Abstract
Purpose
We aimed to evaluate the extent of collagenase clostridium histolyticum (CCH) diffusion in Dupuytren’s contracture (DC) for tissues outside of the contracture cord using Magnetic Resonance Imaging (MRI) immediately after CCH injection.
Keywords
Introduction
Dupuytren’s contracture (DC) is a progressive flexion deformity of the fingers caused by thickening and shortening of the palmar fascia. 1 Collagenase clostridium histolyticum (CCH: Xiaflex®) is considered a nonsurgical option for patients with DC. Compared to surgical treatment, CCH is less invasive, 2 allows earlier return to work, costs less, and provides equivalent efficacy. 3
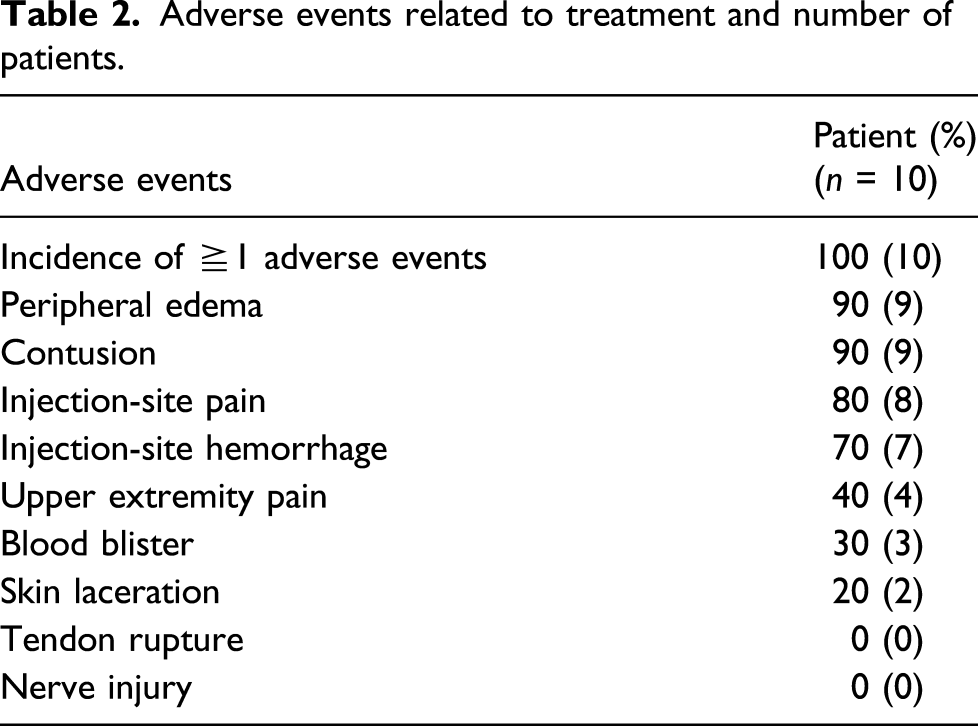
Randomized phase 3 clinical trials on CCH treatment have reported peripheral edema of the affected finger (72.5–77.8%), contusion (51.0–73.3%), injection-site pain (32.4–37.8%), and skin laceration (10.8%) as common adverse events.4,5 Most of these adverse events are mild to moderate in severity, local to the injection site, and transient. 6 In contrast to common adverse events, severe adverse events such as tendon rupture, pulley rupture, ligament injury, vasospasm, and vascular injury have been reported in the literature.7,8 Although the incidence of severe adverse events is less than 1%, these injuries may require surgical treatment and place considerable burden on patients.
Collagenase clostridium histolyticum is made of a purified mixture of AUX-I and AUX-II collagenases that can potentially affect normal tissue or dissolve collagen-containing structures indiscriminately; however, little is known about the diffusion of CCH solution after injection in normal tissues, including tendons and neurovascular bundles around the contracture cord. Yacoe et al. 9 described the appearance of DC in Magnetic Resonance Imaging (MRI) and demonstrated a good visualization of its location and extent. In a previous study, MRI demonstrated cord discontinuity, reduction of cord volume, a significant increase in cord signal intensity, and a slight increase in flexor tendon signal intensity with no significance after 1 month of CCH injection. 10
This study aims to radiologically evaluate diffusions of CCH and quantitative changes around the contracture cord after injection of CCH. Using MRI as soon as possible after CCH injection for DC, we investigated the characteristic changes and diffusion of CCH injections. Our central hypothesis is that leakage of CCH from inside the cord could be a causative agent of damage to healthy tissue, tendon, and neurovascular bundle.
