Abstract
Background:
Synovial sarcoma (SyS) is a rare malignancy that is typically located on the limbs and occurs predominantly in adolescents. A study in a large population for SyS comparing subtypes has not yet been reported.
Methods:
National Cancer Institute’s Surveillance, Epidemiology, and End Results database was queried for patients diagnosed with SyS between January 1975 and December 2016. Patients were classified demographically according to gender, age, race/ethnicity, and marital status, and they were also classified by tumors, subtypes, localization, grade, year of diagnosis, laterality, type of treatment, and follow-up results.
Results:
A total of 3228 patients were included, with a mean age of 39.3 ± 18.8 (range: 1–94), of which 1521 (47.1%) were females and 1707 (52.9%) were males. According to its subtypes, 47.2% were SyS not otherwise specified, 32.3% were spindle cell, 19.9% were biphasic, and 0.6% were epithelioid type. The overall survival period is 138.0 (95% confidence interval: 113.2–162.8) months. Survival duration was found to be significantly different between groups according to gender (log-rank test; p < 0.001), age groups (log-rank test; p < 0.001), race (log-rank test; p = 0.001), marital status (log-rank test; p < 0.001), tumor subtypes (log-rank test; p < 0.001), tumor location (log-rank test; p < 0.001), tumor laterality (log-rank test; p < 0.001), date of diagnosis (log-rank test; p = 0.025), tumor grade (log-rank test; p < 0.001), historic stage (log-rank test; p < 0.001), state of chemotherapy (log-rank test; p < 0.001), state of radiotherapy (log-rank test; p < 0.001), presence of metastasis (log-rank test; p < 0.001), and total number of malignant tumors (log-rank test; p < 0.001).
Conclusion:
Male gender, being colored individual, being over 35 years at the time of diagnosis, epithelioid type, non-head and neck region localization is associated with poor prognosis. While radiotherapy improves survival, benefit of chemotherapy is unclear.
Level of Evidence:
III retrospective analysis.
Introduction
Soft tissue sarcomas are a group of heterogeneous mesenchymal tumors, 1 each exhibiting variable clinical behavior and treatment response. In recent years, it has become increasingly common to determine specific treatment modalities for subtypes of soft tissue sarcomas. 2 However, studies on very rare subtypes are limited due to case scarcity.
Synovial sarcoma (SyS) is a rare soft tissue sarcoma 3 that occurs mostly in adolescents and young adults, accounting for about 5–10% of soft tissue sarcomas. A commonly believed etiological cause for SyS is a chromosome abnormality known as T (x; 18) (P11.2; q11.2), which results in the formation of SS18-SSX Fusion oncogenes. This chromosome anomaly can be seen in more than 90% of SyS, so it is believed to be the cause of SyS. 4
SyS is typically located at the lower limbs, especially the thigh area, and its classic treatment is resection with wide borders, chemotherapy or combinations. This tumor is generally considered a high-grade tumor with a poor prognosis. 5,6 In SyS, tumor size, tumor depth, tumor localization, and the ability to achieve a complete resection are factors affecting prognosis. 2,7
Since extensive data from clinical trials and outcome studies are often not available to guide evidence-based management, this poses a problem in the treatment of patients with rare subtypes. 2 Data on the clinical outcomes of SyS, which are also based on large sample size, are quite limited due to its low incidence. The aim of this study was to clarify the incidence and survival changes of the SyS in a large sample size using data from the SEER database. In addition, it was aimed to identify factors related to survival.
Methods
A retrospective review of SyS cases was performed using the latest version of the Surveillance, Epidemiology, and End Results (SEER) database published in November 2018. This release covers the dates between January 1975 and December 2016. The SEER database is the most comprehensive registry on cancer incidence and survival in the United States. 8 It has previously been approved and used in the study of cancer in many surgical subbranches. SEER database is a compilation of cancer data collected from nationwide registries beginning in 1973 and currently represents 28% of the US population. 8,9 The SEER database provides multi-institutional data for population-level representation of disease and statistical power in the study of diseases such as SyS; it is not possible to replicate this scale outside the database.
