Abstract
Non-union is a taxing complication of fracture management for both the patient and their surgeon. Modern fracture fixation techniques have been developed to optimise the biomechanical environment for fracture healing but do not guarantee union. Patient biology has a critical role in achieving union and stem cell therapy has potential for improving fracture healing at a cellular level to treat or avoid non-union. This article reviews the current understanding of non-union, concepts in bone healing and the current literature on the application of stem cells in non-union.
Introduction
Fracture non-union continues to pose a heavy burden on patient quality of life and the health system. Fractures are the most common cause for hospital admission following injury in Australia 1 with non-union affecting 5–10% of all fractures. Even higher rates of non-union are encountered in specific injury patterns and locations. 2 A recent analysis of lower limb related trauma-centre admissions found that 41% of patients with non-union were still unable to return to work after 12 months from injury despite modern fracture management techniques. 3 The median cost to the health system for managing an inpatient with non-union was 14,957 AUD after initial costs of acute fracture management. 1
Stem cells represent a new biological therapy that theoretically promises to revolutionise management of fracture non-union. We can expect benefits such as improved successful treatment rates, quicker recovery times, reduced patient morbidity and potentially reduced cost of managing non-union.
Current stem cell therapy for non-union is still experimental. Prior to using stem cell therapy for non-union management on a broad scale, it is essential to clearly understand how stem cells affect the natural history of non-unions, determine the side effects of this specific stem cell therapy, standardise the treatment parameters and systematically report the outcomes of therapy. Given the complexity of patient and non-union factors (i.e patient biology, anatomical area, injury specifics and initial treatment specifics), stem cell research must be investigated on meticulously selected patients with non-union. Unselected use of stem cell therapy for simply any non-union indication is unethical and likely leaves the outcome to chance with increased cost. This practice is potentially dangerous to the patient with no clear understanding of the benefit to be gained. It is worthy to note that autologous bone grafting with cancellous bone, which represents the gold standard of non-union management, does contain all potential active cells considered for non-union and a proven form of ‘stem cell therapy’ but not the focus of this review.
This paper reviews the current understanding of how stem cells can augment fracture healing and analyses the available literature on stem cell therapy for human fracture non-union management.
Defining non-union
Non-unions occur when the physiological process of fracture healing is unable to overcome the local biological and mechanical stresses related to the fracture. 4 We define a non-union as a fracture that has failed to heal in the expected time frame and will not heal without intervention. The time component in the definition is necessary purely because there is no available test to determine the fate of a fracture without first giving it a chance to heal. The expected duration of healing for long bone fractures has been approximated by the Perkin’s Timetable. This states that spiral fractures of the upper limb consolidate (visible union on radiographs and resolution of pain when fracture is stressed externally) in 6 weeks. The time is doubled lower for limb fractures and doubled again for transverse fracture patterns. 5
The United States Food and Drug Administration (FDA) defines non-union as a fracture that has not healed in 9 months or when there has been no radiological progression of healing for 3 consecutive months. The problem with this definition is the long duration prior to classification as a non-union. Others have endeavoured to classify non-unions too. Calori et al have created a non-union severity score to aid management and research. Their Non-Union Scoring System (NUSS) aims to evaluate the cause of the non-union and guide management. 6 The NUSS evaluates 16 different aspects of bone quality, soft tissue status, surgical fixation factors and patient comorbidities to give four grades of non-union. Each grade escalates further in management complexity ranging from improving fixation stability, use of biological augments to limb amputation. 6,7
The Weber-Cech classification divides non-unions based on radiographic features and vascularity of the fracture margins into three groups. Hypertrophic non-unions have abundant callus formation with bulging of the fracture margins. These non-unions have good vascularity of the fracture margins and need only more mechanical stability to progress to union. Atrophic non-unions demonstrate a lack of callus formation with resorption of bone at the fracture margins. These non-unions imply a failure of biology with poor vascularity of the fracture margins. Some of these atrophic non-unions develop a fibrous capsule around the fracture site and may be painless despite persistence of movement across the fracture site. The third group in this classification is the oligotrophic non-union which has vascularised fracture margins but little to no callus formation. 8
Biology of fracture healing, pathophysiology of non-union
