Abstract
Purpose:
Patients with haemophilia suffer from recurrent joint haemarthrosis. This can progress to symptomatic arthropathy commonly affecting the knee. While modern coagulation strategies have reduced those proceeding to end-stage arthropathy, total knee arthroplasty (TKA) remains the optimal treatment for some patients. Despite innovation in perioperative haematological management, concerns about the potential for excessive haemorrhage still exist. The aim of this study is to quantify immediate postoperative blood loss and haematological complications in haemophiliacs following TKA.
Methods:
A retrospective study of patients with haemophilia types A or B who underwent a TKA over a 12-year period at a single institution was conducted. These patients were compared to both a non-haemophiliac control group and to published standards in non-haemophiliacs undergoing TKA.
Results:
Twenty-one TKA procedures in 18 patients (72% haemophilia A, 28% haemophilia B) were suitable for inclusion with a mean age of 44 years. The mean haemoglobin drops at 24 and 48 h postoperatively were 2.7 g/dl and 3.8 g/dl respectively. There was no significant difference in haemoglobin drop at 48 h postoperatively when compared to the non-haemophiliac control group (P = 0.2644). There were no immediate perioperative complications and two patients (9.6%) required postoperative transfusion.
Conclusion:
Haemophiliacs undergoing a unilateral primary TKA in a specialised tertiary referral centre appear to have comparable rates of perioperative blood loss when compared to both a non-haemophiliac control group as and published haemostatic standards in non-haemophiliac patients following TKA. Perioperative management with expert orthopaedic and haematological input is recommended to optimise outcomes in this complex patient group.
Introduction
Degenerative joint arthropathy can occur more frequently in patients with rare haematological disorders such as haemophilia, with the knee joint commonly involved. 1 Despite progress in prophylactic clotting factor management, haemophilic arthropathy continues to occur in up to 50% of patients. 2 Spontaneous haemarthrosis and subsequent synovial hypertrophy can damage intra-articular structures, ultimately leading to cartilage and bony destruction. 2 The resultant arthritis tends to present at a younger age, 3 and with the advent of safer haematological therapies and increased life expectancy, has become increasingly prevalent in ageing haemophiliac populations. 2
While coagulation factor strategies have reduced progression to end-stage knee haemophilic arthropathy, total knee arthroplasty (TKA) remains the treatment of choice when conservative management is no longer successful. 4 TKA in haemophiliacs is a technically challenging procedure due to the presence of anatomical deformities, arthrofibrosis and poor bone quality. 5 The perioperative course is further complicated by the need for monitored therapy of the underlying coagulation disorder, 6 while still allowing early postoperative rehabilitation. 7 Despite this, TKA has been shown to achieve excellent outcomes in this patient group, with significant improvement in pain scores, flexion contractures, range of movement and functional scores. 5,7
Nevertheless, the complication rate appears to be higher in patients with inherited bleeding disorders compared to those with adequate haemostasis. 3 It has been suggested that haemophiliacs have an increased incidence of both periprosthetic joint infection (PJI) and aseptic loosening following TKA, 5,8 with a higher risk of pseudoaneurysm development in the early or delayed postoperative period. 9 While the long-term outcomes and survivorship following TKA in haemophiliacs are well described, 4,10,11 few studies have reported on early haemorrhagic outcomes 12,13 and concerns persist in regards the risk of increased blood loss in this patient cohort during the perioperative period. 1,5,12,14 –16 To our knowledge, there is a relative paucity of evidence evaluating immediate postoperative haemorrhagic outcomes using serological markers in haemophiliacs and comparing these to non-haemophiliac patients.
The objective of this study, was therefore to quantify immediate postoperative blood loss and haematological complications in a cohort of haemophiliacs following TKA and compare this group’s outcomes to those in a non-haemophiliac population.
