Abstract
Background: Essential amino acid (EAA) supplementation, including conditionally essential amino acid (CEAA) and branched-chain amino acids (BCAA) supplementation, has been suggested as a mechanism to optimize patient outcomes by counteracting the atrophy associated with orthopedic procedures. Purpose: We sought to investigate the effect of EAA supplementation in the perioperative period on patients undergoing orthopedic and spine surgery, specifically whether it is associated with (1) reductions in postoperative muscle atrophy and (2) improved postoperative function including range of motion, strength, and mobility. Methods: We conducted a systematic review of the literature. The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines were used, and the protocol was registered in the Prospective Register of Systematic Reviews (PROSPERO) database (CRD42023447774). Studies of interest were prospective, placebo-controlled, randomized clinical trials (RCTs) published between 2002 and 2023 evaluating the impact of EAA supplementation on patients undergoing orthopedic and spine surgery. Results: Ten RCTs evaluating EAA supplementation in trauma, adult reconstruction, and spine surgery were identified; half of these focused on adult reconstruction. The EAA supplementation dose (3.4-20 g), frequency (daily to 3 times per day), and duration (14-49 days) varied widely across studies. Seven studies reported parameters relating to muscle size and/or composition, with 3 studies reporting superior muscle size/composition in patients receiving perioperative EAA supplementation, when compared with controls. Three studies reported favorable mobility outcomes for patients receiving EAA. Meta-analysis was prohibited by variation in measurement and outcome variables across the studies. Conclusions: Pooled data from level I studies supports the use of EAA, BCAA, and CEAA supplementations across several orthopedic subspecialties. However, significant heterogeneity exists in the quantity, duration, and content of EAA administered. Further prospective studies are needed to determine optimal/standardized parameters for supplementation.
Introduction
Surgical outcomes in orthopedic surgery are increasingly understood to be influenced by myriad factors: degree or severity of underlying pathology, surgical technique, implant design, mental health, socioeconomic status, comorbid conditions, nutritional status, physiologic reserve, and perioperative management [3,15,25,27,28,40,47]. Recent focus has been placed on perioperative optimization to reduce surgical complications and improve outcomes of orthopedic procedures, particularly elective surgery [35]. For example, numerous studies have demonstrated that smoking cessation, deep vein thrombosis (DVT) prophylaxis, and early mobilization lead to improved outcomes across orthopedic subspecialties [44]. In addition, each subspecialty has identified factors unique to procedures or patient population, such as improving bone mineral density prior to posterior spinal fusion or spinal deformity corrections [5,8,13,29,37,46], establishing body mass index (BMI) and hemoglobin A1c thresholds prior to total joint arthroplasty [1,9], and implementing preoperative nerve blocks for geriatric patients with femoral neck fractures [27].
In recent years, increased attention has been paid to nutrition and metabolic health in the perioperative period; Fig. 1 shows publications by subspecialty on essential amino acid (EAA) supplementation. Surgery stresses the body, resulting in a catabolic state associated with increased protein mobilization, lipid oxidation, hepatic glucose production, and insulin resistance [10,18,19,45]. Malnutrition has been associated with delayed mobilization, longer hospital stays, and increased mortality rates [17,20,22,33]. Although malnutrition is less prevalent in the United States than in some other countries, protein malnutrition in older adults can lead to low muscle mass, decreased function, and increased frailty [30]. Moreover, this population is at increased risk for sarcopenia, a diffuse muscular disease (or failure) resulting from advanced age or chronic disease [6]. Several studies have highlighted the importance of perioperative nutrition management, including preoperative nutritional assessments and amino acid and carbohydrate loading and early postoperative enteral nutrition [18,19,34,38]. Optimizing perioperative nutritional status has been associated with improved surgical outcomes and, as a result, has been incorporated in protocols across specialties.
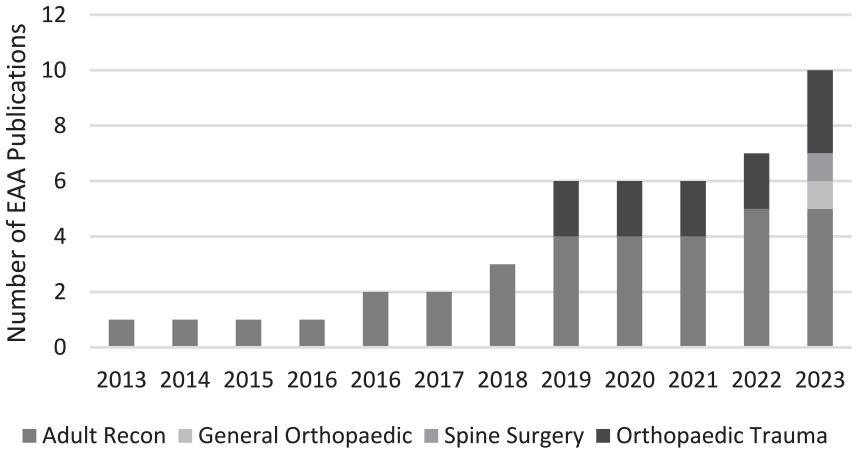
Cumulative publications by subspecialty on EAA supplementation.
Amino acid mobilization is a hallmark of the catabolic state associated with surgery, even in patients without evidence of malnutrition or caloric deficit [21,26]. To mitigate the effects of this catabolic state and the associated skeletal muscle wasting, numerous authors have examined perioperative supplementation of EAAs, which are those that cannot be synthesized by humans and must be obtained through dietary intake [21,41]. Intake of EAAs, leucine in particular, is necessary for muscle protein synthesis, which leads to increased muscle mass and quality [14,36]. The low cost and presumed safety of EAA supplementation make this intervention amenable to prospective clinical trials. Adult reconstruction, with its protocolized workflows and predominantly elderly patient population, has served as a common testing ground.
We sought to summarize prospective, placebo-controlled, randomized clinical trials (RCTs) evaluating EAA, including branched-chain amino acids (BCAAs) and conditionally essential amino acid (CEAA), supplementation in patients undergoing orthopedic and spine surgery. We aimed to assess if available data shows that perioperative EAA, CEAA, and/or BCAA supplementation is associated with (1) reductions in postoperative muscle atrophy and (2) improved postoperative function including range of motion, strength, and mobility.