Non-unions can be broadly divided into infected and sterile non-unions. Stem cell therapy has not been investigated in infected non-unions thus far and this is beyond the scope of this review. A non-union represents the lack of progression through the normal stages of fracture healing. Failure occurs due to factors affecting mechanical and/or biological properties of the fracture. 4
Physiological healing of fractures involves formation of callus, which defines indirect/secondary healing 5,9 as opposed to primary bone healing, which happens after surgical treatment of fractures following anatomical reduction, inter-fragmentary compression and internal fixation with absolute stability principles. Fracture involves displacement of bone with resulting bleeding and haematoma formation. The local environment of the haematoma facilitates recruitment of stem cells, inflammatory cells and procoagulant cells with resulting inflammation and stabilisation of the haematoma. Mesenchymal stem cells (MSCs), adjacent progenitor cells and fibroblasts then give rise to formation of soft callus which is made up of collagen fibres and blood vessels. This process takes days to develop soft callus. Soft callus also contains osteoblasts, chondroblasts and osteoclasts. Over the next few weeks, the soft callus is converted to fibrocartilaginous fibrous tissue. 5,9 Growth factors play a vital role in progression of healing through this phase. This corresponds clinically with gradual resolution of pain from the fracture although there is still some movement and potential strain at the fracture site. 4,5
Subsequent healing sees the osteoblast and osteoclasts convert the fibrocartilage into lamellar bone which greatly reduces strain at the fracture site. This phase of healing is termed consolidation which involves formation of ‘cutting cones’ by the osteoblasts and osteoclasts which traverse across the fracture site, laying down lamellar bone with its Haversian structure. At this stage, there is clinically no pain with stressing of the fracture by hand. Finally, the fracture goes through remodelling over the subsequent weeks to months. The lamellar bone is thickened or thinned depending on the mechanical forces applied across the bone. Radiographic analysis at this stage shows no residual fracture line. 5,9
Non-union arises when mechanical and/or biological deficiencies impair fracture healing. Numerous attempts in the literature are trying to provide a simplified, practical and therapy targeting approach to fracture healing. 10 Giannoudis et al coined the ‘diamond concept’ of fracture healing while Elliott et al described the ‘Unified Bone Healing Theory’ to explain the requirements for fracture union and reciprocally guide management of non-union. 4,11 These concepts highlight our understanding that stem cell therapy alone is unable to facilitate fracture union, rather a holistic approach to tackle mechanical and biological aspects of healing are required.
The ‘diamond concept’, states the need for four elements to co-exist in optimal balance for successful fracture healing. These include the presence of osteogenic cells, generation/presence of an osteoconductive scaffold, availability of growth factors and a conducive mechanical environment for fracture union. 11 All these factors form the microenvironment at a fracture site and stem cells are vital to marrying these elements together. MSC at fracture sites differentiate to ultimately give rise to osteoblasts which are osteogenic. 12,13 They also differentiate into chondroblasts which form the soft callus that acts as an osteo-inductive scaffold at the fracture site. 5,13 This scaffold in turn adds to the mechanical stability at the fracture site, gradually reducing strain and allowing conversion of soft callus to hard callus. 4,5 Mechanical stability regulates the tensile and compressive forces at the fracture site. These mechanical stresses have been shown to regulate MSC activity. 14 The relationship between movement at the fracture site and osteogenesis is explained by the unified bone healing theory, discussed below. 4 Lastly, growth factors directly affect MSC differentiation into osteogenic cells, while MSC secrete some growth factors as well. Bone Morphogenic Proteins (BMP) are essential to fracture healing by regulating osteogenesis, chondrogenesis and MSC proliferation. 15,16 Other growth factors like PDGF has been shown to support MSC proliferation, differentiation and migration 17 while SDF1 is a strong chemoattractant for MSC migration. 18 Fischer et al. measured the serum concentrations of TGF-B1, IGF-1 and PDGF in patients with long bone non-union who underwent successful and unsuccessful treatment with the induced membrane technique compared to control patients who healed their fractures physiologically. Patients with failed treatment of their non-union had lowest levels of IGF-1 and PDGF. TGF-B1 levels in patients with non-union were lower than control. 19
The unified bone healing theory highlights the important relationship of osteogenesis and strain tolerance at a fracture site. Strain refers to the degree of change in length of a tissue when a force acts upon it. The authors describe a biological ‘bone healing unit’ that forms callus provided the suitable mechanical stability. In reference to Perren’s laws, soft callus tolerates 5–10% strain and osteoblasts tolerate 1–5% strain. 4 Thus, it is important to realise that mechanical stability from casts or surgical intervention is necessary for healing. The afforded mechanical stability allows stem cells and the ‘bone healing unit’ to form new bone and return the bone to physiological strength.
