Abstract
Objectives:
The patient of severe psoriatic arthritis (PsA) is mainly treated with oral methotrexate, ciclosporin, and anti-tumor necrosis factor-alpha inhibitors (TNFi). Recently, anti-interleukin-17A inhibitors (IL-17Ai) have been used in the treatment of PsA. This study aimed to evaluate the efficacy and safety of IL-17Ai in Japanese patients with PsA compared with those of TNFi.
Methods:
This was a longitudinal and retrospective study. The study population included 31 Japanese patients with PsA. All enrolled patients fulfilled the Classification Criteria for Psoriatic Arthritis. All patients were treated with TNFi or IL-17Ai. The assessed clinical manifestations were C-reactive protein (CRP)-based Disease Activity Score in 28 Joints (DAS28-CRP), disease activity in psoriatic arthritis (DAPSA), 20% achievement of American College of Rheumatology core set, swollen joint count (SJC), tender joint count (TJC), and visual analog scale (VAS). Functional ability of patients with PsA was analyzed using the modified health assessment questionnaire (mHAQ) score. We evaluated the parameters at baseline and weeks 12, 24, and 52.
Results:
The change in SJC, TJC, VAS, mHAQ, and DAPSA had no significant difference at weeks 12, 24, and 52. The improvements of CRP and DAS28-CRP were significantly higher in TNFi group only at week 12. The biologics retention rate was significantly higher in TNFi group by the log-rank test. No critical adverse events occurred.
Conclusions:
Our study presented that IL-17Ai had treatment effects comparable to TNFi. IL-17Ai might have the potential to become an alternative to the previous drug, but more large-scale studies are expected.
Keywords
Background
Psoriatic arthritis (PsA) is an inflammatory disease with destructive arthritis, enthesitis, sacroiliac arthritis, and ankylosing spondylitis, often resulting in deterioration in patient’s activity. 1 Moreover, patients with high disease activity have high mortality rate due to cardiovascular disease. 2 To prevent cardiovascular complications, it is necessary to provide appropriate treatment as soon as possible from disease onset. Oral methotrexate (MTX), salazosulfapyridine, ciclosporin, and anti-tumor necrosis factor-alpha (TNF-α) inhibitors (TNFi) are used in the treatment of PsA. TNFi dramatically improves the activity of daily life and mortality in patients with PsA. 3 –5 However, TNFi has the risk of adverse events, including interstitial pneumonia and severe pulmonary infection, subsequently increasing mortality rate. 6
Recently, anti-interleukin (IL)-17A inhibitors (IL-17Ai) are available as new treatment reagents for PsA, such as secukinumab, 7 ixekizumab, 8 and brodalumab. 9 Generally, IL-17Ai are significantly useful for plaque psoriasis compared to TNFi. 8,10 However, the equivalency of the effect of IL-17Ai and TNFi in the treatment of arthritis is controversial. 8,11 –14 In contrast, some studies reported that fatal risk and serious adverse events in IL-17Ai treatment is considerably lower than that in TNFi treatment. 15 Therefore, IL-17Ai might provide safer treatments.
The efficacy and safety of IL-17Ai in patients with PsA were demonstrated in several large-scale studies that recruited biologic-naïve patients and patients with biologics-inadequate response. The significant treatment effects of IL-17Ai were demonstrated in the European and North American populations. In contrast, there were only a few studies about the clinical effect of IL-17Ai for the treatment of PsA in Japanese patients. 16,17 In these studies, the evaluation of IL-17Ai treatment effect was mainly performed in patients with plaque psoriasis, and the ratio of participants with PsA was small. Therefore, the treatment effect and safety of IL-17Ai were still undetermined in Japanese patients with PsA, including biological-naïve patients and biological non-responders. The comparison of the treatment effects of IL-17Ai and TNFi were not reported in Japanese patients with PsA. Furthermore, the maintaining remission of patients treated with TNFi followed by IL-17Ai treatment remained to be assessed in Japanese patients with PsA.
