Abstract
Purpose:
To identify the independent risk factors for adverse outcomes and determine the effect of L5-S1 involvement on the outcome of surgical treatment of lumbar pyogenic spondylitis (PS).
Methods:
A retrospective analysis was performed for all consecutive patients who underwent surgery for lumbar PS between November 2004 and June 2020 at a single institution. The patients were divided into two groups based on the outcomes: good and adverse (treatment failure, relapse, or death). Treatment failure was defined as persistent or worsening pain with C-reactive protein (CRP) reduction less than 25% from preoperative measurement or requiring additional debridement. Relapse was defined as the reappearance of symptoms and signs with an elevated white blood cell count, erythrocyte sedimentation rate, and CRP after the first period of treatment. Binary logistic regression analyses were performed to identify the independent risk factors for adverse outcomes.
Results:
Twenty-four (21.2%) of the 113 patients were classified as having adverse outcomes: treatment failure, relapse, and death occurred in 15, 7, and 2 patients, respectively. The involvement of L5-S1 (adjusted odds ratio [aOR] = 6.561, P = 0.004), Methicillin-resistant Staphylococcus aureus (MRSA) infection (aOR = 6.870, P = 0.008), polymicrobial infection (aOR = 12.210, P = 0.022), and Charlson comorbidity index (CCI; P = 0.005) were identified as significant risk factors for adverse outcomes.
Conclusion:
Involvement of L5-S1, MRSA, polymicrobial infection, and CCI were identified as independent risk factors for adverse outcomes after surgical treatment of lumbar PS. Because L5-S1 is anatomically demanding to access anteriorly, judicious access and thorough debridement are recommended in patients requiring anterior debridement of L5-S1.
Introduction
The incidence of spinal infection has been rising recently, possibly due to the increased life expectancy of patients with chronic disease, better diagnostic rate, intravenous drug use, and an increase in health-care-related infection and spinal surgery. 1 –3 Spinal infection represents 2–7% of all cases of osteomyelitis, 1,4 and the incidence has been reported to range from 2 to 24 cases per million annually. 1,3,5
The etiology of spinal infection, or infectious spondylitis, comprises pyogenic, granulomatous (tuberculous, brucellar, or fungal), or parasitic infection. 3,6 Pyogenic spondylitis (PS), the most common type of infectious spondylitis, 7 is a broad term encompassing spondylodiscitis, vertebral osteomyelitis (VO), epidural abscess, and facet arthropathy caused by bacterial species. 3,8 The diagnosis and management of PS remains a challenge due to the insidious onset, non-specific symptoms, and debilitating course. 9 PS is potentially life-threatening as elderly patients with a precarious state of health or chronic debilitating disease are easily affected. 9,10 There have been several studies on the outcome or prognosis of the entire PS. 11,12 However, there is a paucity of literature on the prognosis and risk factors for adverse outcomes after surgical intervention for PS. Patients requiring surgical intervention for PS are generally more vulnerable to adverse outcomes than those cured by conservative treatment because they are more likely to have progressive neurological deficits, impending biomechanical instability, or failed antibiotic therapy. It is more critical to recognize those at a higher risk of adverse outcomes in this surgical population.
The L5-S1 level is anatomically difficult to access anteriorly due to the variation of major vascular structures and larger overlap of the iliac crest. 13,14 Additionally, infectious conditions would cause adhesion between the annulus and iliac vessels, making the identification and dissection of anatomical structures difficult and iliac vessels prone to injury during the anterior approach. Therefore, we aimed to determine whether the involvement of the L5-S1 segment affects the outcome and identify independent risk factors for adverse outcomes after surgical intervention of PS in the lumbar spine, the most common site for PS.
Materials and methods
Patients and surgical treatment
This retrospective study was approved by the institutional review board of our institution, and a waiver of consent was obtained. We reviewed the medical records of patients who underwent surgery for PS between November 2004 and June 2020 at a single institution, a tertiary care referral center. Patients with granulomatous (tuberculous, brucellosis, or fungal) spondylitis, cervical or thoracic infectious spondylitis, or incomplete medical record documentation were excluded. Four surgeons from a single orthopedic department performed surgical treatment for PS. Surgical indications included neurologic deficits or uncontrollable pain, significant progressive bone destruction causing mechanical instability, and failure to respond to conservative treatment. 1,10 The detailed surgical plan including the surgical approach was determined based on the location and extent of infection and can be divided into four techniques: (1) anterior debridement/decompression and fusion (anterior only); (2) posterior debridement/decompression with or without fusion with instrumentation (posterior only); (3) anterior debridement/decompression and fusion, with simultaneous posterior instrumentation with or without fusion, (single-stage combined); and (4) anterior debridement/decompression and fusion, with sequential posterior instrumentation with or without fusion (two-stage combined). 15 Regarding the patients with remnant implants at the time of PS diagnosis, the surgeons removed the implants in case of screw loosening or the infection occurring more than 3 months after the index procedure, and reinserted implants in case of the risk of pseudarthrosis or loss of correction. 16 –18 Antibiotics were administered for at least 6 weeks according to the recommendation of an infection specialist in the internal medicine department.