The simple presence of MSC at a fracture site does not prevent non-union. Ismail et al showed that non-unions contained a similar number of functional MSCs to iliac crest aspirate in patients with non-union, highlighting the importance of the microenvironment to successful osteogenesis. 20 Bajada et al investigated the properties of non-union stromal cells in 8 patients with long bone non-union. They found MSCs present within non-union tissue however their osteogenic potential was reduced and the MSCs showed features of senescence. The impaired osteogenic capacity was attributed to higher levels of Dkk-1, an inhibitor of osteogenesis, compared to control. 21
The number of MSCs present at a fracture site may correlate with the type of non-union. Mathieu et al investigated the MSC profile of 24 patients with atrophic non-union compared to control. Unlike other studies, MSC counts were made from BMA and not from non-union stromal tissue. Patients with non-union had lower MSC counts in bone marrow and these cells had a decreased proliferative and differentiation potential compared to controls. The authors attributed this MSC impairment to differences in serum concentrations of key cytokines and growth factors. 22 It remains to be seen whether these serum assays could predict risk of non-union. The panel included PDGF, Insulin-like growth factor-1, Interleukin-6, interleukin-8, transforming growth factor beta-1, vascular endothelial growth factor, Dickkopf-related protein-1 (Dkk-1), stromal derived factor, angiopoietin 1 and angiopoietin 2. 22
Conversely, Iwakura et al found abundant MSC in hypertrophic non-union stromal tissue. Seven patients with hypertrophic non-union of long bones underwent revision surgery where non-union stromal tissue was sampled. MSC counts were compared to that of healthy controls undergoing fixation of acute fractures, where fracture haematoma was sampled. The MSC from non-union and fracture haematoma were found to have similar osteogenic, chondrogenic and adipogenic capacity. 23 This strengthens the belief in the ‘diamond concept’ and unified bone theory in that stability plays a big part in facilitating fracture union.
Stem cell therapy: Cellular factors
The ideal stem cell preparation for non-union management should be obtainable minimally invasively with a reproducible and well defined yield of stem cells with little to no cell loss, require minimal/rapid laboratory processing without compromising the principles of asepsis, be easily administered and at low cost.
We must also understand that there are cellular factors that affect the efficiency of MSC in stem cell therapy. Older donors have a smaller pool of MSC in their bone marrow making it harder to obtain an adequate volume for stem cell therapy. 24 Although there is no consensus on the minimum threshold of number of MSCs for successful stem cell therapy, observations by Hernigou et al demonstrated a positive relationship between number of osteogenic progenitor cells within injected bone marrow aspirate (BMA) and volume of callus formation after 4 months and patients with a total of <30,000 injected progenitors had treatment failure. 24 This suggests a minimum effective MSC count for stem cell therapy and the potential need for in vitro culture expansion prior to re-implantation. Interestingly, Mathieu et al demonstrated that BMA performed on patients with non-union yielded fewer MSC than healthy control, suggesting these patients had an overall smaller pool of MSCs that contributed to non-union. 22 Perhaps such patients will benefit from allogenic MSC therapy from young, healthy donors but there are currently no human studies utilising allogenic MSC therapy for non-union. MSC from aged donors have also been shown to have reduced proliferative capacity. 25 This was a finding made by Stenderup et al when they compared proliferative and bone-matrix-synthetic function of MSC from healthy young and healthy aged donors. This study also found that synthetic function of MSC from aged donors was similar to young donors when implanted into a mouse in vivo model. These findings led authors to infer that the impaired proliferation and smaller number of osteoblasts in elderly donors caused the impaired bone formation in the elderly. However, laboratory processing has been shown to negatively affect the osteogenic capacity of MSCs. Agata et al demonstrated that MSC begin to lose in vivo osteogenic potential after second passage. 26 Bruder et al showed that MSCs became senescent after 38 ± 4 population doublings. 27 Hence over-expansion in culture can impair the osteogenic potential of transplanted MSCs. A recent study by Carlock et al investigated the effectiveness of iliac crest bone grafting in patients above the age of 65 and below. The study prospectively followed the progress of 242 patients treated for non-union and measured rates of radiological union, pain scores and functional scores. Iliac crest bone grafting was found to be equally effective in elderly patients and in younger patients (<65 years). 28 This finding was made from a sample population that consisted of various types of non-union, infected non-unions and various long bones. Some patients also received supplemental graft such as allogenic bone chips and bone morphogenic protein-seven.
