Abstract
Background:
Due to endogenous neuronal deficiency and glial scar formation, spinal cord injury (SCI) often leads to irreversible neurological loss. Accumulating evidence has shown that a suitable scaffold has important value for promoting nerve regeneration after SCI. Collagen/heparin sulfate scaffold (CHSS) has shown effect for guiding axonal regeneration and decreasing glial scar deposition after SCI. The current research aimed to evaluate the utility of the CHSSs adsorbed with mesenchymal stem cells (MSCs) on nerve regeneration, and functional recovery after acute complete SCI.
Methods:
CHSSs were prepared, and evaluated for biocompatibility. The CHSSs adsorbed with MSCs were transplanted into these canines with complete SCI.
Results:
We observed that MSCs had good biocompatibility with CHSSs. In complete transverse SCI models, the implantation of CHSS co-cultured with MSCs exhibited significant improvement in locomotion, motor evoked potential, magnetic resonance imaging, diffusion tensor imaging, and urodynamic parameters. Meanwhile, nerve fibers were markedly improved in the CHSS adsorbed with MSCs group. Moreover, we observed that the implantation of CHSS combined with MSCs modulated inflammatory cytokine levels.
Conclusions:
The results preliminarily demonstrated that the transplantation of MSCs on a CHSS could improve the recovery of motor function after SCI. Thus, implanting the MSCs-laden CHSS is a promising combinatorial therapy for treatment in acute SCI.
Keywords
Introduction
Spinal cord injury (SCI) results in high mortality rates, deficiency of autonomic, and sensory-motor functions. 1 SCI brings about huge impact on property and mentality to patients, and seriously restricts social stability. 2 Although current interventions including the administration of anti-inflammatory agents, and surgery of the vertebral column can extend the lifespan of patients, no effective strategies that substantially promote recovery of functional loss have been established. 3 Thus, one potential therapy is to rebuild the loss of spinal cord tissue, and provide a supportive substrate to guide neurons. Implantation of biological scaffolds combined with stem cell may be an optimal alternative for SCI treatment.
Due to the complex microenvironment in the spine cord and its limited ability of nerve regeneration in human, there is currently no effective strategy to treat SCI. 4 Experimental strategies utilizing different types of cells are being studied extensively. 5,6 Adult mesenchymal stem cell (MSCs) can secrete cytokines, and restrain inflammation response. 7 –9 However, stem cells implantation alone do not show satisfactory results in clinical trials. 10 Therefore, the implantation of MSCs combined with a biological scaffold will facilitate the functional restoration after SCI. It is necessary to assess these approaches on larger animals whose anatomical structures and immune responses are closer to humans, and which allow us to have a long follow-up period.
At present, collagen scaffolds were suitable biomaterial for SCI treatment in terms of preparation cost, degradation products and mechanical properties. 11 Collagen possesses low antigenicity, and excellent biocompatibility and biodegradability. 12 However, many studies have shown that it is not ideal to use collagen scaffold alone to improve neurological function, which might be related to the lack of strategies to alleviate secondary injury, such as inflammation. 13 Furthermore, transplanting collagen scaffolds alone without any functional modification into the lesion sites could significantly inhibit formation of glial scar within the lesion center. 11,14,15 Heparin sulfate is a glycosaminoglycan that consist of neuronal basement membrane, which plays an important role in neural regeneration. 16 Previous researches have showed that in the fabricating process of the biomaterial scaffold, a cross-linking reaction between collagen and heparin sulfate could significantly increase the mechanical strength of the scaffold. 16 Newly researches reported that the CHSSs have good biocompatibility with olfactory ensheathing cells and bone marrow mesenchymal stem cells. 10,17,18 In recent years, therapeutic strategy for SCI has been developed from simple stem cell implantation to a combined therapy based on biomaterial combining various stem cells, and has been obtained certain effect. 17 Interestingly, whether CHSSs modulate MSCs growth by regulating the above or another potential signaling pathway in acute SCI is attractive.
In this study, our objective was to develop a functionalized scaffold using a conceptually new technology with synergistic incorporation of collagen and heparin sulfate. The experiments were to assess the effects of transplanting CHSSs seeded with MSCs in canines after SCI.