This study aimed to evaluate the efficacy and safety of IL-17Ai in Japanese patients with PsA compared with TNFi, including patients switching biological drugs. We also assessed the remission status without reflaring the disease activity by IL-17Ai after TNFi treatment.
Methods
Patients
This was a longitudinal and retrospective study. The protocol for this retrospective study was approved by the Institutional Review Board at of the author’s institutions. All patients provided written informed consent. The study population included 31 Japanese patients with PsA. The enrolled patients fulfilled the Classification Criteria for Psoriatic Arthritis 18 and were treated at author’s hospital from 2015 to 2019. All patients had one of the following characteristics: distal interphalangeal joint involvement, polyarticular arthritis, oligoarthritis, and predominant spinal and sacroiliac joint involvement. Patients with spondyloarthlitis other than PsA were excluded. All patients were treated with TNFi or IL-17Ai. In the course of treatment, physicians decreased the dose of MTX from baseline in the patients with low disease activity and remission.
Patient and public involvement
All patients and public were not involved in the design, conduct, reporting, dissemination plans of the research.
Outcome measurement
The assessed clinical manifestations were assessed by CRP-based Disease Activity Score in 28 Joints (DAS28-CRP), 19 disease activity in psoriatic arthritis (DAPSA), 20 20% achievement of American College of Rheumatology core set (ACR20), 21 swollen joint count (SJC, 0–68), tender joint count (TJC, 0–66), and visual analog scale (VAS). Functional ability of patients with PsA was analyzed using the modified health assessment questionnaire (mHAQ) score. 22 We evaluated the parameters at baseline and weeks 12, 24, and 52.
Safety was evaluated based on adverse events. Adverse events were defined, with reference to previous literature, as infections, malignancies, Pneumocystis pneumonia, cerebrocardiovascular events, cytopenia, liver function test changes, enzyme level elevations, injection site reactions, depression, and Crohn’s disease/ulcerative colitis. 8
Statistical methods
The Mann–Whitney’s U test was used to compare baseline characteristics and change in each clinical parameter from baseline to weeks 12, 24, and 52 after TNFi and IL-17Ai treatment. The log-rank test was performed to compare the difference of retention rate between two treatment groups. Statistical tests were two-sided, with the statistical significance level set at 0.05. Dropped out patients were excluded of the statistics. The results were shown as the mean ± standard deviation. All analyses were performed using JMP version 14 (SAS Institute, Cary, NC, USA).
Results
Patient characteristics
The baseline characteristics of patients are shown in Table 1. Thirteen patients were enrolled in the TNFi treatment group, and 18 were enrolled in the IL-17Ai treatment group. The mean age of the TNFi and IL-17Ai groups were 44.3 and 55.2 years, respectively. In the TNFi group, there were 9 (69%) men and 4 (31%) women. In the IL-17Ai group, there were 9 (50%) men and 9 (50%) women. The mean disease durations were 57.7 months in the TNFi group and 85.6 months in the IL-17Ai group. There were seven patients treated with MTX before TNFi administration, and the mean dose was 6.6 mg/week. There were seven patients treated with MTX before IL-17Ai administration, and the average dose was 7.1 mg/week. In the TNFi group, five patients had been treated with biological drugs before the present TNFi treatment. The detail of the previous biological drugs was as follows: TNFi (four patients) and IL-17Ai (one patient). Sixteen patients had been treated with biological drugs before IL-17Ai treatment, and one patient had been treated with apremilast. In the IL-17Ai group, the details of the previous biological drugs was as follows: TNFi (15 patients) and ustekinumab (one patient). In the TNFi and IL-17Ai groups, the mean DAS28-CRP scores were 3.05 and 2.98, respectively, and the mean DAPSA scores were 18.7 and 18.5, respectively. The mean mHAQ scores were 0.26 and 0.47 in the TNFi and IL-17Ai groups, respectively. In the IL-17Ai group, seven patients demonstrated low disease activity or remission in DAS28-CRP at baseline. These patients performed drug changes from TNFi to IL-17Ai for the improvement of skin lesions and the safety of the IL-17Ai.