To further highlight the complexity of bone healing, in vitro studies on MSC from young, healthy donors showed impaired proliferation and differentiation when cultured with serum from aged females. 29 MSC depend upon growth factors for osteogenesis. Pro-osteogenic growth factors include PDGF, IGF-1 and Bone morphogenetic peptides (BMP). Of the 13 BMPs, BMP2 and BMP7 are the strongest osteoinducing proteins and are synthesised throughout different timepoints in physiological fracture healing. 13,16,30 BMPs have been made into commercial products to stimulate osteogenesis. 16 Numerous other growth factors affect MSC differentiation and proliferation but this is beyond the scope of this review.
Stem cell harvesting
At present, research on stem cell therapy for non-union largely involves bone marrow aspirates and bone marrow concentrates, with or without ex vivo expansion. This involves percutaneous aspiration of the iliac crest, anterior superior iliac spine(ASIS) or the posterior superior iliac spine(PSIS). The difference in iliac crest harvesting from ASIS/PSIS harvesting is the anatomical site. This implies technical differences in position of the patient; supine for iliac crest and ASIS whilst lateral or prone positioning is required for PSIS harvesting. The iliac crest technique also involves passing a needle through multiple sites of the iliac crest outer cortex as opposed to a single puncture site for the ASIS/PSIS technique. Although other anatomical regions such as the sternum, ribs and vertebral bodies also contain bone marrow and MSCs for aspiration, the pelvic source is best described, most researched and promises a higher yield of MSCs. 31
Piuzzi et al have published evidence based recommendations on bone marrow aspiration from the iliac crest and iliac spines. Iliac crest bone marrow aspiration involves multiple advancements with a large bore needle, in the sagittal plane, starting from the lateral aspect of the iliac crest, directed towards the centre of the bone. Each time advancing the needle 3–4 cm without penetrating the medial cortex. The authors recommend a rotational movement while advancing the needle 5–10 mm at a time. Each advancement followed by a corresponding withdrawal of the plunger of a 10 ml syringe to generate negative pressure and fill the syringe. A 5 mm gap between each entry point is enough for aspiration of independent sections of bone marrow. Of note, a total aspirate volume per-syringe is recommended to be less or equal to 2 ml. Further volume was found to dilute the bone marrow with peripheral blood.
Aspiration of the iliac spines should be done with the needle advanced from anterior to posterior, directed to stay within the inner and outer tables of the iliac crest, then likewise, posterior to anterior. Only one entry point is required for each iliac spine as the needle can be fanned out superiorly to inferiorly for a wide area of bone marrow aspiration. The same technique of aspirating 5–10 mm at a time are advised for a total depth of 6–8 cm before redirecting the needle from its apex at the entry point.
These two anatomical sites usually generate 30 ml of bone marrow. 31
Bone marrow can also be aspirated with intramedullary reamer-irrigator-aspirators (RIA). 32,33 This method requires invasive surgical access to the intramedullary canal of the tibia or femur. A proprietary device simultaneously reams the intramedullary canal while flushing the reaming with sterile normal saline. Bone fragments are filtered and the wash/liquid fraction is collection in a sterile reservoir. 32 Porter et al were able to isolate a large number of MSCs following RIA and culture of five patients aged 71–78 years old. 33 RIA offers a viable route of MSC harvesting in patients who require intramedullary nailing/exchange nailing of their non-union. In such patients, morbidity from pelvic bone marrow aspiration can be avoided.
A number of devices for real-time concentration of MSCs from BMA in theatre are available, using principles of low speed centrifugation, Ficoll separation and isolation with magnetic nanobeads. 24,34 –36 These produce a crude preparation that contains a number of haematopoietic progenitors and stromal cells, however only a small proportion (1 in 30,000) are MSCs (Hernigou 2005), even after concentration. Samples from such preparations can be characterised post-injection for total nucleated cell number, MSC number, viability or the ability to form plastic-adherent colonies (CFU-F). CFU-F has the advantage of providing evidence of proliferative capacity and viability, while other characterisation methods such as flow cytometry can provide quantitative results, however this is not necessarily an indicator of cell function as senescent cells may be present. Alternatively, MSCs can be cultured from BMA in the laboratory, based on their ability to adhere to cell culture flasks and form colonies. This specifically enriches for cells that will ultimately form bone, with expansion taking in the order of weeks. As mentioned previously, MSC yield and purity comes at the cost of loss of osteogenic potential and increased senescence, so while the number of injected MSCs is an important factor in BMA concentrate (Hernigou 2005), for cultured MSCs, the number of doublings/time spent in culture is another factor that may influence outcome. As described below, the published clinical studies vary in their reporting of these factors.