Materials and methods
In this study, we mainly aimed to assess the therapeutic effects of CHSSs functionalized with MSCs on promoting axonal regeneration, and functional recovery after SCI. In total, 12 Beagle dogs that underwent complete spinal cord transection were randomly divided into two groups: control group and CHSS functionalized with MSCs group. The CHSS alone was not utilized as a separate group, as CHSS transplantation alone without any functional modification into the lesion sites in rats and dogs has already been repeatedly validated to notably restrain deposition of chondroitin sulfate proteoglycans within the lesion center. 11,14,19,20 Furthermore, the scaffold alone showed very limited effect on promoting nerve regeneration. 21 Therefore, CHSS is a proper scaffold only to guide axonal regeneration and reduce glial scar formation.
Ethics statement
All experimental procedures were performed according to the care and use of laboratory animals form US National Institute of Health (NIH), and approved by the Ethics Committee of Characteristic Medical Center of Chinese People’s Armed Police Force (approval numbers 02688049).
CHSS preparation
The fresh bovine aponeuroses were obtained from a slaughterhouse (Wanlifa, Beijing, China) and repeatedly rinsed with cold distilled water, the fat tissue was removed, and the tendon was crushed and soaked in 0.05 M Tris buffer for 24 hours to remove soluble impurities. After centrifugation, the precipitate was added to an acetic acid solution containing pepsin. After full swelling, the supernatant was collected. The salting precipitate was collected after centrifugation and dialyzed in deionized water for 5 days to obtain a purified collagen gel. Previous study had reported that a mass ratio of 20:1 (collagen/heparin sulfate) could bring the best biocompatibility and mechanical properties for collagen-heparin sulfate composite scaffold. 22 Therefore, the collagen and heparin sulfate were dissolved into 0.05 M acetic acid solution according to the above proportion, and then the solution was uniformly mixed using magnetic stirrer. The mixed solution was irradiated to ultraviolet lamp for 10 min (365 nm, 18 W cm−2) to induce a cross-linking reaction. The mixture was freeze-dried with a vacuum freeze dryer for 2 days, and immersed in 1% NaOH solution for 12 h. Finally, the composite material of collagen and heparin sulfate was douched in deionized water repeatedly. The samples were sterilized by Co60. The product was a standard CHSS.
Isolation, culture, and identification of human mesenchymal stem cells
Human MSCs were derived from umbilical cord according to the protocols reported before, 16,23 MSCs were cultured in low-sugar Dulbecco’s Modified Eagle medium containing 10% Fetal bovine serum. After 24 h, nonadherent cells were removed. MSCs were identified by a Cytomics FC 500 flow cytometer (BD Biosciences, San Jose, CA) of CD73, CD90, and CD105 (FITC-conjugated antibodies, Cambridge, Abcam, U.K.). The third passage MSCs were adsorb into the CHSSs in vitro.
Scaffold biocompatibility
The experiment was divided into co-culture group and control group without CHSSs. The CHSS was cut into 5-mm × 5-mm specimens for co-culture. For cell seeding, 100 µl of MSCs suspension (1 × 105 cells/ml) was seeded onto CHSSs, and co-cultured for 7 days. Then the growth of the MSCs were observed under an inverted phase-contrast microscope and a scanning electron microscope (SEM) (Hitachi, Tokyo, Japan). Finally, the CHSSs co-cultured with MSCs were coated with gold, the MSCs growth were observed under a SEM. The CHSSs without MSCs were also analyzed by SEM.
At 1, 3, 5 and 7 days after seeding MSCs, Cell Counting Kit-8 (CCK-8, Solarbio Science & Technology Co., Ltd.) was performed to assess the proliferation of MSCs co-cultured with CHSSs. The experiment was divided into co-culture group and control group without CHSSs. Five replicate wells were prepared in each group. Briefly, according to manufacturer’s instruction, 30 µl serum-free medium containing 10% CCK-8 reagent were added to each well, and then the samples were incubated for 4 h. Finally, the solution (100 µl) was transferred into a 96-well plate. The optical density (OD) at 450 nm were measured by a multi-mode microplate reader (Bio Tek, Synergy2, USA).