For this study, the International Classification of diseases for oncology, third edition (ICD-O-3) morphology 10 code system was employed, and the data were investigated using SyS not otherwise specified (NOS) (9040/3), SyS, spindle cell (9041/3), SyS, epithelioid cell (9042/3), and SyS, biphasic (9043/3) codes. Patients were classified according to gender, age, race/ethnicity, and marital status. Tumors were classified according to subtypes, localization, grade (TNM staging and grade classification were used (I-IV) 11 ), year of diagnosis, laterality, type of treatment, and follow-up results. The ages of the patients were grouped into 5-year time frames. Date of diagnosis was reported in three different groups. Group 1 from January 1975 to December 1997, group 2 from January 1998 to December 2003, and group 3 from January 2004 to December 2016. Follow-up results were examined in two groups as living and deceased. Patients were divided into two groups: those with localized tumors and those with metastatic tumors. Types of treatment were surgical treatment, chemotherapy, and radiotherapy. In addition, patients were classified according to number of tumors as having one tumor, two tumors, three tumors, or four tumors.
All patient identifiers and all data in this study were removed from the SEER database, and as a result, studies using the SEER database were exempt from the institutional review board approval.
Statistical analysis
Statistical analysis was performed by SPSS 22.0 (Chicago, Illinois, USA) computer program. In statistical analysis, categorical variables were given as numbers and percentages and continuous variables were presented with mean ± standard deviation and median (min–max value) for descriptive analyses. χ 2 tests were used for comparison of categorical variables between groups. The conformity of continuous variables to normal distribution was evaluated using visual (histogram and probability graphs) and analytical methods (Kolmogorov–Smirnov/Shapiro–Wilk tests). Kruskal–Wallis test was used for comparison of data sets which had variables that are not normally distributed. Survival analyses were performed by Kaplan–Meier methods and log-rank test. Variables with p < 0.05 as determined by log-rank testing were entered into multivariate cox regression analysis. Multivariate cox regression analysis was used to determine the independent prognostic factor for overall survival (OS) and cancer-specific survival (CSS). The hazard ratios (HRs) with corresponding 95% confidence intervals (CIs) were used to show the effect of factors on OS and CSS. A p value <0.05 was considered to be statistically significant.
Findings
A total of 3228 SyS patients were included in this study from the SEER database, of which 1521 were females (47.1%) and 1707 were males (52.9%) with an average age of diagnosis of 39.3 ± 18.8 (range: 1–94); 45.5% of patients were married and 80.8% were Caucasians (Table 1). According to its subtypes, 47.2% were NOS, 32.3% were spindle cell, 19.9% were biphasic, and 0.6% were epithelioid type. The age distributions of the patients according to SyS subtypes are presented in Figure 1. In this study, 1974 patients (61.2%) received a diagnosis after 2004; 46.1% of tumors were located at lower extremity, while 16.9% were located at left extremity (Table 1).
Baseline demographics.

SD: standard deviation; NOS: not otherwise specified.
a American Indian/Alaska native/Asian or Pacific Islander.

Age distribution of patients.