Data collection
The medical records were reviewed for demographic data, comorbidities, preoperative laboratory data (white blood cell [WBC] count, erythrocyte sedimentation rate [ESR], and C-reactive protein [CRP]), microbiological and histological data, involved vertebral levels, preoperative neurologic status, radiographic characteristics (presence of paravertebral and/or epidural abscess), the number of levels operated, and surgical procedures.
The American Society of Anesthesiologists (ASA) physical status classification system and Charlson comorbidity index (CCI) were collected to assess the burden of comorbidities. ASA grades for each patient were obtained from the preoperative anesthesia record in our electronic medical record system, which was documented by the anesthesiologist at our institution. The CCI grade was classified into three categories according to previous studies: 0, 1, and 2+. 19
Diagnosis of pyogenic spondylitis
In this study, PS was defined when all the following criteria were met: compatible symptoms or signs, including fever, neurological signs, or pain in the affected area; radiologic evidence on magnetic resonance imaging; and biological evidence, including laboratory data showing infectious conditions and/or isolation of microorganisms from an image-guided percutaneous or surgical tissue culture, or blood culture performed at the time of illness. 20 –22 When common resident skin flora grew in the tissue or blood culture, it was considered as a true pathogen only when the same organism was identified in two or more separate specimens. 20,21 In patients with negative culture results, PS was diagnosed when all the following criteria were satisfied: no histologic evidence of granulomatous inflammation or fungal infection; negative results of a prolonged cultures for Mycobacterium tuberculosis and the polymerase chain reaction for Mycobacterium complex; and intraoperative findings consistent with pyogenic infection such as purulent discharge. 22,23 Polymicrobial infection was defined as infection caused by at least two distinct organisms identified from culture studies. 23 Moreover, we defined postoperative PS as patients with demonstrated infection appearing within 1 year after spinal surgery at the same location or who had residual implants at the time of diagnosis. 21
Outcome assessment
Outcome was determined based on changes in symptoms and signs, laboratory data, and overall mortality. The patients were divided into the good outcome and adverse outcome groups. Patients with treatment failure, relapse, or infection-related deaths were assigned to the adverse outcome group. Treatment failure was defined as persistent or worsening pain with reduction of CRP less than 25% from preoperative measurement, or requiring additional surgical debridement despite thorough surgical debridement and administration of appropriate antibiotics for more than 1 month according to consultation answers from an infection specialist. Relapse was defined as the reappearance of symptoms and signs with an elevated WBC, ESR, and CRP after completion of the first period of treatment. 21,24 Only in-hospital deaths related to infection or consequent complications were regarded as infection-related deaths in this study. 25
Statistical analysis
Continuous variables were compared using the Student’s t-test or Wilcoxon rank sum test, and categorical variables were compared using the χ2 test or Fisher’s exact test. Univariate analyses were performed for all variables using binary logistic regression analysis. Multivariate binary logistic regression was applied to identify independent risk factors associated with adverse outcomes using variables with a P-value < 0.1, by univariate analysis. To manage any issues with multicollinearity, we used a stepwise method and applied an entry condition of P < 0.05, removal condition of P > 0.1, and the final model included only significant variables with a P-value < 0.05. Adjusted odds ratios (aORs) and 95% confidence intervals were calculated for all variables. All statistical analyses were performed using the SPSS system for Windows the SAS system for Windows, version 9.4 (SAS institute, Cary, NC, USA). Statistical significance was set at P < 0.05.