CHSS co-cultured with MSCs and the control group without CHSS were incubated for 1 week in neural differentiation medium containing 100 ng/ml Dulbecco’s modified eagle medium (DMEM/F12) (Gibco, New York, USA), 2 ng/ml EGF (Amresco, St. Louis, Washington, USA), 10% fetal bovine serum (MRC Biotechnology Co., Ltd.), 2%N2/B27 (Invitrogen, New York, USA), and 100U/ml penicillin/streptomycin (Beijing Solarbio Science & Technology Co., Ltd). Cells differentiation were identified by immunofluorescence. The cells were incubated with primary antibodies against NSE (1:200, Chemicon, USA) and GFAP (1:200, Chemicon, USA) at 4°C overnight. The slides were washed with PBS (three times, 5 min each), followed by incubation with a secondary antibody against rabbit IgG conjugated to FITC (1:200, Life Technologies, USA) at room temperature for 1 hour in the dark. The samples were analyzed under an OLYMPUS inverted fluorescent microscope. Four samples from each group were performed quantitative analyses, six random areas of each sample were chosen to record the positive NSE and GFAP at 200× magnification. The percentage of positive NSE and GFAP (positive staining area/total area) was analyzed by Image-Pro Plus software (Media Cybernetics).
Spinal cord injury and transplantation
Twelve healthy canines (beagle, female, 1 year old, weighing 11–14 kg) were supplied from Beijing Fang Yuan Yuan Animal Center (animal license number SCXK 2019-0012, animal batch number No.12415315715834). The animal raising conditions were as follows: animals were fed in single cage with appropriate temperature and relative humidity (T = 22–23°C, H = 50–60% RH). The canines were randomly divided into control group (undergoing spinal cord transection without MSCs/CHSS implantation, n = 6), and CHSS + MSCs group (undergoing spinal cord transection followed by implantation of the CHSS loaded with MSCs, n = 6).
The Beagle canines were received complete spinal cord transection as previous reported. 24 A 4–5 mm long section of the spinal cord at T8–11 levels was completely transected. After complete hemostasis, the CHSS co-cultured with 1 × 107 MSCs was transplanted for the CHSS + MSCs group. The dura mater was sutured tightly, and the paraspinal muscles and skin were sutured separately. The beagles were kept warm by using a blanket and an electric heater until the beagles recovered completely from anesthesia. The bladder was squeezed six times a day. Beagles with dysuria underwent indwelling catheterization until spontaneous urination was restored. The beagles were intramuscularly injected with gentamicin (80000 units/day) once a day for 7 days after SCI to prevent infection. To prevent complications such as hypothermia and hypotension, the beagles were injected intravenously with ringer’s lactate solution (200 ml/day) once a day for 3 days after surgery.
Assessment of neurological function
In canines, the Olby scoring system was used to evaluate the movements of the hindlimbs before implantation and at 0.5, 1, 3, and 6 months after implantation. Two researchers blinded to the experimental groups observed the videos to give each canine a corresponding score.
At 6 months after SCI, the motor evoked potential (MEP) was measured in each canine as described previously, 24 The MEP signals of left and right hind limbs were recorded using Nicolet Viking Quest evoked potential equipment (Nicolet Biomedical Inc., San Carlos, CA, USA).
At 6 months after SCI, a 3.0T Magnetic resonance imaging (MRI) scanner (Magnetom Verio, Siemens, Germany) was performed in each canine. The workstation (Advantage Windows, version 4.2; GE Healthcare, Waukesha, Wisconsin, USA) was used to obtain diffusion tensor imaging (DTI).
Urodynamics assessment
Urodynamics assessment (ELLIPSE CHAIR, ANDROMEDA, Germany) was performed in all groups at 6 months postoperatively. The urinary meatus in female beagles located at the distal end of the cervical opening of the ventral wall (4–6 cm away from the vaginal outdoor). 25 After general anesthesia, vaginal outdoor was propped up with a ring forcep to expose the urinary meatus. A 7F bladder manometer catheter was inserted into the bladder. Residual urine volume was emptied with a syringe. A 9F rectal manometer balloon catheter was placed into the rectum and filled the balloon with saline (37°C). 0 kPa was defined as the level of pubic symphysis at atmospheric pressure, then vented the gas in the catheter.
Urethral pressure profile
The 7F bladder manometer catheter was connected via a three-way stopcock to a pressure transducer and a micro-infusion pump, then the saline was infused uniformly into the bladder (3 ml/min) through 7F bladder manometer catheter with the micro-infusion pump. 26 Urine appeared at the urinary meatus as a beginning of voiding. Bladder threshold volume, detrusor pressure (Pdet) and bladder compliance (BC) were recorded subsequently.