Materials and methods
This study was conducted as an open-label prospective cohort trial. The recruitment period was between April 2016 and March 2017, and 10 patients with DC were enrolled in this study. The inclusion criteria were patients with DC over 20 years of age with a subcutaneously palpable contracture cord in the metacarpophalangeal (MCP) joints. Patients were excluded if they 1) could not undergo MRI, 2) presented a history of hypersensitivity to the components in CCH, 3) could not provide informed consent, or 4) opted for surgical treatment. This research was performed in compliance with the Helsinki Declaration and has been approved by the IRB of the Institutional Ethics Review Board of the Shinshu University Hospital (approval number 3462). Informed consent was obtained from all patients prior to the study.
Subjects
Between April 2016 and March 2017, a total of 22 patients (male, 20; female, two) were diagnosed with DC during the recruitment period. The following patients were excluded: five patients (male, four; female, one), did not require treatment due to lack of extension deficit of the affected finger; two patients, opted for surgical treatment due to multi-finger contractures; one patient, preferred treatment at a local hospital; one patient, unaffected MCP joint; one patient, unable to undergo MRI due to pacemaker implantation; two patients, opted not to participate in the study. Following these exclusions, 10 male patients with a mean age of 70.7 years (range, 57–79) who presented a palpable cord of the MCP Joints were included in this study. The joints that underwent treatment were as follows: little finger, five cases; ring finger, three cases; middle finger, two cases. Extension deficit were as follows: MCP joint, 10–60° (mean, 34.3°); proximal interphalangeal joint (PIP) joint, 0–60° (mean, 26.6°). All patients had no past history of peripheral vascular disease, bleeding problem, anticoagulant intake, or trauma and surgical history of the affected finger. Patients only underwent treatment by CCH injection once during the three-month observation period.
Injection technique
Injections were performed by two board-certified hand surgeons according to the technique recommended by the manufacturer. A 0.58 mg dose of the enzyme (diluted solution; 0.25 mL) was injected into the DC cord of the MCP joint in all patients. A 27G needle was used for CCH injection. In order to prevent the injection needle from penetrating into the flexor tendon, the depth of penetration was set within 3 mm of the subcutaneous. One-third of the dose was administered thrice in close but separate needle insertions. Following the initial injection, the second and third injection sites were moved approximately 2–3 mm from opposing sides of the initial injection. Stretching maneuvers were performed at 24 h after injection.
Night splint therapy was performed up to 4 months after drug administration in all cases. Evaluations were conducted at pre-injection, post-passive extension maneuver, one-week post-injection, one-month post-injection, and three months post-injection. Affected fingers were assessed for their extension, fixation angle, and adverse events at each visit.
MRI evaluation
Scan parameters of each MR sequence.

TR: repetition time; TE: echo time; TI: inversion time; FOV: field of view; CCH: collagenase clostridium histolyticum; STIR: short-T1 inversion recovery.
The presence of a signal change inside or outside of the cord was evaluated. When a signal change was subsequently observed outside of the cord, the intensity, broad pattern, and contact with the surrounding tissues such as the neurovascular bundle and flexor tendon were noted and included in the evaluation criteria. In addition, we evaluated the presence of signal changes inside the joint, across the joints, on the dorsal side of the subcutaneous tissue, perineurovascular bundle, and inside the flexor tendon. The presence of signal change was evaluated by comparing the pre- and post-injection images of each patient, investigating their signal intensity, contrast, edematous changes, and inflammatory changes.
Results
Active extension of the joint
After 1 week of injection, the mean change in contracture was 32.5 (range, 10−50°; SD 12; 94.7%) degrees for the MCP joint and 19.8 (range, 0−45°; SD 22.3; 74.4%) degrees for the PIP joint. As CCH was injected into the DC of the MCP cord in all cases, we believe that the contracture of the PIP joint changed only as a secondary effect. In nine out of 10 cases, the extension deficit decreased 0–5°of full extension for the MCP joint. At 3 months after injection, two of 10 cases were confirmed with decreased extension angle of the MCP joint; however, all cases were within five degrees of extension loss. All cases demonstrated an increased arc of motion.
Adverse events
Adverse events related to treatment and number of patients.
MR findings
On MR images before CCH injection, the contracture cord of the affected finger showed low to intermediate intensity on T1- and T2-weighted images. The contracture cord was positioned subcutaneously parallel to the flexor tendon. The mean distance between the contracture cord and flexor tendon at the injection level was 7.4 mm (7.0–7.9) on the middle finger, 4.1 mm (1.6–6.1) on the ring finger, and 4.4 mm (2.6–6.3) on the little finger. There were neither mass lesions nor aneurysms at the injection level in all cases.