Methods
Adhering to the guidelines set by Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA), a systematic review of previously published literature was conducted [32]. This study was prospectively registered on PROSPERO on September 4, 2023 (CRD42023447774). The literature search was performed by a trained medical librarian (L.E.B.) in PubMed/MEDLINE, Embase, Web of Science, Scopus, and EBSCO/Cumulative Index to Nursing and Allied Health Literature using key terms and controlled vocabulary around the topics of amino acids, orthopedic surgery, and supplementation. An English language limit was used on all databases. An example search can be viewed in Appendix A. Studies of interest were prospective RCTs evaluating the impact of EAA supplementation for patients undergoing orthopedic and spine surgery. The primary outcome of interest was a change in postoperative muscle mass for patients who received perioperative EAA supplementation compared with controls who did not receive EAA supplementation. Secondary outcomes included specific formulation, dose, and duration of EAA supplementation as well as the impact of EAA supplementation on postoperative outcomes, complication rates, and adverse events (reoperation, wound, or infectious complications). Abstracts, proceedings, letters, and editorials were excluded, as were studies not related to orthopedic outcomes or interventions and those not published in the last 20 years (2002 to 2023).
After the initial literature query, 3 authors (P.M.I., E.B., and S.A.M.) independently reviewed the article titles and abstracts to identify articles for full-text review. Review was performed with Covidence systematic review software. In cases of any discrepancy, the abstract was included in full-text review. After abstract review, full texts were reviewed by 2 independent reviewers (E.B., S.A.M.). In cases of discrepancy, the senior author (P.M.I.) made the final decisions on inclusion or exclusion. To assess inter-rater reliability, a Cohen’s κ coefficient was calculated (0 no agreement, 0.10-0.20 slight agreement, 0.21-0.40 fair agreement, 0.41-0.60 moderate agreement, 0.61-0.80 substantial agreement, 0.91-0.99 near perfect agreement, 1 perfect agreement) [7].
Data Extraction and Statistical Analysis
Next, 2 authors (E.B., S.A.M.) independently extracted data from the studies including manuscript details, population of interest, details of supplementation administered (duration, frequency, type, and dose), control treatment, procedure performed, and clinical outcomes with their significance. Studies were independently evaluated by a single reviewer (E.B.) for adherence to Consolidated Standards of Reporting Trials (CONSORT) statement guidelines [39]. All studies included were prospective RCTs, although some of the studies were not blinded, had limited patient subject numbers, and the majority were conducted at a single institution, potentially adding bias to every study included. In addition, there was a high amount of heterogeneity between studies in amount of time, volume, and composition of EAA supplementation.
Microsoft Excel was used to generate summary statistics including mean, standard deviation, median, and interquartile ranges. Forest plots were performed, where appropriate, using OpenMeta [Analyst] software, version 10.10. Statistical significance was defined as P < .05.
Results
Study Screening and Inclusion
An initial query was performed on August 11, 2023, yielding 1489 studies for abstract review. Following exclusion of duplicates, 1176 abstracts were manually reviewed by 3 reviewers. Proportionate agreement between reviewers 1 and 2 was 95.6% (κ = 0.54, moderate agreement) and between reviewers 1 and 3 was 94.8% (κ = 0.31, fair agreement), whereas reviewers 2 and 3 did not review common texts [7]. Forty manuscripts were included in full-text review, with 10 studies ultimately being deemed appropriate for inclusion (Fig. 2).
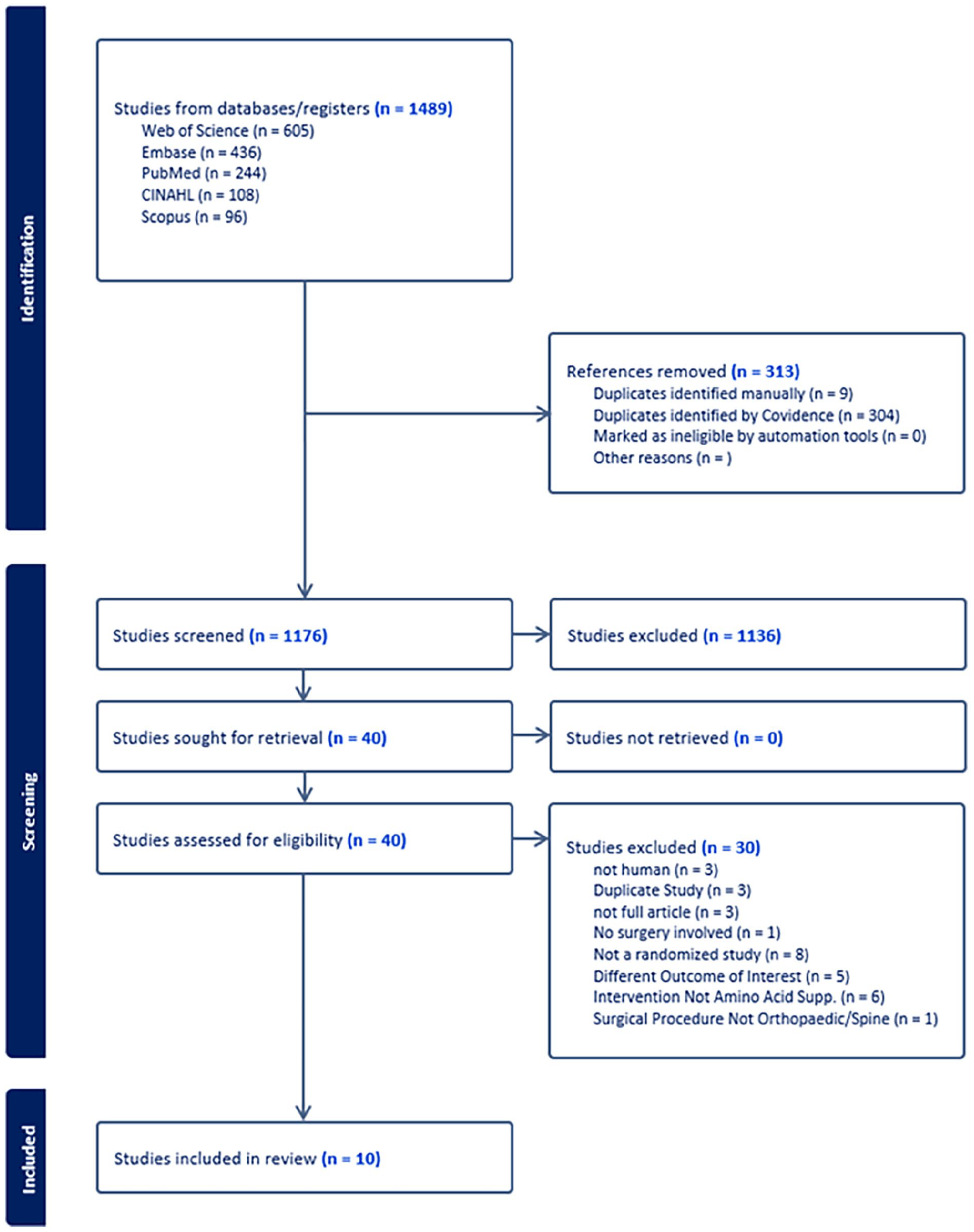
PRISMA flow chart.
