Abstract
Background:
The purpose of this study was to compare the frequency of postoperative stiffness requiring manipulation under anesthesia (MUA) before and after switching from the intraoperative use of liposomal bupivacaine (LB).
Methods:
This was an institutional review board (IRB)-exempt retrospective cohort study (IRB#71733) on all patients who underwent primary total knee arthroplasty (TKA) by a single surgeon between April 2016 and December 2017. We compared 169 knees that received LB group to 167 knees that received a modified Ranawat cocktail (MR group). Perioperative care pathways remained consistent during the study period, as were requirements for MUA which included flexion range of motion below approximately 90 at 6–12 weeks. To compare the incidence of MUA between the groups, a population-averaged Poisson regression analysis was used.
Results:
The two groups were similar, with mean age of 63 (range 31–91) in the LB group and 64 (range 42–84) in the MR group, a preponderance of females in both groups (p = 0.866), similar preoperative knee flexion between groups (p = 0.162), and similar Patient-Reported Outcomes Measurement Information System scores. The frequency of MUA, however, was significantly lower in the MR group (LB: 7.7% [95% CI 3.7–12%] vs. MR: 2.4% [95% CI <1–4.7%] [IRR 3.2, 95% CI 1.08–9.76, p . 0.037]).
Conclusions:
In summary, this is a novel report of a potentially previously unrecognized increased incidence of MUA associated with the use of LB compared with a MR cocktail. Given no other notable changes to the perioperative care or MUA thresholds following TKA, the reasons for these findings are unclear but deserve additional investigation.
Keywords
Introduction
Postoperative stiffness following total knee arthroplasty (TKA) is a complication that continues to affect arthroplasty patients at a troublingly high rate, reported at a frequency of 3–10%. 1 This can be frustrating for both the patient and the surgeon alike and may have dramatic impacts on postoperative satisfaction and perceived patient outcomes. Postoperative stiffness can be quantified, graded based on severity, 2 and from biomechanics studies, the minimum functional range of motion (ROM) requirements are 90° for most activities of daily living. 3 This becomes significant when considering the arthroplasty demographic and postoperative goals, and primarily restoration of function and independence. Patient and surgeon factors influence the underlying pathogenesis of postoperative stiffness making identification of the causal etiology multifactorial and difficult to identify. Gu et al. 4 published a systematic review in December 2017 summarizing these influences. However, despite technically flawless surgery in well-indicated patients, the issue of stiffness persists, indicating that there are likely unrecognized variables.
Recent trends in health care, and particularly in joint replacement, have further increased the focus in the areas of patient satisfaction, expedited rehabilitation and recovery, lower length of stays (LOS), and care pathway redesigns. 5 –7 Much of this is centered on improved perioperative pain management which has seen recent evolution, including the utilization of a multimodal analgesic approach, which may introduce new variables into a patient’s postoperative course. 8 Significant effort has gone into finding ways to both decrease perioperative pain and lessen the use of narcotics, and recently a shift toward regional, and now, periarticular anesthesia has become more popular. 9 –11 In an evolving care pathway improvement process, similar to that seen in centers across the country, at our center, we have worked toward the improvement initiatives listed above with several iterations over the last decade of perioperative pain control practices. Early efforts included multimodal oral analgesics 12 and added regional anesthesia (ultrasound-guided femoral nerve catheters). 13 This progressed to adductor canal blocks to minimize the proximal muscular involvement associated with femoral nerve blocks. We then transitioned to the use of periarticular infiltration (PAI) with a liposomal bupivacaine (LB) cocktail (Exparel, Pacira Pharmaceuticals, Inc Parsippany, New Jersey, USA) including Exparel and, most recently, the use of a periarticular modified Ranawat (MR) cocktail. While notably not changing other major perioperative variables during this timeframe, the single recent change in our care pathway to stop the use of the Exparel solution and move to the MR Cocktail offered an opportunity for us to look at the potential influence of the PAI injectate as a possible factor that may influence the rate of symptomatic arthrofibrosis and manipulation under anesthesia (MUA), which we anecdotally felt had lowered following the change. The purpose of this study was to compare the frequency of postoperative stiffness requiring MUA before and after switching from the intraoperative use of LB.