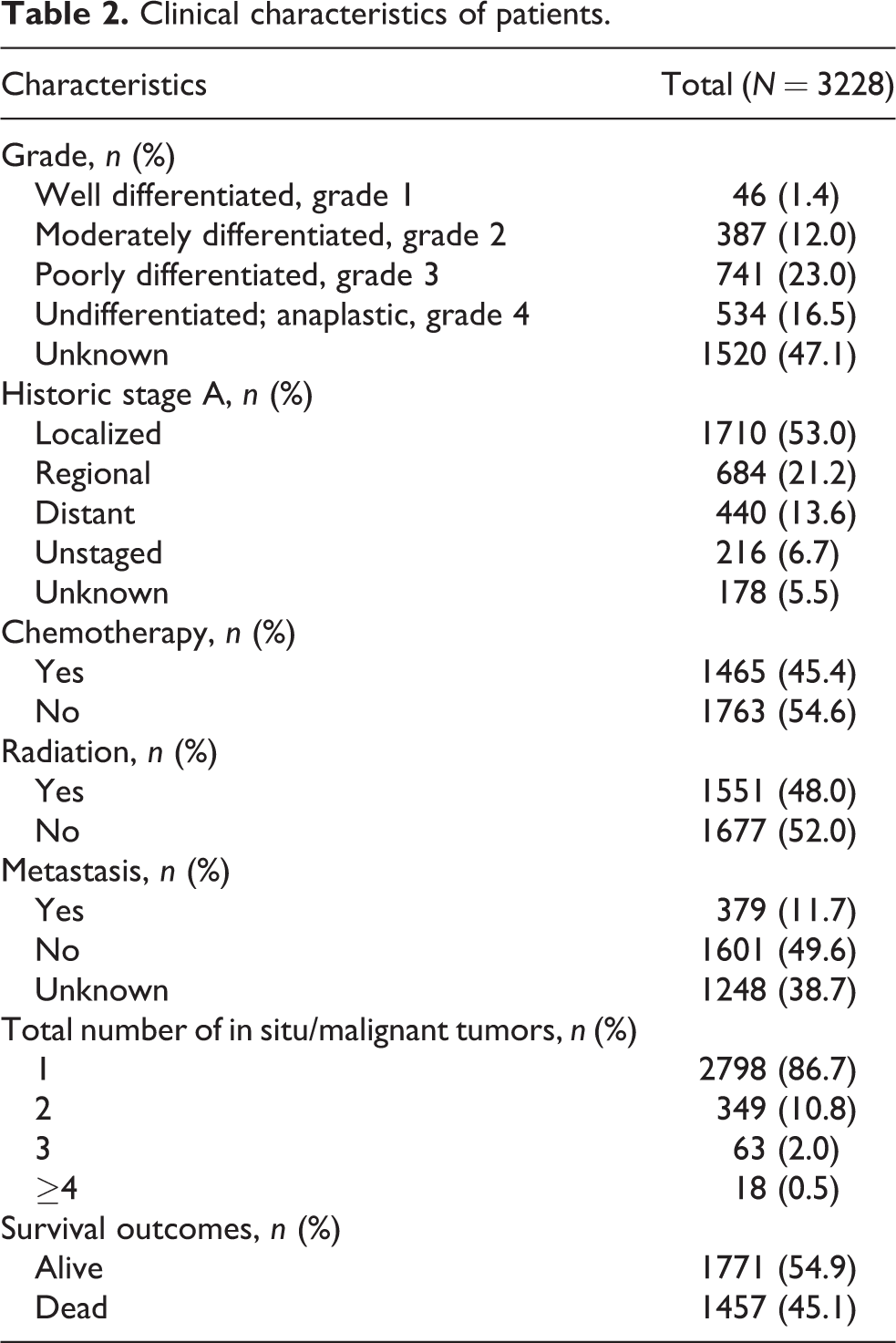
In Table 2, clinical and pathological characteristics of patients are presented. Histologically, 23.0% of patients were observed to be grade 3, 16.5% to be grade 4, and for 47.1% grade status was unknown. Most of the patients have historical stage information (95.5%) and the majority have localized tumors (53.0%). It was observed that 1465 patients received chemotherapy (45.4%) and 1551 patients received radiotherapy (48.0%). While 11.7% of patients develop metastases, in 38.7% metastasis status was unknown. The total number of tumors was 1 (86.7%) in the majority and 2 in 10.8%. A total of 1457 (45.1%) patients died (Table 2); 1294 patients died due to cancer (40.1%), 127 died due to noncancerous reasons (3.9%), while 36 patients (1.1%) had no cause of death registered. The distribution of causes of death sorted by sex is presented in Figure 2. The cause of death for 36.6% of women and 43.2% of men was cancer; this difference between the distribution of causes of death by sex was found to be significant (p < 0.001).
Clinical characteristics of patients.

Causes of death distribution of patients according to sex (p < 0.001).