Patients’ characteristics at baseline.

Results are expressed as mean ± standard deviation (SD). **p < 0.01 by Mann–Whitney’s U test.
CASPAR: classification criteria for psoriatic arthritis; CRP: C-reactive protein; DAPSA: disease activity in psoriatic arthritis; DAS: disease activity score; ESR: erythrocyte sedimentation rate; IL-17Ai: interleukin-17A inhibitor; mHAQ: modified health assessment questionnaire; MMP: matrix metalloproteinase; SJC: swollen joint count; TJC: tender joint count; TNFi: tumor necrosis factor-alpha inhibitor.
Clinical efficacy and comparison of IL-17Ai and TNFi
Tables 2 to 4 demonstrate an overview showing a comparison of TNFi and IL-17Ai groups for each week. The changes in SJC and TJC from baseline are shown in Figure 1. At weeks 12, 24, and 52, the mean improved SJCs (standard deviation [SD]) were −1.5 (2.1), −2.3 (2.7), and −2.2 (2.9), respectively, in the TNFi group and −3.5 (9.1), −4.7 (9.8), and −1.7 (7.1), respectively, in the IL-17Ai group. At weeks 12, 24, and 52, the mean improved TJCs (SD) were −3.3 (4.1), −4.4 (3.3), and −3.0 (4.3), respectively, in the TNFi group and −5.6 (12.4), −7.1 (13.6), and −1.8 (8.0), respectively, in the IL-17Ai group. There was no significant difference in SJC and TJC between the two groups. The VAS score improvement rate is shown in Figure 1. At weeks 12, 24, and 52, mean improved VAS scores (SD) were 51.4 (42.8), 42.8 (51.1), and 35.6 (52.0), respectively, in the TNFi group and 4.66 (117.5), −44.3 (213.4), and −31.8 (233.4), respectively, in the IL-17Ai group. No significant differences in VAS improvement were noted between the two groups.
Overview of outcome parameters at week 12.

Results are expressed as mean ± standard deviation (SD). *p < 0.05 by Mann–Whitney’s U test.
ACR: American College of Rheumatology; CRP: C-reactive protein; DAPSA: disease activity in psoriatic arthritis; DAS: disease activity score; mHAQ: modified health assessment questionnaire; SJC: swollen joint count; TJC: tender joint count; VAS: visual analog scale.
Overview of outcome parameters at week 24.

Results are expressed as mean ± standard deviation (SD).
ACR: American College of Rheumatology; CRP: C-reactive protein; DAPSA: disease activity in psoriatic arthritis; DAS: disease activity score; mHAQ: modified health assessment questionnaire; SJC: swollen joint count; TJC: tender joint count; VAS: visual analog scale.
Overview of outcome parameters at week 52.

Results are expressed as mean ± standard deviation (SD).
ACR: American College of Rheumatology; CRP: C-reactive protein; DAPSA: disease activity in psoriatic arthritis; DAS: disease activity score; mHAQ: modified health assessment questionnaire; SJC: swollen joint count; TJC: tender joint count; VAS: visual analog scale.

Improvement in swollen joint count (SJC, 0–66) and tender joint count (TJC, 0–68) and improvement rate in the visual analog scale. Results are expressed as mean ± standard deviation. No significant difference was noted in the Mann–Whitney’s U test.
The CRP improvement rates from baseline are shown in Figure 2. At weeks 12, 24, and 52, the mean improved CRP scores (SD) were 52.1 (40.8), 41.0 (58.0), and 18.2 (103.5), respectively, in the TNFi group and −102.5 (405.6), 2.35 (81.5), and −17.1 (101.2), respectively, in the IL-17Ai group. There was a significant difference only in week 12 (p = 0.031), the improvement ratio of CRP was higher in TNFi group. The mHAQ score gradually improved in the IL-17Ai group (Figure 2). At weeks 12, 24, and 52, the mean improved mHAQ scores (SD) were −0.077 (0.22), −0.11 (0.29), and −0.029 (0.39), respectively, in the TNFi group and −0.0069 (0.29), −0.025 (0.28), and −0.083 (0.30), respectively, in the IL-17Ai group. No significant differences were noted in both groups.