Of the 10 studies that met inclusion criteria, 5 (50%) focused on adult reconstruction. Procedures of interest included total knee arthroplasty (TKA, n = 3), total hip arthroplasty (THA, n = 2), operative management of hip fractures (n = 2), pelvic and lower extremity fracture fixation (n = 1), lumbar spine surgery for spinal stenosis (n = 1), and multiple orthopedic surgeries (n = 1).
Amino Acid Formulation, Dosing, and Placebo Supplementation
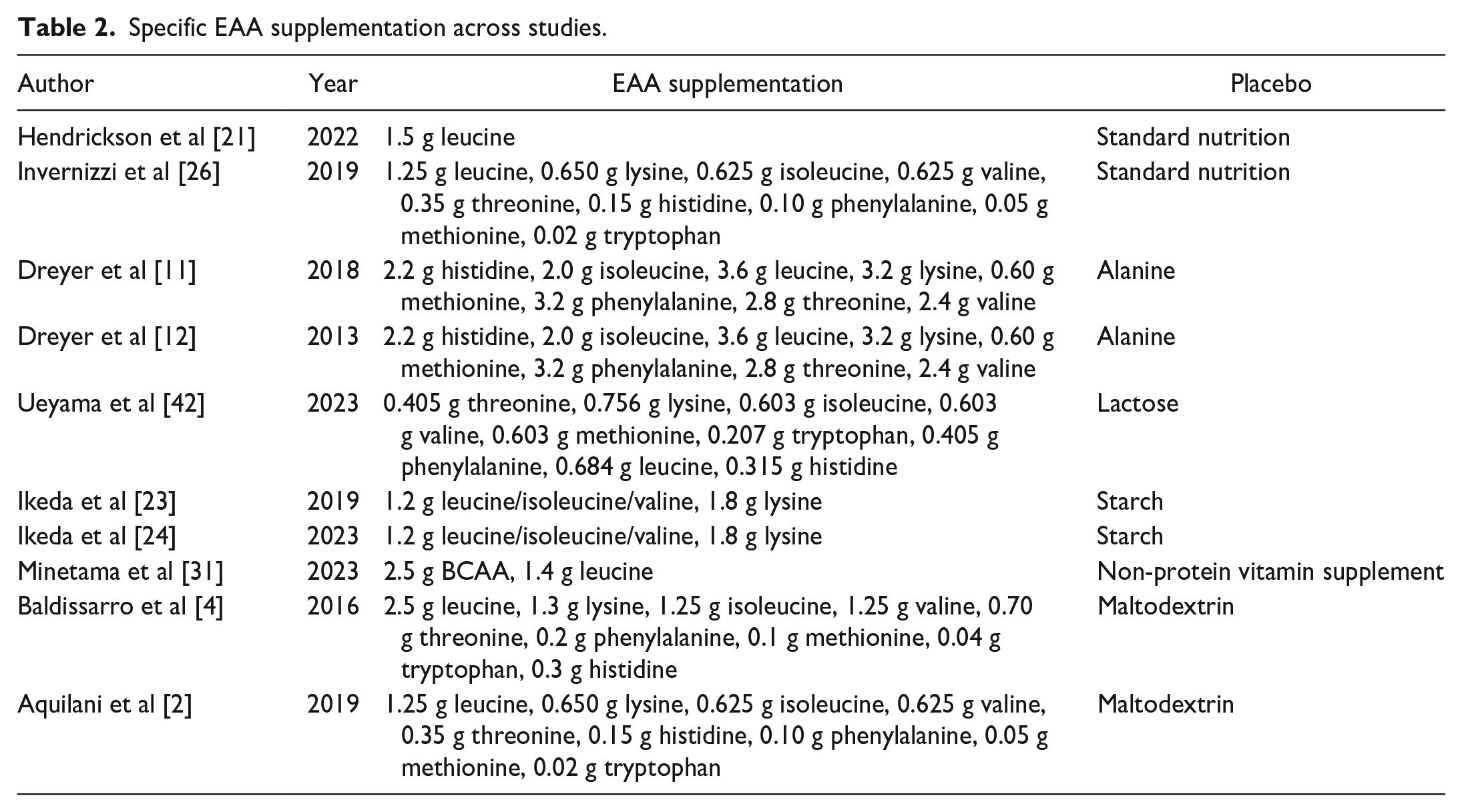
Of the 10 studies reviewed, 3 initiated EAA supplementation 7 days prior to elective orthopedic procedures (Table 1) [11,12,42]. Unweighted average daily dose was 15.8 ± 15.1 g (mean ± standard deviation), with an average treatment duration of 28.1 ± 16.4 days perioperatively. Composition of amino acids varied widely (Table 2). Placebo supplementation was maltodextrin, alanine, or starch in 2 studies each. Other studies used lactose powder, “standard nutrition,” “non-protein” supplement, or no placebo (ie, randomized to standard postoperative care).
Characteristics of amino acid supplementation for included studies.
