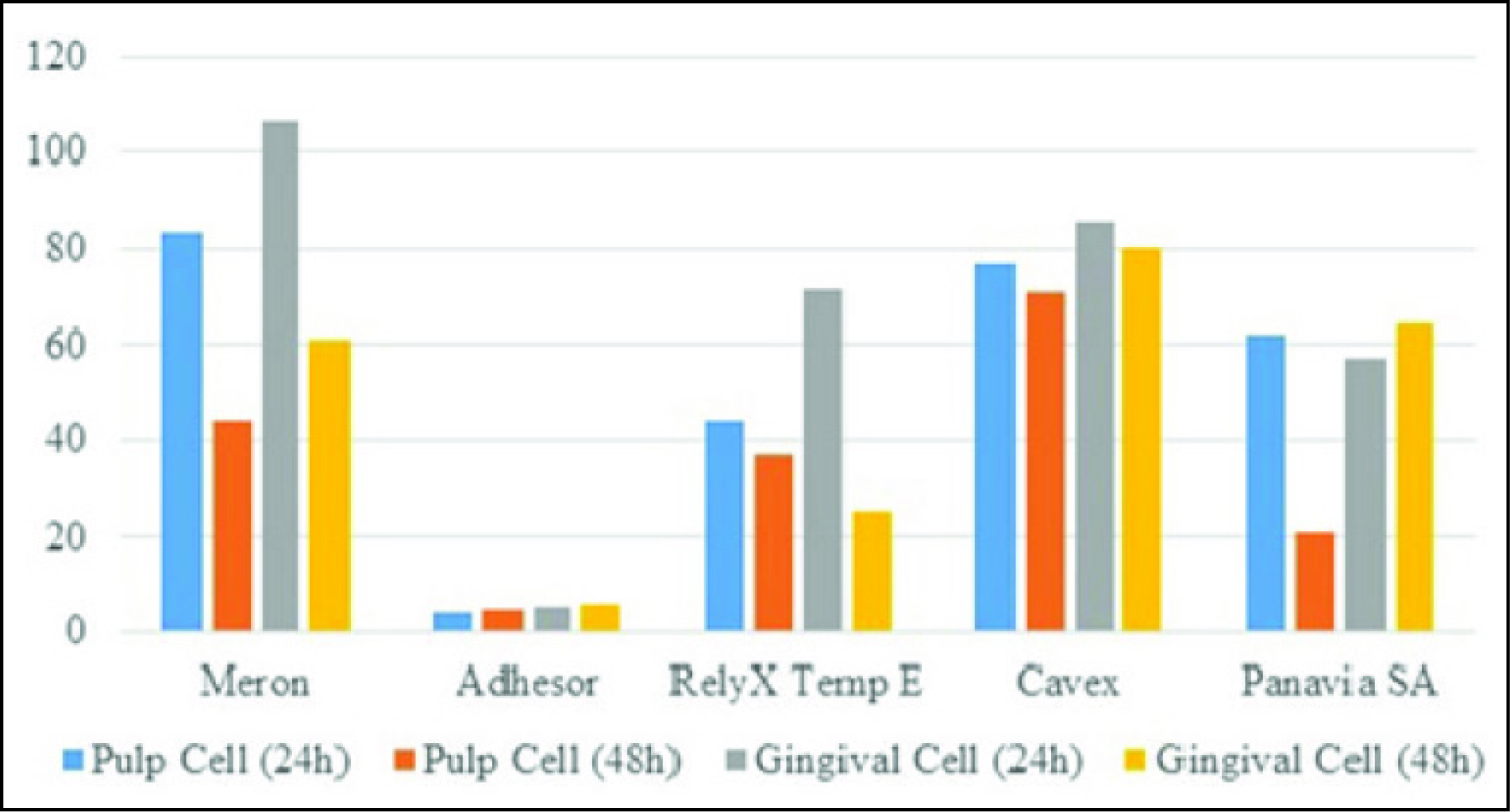
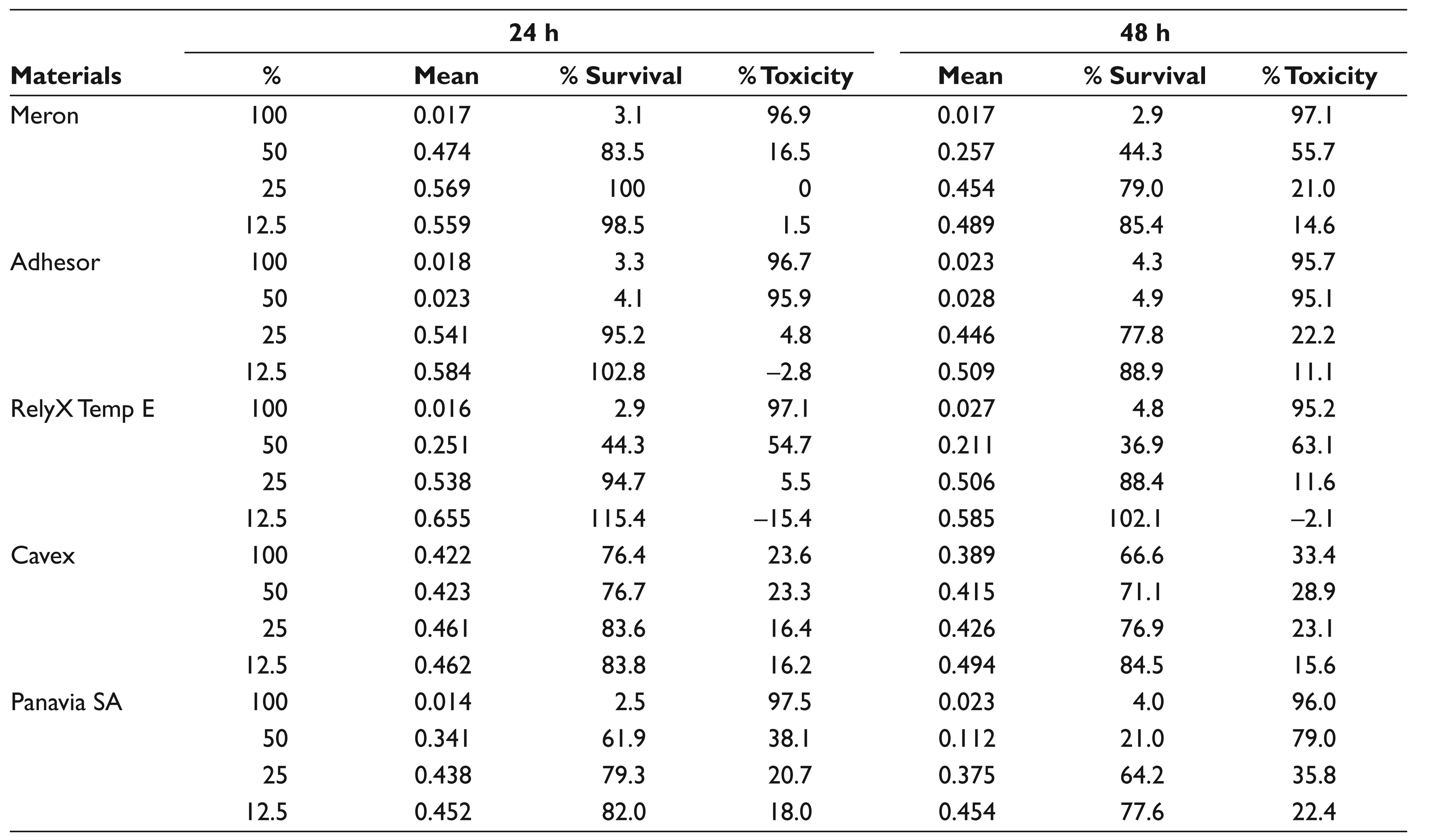
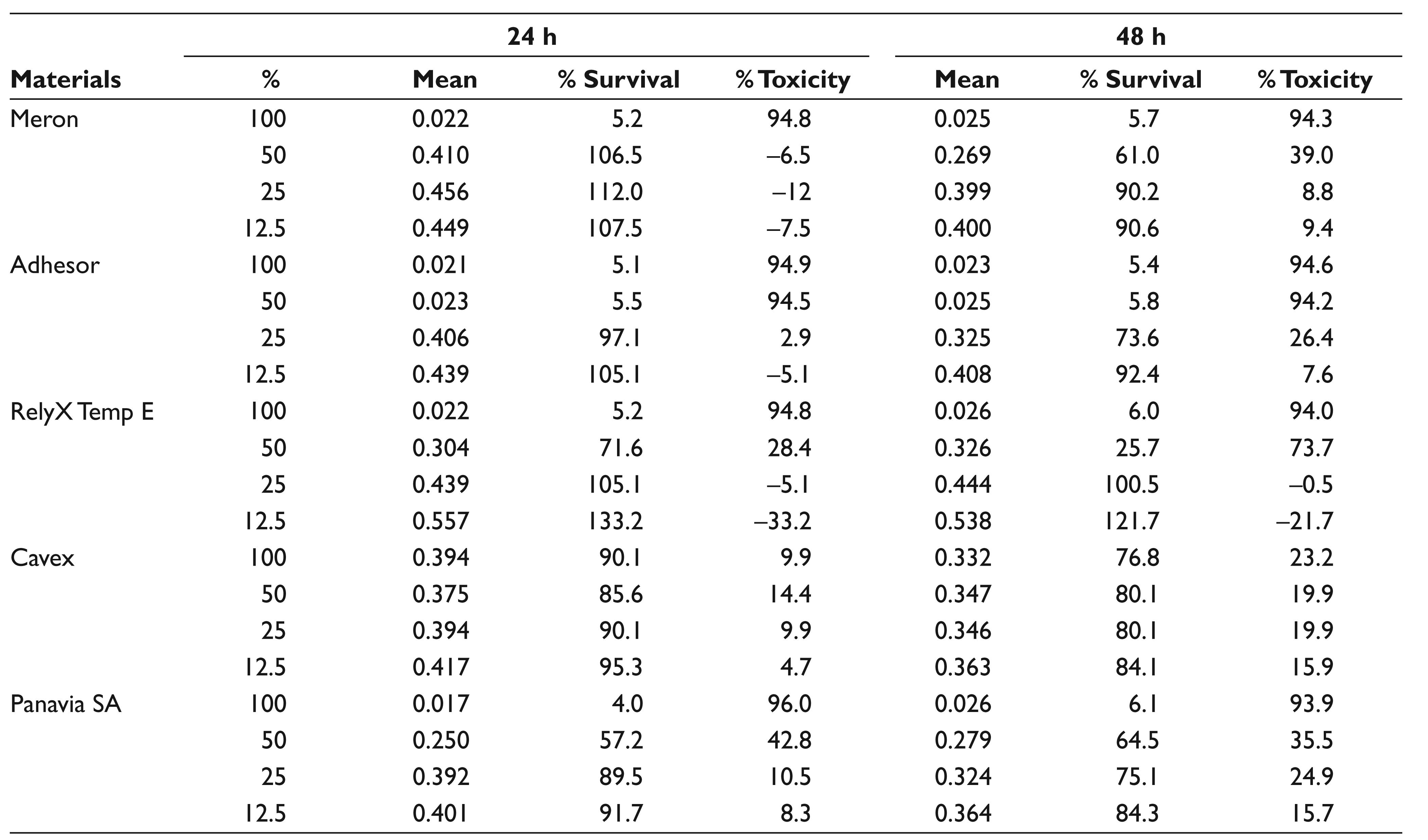
The OD values measured at 550 nm of the cytotoxic effects of the cements at 100% concentration against pulp and gingiva cells are shown in Table 3. Cytotoxicity values of 100% and 50% are shown in Figures 4 and 5, respectively. Moreover, the pulp cell and gingival cell viabilities at different concentrations of the material are shown in Tables 4 and 5, respectively.