We hypothesized that there would be a greater incidence of postoperative stiffness requiring MUA in patients who received LB as part of the periarticular injection. Secondarily, we sought to compare early patient-reported outcomes between the groups, with an assumption that other than the rate of MUA, the groups would otherwise be similar in terms of preoperative and postoperative pain and Patient-Reported Outcomes Measurement Information System (PROMIS) scores. Given the low incidence of MUA overall, we felt there would be no difference in 6-week outcomes between the groups.
Materials and methods
Subjects
This was an institutional review board (IRB)-exempt retrospective cohort study (IRB#71733) on all patients who underwent primary TKA by a single orthopedic surgeon between April 2016 and December 2017 at a single academic medical center. Data were obtained by querying an electronic database, performing chart reviews, and collecting outcomes from routine telephone follow-up. Patient demographics including age, sex, and body mass index (BMI) along with the American Society of Anesthesiologist’s physical status classification (ASA score) were collected (Table 1). The surgeon performed 353 primary TKA procedures in 307 patients. A minimum 90-day follow-up was required for inclusion in the study. Seventeen knees lacked a minimum 90-day follow-up and were considered lost to follow-up. Ultimately, 169 knees received LB and were compared with 167 knees that underwent primary TKA after the surgeon discontinued using LB in April of 2017 at which time the use of a MR cocktail group was implemented. All patients were evaluated, examined, indicated, and offered MUA by a single surgeon. The requirements for MUA remained constant throughout the study period and included decreased flexion ROM below approximately 90° at a 6- to 12--week follow-up visit or failure to progress with therapy with an unacceptable flexion arc.
Patient characteristics.
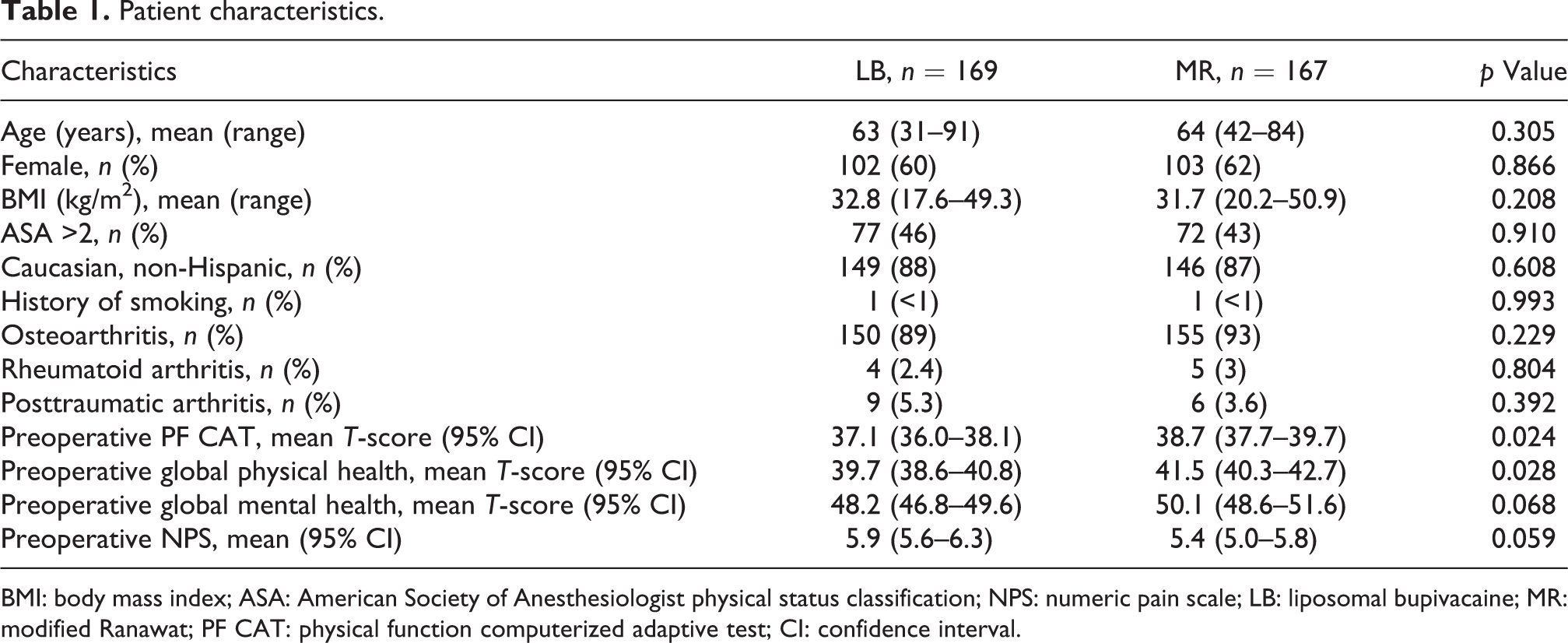
BMI: body mass index; ASA: American Society of Anesthesiologist physical status classification; NPS: numeric pain scale; LB: liposomal bupivacaine; MR: modified Ranawat; PF CAT: physical function computerized adaptive test; CI: confidence interval.
Although this was a system-wide change and care-pathway redesign, the series of patients included was intentionally gathered from only a single surgeon’s operative cohort intentionally to limit the introduction of added variability. By looking at data from the single surgeon, the variation in surgical technique, surgical implants, and the indications for MUA were minimized, allowing for a more standardized patient group. In addition, this surgeon was the only provider from this