Materials and methods
A retrospective, observational single-centre study of patients undergoing elective TKA was performed utilising STROBE guidelines. 17 This study took place in a large metropolitan university teaching hospital which also acts as a national tertiary referral centre for hereditary coagulation disorders, thus providing dedicated on-site haematological and orthopaedic services for this particular patient subgroup. Ethical and Data Protection Impact Assessment approvals for both the study and its methodology were obtained from the hospital’s Research and Ethics Committee and Research and Innovation Department respectively.
All patients undergoing elective TKA between 01 January 2005 and 30 December 2016 were identified via a prospectively maintained hospital local database. Patients with an underlying diagnosis of either Haemophilia A (Factor VIII deficiency) or Haemophilia B (Factor IX deficiency) were selected from this group. This time period was selected in an attempt to minimise potential heterogeneity in perioperative haematological therapy by assessing those who underwent contemporary coagulative regimens. Patients suffering from other haematological disorders or undergoing revision knee arthroplasty procedures were excluded.
In addition to routine perioperative TKA workup, all patients underwent a standardised approach to perioperative haematological control in accordance with institutional haemostatic guidelines as directed by the hospital coagulation centre. This is illustrated further in Figure 1. A typical regimen would involve bolus infusion of either FVIII concentrate (Advate®) or FIX concentrate (Benefix®) for Haemophilia A or B, respectively, on the morning of planned surgical intervention, with factor levels tested within 30 min following infusion. A postoperative target of 1.0 IU was then set and each patient underwent subsequent continuous perioperative factor infusion, with the postoperative duration and rate of infusion determined by the senior haematologist in conjunction with daily serological factor levels. Bolus factor administration was additionally utilised upon review by the consulting haematology service. For patients with a positive haemophiliac inhibitor status, this standard regimen would also be used in those with low-responding inhibitors, with bypass agent therapy considered on a case-by-case basis in those with high-responding inhibitors.
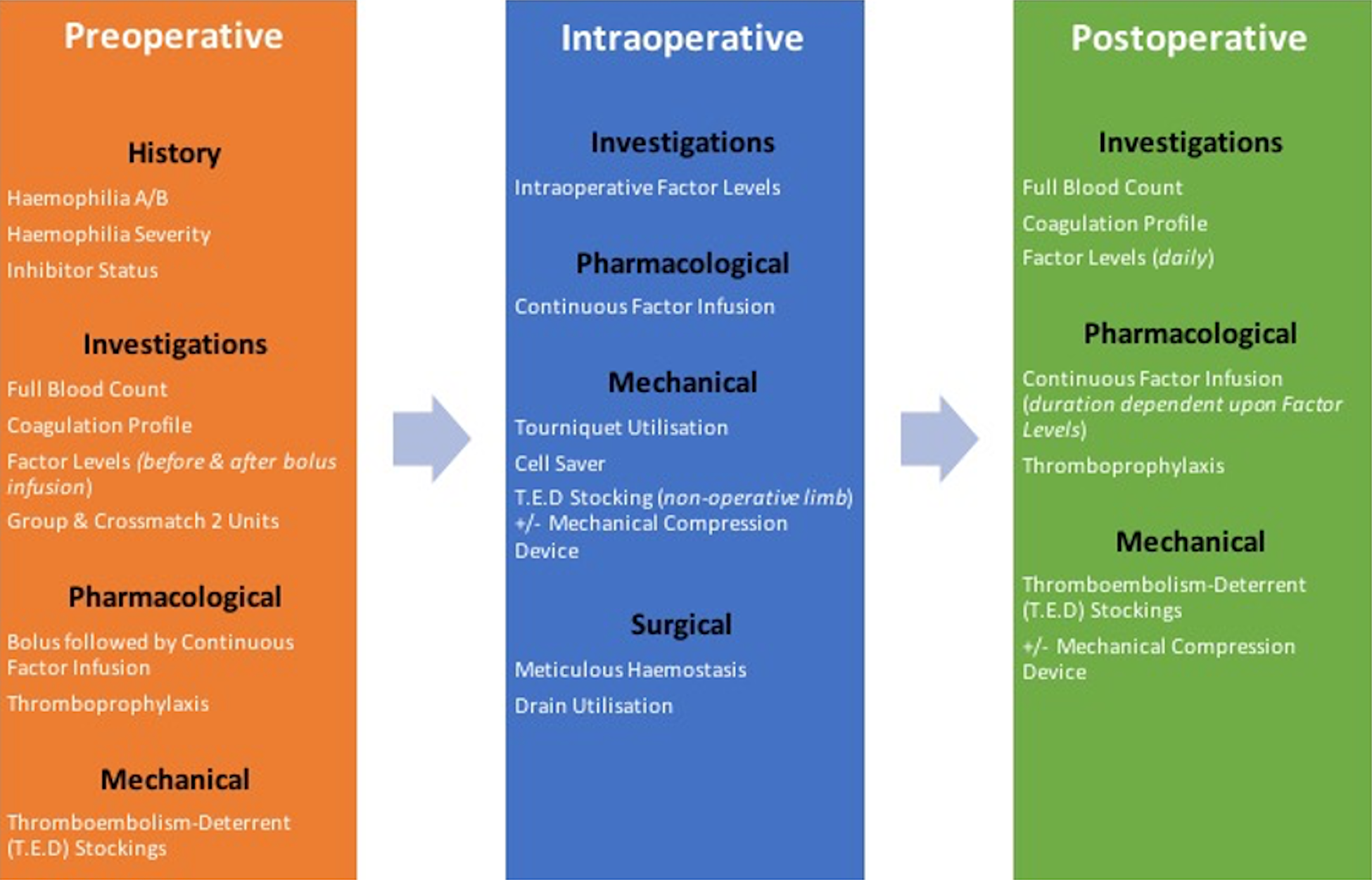
Perioperative haematological workflow for TKA in haemophilia.