Results
Patients’ clinical characteristics
A total of 113 consecutive patients who underwent surgical intervention for pyogenic lumbar spondylitis were identified (63 male; mean age: 63.5 ± 13.1 years). Of these patients, pyogenic infection involved the L1-2 segment in 11 patients (9.7%), L2-3 in 21 patients (18.6%), L3-4 in 26 patients (23.0%), L4-5 in 56 patients (49.6%), and L5-S1 in 25 patients (22.1%). Twenty-four patients (21.2%) were classified as having adverse outcomes: treatment failure, relapse, and death occurred 15, 7, and 2 patients, respectively. The average follow-up duration was 34.5 ± 29.5 months.
Microbiological results
Among the 113 patients, microorganisms were identified in 97 cases (85.8%). The most commonly identified organism was Staphylococcus aureus (n = 29, 25.44%), followed by coagulase-negative Staphylococci (n = 22, 19.47%). Methicillin-resistant Staphylococcus aureus (MRSA) was significantly more common in patients with adverse outcomes than in those with good outcomes (29.17% vs. 8.99%, P = 0.017). All microbiological study results are described in Table 1.
Identified micro-organisms in patients with good and adverse outcome.

*P-value derived by Fisher’s exact test.
Comparison of patients with good and adverse outcomes
When the patients with good (n = 89) and adverse outcomes (n = 24) were compared, patients with adverse outcomes had a significantly higher proportion of PS at L5-S1 (41.67% vs. 16.85%, P = 0.009), MRSA infection (29.17% vs. 8.99%, P = 0.017), polymicrobial infection (20.83% vs. 2.25%, P = 0.005), and preoperative paresis (41.67% vs. 21.35%, P = 0.043) (Table 2). Additionally, preoperative WBC (×103/mm3) was significantly higher in patients with adverse outcomes than in those with good outcomes (10.05 ± 3.91 vs. 7.95 ± 3.00, P = 0.017). Regarding the burden of comorbidities, patients with adverse outcomes had significantly higher ASA class (P = 0.016) and CCI scores (P < 0.001).
Comparison of demographics and clinical characteristics between patients with good and adverse outcome.

ASA, American Society of Anesthesiologists Physical Status Classification System; CCI, Charlson Comorbidity Index; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; MRSA, Methicillin-resistant Staphylococcus aureus; WBC, white blood cell counts.
* Statistically significant (P < 0.05).
Logistic regression analysis: univariate and multivariate analyses
Univariate analysis identified preoperative WBC (crude odds ratio [cOR] = 1.207, P = 0.008), PS at L5-S1 (cOR = 3.524, P = 0.012), MRSA infection (cOR = 4.169, P = 0.014), polymicrobial infection (cOR = 11.447, P = 0.005), CCI (P = 0.003), ASA (P = 0.027), and preoperative paresis (cOR = 2.632, P = 0.048) (Table 3). Using the stepwise method, PS at L5-S1 (aOR = 6.561, P = 0.004), MRSA infection (aOR = 6.870, P = 0.008), polymicrobial infection (aOR = 12.210, P = 0.022), and CCI (P = 0.005) were identified by the multivariate analysis as significant risk factors for adverse outcomes after surgical treatment (Table 4).
Univariate analysis of risk factors for adverse outcome after surgical treatment of lumbar pyogenic spondylitis.

ASA, American Society of Anesthesiologists Physical Status Classification System; CCI, Charlson Comorbidity Index; CI, confidence interval; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; MRSA, Methicillin-resistant Staphylococcus aureus; WBC, white blood cell counts.
* Statistically significant (P < 0.05).
Multivariate analysis of risk factors for adverse outcome after surgical treatment of lumbar pyogenic spondylitis.
