Abstract
Purpose:
Evidence on the incidence, prevalence, and outcomes of bone metastases among patients with systemic malignancy is limited. This study aimed to evaluate it using the Surveillance, Epidemiology, and End Results (SEER) database.
Methods:
We collected patients diagnosed with solid malignant tumors deriving outside of the bone, hematologic malignancies, Kaposi sarcoma, lymphoma, and myeloma from the SEER database (from 2010 to 2013). The incidence, prevalence, and outcomes of these systemic malignancies with bone metastases were then analyzed.
Results:
A total of 67,605 patients with bone metastases at cancer diagnosis were included. The highest rate of bone metastases was observed in patients with small-cell lung cancer at the time of alternative primary site cancer diagnosis. Among 226,816 cases with metastatic disease, cases with breast cancer (65.58%), and prostate cancer (89.60%) had a high incidence proportion (>10%) of identified bone metastases. Patients with additional bone metastases resulting from prostate cancer, breast cancer, and testis cancer presented the best survival time.
Conclusions:
Incidence and prognosis differ considerably among bone metastases with different primary malignancy sites. These results may encourage appropriate application of bone imaging.
Introduction
Malignant tumors have become a major public health problem worldwide. Cancer accounted for nearly one-fourth of the deaths in the United States, the second largest cause of death after heart disease. 1 Additionally, as the leading cause of death, cancer seriously affects patient quality of life globally. Metastasis is a critically insidious biological feature of malignant tumors. Most patients with cancer do not die from primary cancer but from secondary metastatic disease.
The bone is the most common site of such secondary metastases, secondary to the lung and liver. 2 Bone metastasis is a malignant tumor of the extraosseous organ or tissue. 3 These metastatic cells are transferred by the lymphatic blood system to the bone, which then continues to grow and form a tumor. According to the American Cancer Society, about 400,000 new cases of malignant bone metastasis are diagnosed in the United States each year. 4 The incidence of advanced malignant tumors with bone metastasis is 30–75%, especially common in patients with advanced prostate cancer and breast cancer. 5 –7 Importantly, bone metastases often cause limb dysfunction, pathological fractures, spinal cord compression, and severe pain, seriously affecting the quality of life of patients with advanced cancer and poor prognoses. 8
Despite the influence of bone metastases on cancer patients, large-scale research studies examining the incidence, prevalence, and outcomes of patients with bone metastases remain lacking. In this study based on the Surveillance, Epidemiology, and End Results (SEER) database, we calculated estimates for the incidence and prognosis of systemic malignancies with bone metastases according to tumor type, age, gender, income, and lymph node involvement among others.
Methods
Selection of the study cohort
The SEER publishes the largest publicly available cancer data set, representing approximately 28% of the US population. 9 Worldwide, there was acquiescence for research works from the SEER database. Information related to bone metastases was available for patient registered between 2010 and 2013. Patients were excluded from the study if data relating to bone metastases, survival time (months), and presence of other distant metastases were incomplete. Finally, 1,356,371 patient records were included in the final cohort.
Statistical analysis
Patient data were stratified by both tumor and histology type. The absolute number and incidence of bone metastasis were calculated according to tumor type, age, sex, tumor stage, race, and lymph node stage. 10 Race representation among our study cohort included those patients identified as Caucasian, Hispanic, African American, Asian American, unknown race, and other race. Tumor, as well as lymph nodes, was staged according to the guidelines outlined in the American Joint Committee on Cancer tumor–node–metastasis staging system (seventh edition). 11
We used the SEER database to collect cases between 2010 and 2013. A total of 1,356,371 patients, above the age of 18 and those diagnosed with solid malignant tumor deriving outside of the bone, hematologic malignancies, myeloma, lymphoma, and Kaposi Sarcoma, were included in this study.
Results
Study population and incidence
Data from 1,401,813 patients diagnosed with a malignancy between 2010 and 2013 and 226,816 patients with distant metastasis were included in the SEER database. A total of 67,605 patients, 29.81% of patients with distant metastasis, were finally found to have bone metastases and were included in this study (Figure 1).