Cytotoxic Effects of the Cements at 100% Concentration
Different uppercase letters indicate significant difference among cements and control group.
Different lowercase letters indicate significant difference among cell types (gingiva and pulp cells).
Different symbols indicate difference among times (24 and 48 h).
Percent Cell Viability (100% Concentration)
Percent Cell Viability (50% Concentration)
In Table 3, the 3-way ANOVA) results of the cytotoxic effect of the cements at 100% concentration against pulp and gingiva cells are shown; Meron, Adhesor, Panavia SA luting, and RelyX Temp E and Cavex eugenol-free temporary cements showed a significant difference when compared with the control group (P < .01). There is a statistically significant interaction between cements and cell type and that between cements and time (P < .01), whereas there is not a statistically significant interaction between the cell type and time. The results of the posthoc Tukey test, cell type, and time were showed statistically significant within the Cavex eugenol-free temporary cement group.
In Table 4, pulp cell viability at different concentrations of the material is shown; Meron, Adhesor, Panavia SA luting, and RelyX Temp E temporary cements showed the highest toxicity at 100% concentration; the toxicity was above 95%. At 100% concentration, Cavex temporary cement showed the least toxicity (23.6%) and highest cell viability (76.4%) in 24 hours. At 50% concentration, Adhesor luting cement indicated the least cell viability (4.1%) in 24 hours, while Meron luting cement displayed the highest cell viability (83.5%) in 24 hours. At 25% con-centration, Meron luting cement demonstrated high-er cell viability (
Pulp Cell Viability at Different Concentrations of the Material (Cytotoxic Effects)
In Table 5, gingival cell viability at different con-centrations of the material is shown; Meron, Adhesor, Panavia SA luting, and RelyX Temp E temporary cements showed the highest toxicity (above 94%) at 100% con-centration. At 100% concentration, Cavex temporary cement showed the least toxicity (9.9%) and the highest cell viability (90.1%) in 24 hours (Figure 4). At 50% con-centration, Meron luting cement displayed the highest cell viability (106.5%) in 24 hours, and Adhesor luting cement showed the least cell viability (5.5%) in 24 hours (Figure 5). At 25% concentration, Meron luting cement demonstrated a higher cell viability (112%) and less toxicity than the other cements in 24 hours. At 12.5% concentration, RelyX Temp E temporary cement exhibited the highest cell viability (133.2%) in 24 hours.
Gingival Cell Viability at Different Concentrations of the Material (Cytotoxic Effects)
Discussion
Zinc polycarboxylate luting cement showed the strongest antibacterial effects, whereas zinc-containing zinc oxide eugenol and noneugenol temporary cements showed a limited antimicrobial effect. Thus, the first hypothesis that 5 commercially different luting and temporary dental cements have no antimicrobial activity against 4 different microorganism was partially rejected.
Meron, Adhesor, Panavia SA luting, and RelyX Temp E temporary cements showed the highest toxicity, whereas Cavex temporary cement showed the least toxicity and highest cell viability. Thus, the second hypothesis that the 5 commercially different luting and temporary dental cements induce cytotoxic effects on the gingiva and pulp living cells was partially accepted.