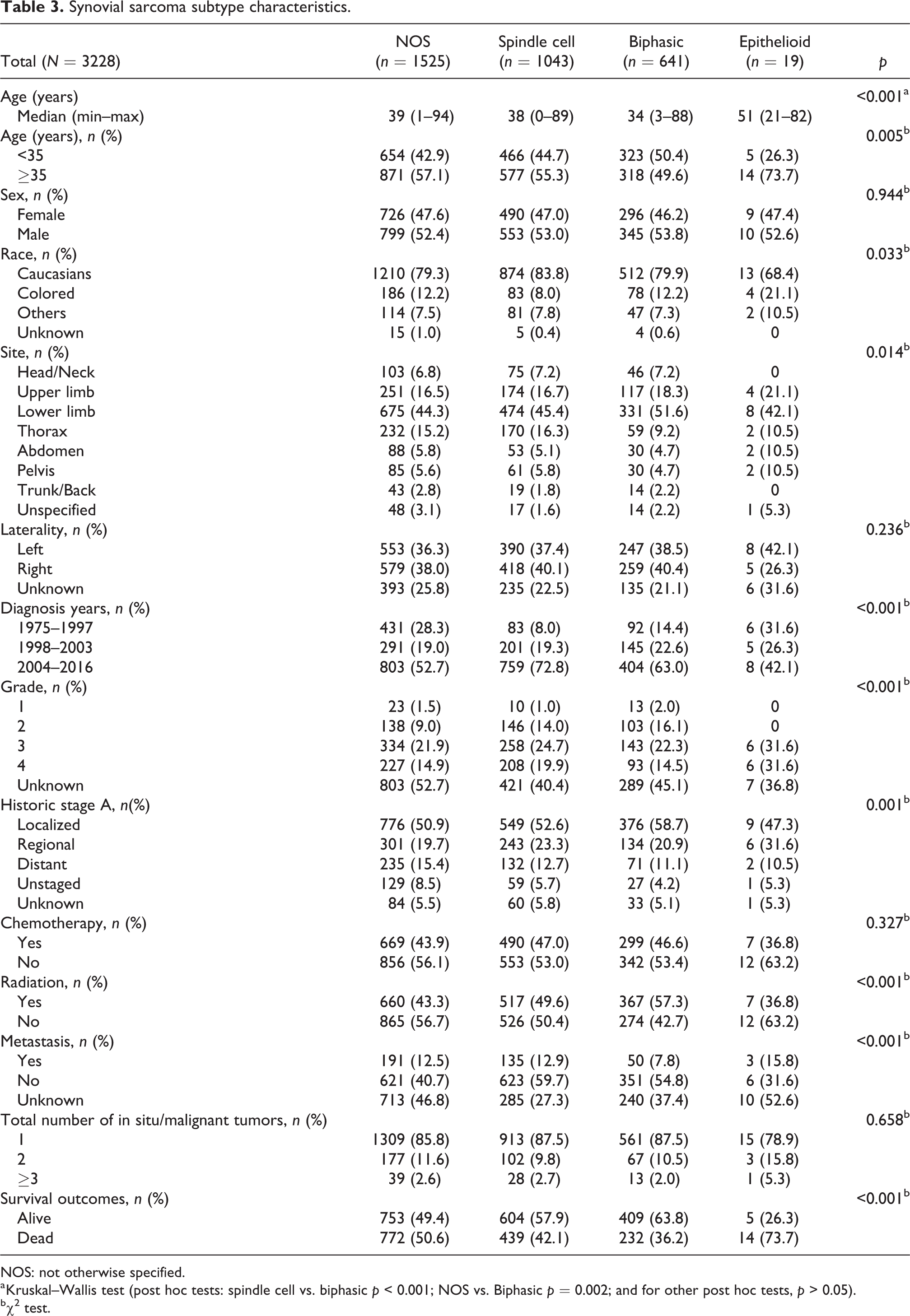
Some demographic and clinical characteristics of SyS subtypes are evaluated in Table 3. When median ages were evaluated among the types, the epithelioid type had the highest median age (median age = 51), while the biphasic type had the lowest median age (median age = 34) (p > 0.001). The Caucasian rate (68.4%) observed in the epithelioid type was less than in the other three types (79.3%, 83.8%, and 79.9%) (p = 0.033). The distribution of sex, laterality, chemotherapy status, and total tumor number among subtypes was similar (p > 0.05); 42.1% of epithelioid subtypes, 52.7% of NOS subtypes, 63.0% of biphasics, and 72.8% of spindle cells were diagnosed after 2004 (p < 0.001). In all subtypes, biphasic type had the rate of radiotherapy with 57.3% and epithelioid type had the lowest rate of radiotherapy with 36.8% (p < 0.001); 73.7% of epithelioid subtypes, 50.6% of NOS subtypes, 42.1% of spindle cells, and 36.2% of biphasics died (p < 0.001).