Improvement rate in C-reactive protein (CRP) and modified health assessment questionnaire (mHAQ). Results are expressed as mean ± standard deviation. *p < 0.05 by Mann–Whitney’s U test.
The change in DAS28-CRP from baseline is presented in Figure 3. At weeks 12, 24, and 52, the mean improved DAS28-CRP scores (SD) were 41.3 (24.5), 38.1 (30.4), and 28.6 (35.0), respectively, in the TNFi group and 10.8 (44.1), 17.2 (39.7), and −0.39 (1.77), respectively, in the IL-17Ai group. The improvement ratio of DAS28-CRP was significantly higher in TNFi group at week 12 (p = 0.026). The DAPSA improvement rate from baseline is presented in Figure 3. At weeks 12, 24, and 52, the mean improved DAPSA scores (SD) were 59.1 (41.9), 57.0 (41.8), and 49.2 (46.8), respectively, in the TNFi group and 11.4 (118.0), 12.8 (78.9), and −2.29 (100.5), respectively, in the IL-17Ai group. There was no significant difference at weeks 12, 24, and 52.
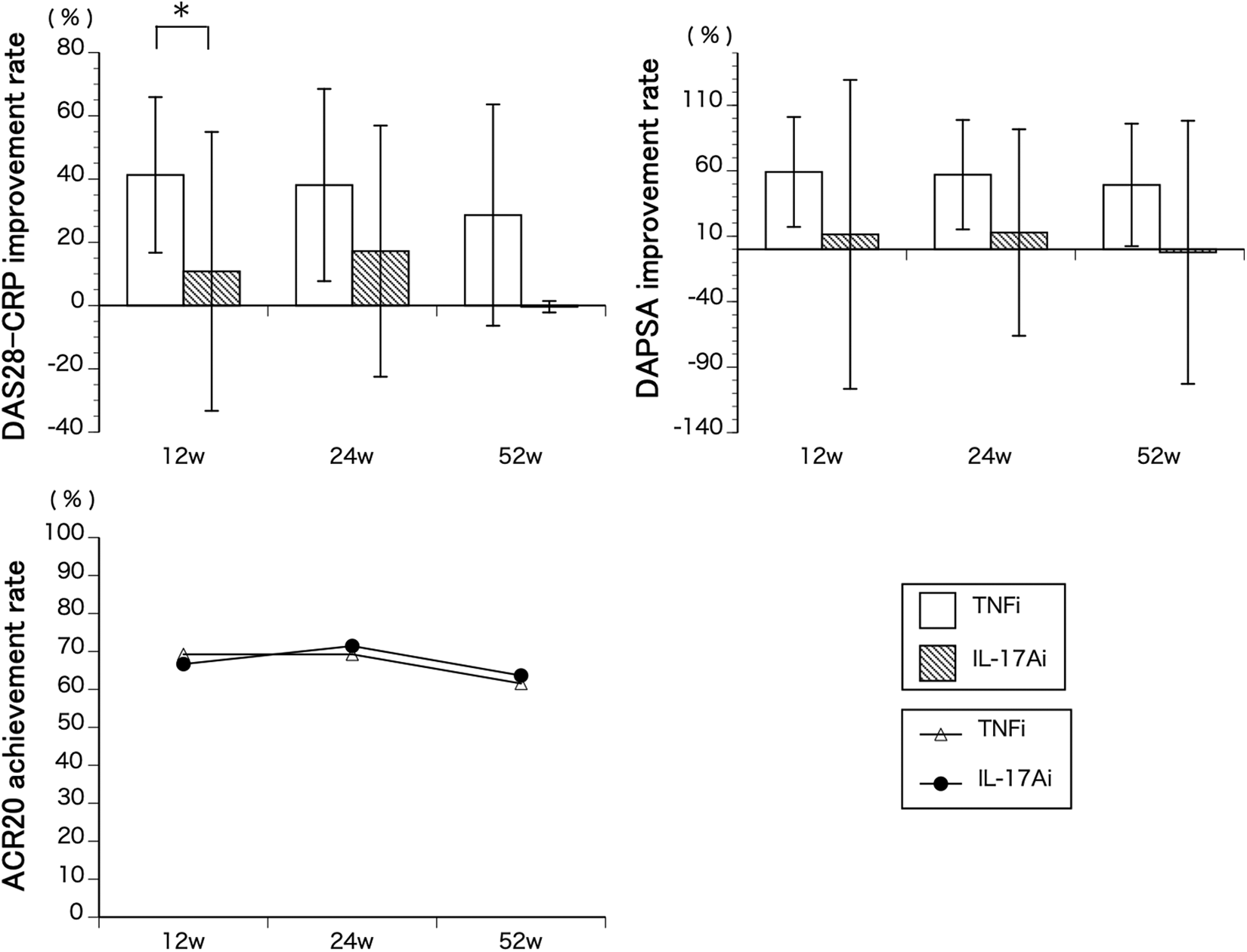
Improvement rate in Disease Activity Score (DAS)28-C-reactive protein (CRP), disease activity in psoriatic arthritis (DAPSA), and Achievement rate of American College of Rheumatology (ACR) 20. Results are expressed as mean ± standard deviation. *p < 0.05 by Mann–Whitney’s U test.
The ACR20 achievement rate is shown in Figure 3. At weeks 12, 24, and 52, the achievement rates were 69.2, 69.2, and 61.5, respectively, in the TNFi group and 66.7, 71.4, and 63.6, respectively, in the IL-17Ai group. There was no significant difference at weeks 12, 24, and 52. The biologics retention rate is shown in Figure 4. The retention rate in the TNFi group was 100% throughout the observation period. However, the retention rates in the IL-17Ai groups decreased to 61.1% over time. There was a significant difference at week 52 in the log-rank test (p = 0.013).

Drug retention rate of anti-tumor necrosis factor-alpha inhibitors and anti-interleukin (IL)-17A inhibitors. Results are expressed in Kaplan–Meier curve. P = 0.013 by log-rank test.