Incidence proportion of patients diagnosed with bone metastases. (a) Proportion within entire cohort, (b) proportion within subset with metastatic disease, and (c) median survival of patients with identified bone metastases, stratified by primary cancer site.
The proportion of patients who had bone metastases identified varied considerably according to the cancer type (Table 1 and Figure 1(a)). The three largest incidence proportions of bone metastases among this sample population were observed in patients with small-cell lung cancer, bronchioloalveolar carcinoma, and non–small-cell lung cancer (NSCLC) (adenocarcinoma), respectively (22.99%, 21.37%, and 19.16%, respectively). Contrastingly, brain cancer, penis cancer, and thyroid cancer had the lowest proportion of bone metastases (0.11%, 0.37%, and 0.69%, respectively). In ascending order, patients with renal pelvis (39.61%), breast (65.58%), as well as kidney and prostate (89.60%) cancers were observed to have the top three incidence proportion of bone metastases.
Incidence proportion and median survival of patients with identified bone metastases at diagnosis by primary site.

SCLC: small-cell lung cancer; BAC: bronchioloalveolar carcinoma; NSCLC: non–small-cell lung cancer; GYN: Gynecologic; AND: Andrology.
Survival estimates
Median survival in patients with primary bone metastases is presented in Table 1. The three longest median survival times among patients were 12 months (prostate cancer), 11 months (breast cancer), and 11 months (testis cancer). Median survival was also stratified by age, race, and sex in patients with bone metastases from malignant tumors, as presented in Table 2.
Incidence proportion of patients with identified bone metastases at diagnosis by sex, race, and age, respectively.
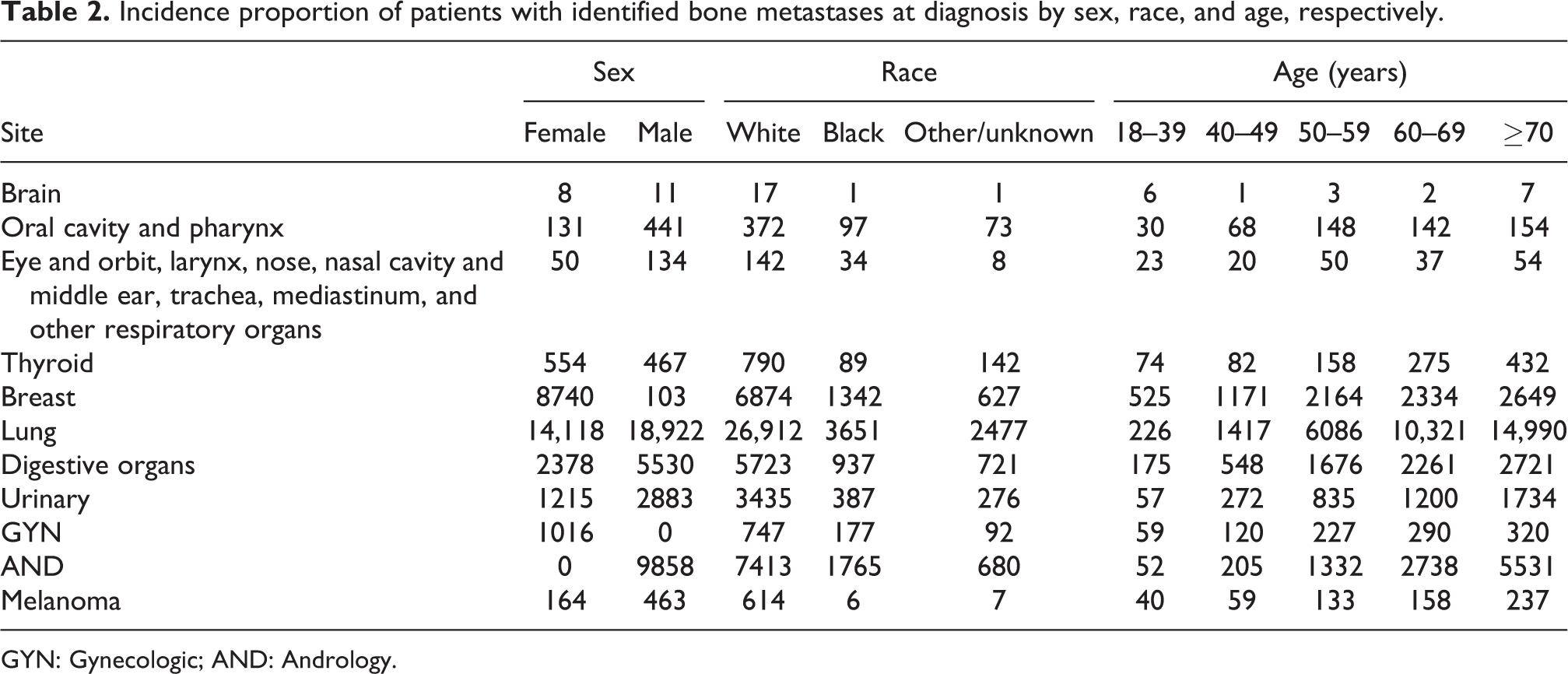
GYN: Gynecologic; AND: Andrology.
Discussion
The prognosis of malignant tumors with bone metastasis is demonstrably poor. 12,13 Devastating consequences of bone metastases include pain, hypercalcemia, pathological bone fractures, and nerve-compression syndromes. 14 With the development of surgical, radiotherapeutic, and chemotherapeutical clinical applications in recent years, patient quality of life has improved and survival times have been prolonged. 15 –17 Unfortunately, most of these patients still die due to their bone metastasis. 18 In fact, factors affecting the outcomes of patients with bone metastases are complex and include not only the treatment methods but also patient age, gender, ethnicity, and cancer type. 19