Patients identified as suitable for inclusion underwent review of both their electronic patient and medical chart records. Demographic data included age, gender, haemophilia type and inhibitor status. Patients’ underlying hepatitis and HIV status was also recorded and the American Society of Anaesthesiologists Physical Status Scale (ASA-PS) was documented for each patient. Haemophilia severity was quantified as per the World Federation of Haemophilia classification. Arthritis severity was graded on preoperative AP weight-bearing knee radiographs using the Kellegren-Lawrence Classification system.
Operative data included operating consultant, side of surgery, implant type, tourniquet use, overall tourniquet time, estimated blood loss (EBL) and postoperative drain usage. In all patients, a tourniquet was inflated prior to the initial skin incision and deflated following wound closure and application of the postoperative dressing, and as such is used as a surrogate marker of overall operative time. EBL was measured by calculating the difference between the volume of fluid in a suction cannister and the cumulative volume of utilised surgical gauze swabs and utilised intraoperative irrigation fluid. Postoperative drain usage, cumulative output and removal time was also recorded based on the clinical notes. An intraoperative cell-saver was not employed in any case due to the usage of an intraoperative tourniquet.
Evaluation of patients’ perioperative haematological status was primarily through assessment of serological markers obtained via the institution’s electronic patient record system; the parameters collected are displayed in Table 1.
Recorded haematological data parameters.

The above information was recorded into an electronic data spreadsheet, with subsequent assessment involving primarily a quantitative analysis of trends. Descriptive statistics for qualitative data are presented in terms of number (n) and percentage (%).
Quantitative analysis was twofold and in both instances was undertaken using an independent T-test, with a P-value < 0.05 considered statistically significant (SPSS Version 26 for Windows ®). Firstly, a group of non-haemophiliac patients who underwent TKA over the same time period were collated to serve as a control group in order to compare postoperative blood loss. Control group patients underwent surgery by the same surgeons as the haemophiliac group with an identical intraoperative protocol. As per institutional protocol for non-haemophiliac patients undergoing TKA, each of these patients had a Hb level measured at 48 h postoperatively, and as such were compared directly to the 48 h postoperative Hb levels in the in the haemophiliac group.