institution using LB consistently prior to the switch. The indications for manipulation remained consistent before and after the change in periarticular injectate. The surgeon had noted rates of MUA that were higher than other’s in the group, allowing for any significant change in MUA rates to be easily analyzed. The surgeon continues to offer MUA to patients when meeting the above indications and has not made efforts to avoid the procedure due to concerns of quality metrics or other external pressures given that patients do benefit from the intervention. Finally, the interest in performing this study was not related to a preconceived concern about MUA rates and the use of LB but rather the surgeon and his staff noted a significant decrease in MUA rates anecdotally in the clinical setting. After consideration of the timing of the drop in MUA rates, the team realized that the only change made in the care pathway during this period was the change in the periarticular injectate.
Care pathway and in-hospital recovery
In addition to the perioperative pain control practices mentioned above, our surgical care pathway has evolved over time and we have reported on these changes. 7,14,15 The majority of these changes were driven by improving value at our institution as well as the initiation of the bundled payment for care improvement program. Changes to our inpatient care pathway began in the fall of 2012, where a multidisciplinary team identified improved early ambulation following TKA as a target for improvement. 7 Inpatient physical therapy (PT) staffing was adjusted and resulted in an increase of patients ambulating on the day of surgery from 64% to 85%. This pathway change has remained consistent throughout the course of the present study. Therapy utilization was not different at the beginning or end of the study period creating similar groups for postoperative mobilization. ROM exercises were included in the initial therapy visits and, in the majority of patients, was initiated on the day of surgery and continued through the duration of their hospital stay. Beginning June of 2015, a comprehensive patient education program and care navigation process was initiated. 15 It is important to note that these changes were completed by November 2015 and remained constant throughout the duration of this study. Preoperative education included an emphasis on postoperative placement and discharge criteria which were the same through the duration of the study period. Additionally, the majority of patients (74%) were discharged from the hospital on the first postoperative day. No other known changes to care pathways, medications, discharge, PT use, or rehabilitation protocols occurred during the study time frame.