On MR images within 15 min after injection, the signal change was observed outside of the cord in all cases. The area of signal change showed a high intensity that extended outside of the contracture cord on STIR images. A closer injection site demonstrated a higher contrast and greater density in the area of signal change. The signal change area tended to spread concentrically around the cord in axial view. In the signal change area, the continuity from the injection site was confirmed, and the area was in contact with the flexor tendon and neurovascular bundle in all 10 cases. In axial view, the signal change area spanned a mean of 7.9 mm (6.9–9.2 mm) in depth and 11.4 mm (7.5–18.0 mm) in width when measured from the injection site. In sagittal view, the signal change area located between the flexor tendon and the layer of palmar fascia measured a mean of 25.4 mm (21.3–35.4 mm) longitudinally and spanned distally and proximally beyond the injection level. T1-weighted and T2*3D MEDIC images show no clear signal alteration around the cord but demonstrated a good visualization of the location of cord, flexor tendon, and neurovascular bundle (Figure 1). MR images within 15 min after collagenase clostridium histolyticum (CCH) injection. Injection site: block arrow. Flexor tendon and neurovascular bundle: arrow. (a) Axial short-T1 inversion recovery (STIR) image. (b) Sagittal STIR image. (c) Axial T2*3D MEDIC image. (d) Axial T1-weighted image.
The signal change area expanded along the tendon sheath but no signal changes were observed beyond the tendon sheath nor inside the flexor tendon, suggesting that the tendon sheath may serve as a protective barrier from CCH reaction. In all cases, no signal changes were observed inside the joint or in the dorsal aspect beyond the joint. Furthermore, no signal changes were observed within the flexor tendon or perineurovascular bundle.
In the affected hand, 24 h after injection of CCH, the area of contusion was more extensive than the signal change area in MRI (Figure 2). In all cases, contusion or appearance and location of swelling did not correspond to the signal change area on MR images. 74 year-old male with DC in ring finger. (a) Axial STIR image within 15 min after injection. (b) Sagittal STIR image show high-intensity area mainly located at the deeper layer of the cord. (c) 24 h after CCH injection.
On MR images after 3 months of injection, these signal changes were no longer observed. Additional signal changes were not observed within the flexor tendon nor the neurovascular bundle (Figure 3). (a) Magnetic resonance imaging (MRI) within 15 min after injection. STIR images (middle, right) show high-intensity signal changes around the cord/injection site (block arrow). (b) MRI after 3 months injection. STIR high signal changes were no longer observed.
Discussion
The principal result of our study is that high-intensity signal change on STIR images was observed outside of the cord in all 10 cases, and a signal change expanded spherically and eventually contacted the tendon and neurovascular bundle. Our results revealed the CCH solution diffuses outside the cord and expands towards the surrounding normal tissues, tendon, and neurovascular bundle in the early phase after administration, suggesting that CCH could persistently affect healthy tissues until CCH inactivates its enzyme process. To our knowledge, this is the first report to evaluate the diffusion area after CCH injection for DC patients. A previous report has described that edema and plasma protein extravasation changes were observed following CCH injection into the subcutaneous tissue of a rat hind paw model and reached its maximum change within 30 min of injection. 11 STIR sequences have been shown to be the most sensitive imaging modality for accentuating fluid and edema in detecting pathologic changes of bone and soft tissue for the hand and wrist. 12 Therefore, in the current study, we interpret the areas of signal change on STIR as either inflammatory edema of the soft tissue, the CCH solution itself, or a mixture of the two.
No serious adverse events of tendon rupture or neurovascular injury were observed in this study. The mean decrease of contracture was 32.5° (94.7%) for the MCP joint and 19.8° (74.4%) for the PIP joint. Hirata et al. reported that the mean percentage decrease was 97% for the MCP joint, and that no tendon rupture was observed for primary MCP joints in 77 non-Caucasian patients. 13 Our clinical efficacy was comparable to previous reports. Our results also revealed that the occurrence of severe adverse events was neither inherent to the diffusion of CCH solution nor its subsequent contact with the tendon or neurovascular bundle; thus, this study also provided some confirmations about the safety of CCH, and all cases that underwent CCH injection likewise suggested the potential risk of tendon and neurovascular damage.