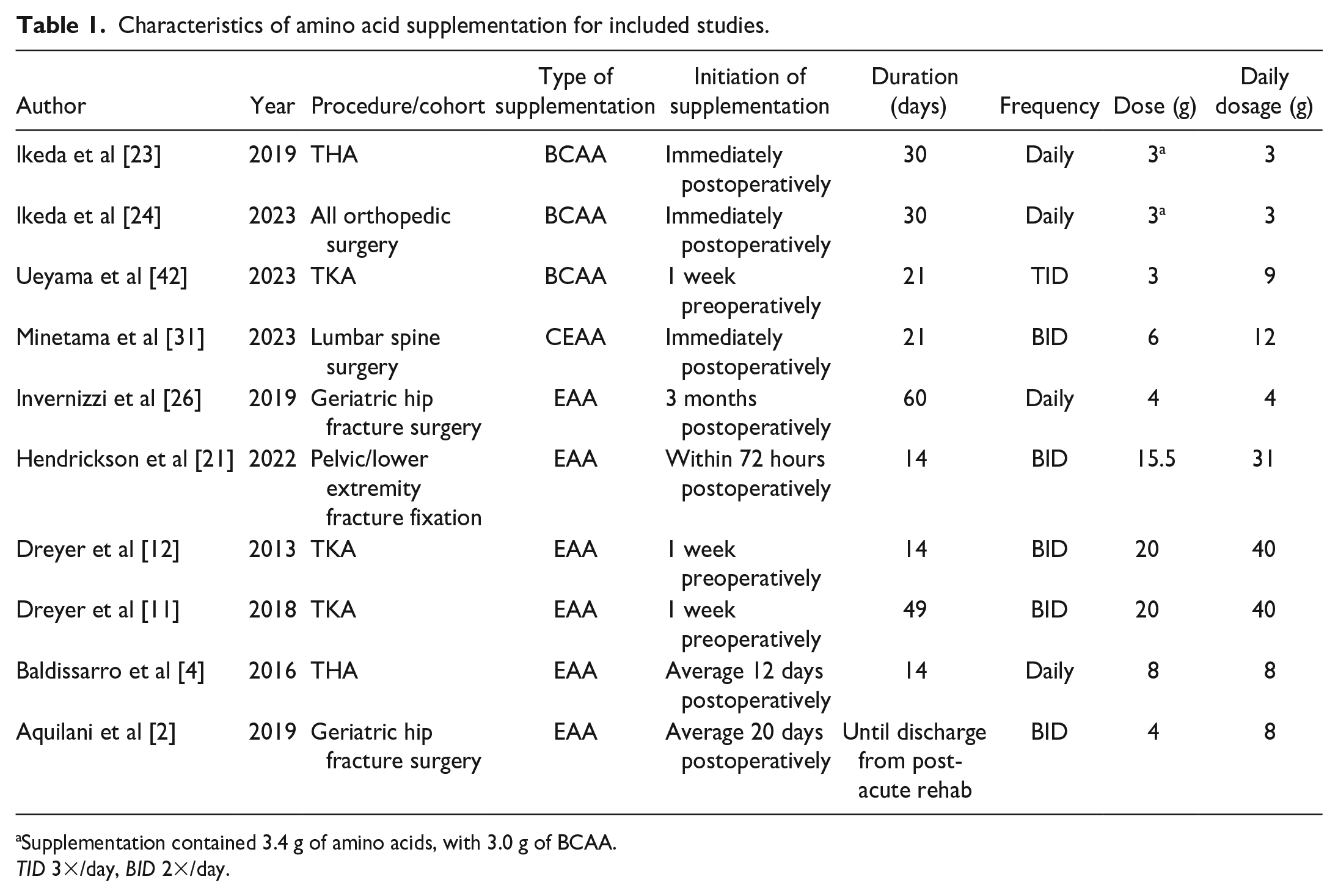
Supplementation contained 3.4 g of amino acids, with 3.0 g of BCAA.
TID 3×/day, BID 2×/day.
Specific EAA supplementation across studies.
Supplementation and Muscle Size and/or Composition
Seven studies reported parameters relating to muscle size and/or composition [11,12,21,23,24,31,42]. Three of the studies found significantly greater quadriceps muscle volume after TKA in patients who received perioperative EAA supplementation compared with those given a placebo, with final follow-up durations ranging from 6 weeks to 2 years (Fig. 3) [11,12,42]. All 3 studies concluded that perioperative EAA supplementation for 1 week preoperatively and 2 to 6 weeks postoperatively reduced muscle loss in older patients after TKA. One study analyzing muscle strength after THA showed a significant increase in upper arm muscle mass but did not look at muscle volume/composition of quadriceps postoperatively [23]. Conversely, 3 studies examining muscle size and/or composition after open reduction and internal fixation of long bone and pelvic fractures or after spine surgery did not find a statistically significant difference between those who received EAA supplementation and those who did not [21,24,31]. However, 1 of these studies noted that the placebo group lost a significant amount of fat-free mass compared with the EAA group after 6 weeks, suggesting that EAA supplementation in the perioperative period prevented global skeletal muscle loss [21] (Table 3).
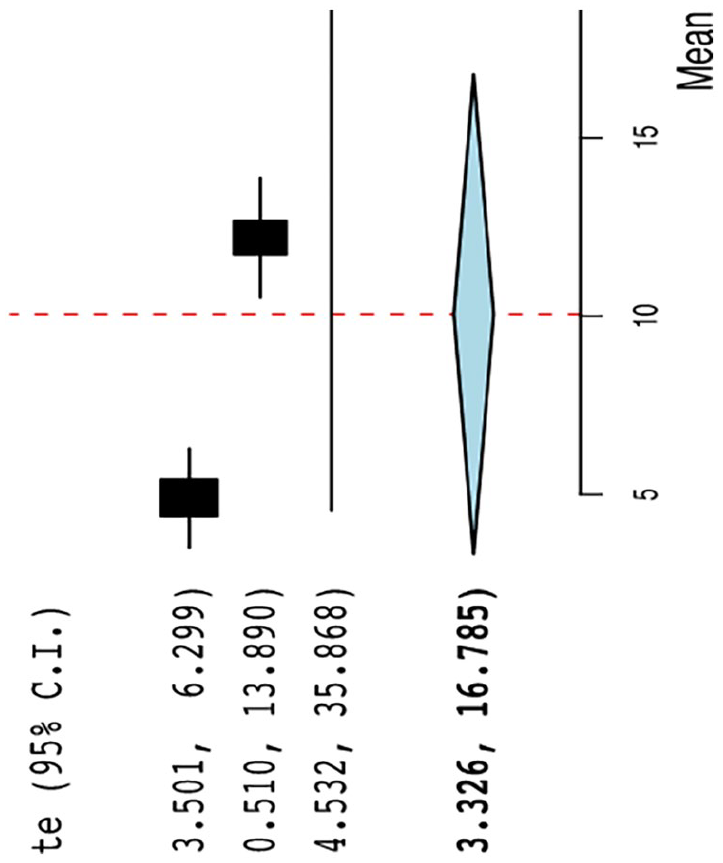
Forest plot of significant mean difference in muscle size at final follow-up in patients who underwent TKA.
Studies analyzing the mean change in muscle mass/volume.