Antimicrobial and cytotoxic properties of cements prevent pulpal and periodontal tissue damage and formation of secondary caries after microleakage during restorations after permanent prosthetic treatment.7,8 The 2 most important tests for examining the antimicrobial effects of restorative materials are the agar-diffusion test and the direct contact test. 9 Although the agar-diffusion test is an effective method for examining the antimicrobial efficacy of restorative materials, it has several disadvantages.10,11 Its biggest disadvantage is that it is difficult for the test material to dissolve and diffuse into the study medium. In general, the dissolution of restorative materials and their minimum level of diffusibility make it difficult to study antimicrobial effects with the agar-diffusion test. 11
In this study, we used 4 different bacterial species: S. salivarius, S. sanguis, S. mutans, and L. casei. S. salivarius and S. sanguis are common inhabitants in the healthy oral cavity and initial colonization of dental surfaces related to low caries risk. However, S. mutans and L. casei species are considered to be related to dental caries due to acid production.12,13
Farret et al 14 investigated the antimicrobial effects of chlorhexidine gluconate solutions added at different concentrations in liquids of KetacCem Easymix and Meron glass ionomer cements on cements by the agar-diffusion test. In their study, they have reported that Meron cement does not cause any antibacterial effects on S. mutans without the addition of chlorhexidine gluconate into its liquid. Lewinstein et al 11 examined the antimicrobial effects of 3 different dental adhesive cements by using both the agar-diffusion test and the direct contact test. They reported that glass ionomer and zinc polycarboxylate dental adhesive cements did not show any antimicrobial effect in the agar-diffusion method. Researchers have attributed antimicrobial effects of glass ionomer cements to fluoride release properties and reported that the remineralization of dentin that is affected from caries is also responsible for fluorine release.15,16 Vermeersch et al 15 have reported that the antimicrobial effects of glass ionomer cements originate from the acidic area formed by the diffusion of F–, Ca+2, Al+3, and OH– ions from the material. Seppa et al 16 have shown that glass ionomer cements can affect electrolytic metabolism of S. mutans by acid formation. In some studies, it has been reported that the conventional glass ionomer cement shows an antimicrobial effect within 24 hours.17,18 However, as the result of this study, we observed that the antimicrobial effects of Glass ionomer cement (Meron) and self-adhesive resin cement (Panavia SA) were not sufficient for bacterial strains. This may be caused by the fact that the cements did not dissolve during the test and could not diffuse into the blood agar.
Various kinds of dental luting materials such as zinc polycarboxylate, zinc phosphate, and zinc oxide eugenol contain different proportions of zinc ions due to inhibiting the grown of bacteria ability. 19 Release zinc ions enhance the proton permeability of the bacterial cell membrane and inhibit glycolytic enzyme in bacterial cells. Zinc poly-carboxylate cement has a high portion of zinc than zinc oxide eugenol and noneugenol cements. 20 In this study, zinc polycarboxylate luting cement showed the strongest antibacterial effects, but it has an effect on only S. sangius and L. casei. Less zinc-containing zinc oxide eugenol (RelyX Temp E) and noneugenol temporary cements (Cavex) showed a limited antimicrobial effect. Zinc oxide eugenol temporary cement exhibited a weak antibacterial effect on S. sangius and L. casei, while the noneugenol temporary cement showed a weak antibacterial effect only on S. sangius.
Some in vitro studies have shown that glass ionomer cement releases a highly toxic chemical material to cell after mixing.21,22 Also, Kanjevac et al 23 concluded that highly released flour ions cause a highly cytotoxicity effect on cells. In this study, glass ionomer luting cement (Meron) showed a highly toxic effect on the gingiva and pulp cells at 100% concentration. However, Meron luting cement displayed the highest cell viability (106.5% gingival cell and 83.5% pulp cell) in 24 hours at 50% concentration. At 25% concentration, Meron luting cement demonstrated a higher cell viability (112% gingival cell and 100% pulp cell) and less toxicity than the other cements in 24 hours. The highest cell viability at 50% and 25% concentrations may be related to releasing the high-level flour ions that cause an effect on the gingival or pulp cells. Highly toxic effect on the gingiva and pulp cells at 100% concentration may be related to a high pH level due to the acidic property of the material.