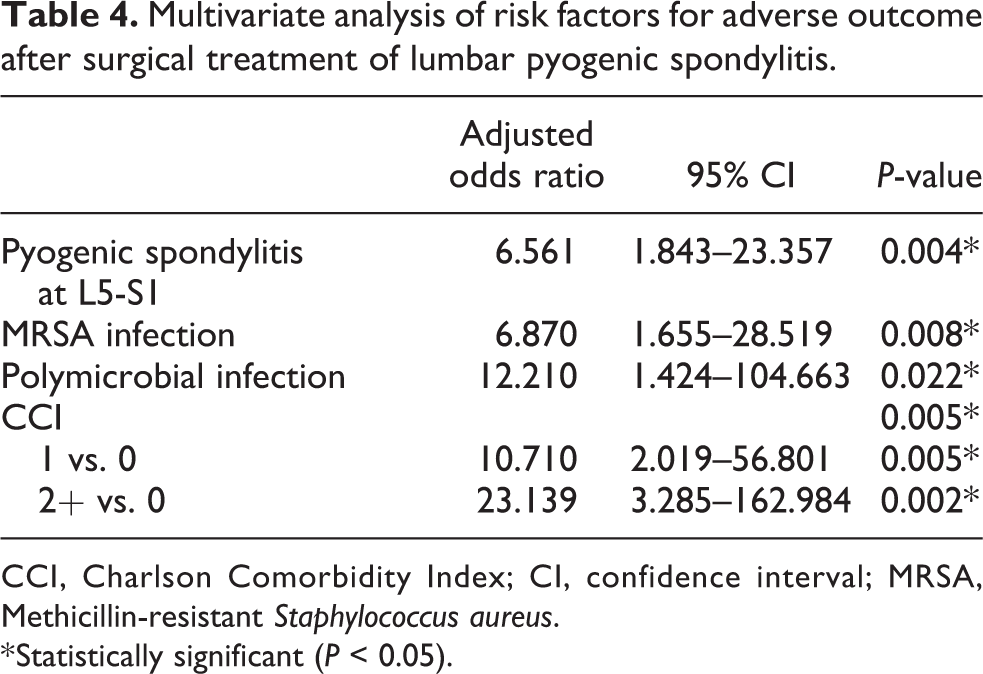
CCI, Charlson Comorbidity Index; CI, confidence interval; MRSA, Methicillin-resistant Staphylococcus aureus.
* Statistically significant (P < 0.05).
Discussion
While conservative treatment, comprising antibiotic therapy and immobilization of the affected segment with bed rest and/or orthosis, is considered the first treatment choice for PS, 9,10 surgical intervention might be required in some cases. Accordingly, it is reasonable to distinguish the surgical treatment of PS from conservative treatment with respect to the outcomes and prognosis, since the indications of these treatments are different, and surgery would also be required when conservative treatment alone appears to be insufficient. Therefore, we only included patients managed by surgical treatment to improve the homogeneity of the study population and specify the surgery outcome.
The definitions of PS have varied in previous studies. 20,21,26 While the diagnosis of spinal infection can be confirmed by microbiological studies, several studies have reported culture-negative cases of spinal infection. 22,27,28 Sixteen cases (14.16%) in this cohort had negative cultures, which was lower than that reported in previous studies. 8,22 While patients with negative culture are reportedly less likely to have pyrexia, lower WBC, ESR, and CRP, culture-negativity had no significant impact on the final outcomes in this study, which is consistent with the results of previous studies. 22,28
There have been several studies on the outcomes of PS and related factors. 21,22 In this study, independent risk factors for adverse outcomes after surgical intervention of PS in the lumbar spine were involvement of the L5-S1 segment, MRSA infection, polymicrobial infection, and CCI. To our knowledge, this is the first study to suggest that L5-S1 infection is an independent risk factor for poor outcomes of spinal infection. In previous studies, several anatomical variations of vascular structures have been reported around the L5-S1 level, and these great vessels might block anterior access to the disc space. 13,29 Moreover, it is difficult to secure a working window because the iliac vessels, peritoneal contents, and nerves are prone to injury during the approach due to adhesion in the infectious condition, although wide exposure of infectious lesions is necessary for thorough debridement.
Among the 25 patients with L5-S1 involvement in the current cohort, 9 patients had facet joint infection and/or epidural abscess and underwent only posterior debridement. While only 1 (11.1%) of these 9 patients had adverse outcomes; however, 9 (56.3%) of 16 patients who underwent anterior debridement had adverse outcomes (P = 0.027). These results indicate that the presence of spondylodiscitis necessitating anterior debridement at the L5-S1 level can be associated with adverse outcomes after surgical management. Therefore, spine surgeons should secure a sufficient working window during the anterior approach to the L5-S1 level for meticulous debridement, and assistance by a vascular surgeon should be requested whenever necessary (Figure 1).

A 40-year-old male patients who underwent anterior debridement and iliac bone graft with vascular surgeon’s assistance with sequential posterior instrumentation (two-stage combined) surgery for L5-S1 spondylodiscitis due to the failure to respond to antibiotics for more than 1 month. (A) Preoperative sagittal T2-weighted MR image showing intradiscal fluid-like signal and bone marrow edema involving L5 and S1. (B) Preoperative axial T2-weighted MR image revealing the adhesion between iliac vessels and infectious tissue. (C, D) Postoperative T2-weighted MR image showing partial improvement of pyogenic spondylitis. (E, F) The last follow-up X-ray showing well-maintained status without structural change.