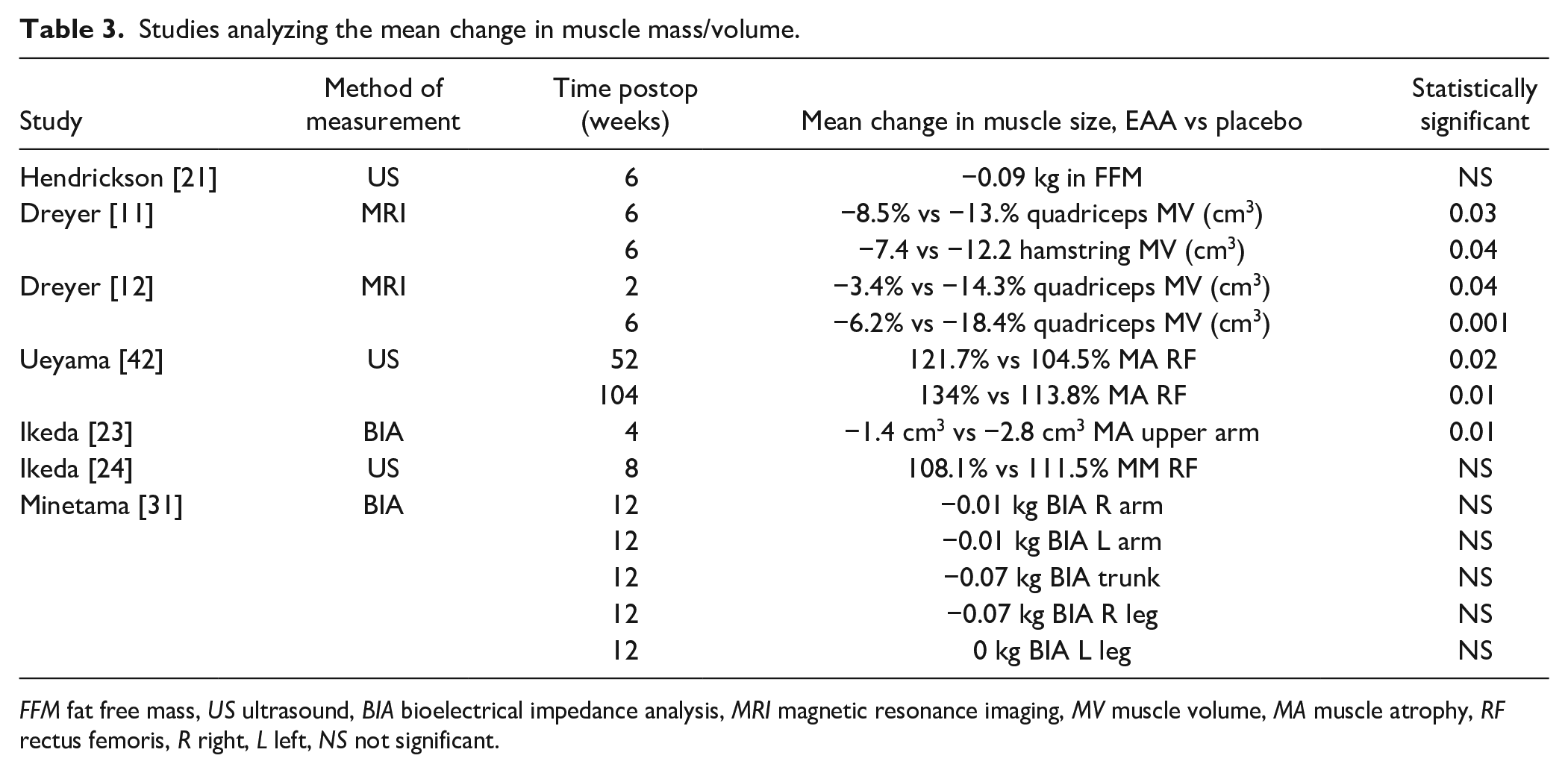
FFM fat free mass, US ultrasound, BIA bioelectrical impedance analysis, MRI magnetic resonance imaging, MV muscle volume, MA muscle atrophy, RF rectus femoris, R right, L left, NS not significant.
Impact of Essential Amino Acid Supplementation on Grip Strength
Of the 10 studies included, 7 analyzed changes in grip strength (Table 4) [12,21,23,24,26,31,42]. Only 1 study found a significant improvement in grip strength between patients treated with EAA supplementation compared with controls [12], whereas the other 6 did not show an intergroup difference in grip strength, 2 found a significant difference in grip strength within groups over time [21,26]. However, for sarcopenic patients, there was a significant increase in mean grip strength only in the EAA-supplemented group and not in the placebo group [26].
Mean differences in grip strength.