Urethral function study
The manometer catheter was connected to an infusion pump and a pressure transducer, respectively, then the saline was infused (3 ml/min) uniformly and the catheter was pulled out (2 mm/s) uniformly to measure urethral pressure (UP) simultaneously.
Measurement of cytokines level
The levels of inflammatory cytokines were measured to estimate the effects of CHSS combined with MSCs transplantation on inflammation in the cerebrospinal fluid at 7, 14, and 28 days after SCI according to previously reported method. 27 Under general anesthesia with isoflurane inhalation (volume concentration 2.0%, oxygen flow 2 l/min), the cerebrospinal fluid was drawn from the cisterna magna in the canines. The levels of TNFα, IL-10, IL-1β, and TGF-β1 were determined according to the instructions of ELISA kit (Xin fan Biotechnology Co., LTD, Shanghai, China).
Statistical analysis
Data are presented as the mean ± standard deviation (SD). Data were analyzed using the SPSS 15.0 package (SPSS, Chicago, IL, USA). One-way analysis of variance (ANOVA) was analyzed for multiple-group comparisons. Statistically significant difference in two groups comparison was determined using Mann-Whitney U test Fisher’s exact test, or t-test. P values less than 0.05 were statistically significant.
Results
Structure and biocompatibility of the CHSS
The CHSS was fabricated using freeze drying technology (Figure 1(a)).The SEM images revealed that the CHSS had a three-dimensional porous structure and that the pores were connected (Figure 1(b)). MSCs were mostly fusiform under a phase-contrast microscopy at 3 days after culture (Figure 1(c)). After the MSCs and scaffolds were co-cultured at 7 days, SEM results showed that MSCs adhered firmly on the surface of the CHSS, and the cells grew inside the pores (Figure 1(d)). The special biomarker CD90 and CD105 of MSCs were detected by immunofluorescence staining (Figure 1(e)–(h)). The OD values showed no significant differences between the two groups at 1–7 days (P > 0.05) (Figure 1(i)).

Morphology and characterization of human mesenchymal stem cells (MSCs) and the collagen/heparin sulfate scaffold (CHSS). (a) Photograph of the CHSS. (b) Scanning electron microscope images of the CHSS. (c) MSCs morphology observed by phase-contrast microscopy. (d) Morphology of the CHSS co-cultured with MSCs as visualized by scanning electron microscopy: the red arrows indicated MSCs. ((e)-(h)) Immunofluorescence images of MSCs immunostained with CD90, and CD105: blue indicates DAPI, green indicates CD90, and red indicates CD105. (i) Cell Counting Kit-8 assay of the MSCs cultured with the CHSS after 1, 3, 5, and 7 days of co-culture. Scale bars = 5 mm in (b); 100 mm in (c, e, f, g, h); 50 mm in (d). OD: optical density. CD: Cluster of differentiation; DAPI: 4,6-diamino-2-phenyl indole. Data are shown as the mean + sd; SD: standard deviation.
Compare with the control group, CHSS co-cultured with MSCs group significantly promoted expression of NSE and inhibited expression of GFAP (Figure 2(a)–(e)).

Positive cell ratio of GFAP, and NSE in differentiated MSCs. Compare with the control group, CHSS co-cultured with MSCs group significantly increased expression of NSE and inhibited expression of GFAP. * P < 0.05, ** P < 0.01 vs control group. Scale bars = 50 µm in panels (a, b, c, d) and Positive cell ratio of GFAP, and NSE in control group and CHSS co-cultured with MSCs group (e). Data are shown as the mean + sd; SD: standard deviation.
Recovery of motor and electrophysiological function in canines
At 1, 3, and 6 months after transplantation, the Olby scores of the CHSS + MSCs group were significantly better than those in the control group (P < 0.05 or P < 0.01; Figure 3). After 6 months, the hind limbs of the canines in the CHSS + MSCs group showed some standing and ambulating abilities, while the control group displayed no voluntary movement (video. S1–2). In addition, compared with the control group, the amplitude and latency of MEP in the CHSS + MSCs group were noticeably improved (Figure 4(a)–(c)).