Synovial sarcoma subtype characteristics.
NOS: not otherwise specified.
a Kruskal–Wallis test (post hoc tests: spindle cell vs. biphasic p < 0.001; NOS vs. Biphasic p = 0.002; and for other post hoc tests, p > 0.05).
b χ 2 test.
OS rate
The OS for this study is 138.0 (95% CI: 113.2–162.8) months. One, five, ten, and twenty-year survival rates were found to be 87.3%, 59.4%, 50.8%, and 42.8%, respectively (Table 4 and Figure 3).
Overall survival rates according to factors.

CI: confidence interval; NOS: not otherwise specified; NA: not available.

Kaplan–Meier curve for overall survival.
Survival duration found to be significantly different between groups according to gender (log-rank test; p < 0.001), age groups (log-rank test; p < 0.001), race (log-rank test; p = 0.001), marital status (log-rank test; p < 0.001), tumor subtypes (log-rank test; p < 0.001), tumor location (log-rank test; p < 0.001), tumor laterality (log-rank test; p < 0.001), date of diagnosis (log-rank test; p = 0.025), tumor grade (log-rank test; p < 0.001), historic stage (log-rank test; p < 0.001), state of chemotherapy (log-rank test; p < 0.001), state of radiotherapy (log-rank test; p < 0.001), presence of metastasis (log-rank test; p < 0.001), and total number of malignant tumors (log-rank test; p < 0.001). Twenty-year survival rates are presented in Table 4. The median OS survival time of male patients was 89.0 months (95% CI: 10.9–67.5) and was observed to be shorter than that of females. The median OS survival time of patients over 35 years of age was 60.0 months (95% CI: 52.3–67.7) and was shorter than those under 35 years of age. The median OS survival time of the colored individuals was 89.0 months (95% CI: 10.9–67.5) and was observed to be shorter than that of Caucasians (median = 147.0; 95% CI: 116.7–177.3). Median OS survival times for epithelioid type tumors, NOS, spindle cell type, and biphasic type were 15.0 (95% CI: 0–39.2), 96.0 (95% CI: 69.4–122.6), 117.0 (95% CI: 74.7–159.3), and 369.0 (95% CI: 183.8–554.2) months, respectively (Figure 4). The 1-year, 5-year, and 20-year survival rates of the epithelioid type were 57.9%, 31.6%, and 26.3%, respectively. Biphasic type 1-year, 5-year, 10-year, and 20-year survival rates were 91.6%, 68.5%, 60.3%, and 50.7%, respectively. The median OS survival time of metastatic patients was 17.0 (95% CI: 14.5–19.5) months, with a 1-year survival rate of 59.5% and a 5-year survival rate of 19.6%. The median OS survival time of patients receiving chemotherapy was 65.0 (95% CI: 53.6–76.4) months, while it was 231.0 (95% CI: 187.3–274.7) months for patients who did not receive chemotherapy (Table 4).

Kaplan–Meier curves for overall survival according to subtypes.