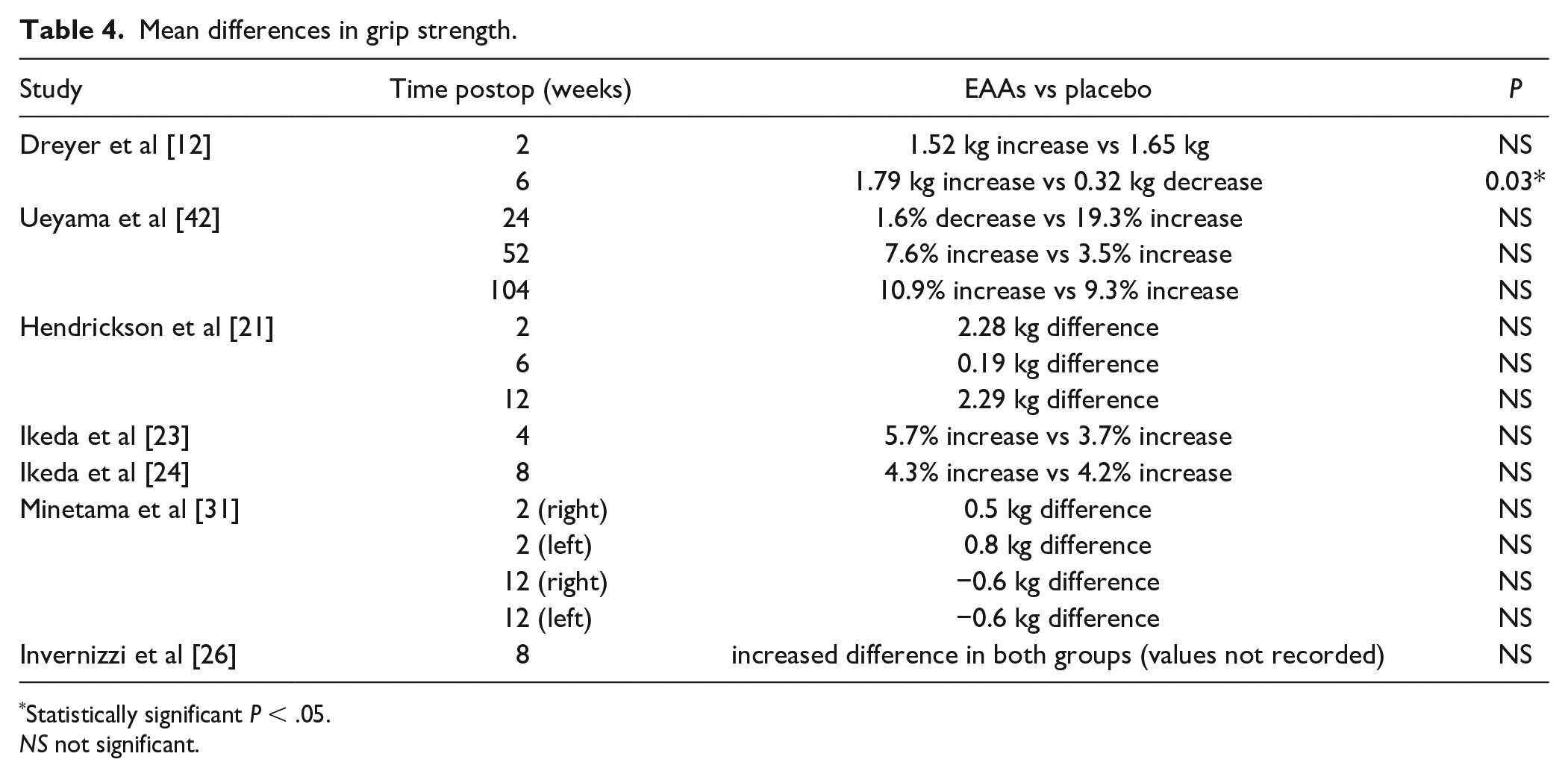
Statistically significant P < .05.
NS not significant.
Impact of Essential Amino Acid Supplementation on Mobility
The effect of EAA supplementation on mobility was evaluated in 6 studies; meta-analysis of study results was prohibited by variation in instruments used and the timeframe in which changes were observed. Measures of patient mobility included stair climb up/down, timed up and go (TUG) test, maximum gait speed, and timed short distance walk. Three studies (2 evaluating patients with TKA and 1 evaluating geriatric hip fracture) reported significant improvements in lower extremity mobility including TUG test, timed walks, and stair assessment for EAA groups compared with controls [11,12,26] (Table 5).
Differences in mobility outcomes.
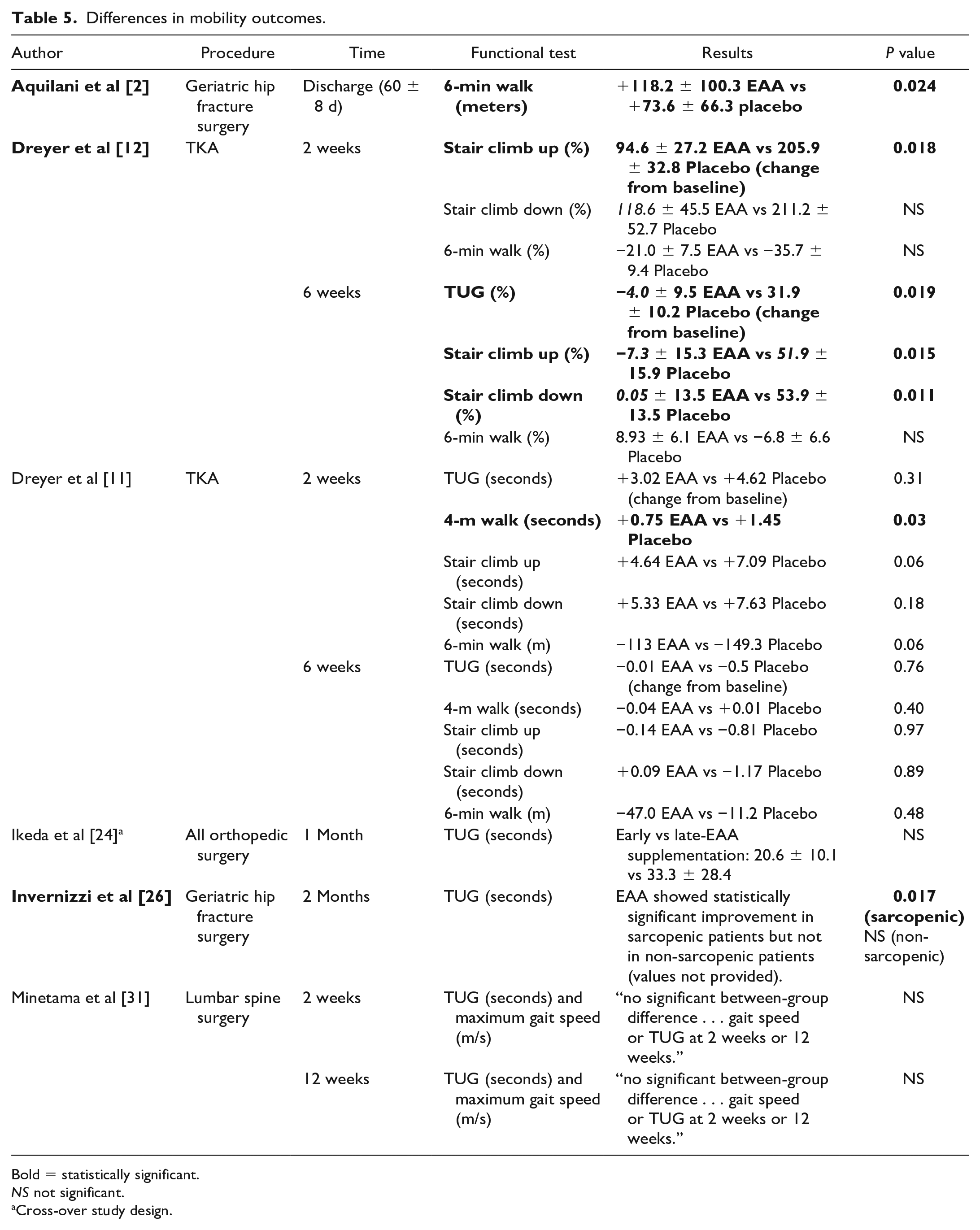
Bold = statistically significant.