The DAS28-CRP score of patients in remission status changed from TNFi to IL-17Ai is shown in Figure 5. Six of seven patients could sustain the remission status but one patient dropped out at week 12 due to deterioration in disease activity.

Disease Activity Score (DAS) 28-C-reactive protein (CRP) in remission status when changed from anti-tumor necrosis factor-alpha inhibitor to anti-interleukin-17A inhibitor. Only one patient had deterioration in disease activity.
Safety
None of the patients were unable to continue the biologics due to adverse events. The reason of all patients who needed switching biologics was the inadequate effect of biologics.
Discussion
The recommendations of PsA treatment were published in the European League Against Rheumatism (EULAR) and Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) 23,24 in 2015, and the guideline from the American College of Rheumatology (ACR) was published 25 in 2019. The EULAR recommendation prioritizes TNFi over IL-17Ai for use as first biologics, but GRAPPA recommends using TNFi and IL-17Ai equally. The ACR guidelines state that even if first TNFi does not work in severe PsA, another TNFi should be used instead of IL-17Ai. Previous studies reported that IL-17A blockade therapy had good improvement effect equal to TNF-α therapy. 11,26 These studies mainly showed the drastic improvement for biologic-naïve patients with PsA compared to placebo patients or bio-switched patients compared to placebo patients. However, in daily treatment, we might treat patients with PsA having multiple backgrounds and administer the new biologics without washout interval and with or without previous biologics. There were few studies on IL-17Ai treatment resembling clinical practice especially in Japanese patients. Hence, it is controversial which biologic should be used for Japanese patients with PsA. This present retrospective study compared the IL-17A blockade therapy to TNFi blockade therapy without washout interval in Japanese patients. Moreover, this study showed IL-17Ai could maintain the remission of disease activity subsequently to biologics switching from TNF-α inhibitors for the first time.