Independent prognostic factor for OS and CSS
Effective risk factors for OS and CSS are presented in Table 5. Multivariate analyses showed that male sex was associated with a worse OS (HR = 1.27, p < 0.001) and CSS (HR = 1.25, p < 0.001). Similarly, age (≥35 years) was associated with a worse OS (HR = 1.78, p < 0.001) and CSS (HR = 1.70, p < 0.001). For both OS and CSS, the colored individuals were found to be at risk for shorter survival compared to the Caucasians (HR = 1.26, p = 0.004, HR = 1.41, p < 0.001, respectively). Being widowed was associated with a worse OS (HR = 2.32, p < 0.001) and CSS (HR = 2.61, p < 0.001) compared to never being married. Compared to biphasic type SyS, the prognosis of epithelioid type SySs in terms of both OS (HR = 2.88, p < 0.001 and CSS (HR = 2.64, p < 0.001) was the most unfavorable subtype. This was followed by NOS (OS: HR = 1.32, p<0.001; CSS: HR = 1.31, p < 0.001) and spindle cell type SySs (OS: HR = 1.23, p < 0.001; CSS: HR = 1.22, p < 0.001). Compared to head and neck tumors, thorax tumors (OS: HR = 2.04, p < 0.001; CSS: HR = 2.26, p < 0.001), abdomen tumors (OS: HR = 1.84, p < 0.001; CSS: HR = 1.92, p < 0.001), and pelvis tumors (OS: HR = 1.39, p = 0.013; CSS: HR = 1.74, p = 0.004) were found to have worse prognosis for both OS and CSS. It has been observed that the tumor side and chemotherapy status do not affect survival (p > 0.05; Table 5). While the year of diagnosis was not found to be associated with OS, it was observed that those diagnosed in CSS between 1998 and 2003 had a better prognosis than those diagnosed before 1998 (HR = 0.80, p = 0.013). Compared to grade 1 tumors, grade 3 (OS: HR = 2.06, p = 0.009; CSS: HR = 1.98, p = 0.017) and grade 4 tumors (OS: HR = 2.40, p = 0.002; CSS: HR = 2.23, p = 0.005) were found to have worse prognosis for both OS and CSS. Distant tumors for both OS and CSS (HR = 5.87, p = 0.004, HR = 6.39, p < 0.001, respectively) were found to be more at risk for shortened survival than regional tumors (HR = 2.01, p = 0.004, HR = 2.10, p < 0.001, respectively) compared to localized tumors. Patients who did not receive radiotherapy had a worse prognosis than those who did (OS: HR = 1.39, p < 0.001; CSS: HR = 1.39, p < 0.001). The presence of metastasis was also found to be a similarly poor prognostic factor (OS: HR = 1.50, p = 0.020; CSS: HR = 1.50; p = 0.020). While the number of malignant tumors was not found to be associated with OS, those with two and more malignant tumors were found to have a worse prognosis in terms of CSS (HR = 1.18, p = 0.042).
Multivariate cox regression analysis for OS and CSS for patients identified in the SEER Program database.

OS: overall survival; CSS: cancer-specific survival; SEER: Surveillance, Epidemiology, and End Results; NOS: not otherwise specified; CI: confidence interval.
In all patients, gender, age, race, marital status, SyS subtype, location, grade, stage, metastasis, and radiotherapy were identified as independent predictors of OS. Results of multivariate analysis of parameters affecting CSS suggest that gender, age, race, marital status, subtypes of SyS, location, year of diagnosis, grade, stage, metastasis, malignant tumor count, and radiation therapy were independent prognostic factors for CSS.
Discussion
Soft tissue sarcomas are a collection of rare malignancies. It involves several challenges in our understanding and risk classification of this heterogeneous group of diseases. 12 This study included the largest series on SySs, with a total of 3228 patients included. Many meaningful results were found in this study. In just over half of the patients, localized disease was present at the time of diagnosis. Male sex and being colored individual were associated with poor prognosis. It was also observed that those with a widowed marital status had higher mortality. When examined in terms of age groups, patients over 35 were found to have a less favorable prognosis. It was found that prognosis of subtypes are different from each other and epithelioid type is associated with worst prognosis among them. It was also found that tumors with head-and-neck placement were associated with better prognosis. Data about surgical treatments were missing, and it remains unclear whether radiotherapy had a positive effect on survival and benefit of chemotherapy.