NS not significant.
Cross-over study design.
Assessment of Methodologic Quality
The CONSORT scores for each included study are available in Supplemental Table 1.
Discussion
This systematic review of RCTs that assessed EAA supplementation in the perioperative period in patients undergoing orthopedic and spine surgery identified 10 RCTs; half focused on patients undergoing elective adult reconstruction procedures. Notably, the dose, duration, and timing of interventions varied widely across studies. For example, the daily dosage provided ranged from 3 to 40 g/day, a greater than 10-fold difference. Similar variation was seen with regard to the initiation of dosing (ie, preoperatively or postoperatively) and its duration. In addition, the formulations in each study varied and included EAA alone, EAA with non-EAA (glycine with leucine, isoleucine, valine, and lysine), BCAA, and CEAA (such as glutamine and arginine) [43]. As with many medications and interventions, there is likely a maximally efficacious dose and formulation of EAA supplementation, which cannot be determined given the varying procedural interventions and supplementation strategies in these orthopedic studies. Further study may consider providing multiple dose sizes of supplements to better define optimal dosing strategies.
Numerous limitations exist in this systematic review. Differing patient populations, surgical settings, and timing of intervention/outcome measures prevented meta-analyses of our outcomes of interest. Future studies may better define the impact of these variables on the efficacy of EAA supplementation. In addition, varying placebo controls, or lack thereof, also likely affected the relative between-group differences in patients receiving EAA and those receiving control interventions. Moreover, we elected to include studies in which both BCAA and CEAA were given, which deviates from the strictest definition of EAA supplementation, with large variations in dose.
Our primary outcome of interest was a priori determined to be the impact of amino acid supplementation on postoperative muscle volume. Three studies evaluating quadriceps atrophy in patients undergoing TKA indicated that supplementation limits but does not prevent post-surgical atrophy in the operative extremity, which resulted in improved quadriceps muscle volume at 1 and 2 years (Table 3) [11,12,41]. It should be appreciated that quadriceps atrophy following TKA may be a muscle-specific phenomenon. This could result from either the surgical insult inherent in displacing the patella laterally or the direct violation of the quadriceps muscle (ie, mid-vastus or subvastus approach) to allow exposure for implant placement. The use of a tourniquet and the duration of the procedure may also be of consequence. In addition, these results may be difficult to generalize to patients undergoing other orthopedic procedures. For example, a study on the impact of EAAs on general muscle mass in trauma patients failed to demonstrate significant differences when compared with placebo [21].
Numerous studies used surrogate markers of generalized strength and/or mobility (ie, grip strength, the 6-minute walk test, the TUG test) as outcomes of interest. For grip strength, the majority of studies failed to demonstrate significant differences between EAA and control groups [12,21,23,24,26,31,42]. Given the heterogeneity of supplementation and time-points of evaluation, meta-analyses were not performed; thus, a lack of significant findings may be a result of inadequate power in each study. It should also be noted that numerous studies evaluating grip strength [12,21,23,24,26,31,42] used EAA supplementation doses of less than 10 g/day, whereas 40 g was used in the study that demonstrated a statistically significant change in grip strength. Finally, the varying pathologies and procedures encountered in this review may further obscure a potential relationship between EAA supplementation and grip strength preservation. For example, patients undergoing an upper-extremity or forearm procedure may recover and respond differently to this metric than those undergoing a foot and ankle procedure.
Importantly, when analyzing mobility metrics, several studies identified a positive impact of EAA supplementation with improvements in the TUG test, walk tests, and stair tests (Table 5). It is likely that myriad factors play into metrics of generalized function or strength, which may convolute the specific impact of EAA supplementation in the remaining cohorts. Given that quadriceps volume and patient mobility are directly correlated in older populations [16], studies identifying improved quadriceps volume or mobility are likely reporting the same findings through different vehicles.
In conclusion, perioperative EAA supplementation for orthopedic and spine procedures is a concept that has gained traction across subspecialties, but the current literature is limited by immense variation in amino acid content, dosing, timing, and duration of supplementation. Regardless, evidence exists supporting the use of EAA to prevent quadriceps muscle atrophy in patients undergoing TKA. The impact of EAA in other domains is less clear, although some evidence suggests improvement in patient mobility and function with EAA supplementation. Although well-designed studies with consistent methodology are needed to more definitively describe the role of perioperative EAA supplementation on orthopedic and spine surgery outcomes, our analysis of a small body of high-level evidence supports further investigation into the potential benefits of EAA use.