Temporary cements with and without eugenol are widely used materials in the temporary cementation of prosthetic restorations. Based on several studies, temporary cements, both with and without eugenol, show cytotoxic effects at different levels.24,25 Kwon et al 26 evaluated the temporary cements with and without eugenol for cyto-toxicity against different human fibroblast cells. They have shown that both cements have cytotoxic effects at different ratios. Also, Malkoç et al 24 reported that temporary cements with and without eugenol showed cytotoxic effects at different ratios in their study in which they investigated the cytotoxicity of temporary cements on animal pulp cells. However, Ülker et al 27 have reported that the eugenol-containing temporary cement is not cytotoxic to animal-derived pulp cells without the dentin barrier.
In this study, cytotoxicity values of different luting and temporary cements on the gingiva and pulp cells after 24 hours and 48 hours were evaluated by reading the color OD. Meron, Adhesor, Panavia SA luting, and RelyX Temp E temporary cements showed the highest toxicity (above 94% gingival cell and 95% pulp cell) at 100% concentration. However, Cavex temporary cement showed the least toxicity (23.6% pulp cell and 9.9% gingival cell) and highest cell viability (76.4% pulp cell and 90.1% gingival cell) in 24 hours at 100% concentration.
Based on filled polymers, self-adhesive resin cements are hybrid and do no require a separate etch and bonding step. Methacrylate monomer is a component of self-adhesive cement and used in different dental base materials such as Bis-GMA, UDMA, TEGDMA, HEMA, and others.28,29 However, previous studies claimed that using UDMA and TEGDMA materials have a highly cytotoxic effect and can cause DNA damage on cells. 30 Kleinsasser et al 31 reported that TEGMA has a higher genotoxic effect on human lymphocytes than UDMA using comet assays. On the contrary, Urcan et al 32 and Arossi et al 33 observed that UDMA has a greater genotoxic effect on human gingival fibroblasts than TEGMA using the sensitive γ-H2AX DNA immunofluorescence and somatic mutation and recombination test. Also, Schmid-Schwap et al 34 concluded that self-adhesive resin cement is more cytotoxic than adhesive resin cement. Thus, pulp cell metabolism could change when a direct or indirect contact between pulp tissue and self-adhesive cement occurs. 35 In accordance with previous study results, self-adhesive luting cement (Panavia SA) including the TEGDMA component showed highly cytotoxic effects on the gingival cells (0.017 ± 0.006) and pulp cells (0.014 ± 0.005) at 100% concentration in 24 hours.
Conclusion
Meron, Adhesor, Panavia SA luting, and RelyX Temp E temporary cements may cause cytotoxic effects. This result should be considered when selecting cement for temporary and permanent luting.
Within the limitations, the present study concluded that zinc polycarboxylate luting cement (Adhesor) showed the strongest antibacterial effects, but it has an effect only on S. sangius and L. casei. Less zinc-containing zinc oxide eugenol (RelyX Temp E) and noneugenol temporary cements (Cavex) showed a limited antimicrobial effect. RelyX Temp E temporary cement exhibited a weak antibacterial effect on S. sangius and L. casei, while the Cavex temporary cement showed a weak antibacterial effect only on S. sangius.
At 100% concentration, Cavex temporary cement showed the least toxicity and highest cell viability at 100% concentration in 24 and 48 hours. However, Meron, Adhesor, Panavia SA luting, and RelyX Temp E temporary cement showed the highest toxicity.
At 50% and 25% concentrations, Meron luting cement displayed the highest cell viability and less toxicity in 24 hours.
Footnotes
Acknowledgements
This study was supported by the scientific research projects, Department of Recep Tayyip Erdoğan University (2015/357). It was presented at the 105th Annual World Dental Congress (FDI), Madrid, Spain, August 29 to September 1, 2017.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