Secondly, postoperative results in the haemophiliac group were compared to published standards in non-haematological patients, with this method having previously been used by other authors examining bleeding risk in haemophiliacs. 18,19 In this second analysis, the mean 24-h postoperative haemoglobin (Hb) change in our haemophiliac cohort was also compared to published standards in non-haemophiliacs.
Results
Twenty-eight primary TKAs were identified in 25 patients with an underlying haematological disorder. Of these patients, seven were excluded, with two patients having a haematological disorder other than haemophilia (Factor XI and a bleeding disorder of unspecified origin, respectively) and medical charts being unavailable for analysis in another five cases.
This left 21 TKAs in 18 patients suitable for inclusion, with three patients undergoing bilateral TKAs across two separate hospital episodes. Patient demographics are presented in Table 2 and surgical details in Table 3. Each patient was male, with a mean age of 44 years (range 30–55, SD ±8.8) at time of operation.
Patient demographics.

Patient surgical details.

* Triathlon, Stryker, Kalamazoo, Michigan.
† LCS (Low Contact Stress), DePuy, Warsaw, Indiana.
‡PFC (Press Fit Condylar), DePuy, Warsaw, Indiana.
The underlying haematological disorder was haemophilia A in 13 patients (72%) and haemophilia B in 5 patients (28%). Seventeen patients (94%) were diagnosed with severe haemophilia (12 haemophilia A, 5 haemophilia B) and 1 patient (6%) (haemophilia B) was diagnosed as moderate. Three patients (17%) had underlying acquired inhibition to relevant factor treatment (two haemophilia A, one haemophilia B). Overall ASA grading at time of surgery showed a predominance of mild and severe underlying systemic disease, with 4 (22%) and 14 (78%) of the patients identified as either ASA-PS II or III, respectively. Four (22%), 2 (11%), 7 (39%) and 15 (83%) patients had an underlying diagnosis of HIV, hepatitis A, hepatitis B and hepatitis C, respectively.
Fourteen (67%) left TKA and seven (23%) right TKA procedures were performed, with the primary indication for each being haemophilic arthropathy. At time of surgery, all patients (100%) had underlying Kellegren-Lawrence Grade IV arthritis. Each case was performed using cemented implants and was undertaken by one of three specialist consultant orthopaedic surgeons, with eight, seven and six procedures performed by each consultant, respectively. All cases were done via a medial parapatellar approach under tourniquet control, with a mean tourniquet time of 113 min (range 75–156, SD ±20.8). Primary total condylar knee prostheses were used in all cases with no knee requiring a stem or augment.
A 400 ml negative pressure suction reservoir drain device was used in all 21 cases (100%). Owing to operative surgeon preference, the drain was removed following adequate haemostasis on either the first or second postoperative day in 12 (57%) and 9 (43%) cases, respectively. Postoperative drain output was recorded clearly in 18 (86% of patients), with a mean cumulative drain output at 24 h of 364 ml (range 74–1025, SD ±286) in this group. The mean cumulative drain output over 48 h for the nine patients whose drain was removed at 48 h postoperatively was 664 ml (range 100–1250, SD ±400). There was no significant difference in drain output at 24 h between those who had their drain removed at 24 or 48 h respectively (P = 0.3787).
Haematological data is presented in Table 4. Each patient received preoperative haematological factor replacement, with factor levels subsequently deemed adequate prior to surgery. The mean preoperative factor level was 1.22 IU/ml (range 0.78–1.61) and preoperative APPT level was 35.8 s (range 29.5–53). No patient required an intra-operative transfusion. Estimated intraoperative blood loss (EBL) was recorded in perioperative documentation. This was quantified as ‘minimal’ in 11 (52.4%) procedures, with six patients having an EBL of 50 ml or less, one patient an EBL of 75 ml and three having an EBL of 100 ml or less.
Patient haematological details.

* NR: not recorded.