In terms of SJC, TJC, VAS, and DAPSA, the IL-17Ai showed equal effect to TNFi and tended to result in improvement from even the early days. In the SPILIT and FUTURE studies, a similar tendency was noted. The DAPSA score improved from week 12 in the FUTURE study, and ACR20 and ACR50 improve from week 4 in the SPILIT study. 8,27 The present study showed that data from CRP and DAS28-CRP at week 12 showed a significant improvement in TNFi. This finding may suggest that TNFi may temporarily down-regulate CRP with PsA. On the other hand, CRP temporarily exacerbated early after switching in IL-17Ai by the loss of TNF suppression probably due to no biologic-free period. This may be the reason why there was a difference in DAS28-CRP in the 12th week. IL-17Ai could improve peripheral arthritis early after switching than TNFi (no significant difference) even though CRP were deteriorated. It seems paradoxical, but IL-17Ai were reported to improve the enthesitis, which is one of the main pathological conditions of PsA and more strongly caused by IL-17A than TNF- alpha, 28 accordingly it was possibility that joint symptoms were improved from early after switching. It is no wonder that IL-6 receptor inhibitors, which strongly suppress inflammation, have been shown to be ineffective against PsA and other SpA. 29 –32 The mHAQ score in the IL-17Ai group tended to have slower improvement than the TNFi group. It was different from previous studies in this respect. In SPILIT-P1 study, the mHAQ score of the ixekizumab group were ameliorated from week 12 and was equal to that in the adalimumab group. 8 Secukinumab had shown a similar result; the mHAQ score started to improve 6 weeks after administration. 33 Moreover, brodalumab had also shown similar result, and the SF-36 score was ameliorated at least 12 weeks from administration. 9 The one of possible reasons for explaining this difference was that many patients in the TNFi groups were biologic-naïve, but almost all patients in the IL-17Ai groups started after inadequate response from TNFi (TNF-IR). Even if the patients had TNF-IR, it might appear that their daily activities were better than biologic-free state. Another reason for slower improvement of mHAQ in IL-17Ai group was considered that, although many previous studies established at least 4 weeks of washout period, 8,9 the IL-17Ai group in the present study had no washout period, therefore inflammation is temporarily exacerbated due to the loss of TNF inhibition. Subsequently, IL-17Ai gradually improved inflammation, mHAQ may have improved accordingly. It is suggested that mHAQ score might not improve due to not only joint symptoms but also inflammation.
The IL-17Ai had better effect in ameliorating joint symptoms in anti-TNF-naïve patients than in TNF-IR patients. 8,33 Moreover, a similar trend was shown in biologics for rheumatoid arthritis. 34 The mean changes in the CRP and DAS28-CRP score of the IL-17Ai and TNFi groups had statistically significant difference in 12 weeks. Although there was no significant difference, the DAPSA score, also showed more improvement effect in the TNFi group than that in the IL-17Ai group. Such data might show that, when we change from TNFi to IL-17Ai, it is difficult to observe the improvement effect especially early after the change.
ACR20 achievement rate did not have significant difference even though the DAS28-CRP score was better in TNFi. This reason might be explained from the low persistency rate of IL-17Ai group. The dropped out patients of IL-17Ai groups mainly did not achieved the ACR20. Despite the total number of IL-17Ai groups were gradually decreased, the number of achiever remained, so ACR20 achievement rate of IL-17Ai groups remained high score to 52weeks. Therefore, it could not appear that IL-17Ai has equal potential to TNFi, particularly without drug holiday. The type of background patients who can continue treatment requires further discussion.