Supplemental Material
sj-docx-1-hss-10.1177_15563316241308265 – Supplemental material for Amino Acid Supplementation May Help Prevent Muscle Wasting After Orthopedic Surgery, but Additional Studies Are Warranted: A Systematic Review of Randomized Clinical Trials
Supplemental material, sj-docx-1-hss-10.1177_15563316241308265 for Amino Acid Supplementation May Help Prevent Muscle Wasting After Orthopedic Surgery, but Additional Studies Are Warranted: A Systematic Review of Randomized Clinical Trials by Elizabeth Brown, Samantha A. Mohler, Shiloah A. Kviatkovsky, Lindsay E. Blake, J. Ryan Hill, Jeffrey B. Stambough and Paul M. Inclan in HSS Journal®
Supplemental Material
sj-docx-2-hss-10.1177_15563316241308265 – Supplemental material for Amino Acid Supplementation May Help Prevent Muscle Wasting After Orthopedic Surgery, but Additional Studies Are Warranted: A Systematic Review of Randomized Clinical Trials
Supplemental material, sj-docx-2-hss-10.1177_15563316241308265 for Amino Acid Supplementation May Help Prevent Muscle Wasting After Orthopedic Surgery, but Additional Studies Are Warranted: A Systematic Review of Randomized Clinical Trials by Elizabeth Brown, Samantha A. Mohler, Shiloah A. Kviatkovsky, Lindsay E. Blake, J. Ryan Hill, Jeffrey B. Stambough and Paul M. Inclan in HSS Journal®
Supplemental Material
sj-docx-3-hss-10.1177_15563316241308265 – Supplemental material for Amino Acid Supplementation May Help Prevent Muscle Wasting After Orthopedic Surgery, but Additional Studies Are Warranted: A Systematic Review of Randomized Clinical Trials
Supplemental material, sj-docx-3-hss-10.1177_15563316241308265 for Amino Acid Supplementation May Help Prevent Muscle Wasting After Orthopedic Surgery, but Additional Studies Are Warranted: A Systematic Review of Randomized Clinical Trials by Elizabeth Brown, Samantha A. Mohler, Shiloah A. Kviatkovsky, Lindsay E. Blake, J. Ryan Hill, Jeffrey B. Stambough and Paul M. Inclan in HSS Journal®
Supplemental Material
sj-docx-4-hss-10.1177_15563316241308265 – Supplemental material for Amino Acid Supplementation May Help Prevent Muscle Wasting After Orthopedic Surgery, but Additional Studies Are Warranted: A Systematic Review of Randomized Clinical Trials
Supplemental material, sj-docx-4-hss-10.1177_15563316241308265 for Amino Acid Supplementation May Help Prevent Muscle Wasting After Orthopedic Surgery, but Additional Studies Are Warranted: A Systematic Review of Randomized Clinical Trials by Elizabeth Brown, Samantha A. Mohler, Shiloah A. Kviatkovsky, Lindsay E. Blake, J. Ryan Hill, Jeffrey B. Stambough and Paul M. Inclan in HSS Journal®
Supplemental Material
sj-docx-5-hss-10.1177_15563316241308265 – Supplemental material for Amino Acid Supplementation May Help Prevent Muscle Wasting After Orthopedic Surgery, but Additional Studies Are Warranted: A Systematic Review of Randomized Clinical Trials
Supplemental material, sj-docx-5-hss-10.1177_15563316241308265 for Amino Acid Supplementation May Help Prevent Muscle Wasting After Orthopedic Surgery, but Additional Studies Are Warranted: A Systematic Review of Randomized Clinical Trials by Elizabeth Brown, Samantha A. Mohler, Shiloah A. Kviatkovsky, Lindsay E. Blake, J. Ryan Hill, Jeffrey B. Stambough and Paul M. Inclan in HSS Journal®
Supplemental Material
sj-docx-6-hss-10.1177_15563316241308265 – Supplemental material for Amino Acid Supplementation May Help Prevent Muscle Wasting After Orthopedic Surgery, but Additional Studies Are Warranted: A Systematic Review of Randomized Clinical Trials
Supplemental material, sj-docx-6-hss-10.1177_15563316241308265 for Amino Acid Supplementation May Help Prevent Muscle Wasting After Orthopedic Surgery, but Additional Studies Are Warranted: A Systematic Review of Randomized Clinical Trials by Elizabeth Brown, Samantha A. Mohler, Shiloah A. Kviatkovsky, Lindsay E. Blake, J. Ryan Hill, Jeffrey B. Stambough and Paul M. Inclan in HSS Journal®
Supplemental Material
sj-docx-7-hss-10.1177_15563316241308265 – Supplemental material for Amino Acid Supplementation May Help Prevent Muscle Wasting After Orthopedic Surgery, but Additional Studies Are Warranted: A Systematic Review of Randomized Clinical Trials
Supplemental material, sj-docx-7-hss-10.1177_15563316241308265 for Amino Acid Supplementation May Help Prevent Muscle Wasting After Orthopedic Surgery, but Additional Studies Are Warranted: A Systematic Review of Randomized Clinical Trials by Elizabeth Brown, Samantha A. Mohler, Shiloah A. Kviatkovsky, Lindsay E. Blake, J. Ryan Hill, Jeffrey B. Stambough and Paul M. Inclan in HSS Journal®
Supplemental Material
sj-docx-8-hss-10.1177_15563316241308265 – Supplemental material for Amino Acid Supplementation May Help Prevent Muscle Wasting After Orthopedic Surgery, but Additional Studies Are Warranted: A Systematic Review of Randomized Clinical Trials
Supplemental material, sj-docx-8-hss-10.1177_15563316241308265 for Amino Acid Supplementation May Help Prevent Muscle Wasting After Orthopedic Surgery, but Additional Studies Are Warranted: A Systematic Review of Randomized Clinical Trials by Elizabeth Brown, Samantha A. Mohler, Shiloah A. Kviatkovsky, Lindsay E. Blake, J. Ryan Hill, Jeffrey B. Stambough and Paul M. Inclan in HSS Journal®
Footnotes
Appendix
PubMed search—August 11, 2023.

| Search # | Search terms | Number of results |
|---|---|---|
| #1 | (“amino acids, essential”[MeSH] OR “Amino Acids, Branched-Chain”[Mesh] OR “Arginine”[MeSH] OR “Histidine”[MeSH] OR “Isoleucine”[MeSH] OR “Leucine”[MeSH] OR “Lysine”[MeSH] OR “Methionine”[MeSH] OR “Racemethionine”[MeSH] OR “omega-N-Methylarginine”[MeSH] OR “Phenylalanine”[MeSH] OR “N-Formylmethionine Leucyl-Phenylalanine”[MeSH] OR “Threonine”[MeSH] OR “Phosphothreonine”[MeSH] OR “Tryptophan”[MeSH] OR “Valine”[MeSH] OR “amino acids”[tiab] OR Arginine[tiab] OR Histidine[tiab] OR Isoleucine[tiab] OR Leucine[tiab] OR Lysine[tiab] OR Methionine[tiab] OR Racemethionine[tiab] OR omega-N-Methylarginine[tiab] OR Phenylalanine[tiab] OR N-Formylmethionine Leucyl-Phenylalanine[tiab] OR Threonine[tiab] OR Phosphothreonine[tiab] OR Tryptophan[tiab] OR Valine[tiab]) | 779,022 |
| #2 | (“Orthopedic Procedures”[Mesh] OR “Fracture Fixation”[Mesh] OR “arthroplasty”[Mesh] OR Arthroplasty[tiab] OR fracture*[tiab] OR orthopedic*[tiab] OR orthopaedic*[tiab] OR surgery[ti] OR surgical[ti]) | 1,332,585 |
| #3 | (“Dietary Supplements”[Mesh] OR supplement*[tiab]) | 463,738 |
| #4 | #1 AND #2 AND #3 | 253 |
| #5 | #4 and English language limit | 244 |
CME Credit
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Human/Animal Rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration.
Informed Consent
Informed consent was not required for this systematic review.
Level of Evidence
Level I: Systematic Review of Level I Studies.
Required Author Forms
Disclosure forms provided by the authors are available with the online version of this article as supplemental material.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