Post-injury behavioral evaluations in canines. Olby scores before and 0.5, 1, 3 and 6 months after surgery. * P < 0.05, ** P < 0.01, vs control group. CHSS: collagen/heparin sulfate scaffold; MSCs: mesenchymal stem cells; SCI: spinal cord injury. Data are shown as the mean + sd (n = 6 at each time point); SD: standard deviation.
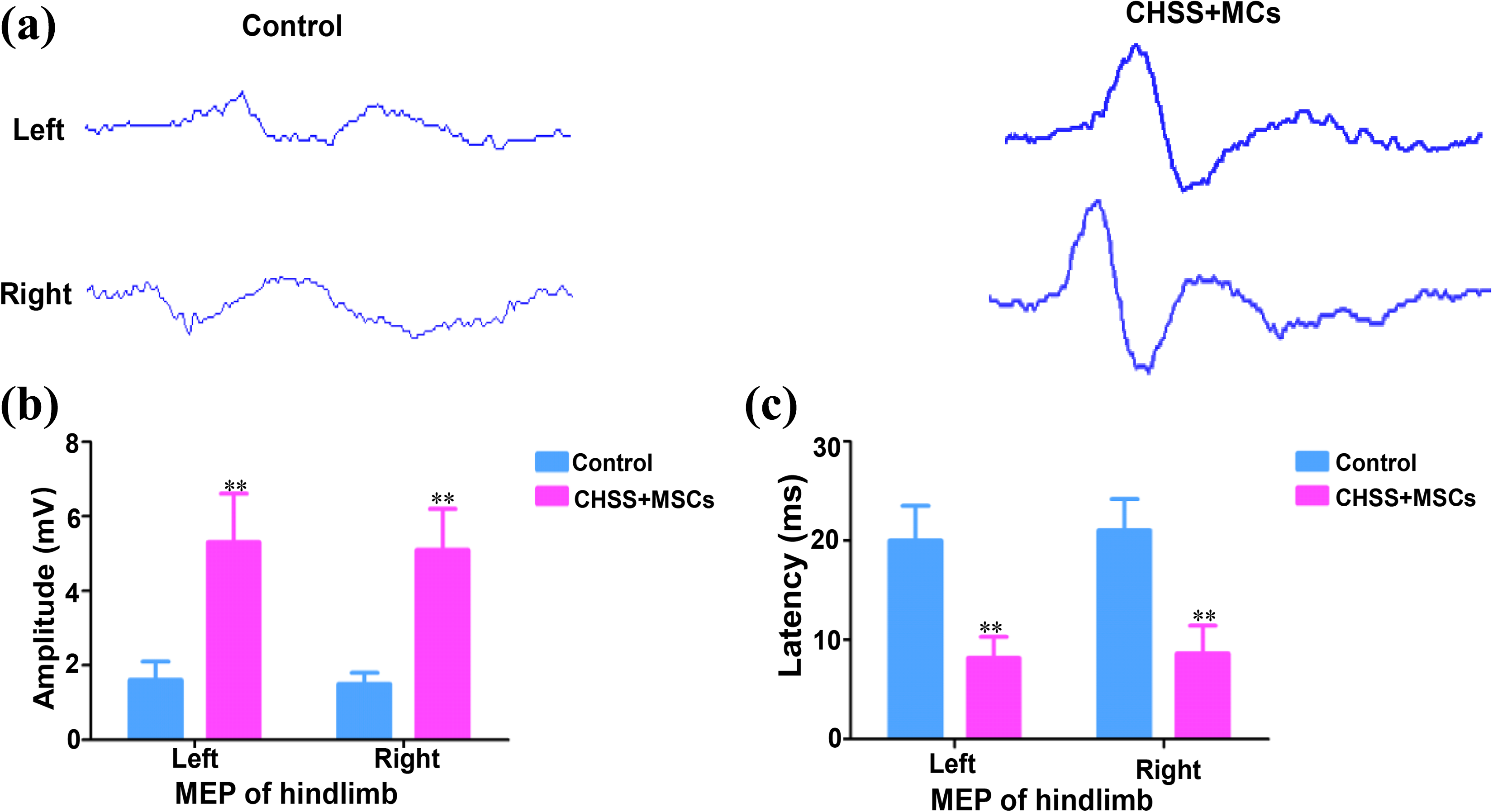
Electrophysiological results for all groups. (a) MEP traces of the left and right hindlimbs of canines at 6 months after surgery. (b, c) Amplitude (b) and latency (c) of the MEPs in the left and right hindlimbs of canines at 6 months after surgery. * P < 0.05, ** P < 0.01 vs. control group. CHSS: collagen/heparin sulfate scaffold; MSCs: mesenchymal stem cells; MEP: motor evoked potential; SCI: spinal cord injury. n = number of animals under each condition (n is expressed as dots in the bars). In (a), (b) and (c), n = 6. Data are shown as the mean + sd; SD: standard deviation.