The mean preoperative Hb level for all included patients was 14.4 g/dl (range 9.3–16.5, SD ±1.7). Nineteen (90%) of these patients had serum Hb levels checked at 24 h postoperatively, with a mean drop in Hb of 2.7 g/dl (range 0.2–5.1, SD ±1.3). Similarly, 19 (90%) patients had serum Hb levels at 48 h postoperatively, with a mean drop of 3.8 g/dl (range 0.4–6.8, SD ±1.9).
By ways of comparison, a control group of 19 primary total knee arthroplasty (TKA) procedures performed within the same time period was identified, with a mean age of 55 years (range 34–60, SD ±8.1). The control group’s mean preoperative Hb level was 13.6 g/dl (range 11.9–15.2) and mean 48 h postoperative Hb drop 3.2 g/dl (range 1.3–6, SD ±1.1). When compared to those patients in the haemophiliac cohort who had a Hb level at 48 h postoperatively (n = 19), there was no significant difference in the preoperative Hb levels (P = 0.1183) or mean Hb drop at 48 h (P = 0.2644) postoperatively.
Two (9.6%) patients required a postoperative transfusion of red cells (RCC) following TKA. Both cases were deemed to be technically challenging TKA procedures, requiring extensive intraoperative soft tissue release and dissection to obtain adequate exposure. Postoperatively, the first of these patients (Patient 8 R) showed symptomatic signs of anaemia with an associated cumulative Hb drop of 5.4 g/dl and 6.7 g/dl on the second and third postoperative day respectively, with improvement in Hb levels and resolution of symptoms following transfusion of two units of red cells on the third postoperative day. The second patient (Patient 14) was clinically asymptomatic in the postoperative period but a drop in Hb of 5.1 g/dl was noted at 48 h postoperatively This patient received a transfusion of two units of red cells with a subsequent improvement in Hb levels.
There were no immediate haemorrhagic sequelae or wound complications in any patient prior to discharge, with no patient suffering from an immediate postoperative wound haematoma requiring return to theatre for evacuation. At a mean follow-up of 9.4 years (range 4.5–14.1, SD ±2.9), no patient has undergone revision TKA surgery.
Discussion
Despite modern advances in haematological factor prophylaxis, haemophiliac patients continue to suffer from recurrent joint haemarthrosis. This can precipitate debilitating haemophilic arthropathy, 20 most frequently seen in the knee. 1 In those refractory to conservative therapy, 4 TKA is associated with both excellent pain relief and functional outcomes. 5,7 However, it has been suggested that there is an increased likelihood of haemorrhage in this patient group, 3,14 with bleeding complications previously reported in up to 40% of haemophiliacs following knee surgery. 15
More recently, Moore et al. reported a bleeding-related complication rate of 8.9% in a meta-analysis of 336 TKA procedures in haemophiliac patients. 1 Cancienne et al. documented a transfusion rate of up to 29.1% in haemophiliac patients in the first three months following TKA 14 and a postoperative haemarthrosis rate of up to 5.34% has also been reported in this patient group. 5 Immediate haemorrhagic outcomes are also of concern, with a single study by Anderson et al. evaluating outcomes following hip or knee arthroplasty in patients with inherited bleeding disorders noting a mean postoperative haemoglobin drop of 4.4 g/dl. 16 Although immediate perioperative haemorrhagic outcomes have been reported in haemophiliacs undergoing TKA within the context of evaluating the effect of tranexamic acid (TXA), 12,13 there are limited reports evaluating immediate postoperative haemorrhagic outcomes in terms of serological markers and comparing these with non-haemophiliac patients. Haemorrhagic outcomes are of particular significance given the potential effect excessive bleeding may have on patient outcomes including cardiovascular events, infection and length of stay. 21
Evaluating perioperative blood loss following TKA can be performed through a number of methods. Changes in Hb levels are thought to closely correlate with EBL across all surgical procedures, 22 and have been shown to be of use in measuring blood loss following TKA. 23,24 Measurement of an objective marker such as haemoglobin minimises potential inaccuracies associated with estimation of intraoperative blood loss, hidden blood loss and other confounding factors such as drain usage. Comparison of 48 h postoperative Hb drop in the primary haemophiliac and a non-haemophiliac control group in this study did not show a significant difference in Hb change between the two groups, indicating a potentially comparable level of postoperative haemorrhage following TKA in haemophiliacs.