Clinical trials on MSC in non-union
Although published scientific data on MSC therapy for human non-union has been available since 1986, 37 to date there is no Level I nor Level II evidence in publication. A clinicaltrials.gov search revealed 13 clinical trials on MSC for non-union however none have been published (Table 1). Five trials are listed as complete, six trials as ‘unknown status’, one still recruiting and one yet to recruit.
Clinical trial database search for MSC therapy in long bone fracture non-union (clinicaltrials.gov).

There are several published case series and case reports on MSC therapy for non-union. This literature search found eight case series with a combined total of 126 patients aged 10–82 (Table 2). Indications included non-unions of the femur, tibia, ulna, humerus, metacarpal, pelvis and long bone defects of the ulna, humerus and tibia. All the studies utilised bone marrow aspirate from iliac crest or iliac spines. MSC preparations differed across the studies with some utilising concentration and/or culture expansion with/without a scaffold. All these studies used radiographic analysis to assess union along with some reporting of clinical function at various timepoints. The results are encouraging with most patients successfully achieving bony union. It should be noted that Table 2 includes two studies that investigated MSC in patients who failed to reach union at 6 weeks and in acute fractures by Kim et al and Liebergal et al respectively. The table also includes a published long term follow-up of patients from a prior publication; these are by Marcacci et al and Quarto et al. As such, the total number of cases from this literature search reflect the duplication of these patients.
Human studies on use of MSC therapy in fracture non-union.

Despite a lack of standardisation across the studies and lack of unified qualitative analysis of the individual stem cell therapies, majority of the patients in these studies achieved bony union.
Practical utility of MSC in non-union management
Each non-union can be seen to have a specific ‘personality’. We will discuss how MSC therapy can be useful in restoring the potential of the bone-healing unit in various combinations of mechanical and biological failure in non-union (Figure 3).
Mechanical failure + biological failure (needs mechanical stability and biological boost)
These non-unions have a combination of inadequate fracture stability (wrong choice of implant/poor fixation) and impaired healing potential (e.g. periosteal stripping, large amount of necrotic bone, inadequate pool of bone marrow MSC, lack of osteogenic growth factors). Management of these non-unions are likely to require revision surgery to increase stability and improve the mechanics of fixation. Revision surgery poses the risk of further devitalising the fracture site. Supplementing revision surgery with MSC therapy increases the osteogenic potential, as achieved to some degree with cancellous bone grafting. Ismail et al compared the effect of MSC and hydroxyapatite granules versus iliac crest bone graft in 10 patients with atrophic non-union of long bones. 40 Both groups underwent plate osteosynthesis, it is unclear whether this was revision surgery or if fractures were previous managed non-operatively. All patients progressed to union. They found that patients in the MSC group achieved union 3 months quicker than the bone graft group, demonstrating augmentation of fracture healing rate with MSC therapy. 40
Mechanical failure + intact biology (needs mechanical stability)
This is the case with fractures in physiological bone that have been managed with insufficient stability (Figure 1). Normally these injuries result in hypertrophic non-union and union is achieved with increasing the rigidity of fixation/optimising the stability of the fracture. The added stability allows for progression of healing through formation of hard callus and eventual fracture consolidation and remodelling. 4 MSC therapy here may not be necessary to ensure union but may accelerate the time to union by augmenting osteogenesis. Conceptually it is very important to protect/maintain optimal biological conditions whilst improved stability is achieved to all progression to union.