At our institution, the decision was made to switch from the LB cocktail to the MR cocktail during the dates described above. This decision was made by the department for several reasons. First, the cost savings potential with each injection delivered was significant, saving $300 US dollars per case (LB injection: 318 US dollars vs. MR injection: 18 US dollars, direct cost). Additionally, there has been recent literature published in support of this periarticular injectate showing good success with postoperative pain relief, narcotic use, and ROM when compared with LB. 16
Surgical technique
The surgical technique and surgical implants remained consistent before and after the implementation of the change as well. Surgery was either performed under general or spinal anesthetic. A standard medial parapatellar approach was utilized for all cases. The femoral preparation utilized intramedullary referencing with a goal of matching the patient’s native femoral valgus angle, as measured as the angle between the femoral mechanical and the anatomical axes based on a preoperative long-standing radiograph, was used to set the valgus angle for the distal cut, with an average of 4–6°. A standard posterior referencing guide with the goal of rotational alignment parallel to the epicondylar axis and perpendicular to the AP axis was used. All cuts were cooled with saline to prevent thermal damage. Femoral component sizing was based on the best fit in the anterior–posterior dimension, ensuring no medial–lateral overhang. Femoral components were a cruciate-retaining (CR) design in the majority of cases (Biomet Vanguard, Zimmer-Biomet Inc., Warsaw, Indiana, USA). In rare instances of ligamentous deficiency, or needed stability, posterior stabilized (PS) or varus–valgus constrained femoral components and their associated bearings were used. For the purpose of this analysis, these were classified as cruciate sacrificing (CS) designs.
The tibial preparation was performed with an extramedullary cutting guide referencing 6–8 mm off the more normal side of the tibial plateau with alignment goal perpendicular to the long axis of the tibia. The tibial implant consisted of a modular titanium baseplate and modular titanium cruciate keel with a grit-blasted titanium surface with surface roughness average (Ra) of 6.8. After pulsatile lavage, all tibial components were cemented with high viscosity Cobalt (DJO Surgical, Vista, California, USA) cement with premixed Gentamycin in all cases.
Selective soft tissue balancing was performed as described in prior publications from our group as needed to achieve symmetric medial and lateral flexion and extension gaps. 17,18 Careful cementation was performed with perforation of any sclerotic bone with a drill bit in multiple drill holes. The bone was then thoroughly irrigated with pulse-lavage irrigation and then dried. Cement was mixed and applied to the clean dry implant prior to any contamination in its most viscous state and then applied to the prepared bone with the use of a wide osteotome with several pressurizing passes made with the osteotome and thumb pressurization with significant focus of maximizing cement interdigitation to the prepared bone. Meticulous cleaning of excess cement was performed. The tibia was cemented first, followed by placement of the final polyethylene insert and then the femoral component was cemented using the same technique under a single batch of cement. The knee is taken to extension and held still with slight constant pressure until the cement had fully cured in the bowl. Only then was the knee moved, inspected and cleaned of any residual extruded cement. The tibial components were paired with anterior stabilized bearings and CR bearings in the majority of cases, and PS or constrained inserts when indicated for the reasons listed above, to match the femoral component.