Nerve fiber regeneration in canines
At 6 months after surgery, T2WI in the CHSS + MSCs group showed continuity between the rostral and caudal stumps of the transected spinal cord (Figure 5(b1), (b2)). However, broken ends of the spinal cord in the control group were far apart (Figure 5(a1), (a2)). Compared with the control group, the DTI images in the CHSS + MSCs group showed more nerve fibers at the sites of SCI (Figure 5(a3), (a4), (b3), and (b4)). Furthermore, transplanting CHSS co-cultured with MSCs resulted in significant increasement in FA value both in 150 µm rostral and caudal away from epicenter lesion (Figure 5(c)).

Magnetic resonance imaging (MRI) and diffusion tensor imaging (DTI) in canines at 6 months after surgery. ((a1–a4), (b1–a4)) T2WI images and DTI in the control group (a1, a2, a3, a4), CHSS + MSCs group (b1, b2, b3, b4). (a2, a4, b2, b4) Amplified images of the yellow boxes in (a1, a3, b1, b3), respectively. (c) Apparent fractional anisotropy (FA) value was measured. All red arrows indicate segments of SCI at T10. * P < 0.05, ** P < 0.01 vs Control group. n = number of animals under each condition (n is expressed as dots in the bars). In a1–a4, b1–b4 and C, n = 6. Data are shown as the mean + sd; SD: standard deviation.
Improvement of bladder in canines
Table 1 showed the results in urodynamic parameters. Compared with the control group, a significant increase in maximum cystometric capacity (MCC), bladder compliance (BC), functional profile length (FPL), and control area (CA) and a striking decrease in detrusor pressure (Pdet), urethral pressure (Pura) and maximum urethral closure pressure (MUCP) were found in the CHSS + MSCs group.
Comparing the urodynamic parameters in all groups at 6 months.

BC: bladder compliance; MCC: maximum cystometric capacity; Pdet: detrusor pressure at max; Pura: urethral pressure; MUCP: Maximum urethral closure pressure; CA: Control area; FPL: functional profile length;*P < 0.05,**P < 0.01 vs Control group.
At 6 months postoperatively, the beagles in control group showed a low BC, MCC was decreased to less than 52 ml and the profile increased rapidly during the bladder-filling phase (Figure 6(a)). What’s more, the detrusor pressure (Pdet), urethral pressure (Pura) increased significantly in the control group (Figure 6(a)). However, the BC and MCC in the CHSS + MSCs group increased significantly but the Pdet, intra-vesicle pressure (Pves) and Pura showed a downward trend (Figure 6(b)).

Representative image of urodynamics for all groups at 6 months postoperatively. (a) In control group, MCC decreased significantly while Pdet and Pves increased remarkably at 6 months postoperatively compared with the CHSS +MSCs group. (b) In the CHSS +MSCs group, MCC augmented significantly while Pdet and Pves declined remarkably at 6 months postoperatively compared with the control group. UPP: urethral pressure profile; UFS: urethral function study; Pdet: detrusor pressure; Pves: vesicle pressure; MCC: maximum cystometric capacity. n = number of animals under each condition (n is expressed as dots in the bars). In (a)–(b), n = 6. Data are shown as the mean + sd; SD: standard deviation.
Implantation of the MSCs-laden CHSS may reduce inflammatory cytokines
Compared with the control group, the levels of IL-1β and TNF-α in the CHSS adsorbed with MSCs group decreased greatly at 7, and 14 days post-SCI (Figure 7(a) and (b)). The levels of IL-10 and TGF-β1 in the CHSS combined with MSCs group were significantly higher than those in the control group at 7 and 14 days after SCI (Figure 7(c) and (d)). At 28 days after SCI, the cytokine levels were not significantly different between the control group and CHSS combined with MSCs group, and a decreasing trend of TNF-α, IL-10, IL-1β, and TGF-β1 were detected (Figure 7(a)–(d)).