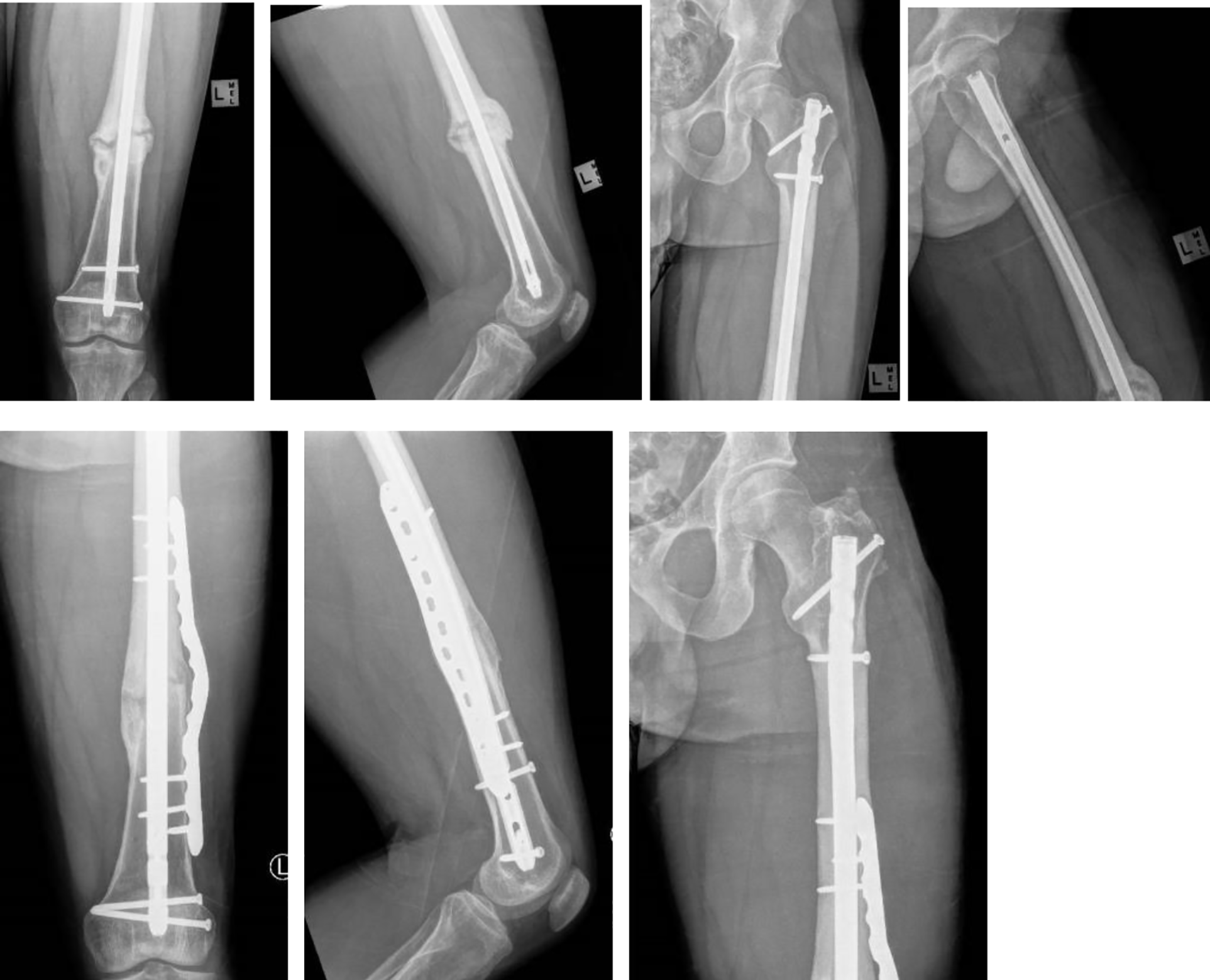
(
Another example of this category of non-union are fractures with rigid fixation and lack of anatomical reduction/compression of fracture fragments (e.g. Compression plating with existing fracture gap > 0.5 mm). Here the construct is too stiff to stimulate callus formation to fill the fracture gap. Eventually the hardware fatigues and fails if put through enough stress. This resembles a small critical size defect and MSC injection in combination with an osteoconductive substance into the fracture site could facilitate formation of bridging callus and progression to union. In other words, MSC therapy may be able to alter the bone formation vs strain graph described by Elliott et al in their paper on the unified bone theory. 4 This application of MSC therapy however, has not been studied specifically in literature. The implications are that union may be achieved without revision invasive surgery, rather percutaneous injection of MSC into the fracture gap.