The study found that the majority of patients were admitted with localized tumors (53.0%). These data show that a large percentage of patients as high as 47% were metastatic at the time of diagnosis. It was also confirmed that metastatic spread was associated with poor prognosis. Like many STS, the regional or remote spread of SyS is known to be associated with poor prognosis. 5,13
It has previously been reported that male sex and being colored individual are associated with poor prognosis in SyS. 2,13,14 Same findings were also confirmed in the current study.
As a result of the multivariate analyzes conducted in this study, we found that those with a widowed marital status were found to have higher mortality rates. Why do widows tend to have higher mortality regardless of age? Lack of care providers, depression, and fear of death brought about by loneliness might be factors that decrease patients’ compliance during the treatment process. 15
Age is known to be one of the most important prognostic factors in SyS. It was reported that cases aged 1–19 had the best prognosis, while those over 70 had high mortality rates. 16 Sultan et al. compared SySs in children and adults and showed that SyS in adults had a worse prognosis. 13 In this study, the patients were divided into two groups: those of ≥35 years and those of <35 years. Patients ≥35 years old were found to be associated with a worse OS.
STSs are a heterogeneous group of mesenchymal tumors and exhibit variable clinical behavior and treatment response. 1 Therefore, specific treatment modalities for subtypes are becoming common practice. 2,17 SyS is one of these subtypes. There are also three subtypes of SyS. Biphasic type is known to have the best prognosis. 14 In this current study, the subtypes of SyS were compared with each other thanks to the high number of patients. And the prognosis of epithelioid-type SySs was observed to be the most unfavorable. This was followed by spindle cell SyS.
SyS is often placed in the extremities but can also be seen in different body parts. The second most frequent location is reported as head and neck area. 6,18 It is known that SyS located in other regions are associated with a worse prognosis than SyS located in extremities. 13 In the current study, compared with head and neck tumors, thorax, abdomen, and pelvis tumors were found to have a worse prognosis.
The traditional treatment of SyS is reported as wide border resection, radiotherapy, and adjuvant chemotherapy. 2,5,7,17 The effect of chemotherapy on pediatric patients is well-defined and is known to have a high response rate. 19 In adults, however, the data are insufficient and its effect is controversial, 20,21 and it is generally defined as low chemosensitive. 12,22
Al-Hussaini et al. found an even higher rate of relapse in patients with localized SyS who underwent chemotherapy than in patients who did not. 20 There are also studies reporting the adverse effects of taking chemotherapy and radiotherapy in turn. 23 In the current study, the survival time of those who received chemotherapy was significantly shorter than those who did not. This can be explained by the fact that patients receiving chemotherapy are usually patients with concomitant metastases and patients with poor prognosis, that is, selection bias on chemotherapy. In addition, when independent influencing factors on survival rates were examined by multivariate cox regression analysis, it was understood that chemotherapy status was not an independent risk factor on survival. The patients’ chemotherapy status was interpreted as a confounding factor on survival. Naing et al. found that radiotherapy had a strong association with increased OS and DSS. 2 Current study also found that patients who did not receive radiotherapy showed a worse prognosis than those who did.
It is important to report that some limitations of this study exist. Firstly, this study was a retrospective analysis and although it involves a period of many years, it is difficult to obtain important information from retrospective studies and limited information is attainable. As this study was conducted on a database, some patients could not be included in statistical analysis due to missing data. Some demographic data and data about disease were not mentioned. No information was provided about surgical treatments and surgical borders. There were no data on the details of preoperative and postoperative treatments.
Despite these limitations, this study contains relatively important information, especially compared to studies involving single center outcomes, as this study is involving data from many centers with very large patient numbers. It has also provided data on chemotherapy and radiotherapy. Given the incidence rates of SyS, it is unlikely that there will be randomized controlled studies based on histology, and information obtained studies like the current one is important to shed light on treatment decisions and guide further studies.
Conclusion
Just over half of SyS patients have localized disease at the time of diagnosis. We conclude that male gender, being colored individual, being over 35 years at the time of diagnosis, epithelioid subtype, non-head and neck localization is associated with poor prognosis. While radiotherapy improves survival, benefit of chemotherapy is unclear.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