The medications injected were as follows. The injectate used in the LB group consisted of LB (Exparel, Pacira, Parsippany, New Jersey, USA) 266 mg/20 mL admixed with bupivacaine HCl 0.5% 20 mL expanded with saline to a total volume of 120 mL. The injectate use in the MR group consisted of ropivacaine 5 mg/mL (ropivacaine weight-adjusted dosing, 270 mg or 54 mL above 80 kg body weight and 180 mg or 36 mL below 80 kg), ketorolac 30 mg/mL (1 mL), epinephrine 1 mg/mL (0.5 mL), clonidine 0.1 mg/mL (0.08 mg = 0.8 mL), and saline to expand the total volume to 100 mL. PAI of the injectate was performed similarly and remained consistent before and after changing the medications used in the periarticular cocktail. The injectate was administered before and after cementation using 60 mL syringes with a 22-gauge needle, with the goal of each injection delivering a small volume of 1–1.5 mL to the area. Injection sites and technique were engrained in our practice during the formal training by the manufacturer when our site participated in a large multicenter prospective randomized controlled trial using this drug in the years prior to this present investigation time frame, with further details available in that publication. 19
Patient-reported outcomes
Patient-reported outcomes were evaluated preoperatively and at 6 weeks postoperatively. As part of our patient’s routine care, we have incorporated instruments for collection from the National Institute of Health’s PROMIS. The PROMIS physical function computerized adaptive test (PF CAT) v2.0, the PROMIS global physical and mental health assessments, and an 11-point numeric pain scale (0–11) were evaluated in this study. The PROMIS PF CAT and global health measures (physical and mental health) are reported as a T-score. A T-score of 50 is equivalent to the mean US General Population (standard deviation (SD) 10) for the measure of interest. Outcome measures at the last follow-up were considered missing if the patient reported complications associated with other body parts at the time of last follow-up (n = 1). Regardless, the patients remained in the analysis if the outcomes of the knee replacement itself were recorded, including history of subsequent MUA. A minimal detectable difference (MDD) of half a SD was calculated preoperatively and at 6 weeks postoperatively (Table 2). 20 A two-unit change in pain on the NPS was considered the MDD for that measure. 21,22
MDD for patient-reported outcome measures.

MDD: minimal detectable differences; PF CAT: physical function computerized adaptive test.
Statistical analysis
Data are summarized using descriptive statistics. To compare the incidence of MUA between the groups, a population-averaged Poisson regression analysis was used. This was done to account for the clustering of knees within patients. Additionally, generalized estimating equations (GEE), with an unstructured correlation matrix, were used to compare dichotomous variables between the groups including sex and ASA >2. Continuous variables were analyzed using a linear GEE model as was the numeric pain scale. Extensive simulation research in biomedicine has demonstrated that analyzing ordered data as continuous variables does not inflate type 1 error. 23 –25 Missing data were imputed using the multivariate imputation by chained equations method. 26 Finally, a subanalysis was performed on bearing types between the groups and if these had an impact on the proportion of MUAs in the LB and MR groups. As a subanalysis, a population-averaged Poisson regression analysis was used to assess whether there was a difference in the frequency of bearings (CR vs. CS) used and whether bearing type was an independent risk factor for MUA. Data were analyzed using commercially available statistical software (Stata v14.2, College Station, Texas, USA) and significance was assessed at p < 0.05.
Theory
Although rare, and not well-described in the literature, it seems plausible that new combinations and concentrations of drugs used in periarticular injection for local analgesia may create unforeseen postoperative complications. LB, administered by local infiltration through the surgical incision, first demonstrated safety in animal studies 27 in 2011 and has shown efficacy in clinical studies. 28 –31 While published results have shown variable efficacy compared with plain bupivacaine or other periarticular cocktails, 19,29,32 reports of adverse events have infrequently been described in the clinical literature. 33,34 Of interest, in preclinical trials, the animal studies mentioned above did describe the findings of local granulomatous reaction, though to our knowledge, any clinical significance of this finding has not been shown.
Results
The adjusted mean age was 63 (range 31–91) in the LB group and 64 (range 42–84) in the MR group and there was a preponderance of female patients in each group (Table 1). The majority of patients (88%) were Caucasian of non-Hispanic decent, and this proportion was not different between groups (Table 1). In addition, the proportion of African Americans was 1% or less in each group. Preoperative PRO scores demonstrated statistically significant differences in both the PF CAT and the global physical health T-scores (Table 1). However, these scores were not greater than the MDD and were unlikely clinically relevant (Table 2). There was no difference in the preoperative pain levels reported (p = 0.059, Table 1). The mean follow-up was 13 months (range 2.8–29.8). There was no difference between the two groups in mean preoperative knee flexion (p = 0.162). The adjusted mean preoperative flexion for the LB group was 121° (95% CI 120°–123°) compared with 123° (95% CI 121°–125°) in the MR group. There was no difference in preoperative diagnosis between the groups (all p > 0.05, Table 1). Interestingly, the LB group had a slightly longer LOS. Patients in the LB group had an adjusted mean LOS of 1.9 days (95% CI 1.8–2.0) compared with 1.3 (95% CI 1.1–1.4) in the MR group (p < 0.001). However, there was no difference in the time to the initial inpatient PT visit following surgery (p = 0.264). The adjusted mean time to PT was 4.9 h (95% CI 4.5–5.3) in the LB group compared with 4.6 h (95% CI 4.2–5.0) in the MR group. Finally, only one patient (<1%) in the LB group reported inadequate inpatient pain control (p = 0.392) compared with none in the MR group.