Mechanical stability + biological failure (needs biological boost)
This group of non-unions have received adequate stabilisation but still fail to unite (Figure 2). Patients with these non-unions have little to no option once bone grafting and vascularised bone grafting has been attempted. Again, the impaired biology may be the result of impaired vascularity, an inadequate bone marrow stem cell reserve or lack of vital growth factors at the fracture site. Stem cell therapy in these patients should augment osteogenesis and stimulate fracture healing. The possibility of stimulating fracture healing by virtue of a percutaneous injection of MSCs into the fracture site, as opposed to lengthy revision surgery and bone grafting, is another appealing factor. Kim et al studied the effect of osteogenic cell therapy in 64 patients with delayed union of long bones (defined as minimal callus formation scores at 6 weeks after surgery). Patients were divided into groups that received osteoblast therapy and a control group with no further intervention. No data on volume of injectate was disclosed, however, a cell concentration of 1.2 × 107/0.4 ml was described. Patients in the osteoblast therapy group demonstrated a significantly larger amount of callus at 2 months. No functional analysis was carried out however. Their study demonstrated augmented callus formation with injection of osteoblasts. 44 The volume of injected stem cell suspension varied from 10 ml to 40 ml in literature. This volume was injected into the site of the non-union. 39,40

Oligotrophic non-union of a clavicle fracture despite adequate fixation. Bone grafting of fracture site resulted in union.
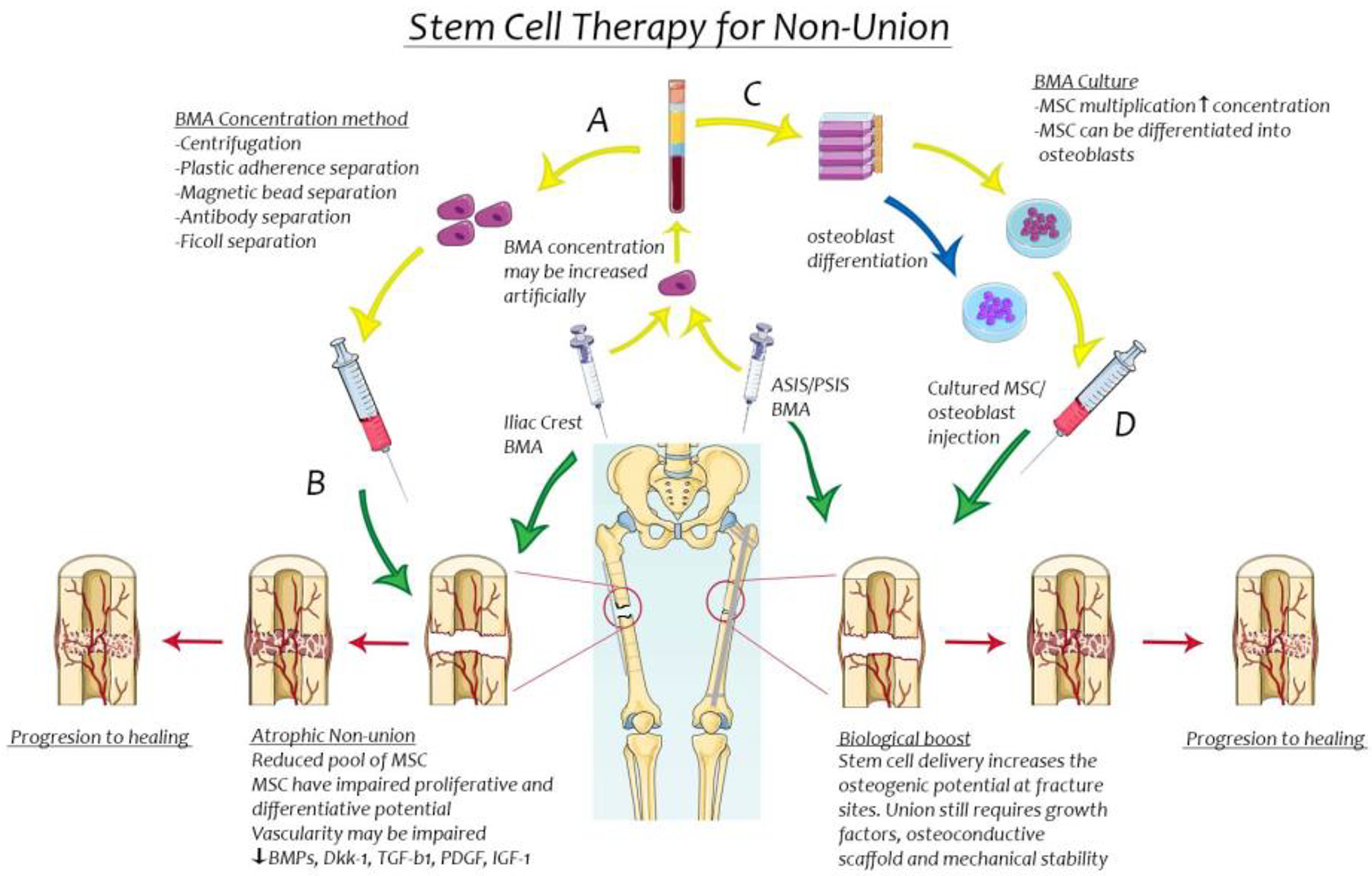
Pictographic representation of the strategies of use of bone marrow aspirate in management of atrophic non-union of long bones. (A) BMA concentration techniques without cell culture. (B) Use of BMA concentrate for atrophic non-union to improve osteogenesis. (C) BMA utilised to harvest stem cells that are expanded with cell culture/differentiated to develop osteoblasts for injection. (D) Stem cell/osteoblast cell therapy as a biological boost in non-union despite mechanical stability.
Mechanical stability + intact biology
This group represents acute fractures that should heal or that have not been deemed non-unions. There still may be a benefit of MSC therapy in this group due to faster recovery as described by Liebergall et al. This group investigated the use of BMA and PRP versus control in 24 consecutive distal tibial fractures that underwent tibial nailing. All fracture progressed to union. Patients in the BMA + PRP group achieved fracture union 3 months quicker than controls. 45
Future direction to standardise research and translate to common practice