Several case reports on complications have suggested that tendons and vascular nerve bundles are vulnerable to CCH. Wozniczka et al. 14 reported a case with a history of Raynaud’s phenomenon that showed a loss of distal circulation and required finger amputation after CCH injection. In another case report, Spiers et al. 8 reported on an arterial injury to the PIP joint of the left little finger, suggesting that an injection into a cord of the middle phalanx from both sides of the digit may compromise circulation. A report on two cases of tendon rupture of the little finger by Zhang et al. 15 described and suggested that CCH should be administered carefully in the PIP of the little finger due to the close proximity between the skin and tendon. Khurana et al. 16 reported on a tendon rupture of the ring finger as a result of being hit on a hard surface at 8 weeks after CCH injection of the little finger and at 2 days after injection into ring finger. In our study, the high signal area spanned a mean of 11.4 mm in width and 7.9 mm in depth in axial view, and 25.4 mm longitudinally in sagittal view. Neither serious adverse effects nor patients with underlying disease were observed in our study; however, the tissues in this high signal area could potentially be vulnerable. A measure of caution should be applied to cases with peripheral vascular disease, trauma, surgical history, as well as cases involving the PIP of the little finger and cases undergoing multiple administrations of CCH. We believe that this study demonstrated results that confirmed the risk of incidence as discussed in prior case reports. If adequate distance is maintained from the contracture cord to the tendon or if the tendon and vascular strength presents no abnormalities, the risk of adverse events after CCH injection may not exceed the benefit of treatment. However, it is important to emphasize that the CCH solution spread outside of the cord can contact the tendon sheath and neurovascular bundle at an early phase after injection. Thus, all practitioners should note the possibility that CCH injections could result in severe complications.
Further studies are warranted to develop measures for reducing the incidence of injury to the tendon and neurovascular bundle after CCH injection. The predominant collagen subtypes in DC contracture cords are collagen type I and III, the former of which is the main component of tendon, dissolved by CCH solution. A number of in vitro and in vivo findings have described the effects of CCH on the neurovascular bundle, which is mainly collagen type IV, but no consensus has been reached.17,18,19,20 Reconsidering an appropriate dosage for the affected finger and joint may lead to reduced risk. Some clinical studies have reported satisfactory clinical outcomes when modified injection techniques were applied, including a higher dose of CCH in one session. 21 Further expertise should be sought to update procedures based on safety and effectiveness. Moreover, the use of MRI before or after injection is beneficial for risk assessment of severe injury. Hurst et al. reports that the distance of the flexor tendon from the skin is 7 mm at the MCP joint and 4 mm at the PIP joint, and determining a suitable depth for insertion of the needle tip is important. 22 Prescribing information recommends that extension maneuvers should be gently performed for 10–20 s with the patient’s wrist in flexion. Wrist flexion provides space for tendinous and neurovascular structures to avoid tension.
The main advantage of this study was that the investigation was conducted within 15 min after injection, which allowed us to directly evaluate the diffusion due to the administration of the CCH and to successfully minimize the effect of diffusion time. Fifteen minutes was the least possible time required to transfer a patient for MRI after CCH injection; furthermore, the duration may provide enough time for the CCH to dissolve the cord with enzyme reaction and diffuse outside of the cord. 23 High signal intensity on STIR images is observed when detecting abnormalities in muscle and soft tissues, indicating inflammation and edema.24,25 STIR is a fluid-sensitive MRI sequence with high signal intensity,26,27 suggesting permeation of the CCH solution. MRI was a useful method to locate the DC cord and its surrounding tissues in this study.
This study has several limitations. First, its small sample size may potentially generate a selection bias. Second, the all-male sample population may introduce a sex bias, and the presence of sexual difference in results remains unknown. However, all patients exhibited a permeation of CCH reaction outside the cord on MR image and subjective improvement of extension deficit without severe complications. Third, a conclusive statement on CCH enzymic reaction cannot be drawn due to the absence of control subjects. Based on previous studies, we believe that a high signal intensity outside the contracture cord describes inflammatory and edematous changes and/or CCH fluid. Fourth, the study duration was limited to 3 months, and future studies should investigate longer-term outcomes. To the best of our knowledge, there have been no previous reports of severe adverse events or flexor tendon ruptures at more than 3 months after a single CCH injection. Lastly, in our study, MR images detected an area of signal change that contacted its surrounding tissue in all cases; however, the peak of the sequential expansion in the signal change was unclear.
In the future, our results should be further examined and verified to address time-dependent changes.
Footnotes
Acknowledgements
We would like to express our gratitude to Sho Steven Sugita at OrthoTranslations for providing professional editing and proofreading services.
IRB approval
This research was performed in compliance with the Helsinki Declaration and was approved by the Institutional Ethics Review Board of Shinshu University Hospital (approval number 3462).
Declaration of conflicting interest
Shinshu University School of Medicine received an honorarium from Asahi Kasei Pharma Corporation. The following authors declare potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Hiroko Iwakawa, Masanori Hayashi, and Jun Takahashi.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by Asahi Kasei Pharma Corporation.