Staphylococcus aureus was the most commonly isolated microorganism, and MRSA was significantly associated with the adverse outcome of PS in this study, consistent with the reported literature. 8,21,26 Kim et al. 21 suggested more aggressive management, including prolonged use of antibiotics, debridement, and implant removal, to achieve favorable outcomes in pyogenic VO caused by MRSA. However, MRSA was still associated with adverse outcomes of PS despite surgical management and appropriate use of antibiotics. Meanwhile, DiPaola et al. 30 recommended multiple debridements to eradicate MRSA infections. In our study population, 5 of 15 patients with MRSA infection had multiple debridements and eventually achieved resolution of infection, although we designated these multiple debridement cases as adverse outcomes.
We identified 7 (6.19%) polymicrobial cases in this study population, which is comparable to the reported incidence ranging from 4.4% to 24.4%. 8,22,31 Polymicrobial infection has been associated with increased mortality in bloodstream infections (BSIs) and high rates of treatment failure in postoperative spinal infections. 17,18,32,33 However, Hadjipavlou et al. 8 found that polymicrobial infection was not associated with the outcome of hematogenous PS. In our study, it was identified as a risk factor for poor outcomes after surgical treatment. Wille et al. 17 suggested that antibiotic therapy of polymicrobial infections often requires the use of multiple antibiotics, thereby increasing the risk of drug toxicity and causing the interruption of adequate treatment. Additionally, several studies have reported that polymicrobial BSIs are associated with anaerobes or high-risk bacteria, such as Pseudomonas aeruginosa, Klebsiella pneumoniae, and Enterobacter faecalis, and excess mortality. 32 –34 In our cohort, 6 (85.7%) of the 7 polymicrobial cases were associated with these microorganisms. However, since there are some discrepancies in the study population and definitions of adverse outcomes in the literature, the direct comparison of these results is limited. A well-designed study regarding polymicrobial infection and outcomes of PS should be conducted in the future.
Comorbidity has been reported to be associated with impaired immunity and infection outcomes. 35,36 Yoon et al. 22 suggested comorbidity as a significant factor in determining the outcome of pyogenic VO. Although the ASA and CCI were originally developed to assess the burden of comorbidities and predict mortality for various medical conditions, they have also been applied to evaluate the outcome and likelihood of complications following spine surgery. 37,38 The CCI was identified as a risk factor for adverse outcomes by multivariate analysis in our study. In addition, ASA was identified as a risk factor by univariate analysis, but not in multivariate analysis. These results imply that not only the comorbidity status, but also the management of those would have an important role in the management of PS.
In several previous studies, relapse rates of postoperative vertebral osteomyelitis (PVO) ranged 0–4%, and there were no significant differences in treatment failure rates between postoperative and native vertebral osteomyelitis (NVO). 24,39 In contrast, Kim et al. 21 reported that the treatment failure and relapse rates of PVO were significantly higher than those of NVO, although more patients with PVO had surgery compared to those with NVO. However, while this report included both conservatively and surgically managed patients, the present study only included patients who underwent surgical debridement and showed no significant difference in the outcome between postoperative and native lumbar PS. Aggressive surgical debridement with or without implant removal has been suggested as an important factor for the successful treatment of postoperative spinal infections. 18,21,40 Consistent with these reports, our results suggest that aggressive surgical debridement should be considered in the management of postoperative PS. Future systematic study to standardize the detailed treatment strategy for postoperative spinal infections.
Our study has several limitations. First, due to its retrospective nature, there may be selection bias and confounding factors that have not been considered. Second, it was difficult to recognize the occurrence of relapse beyond our follow-up period in patients who were thought to have completely recovered from PS. Third, because the decision for surgery and surgical plan depended on each surgeon’s judgment, there might have been a slight discrepancy between surgeons even though there have been commonly agreed indications of surgery for PS. Finally, a clear consensus regarding the diagnosis and outcome of PS is yet to be established. Despite these limitations, to the best of our knowledge, this is the first study to analyze the outcome of the surgical population of the lumbar PS and propose the involvement of L5-S1 as a risk factor for adverse outcomes of PS.
Conclusion
Involvement of L5-S1, MRSA infection, polymicrobial infection, and CCI were identified as independent risk factors for adverse outcomes after surgical management of lumbar PS. Because the L5-S1 level is anatomically difficult to access anteriorly, judicious access and meticulous debridement are recommended in PS patients requiring anterior debridement of L5-S1.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