Theoretically, stem cell therapy has great promise in improving the local biological environment around the non-union site. This is implied by the local delivery of a high concentration of active stem cells to the site of the non-union. The stem cells then facilitate initiation of the fracture healing process by differentiating into osteoblastic cell lines whilst also releasing osteogenic and chondrogenic factors. 46 The current body of evidence shows moderate level of success, low complication rates and no high level of scientific evidence for use of stem cells in management of sterile non-union. Combining basic science and results from the available studies suggests the translational effectiveness of stem cell therapy will depend on the type of non-union being treated, patient age, patient comorbidities, concentration of stem cells injected, nature of laboratory augmentation, supplementation with growth factors and use of scaffold.
Although we are aware of fracture union principles, treatment of non-union involving biological agents is complex. Barriers to high level evidence in this field include vast variation in patient biological factors, complex injury specifics (polytrauma vs isolated injury, shock vs no shock, fracture patterns), variation in initial treatment of the fracture, subjectivity of non-union classification, variation in stem cell techniques, with or without use of adjuvants and lack of publishing of completed studies. Positive and negative findings should be reported to eliminate publication bias and to further the knowledge in the field.
Prior to the widespread use of this stem cell therapy in clinical practice, more knowledge of the factors affecting stem cell function in non-union is essential. We now understand that the mere presence of stem cells at a fracture site is insufficient to guarantee union. Research in this field should be designed to account for these biological variables and study its effects. This should involve standardised reporting of stem cell therapy dynamics such as cell concentration, proliferative and differentiative potential, laboratory handling technique, growth factor analysis and open reporting of results be it positive or negative. The qualitative and quantitative evaluation of stem cell therapy should also incorporate modern stem cell evaluation techniques to minimise error associated with traditional methods. 47 When analysing current literature, clinicians should pay particular attention to which cell lines are being described as some authors use overlapping terminology ie fibroblast colony forming unit to denote osteoblasts. At the American Academy of Orthopaedic Surgeons/National Institutes of Health Optimizing Clinical Use of Biologics Symposium in 2018, a working group was formed to develop a consensus on reporting/communicating cell therapy research. As a result, 34 experts subsequently contributed to the development of a clinical communication tool titled ‘DOSES’. This name is abbreviated from five important aspects of reporting cell therapy research. They suggest reporting of the donor, origin of the tissue, separation method from other cell types, exhibited cell characteristics and site of delivery. 48
This approach favours scientific use of stem cell therapy and can be adapted to also evaluate new advances in the field such as use of novel stem cell harvesting methods and biological augmentation of stem cell function. 38,39,41 –43
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