Perioperative Hb levels are well reported across the literature pertaining to TKA in non-haemophiliacs and thus allowed for a further point of comparison for the primary haemophiliac cohort studied in this paper. Magill et al. evaluated blood loss in 403 primary TKAs and found that up to 90% of perioperative blood loss occurs in the first 24 h postoperatively, making this time period most significant with regards haemostasis, with a mean Hb decrease of 2.6 g/dl at this interval. 25 Other studies by Sehat et al. and Khalfaoui et al. both found similar mean Hb drops of 2.9 g/dl at 24 h post-TKA. 26,27 with Kumar et al. reporting a drop of 3.05 g/dl. 28 Dan et al. reported a mean Hb drop of 2.72 g/dl in 101 patients at 24 h postoperatively, 29 while Cankaya et al. reported a mean drop of 2.4 g/dl. 30 The cumulative mean 24 h Hb drop from the above studies was 2.8 g/dl, with no significant difference found between this value and the 24 h Hb drop detected within the studied haemophiliac cohort (P = 0.8438), further reflecting the trend identified following comparison of the primary haemophiliac group to a non-haemophiliac control group in this study.
Satisfactory haemostasis is additionally demonstrated by the rates of both postoperative transfusion and wound haematoma. A recent systematic review reported an average postoperative transfusion rate of between 7.5% and 22% following unilateral TKA. 31 We recorded a transfusion rate of 9.6% in our cohort of patients, again closely reflecting the range detected in a non-haemophiliac cohort. Similarly, in our study no haemophiliac patients (0%) needed to return to theatre for immediate management of a postoperative wound haematoma, closely reflecting a rate of 0.24% detected in a study of 17,784 primary non-haemophiliac TKA procedures. 32
A variety of factors may have contributed to the apparent acceptable postoperative haemostatic outcomes detected in this haemophiliac TKA group. Each patient was operated upon in a tertiary referral centre for patients with haematological conditions and therefore had timely access to modern haematological therapies delivered by a specialist on-site service. Such an arrangement has previously been associated with reduced perioperative blood loss across other surgical specialities. 33 Additionally, the availability of arthroplasty surgeons with both significant experience and subspecialist interest in bleeding diatheses likely contributed to successful management of this complex patient group, 34 with an increase in usage of agents such as TXA during TKA across all patient groups in this centre now contributing to further improvements in haemostatic outcomes. 12,13
Limitations to our study exist in regards sample size, poorly quantified intra-operative blood loss as well as the retrospective nature of its design .The limited sample size did not allow for an adequately powered comparison of any potential variation in outcomes between Haemophilia A and B subgroups to be drawn with significance and therefore was not included in this study. Furthermore, the small sample size may limit the ability to draw conclusions regarding the true incidence of immediate postoperative transfusion or wound haematoma rates. Nevertheless, we believe this study to be of importance in adding to the current evidence base on management of an infrequently encountered patient group.
Conclusion
Haemophiliacs undergoing a unilateral primary TKA in a specialised tertiary referral centre appear to have comparable rates of perioperative blood loss when compared to both a non-haemophiliac control group as well as published haemostatic standards in non-haemophiliac patients following TKA. Postoperative transfusion and wound haematoma rates also appear to resemble published incidence in non-haemophiliacs following TKA. Perioperative management in a dedicated setting with expert orthopaedic and haematological input is recommended to optimise outcomes in this rare and potentially complex patient group.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Research data
Research Data File not available for dissemination as per Institutional Research and Ethics and General Data Protection Regulation (GDPR) Data Protection Impact Assessment approvals.