Manipulations under anesthesia
The frequency of MUA immediately following primary TKA was 7.7% (13 patients; 95% CI 3.7–12%) in the LB group and 2.4% (4 patients; 95% CI <1–4.7%) in the non-LB group (incidence rate ratio (IRR) 3.2, 95% CI 1.08–9.76, p = 0.037). At 6-week follow-up, the adjusted mean flexion was 118° (95% CI 116°–119°) in those that did not require MUA and 90° (95% CI 84°–96°) in cases that did (β −27.8, 95% CI −34.0 to −21.6, p < 0.001). For those who required MUA, the adjusted mean flexion angle at 6-weeks was 92.5° (95% CI 79°–106°) in the MR group and 89° (95% CI 82°–97°) in the LB group (p = 0.675).
Patient-reported outcomes
There were no significant differences in patient-reported outcome measures at 6 weeks between groups (Table 3). Further, the amount of change from preoperative to 6 weeks postoperative visits was not different between groups. There was a statistically significant difference in the 6-week global mental health T-scores, but this failed to meet the MDD for this measure.
Patient-reported outcomes reported as the adjusted mean (95% CI).

LB: liposomal bupivacaine; MR: modified Ranawat; PF CAT: physical function computerized adaptive test; CI: confidence interval.
Subanalysis: Implants
In an effort to account for the potential influences of implant or bearing utilization between the two groups on the rates of MUA between the LB and MR cohorts, we performed a subgroup analysis on bearing types (Table 4). There was a greater proportion of CR bearings (IRR 4.09, 95% CI 2.06–8.14, p < 0.001) in the LB cohort (37 of 169, 22%) versus the MR cohort (9 of 167, 5%). However, when evaluating bearing type as a risk factor for MUA, we found no difference in the frequency of MUA (IRR 1.92, 95% CI 0.65–5.68, p = 0.237) between CR (4 of 46, 8.7%) and CS bearings (13 of 290, 4.5%). All varus–valgus constrained bearings were used in the MR cohort. There was no difference in the use of PS bearings between the LB (5 of 169, 3%) and MR (6 of 167, 4%) cohorts (p = 0.755).
Subgroup analysis: Implant bearing type.

LB: liposomal bupivacaine; MR: modified Ranawat; CR: cruciate retaining.
Discussion
Pain control following TKA is an important aspect of a successful surgery, and multiple adjuncts to pain control are available, including the use of various periarticular injections. Aside from the perioperative benefits of LB, the early outcomes associated with potential adverse events may not be completely understood. While our two groups were otherwise similar in terms of demographics, preoperative ROM, preoperative and postoperative pain and PROMIS measures, and their exposure to perioperative care pathways, we found a higher of MUA in the LB group. The frequency of both groups falls within the previously cited ranges, 1.3–23%, as reported in the discussion by Yeoh et al. 35
Although there was a slight difference in bearing utilization (CR vs. CS) between groups, we were unable to identify bearing type as a risk factor for MUA in this population. The determination to perform MUA was made by a single surgeon who personally evaluated each patient in a similar fashion within a demographically similar group, thus standardizing the postoperative evaluation of ROM and limiting variability to the extent possible.