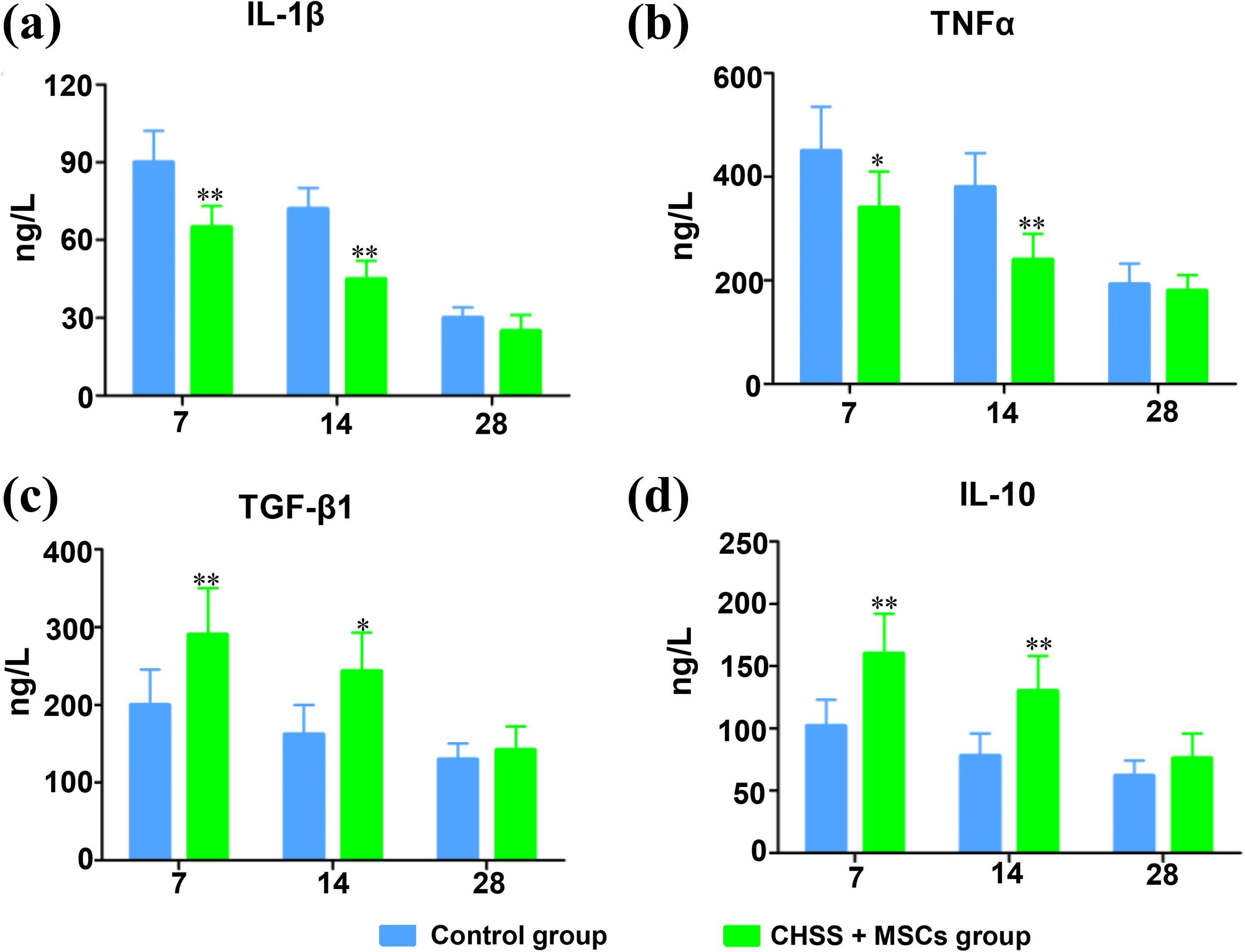
ELISA results of (a) IL-1β, (b) TNF-α, (c) TGF-β1, and (d) IL-10 in cerebrospinal fluid. * P < 0.05, ** P < 0.01 vs control group. Data are shown as the mean + sd; SD: standard deviation.