Our study demonstrated the incidence, prevalence, and outcomes of bone metastases originating in patients with primary malignancies separate from the bone. This is the first epidemiological research of bone metastases using a population-based database. Our results may also lead to the reevaluation of the importance of early bone screening among patients with newly diagnosed cancers. Such an initiative has important clinical significance for clinical trial design as well as treatment options among cancer patients.
As previously highlighted, the life quality of patients with bone metastases is poor. Early diagnosis of bone metastases may reduce mortality and treatment-related toxicity. 15 For example, with NSCLC diagnosis screening for bone metastases is not routinely performed, therefore, bone metastases are usually discovered only due to severe pain or fracture. However, our data show that 17.19% of patients with NSCLC have bone metastases. These relatively high rates of bone metastases this cancer population may warrant the use of density testing of the bone at diagnosis. Conversely, screening has become standard practice for the treatment of brain cancer patients who have a relatively low risk of bone involvement where our data suggest an incidence at diagnosis of 0.11%.
Clinical trial design requires accurate estimates of disease incidence and prognosis. If bone metastasis is a standard exclusion criterion, the data from such studies may help researchers determine the number of patients to be eliminated.
We note that there are many variables independently related to bone metastasis. Patients lacking health insurance, unmarried individuals, and those with poor social status have been shown to have higher risk of bone metastasis. Notably, Hispanic and Asian patients are more likely to suffer bone metastases than Caucasians. Further study needs to be done to explore whether environmental or biological factors are also related to bone metastasis.
There are some limitations that can be identified in our study. Firstly, we could only identify incidences of bone metastases at the primary diagnosis due to a lack of clinical records of disease recurrence. Secondly, we did not have access to information about the number or size of each bone metastases. Thirdly, screening was not employed across all types of malignancies. To better understand the pattern of bone metastases, future research is needed in larger samples with more detailed information.
Footnotes
Author contributions
Wenli Jiang and Youlutuziayi Rixiati contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