One patient of IL-17A group had been treated with ustekinumab that is the IL-12/IL-23 antibody. IL-23 regulates IL-17 expression, so ustekinumab has the possibility to affect the results of IL-17Ai. The detail of patient who treated with it was as follows; The patient was 49 years old male. Ustekinumab was administered from 6 months ago, but the therapeutic effect was insufficient, therefore ustekinumab was switched to secukinumab. The clinical scores at the time of switch were SJC 23, TJC 23, VAS 93, CRP 1.1, DAS28-CRP 7.27, DAPSA 67.7, and mHAQ 1.00. The scores 12 weeks after treatment switch were SJC 0, TJC 0, VAS 80, CRP 0.30, DAS28-CRP 4.25, DAPSA 25.3, and mHAQ 0.875. The scores 24 weeks after treatment switch were SJC 2, TJC 2, VAS 15, CRP 0.40, DAS28-CRP 2.94, DAPSA 8.4, and mHAQ 1.00. The scores 54 weeks after switch were SJC 0, TJC 0, VAS 11, CRP 0.20, DAS28-CRP 1.51, DAPSA 2.40, and mHAQ 0.00. Since the therapeutic effect of ustekinumab was insufficient, but the therapeutic effect of secukinumab was remarkable and disease activity was controlled during treatment. Therefore, the residual effect of ustekinumab was considered to be limited.
We demonstrated that IL-17Ai has the potential to maintain remission of disease activity using TNFi. To the best of our knowledge, there were no other similar reports. In the present study, only one patient could not maintain remission, and the joint symptoms worsened. Subsequently, the patient could not continue IL-17Ai treatment. Since TJC, SJC, and VAS were extremely low, DAS28-CRP of this patient was within remission, but the last CRP score was 2.1, which was relatively high (data not shown). Considering the above, we might require deeper remission to prevent flare in disease activity, especially in highly active cases.
In our study, serious adverse events, such as bacterial infection and inspiratory disorders, did not occur with IL-17Ai therapy. In contrast, in regard to TNFi therapy, some studies reported increasing critical complications. The ADEPT study showed that 5% of patients who had been treated with adalimumab in 2 years developed severe infection. 4 Moreover, Mease et al. reported that patients treated with adalimumab had 5% increase in serious adverse events than patients treated with ixekizumab in their prospective head-to-head trial. 15 Therefore, it seems reasonable for us to use IL-17Ai treatment as alternative therapy in patients with PsA for safety.
It is controversial which TNFi or IL-17A should be used for patients with highly active PsA. Previously, TNFi was the only biological drug for patients with highly active PsA. However, with the advent of IL-17Ai which is more effective for skin conditions 8,35 and almost equal 8,35 or better 15 in terms of incidence of serious adverse event, treatment alternatives of TNFi or IL-17Ai can be selected according to the patient’s age and disease activity of PsA. It may also be possible to use the way that introducing the TNFi at first for obtaining deep remission, and then maintaining it with IL-17Ai in patient with severe skin region. But further study should be needed to determine the advantage in the safety of IL-17Ai because the result of long term observation have not yet reported.
Our study has several limitations. First, we evaluated only joint symptom and did not assess dermatological lesions, such as the Psoriasis Area and Severity Index score. Second, this was a retrospective study, and the number of patients was small. For this reason, it may have been difficult to find a statistically significant difference between the two groups. Lastly, we did not statistically evaluate the structural difference of joint destruction in radiography, computed tomography, and magnetic resonance imaging.
In conclusion, the present study demonstrated that IL-17Ai treatment in patients with PsA had comparable results to TNFi treatment, with few adverse events. Moreover, almost all patients using TNFi who achieved remission could maintain remission after switching to IL-17Ai without washout interval. However, there were a few cases in which the IL-17Ai effect was insufficient and subsequently dropped out. To reveal the reason of the insufficient effect, more large-scale studies are expected.
Footnotes
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
This study was approved by the Institutional Review Board of our hospital. Informed consent was obtained from all patients in accordance with the Declaration of Helsinki before participating in this study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