These results indicated that the implantation of CHSS combined with MSCs reduced inflammatory cytokine levels, that CHSS did not increase the inflammatory response.
Discussion
Recently, although many organizations have observed inspiring results in researches, there are few effective strategies to induce neuronal regeneration after SCI. 28,29 Different methods have been developed to promote the recovery of SCI, such as stem cell technology, immunomodulation and biomaterial-based treatments. 30 In the research, we designed a functionalized scaffold and investigated the effects of CHSSs combined with MSCs on nerve regeneration after SCI. When the CHSSs adsorbed with MSCs were transplanted into the injured site, the composite scaffold provided an ideal microstructure for MSCs proliferation and migration.
In our study, after the transplantation of CHSS combined with MSCs, the canines with SCI showed improvement of neurological function. The Olby score analyses also showed the recovery of some standing and ambulating abilities in the CHSS+MSCs group. As expected, compared with control group, the CHSS + MSCs group showed better recovery in the locomotor function, and MEP, which indicated that the implantation of CHSS loaded with MSCs could further improve the pathophysiological process and provide an effective molecular delivery platform for neurogenesis after SCI. Moreover, the MRI and DTI results indicated that the implantation promoted tissue repair and axonal regeneration.
The improvements in motor and sensory function are considered the primary results in assessing the therapeutic roles of SCI. However, urination is an essential outcome in the assessment of neurological recovery. There is no effective approach for urination dysfunction, which respond poorly to conventional treatment. We observed that compared with control group, the urodynamic parameters in the CHSS loaded with MSCs group were significantly improved. Similarly, Dai et al. reported mild improvement in residual urine volume after MSCs transplantation. 31 The recovery of urinary function is a satisfactory result because it is an important indicator for assessing therapeutic effect after SCI.
After SCI, the extracellular matrix and local microenvironment in spinal cord was destroyed, so the ideal biological scaffold should provide mechanical support for the axonal regeneration and regulate the local microenvironment to facilitate its regeneration in vivo. Collagen and heparin sulfate have excellent mechanical properties and without cytotoxicity. 32 Moreover, CHSSs showed favorable effects on MSCs adhesion, growth and proliferation. In the study, the CHSSs possess a porous spongy structure, which is very suitable for cell adhesion and proliferation. 17 Similarly, many literatures have shown that collagen could guide axonal growth along its orderly fibers, rebuild the injured defect, and serve on a carrier for cells. 33 –35
The aim of tissue engineering is to restore damaged tissue by combining biological scaffolds and stem cell. 36 Human MSCs in the host spinal cord tissue were not observed at either 4 or 24 weeks post-implantation, illustrating that the human MSCs died and vanished within 4 weeks post-implantation. 37 Despite the limited survival time, MSCs implantation improved the microenvironment in the injured spinal cord, such as alterations in the levels of pro-inflammatory cytokines, gene expression, and trophic factors. 38,39 Moreover, compared with control group, levels of IL-1β and TNF-α remained suppressed after 7 and 14 days in the CHSS loaded with MSCs group, while levels of IL-10 and TGF-β1 significantly increased, indicating that the transplantation can up-regulate the expression of anti-inflammatory factors and down-regulate the expression of pro-inflammatory factors. The recovery of neurological function in the CHSS + MSCs group could be interpreted as those MSCs provide a neurotrophic environment for axonal regeneration, regulate inflammation response, and differentiate into neurons in a specific microenvironment in the spinal cord. These results again demonstrated that suitable support and guidance for nerve regeneration should be important in facilitating regeneration.
Our study has some limitations. First, this animal model created complete spinal cord transection is different from normal SCI. However, the complete spinal cord transection is the most serious injuries in vertebral fracture. It may be possible to impact the treatment of SCI for using this animal model. Second, further studies are required to discuss the mechanisms of nerve regeneration. Finally, the histopathology of nerve regeneration is not fully elucidated in this study. Additional histopathological evaluation will be helpful to increase the persuasiveness of experimental results.
Conclusion
In this study, we report that the CHSSs combined with MSCs can promote axonal regeneration, electrophysiological response, and locomotion recovery in canines with acute complete spinal cord transection. Taken together, the transplantation of CHSS loaded with MSCs has shown therapeutic effects for larger animals with severe SCI, and the combinatorial therapy may have great prospects for clinical application.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