Parvizi et al. 36 performed a basic science multicenter case–control study to address the pathophysiology of arthrofibrosis. This study utilized a standardized tissue retrieval protocol to intraoperatively collect tissue samples from patients indicated for revision surgery secondary to arthrofibrosis. After tissue retrieval, the samples were permanently fixed in paraffin, treated with antibody reagents, and studied using immunohistochemistry. Tissue from patients with arthrofibrosis showed increased levels of pro-survival factors and reactive oxygen and nitrogen species (RONS) not present in control tissue. They concluded that the RONS drive inflammatory infiltration and fibroblast proliferation contributing to arthrofibrosis. Through a comparable inflammatory cascade, granulomatous inflammatory reactions produce histologically similar findings. 37,38 Granulomatous reactions are described by James 39 as a nondegradable focus of inflammatory cells, which form as a product of active hypersensitivity. Mononuclear cells are the predominant cell type in this inflammatory reaction which produce proinflammatory cytokines. These granulomatous reactions are, however, often associated with systemic disease and neoplastic processes and are not often associated with postoperative orthopedic complications. As previously mentioned, not all causes of postoperative stiffness after TKA are completely understood. There may be a connection between the histologically similar soft tissue reactions of arthrofibrosis and focal or diffuse granulomatous reactions. The previously mentioned 2011 animal study 27 looked at the safety and tolerability of DepoFoam bupivacaine, a suspension of local anesthetic suspended in natural or synthetic analogs of common lipids and 0.9% sodium chloride. The model simulated an inguinal hernia surgical site and animal subjects were administered different concentrations of DepoFoam bupivacaine. Tissue sample histology was evaluated at days 3 and 15, and upon histological analysis, the authors found 8 of 24 rabbits had developed levels of granulomatous reactions postoperatively. While considered normal in this particular animal group, certain sensitive groups may have heightened reactions to this injected material and trigger an inflammatory cascade. What we cannot answer with our findings is whether this could clinically lead to arthrofibrosis and functional limitations. While we cannot suggest causation nor correlation of the granulomatous findings in the prior animal models to our clinical findings, and certainly without performing histologic analysis of all of these cases, we are left to wonder whether such a phenomenon could explain the differences between the two groups.
Our study focused on very early outcomes following TKA. Given that the window for MUA following TKA is considered to be within the first 3 months, and prior to maturation of the scar tissue, we feel that this is an appropriate time window to report on this for this particular outcome. 40 Further, regarding the timing of outcomes evaluated in this study, a recent review of the recovery of patient-reported outcomes following primary TKA demonstrated no difference in preoperative and 6-week postoperative outcomes as measured by the PF CAT. 41 It further reports that approximately 90% of the recovery occurs by 6 months. This is consistent with prior reports. 42 –45 As expected, our data demonstrated no difference at 6 weeks between PRO measures and suggest that outcomes associated with pain or function were not affected by either treatment option assessed in this study.
This study is subject to the inherent limitations of retrospective cohort studies including both selection and misclassification of outcomes bias. The potential for a selection bias in this cohort study was minimized by including all primary TKA procedures performed by the surgeon during the study period regardless of outcome. This allowed for a more accurate representation of the incidence of MUA in the cohorts. We do acknowledge that relying on a single surgeon’s cohort of patients risks introducing selection bias in addition to the retrospective nature of the study and chart review methodology; however, we felt that providing a standardized group of patients, as mentioned previously, was extremely important to minimize confounding data and variables in the group. In addition, because of the retrospective study design and chart review means of data gathering, much of the granularity related to patient recovery and in-hospital functional assessment is limited and it is possible that these differences may have influenced manipulation rates. Unfortunately, 17 patients (5%) lacked a minimum 90-day follow-up and were excluded. The impact of this is felt to be minimal. It is possible that we failed to classify some patients as having subsequently required MUA. However, we required a minimum 90-day follow-up visit as MUA occurring after this point is unlikely. Multiple methods were used to ensure capture of MUA as the outcome including a report from the institution’s enterprise data warehouse on all MUA cases within our system, regardless of the provider, and an extensive chart review for any reference to a subsequent MUA within our electronic medical records, care navigation notes, or obtained via routine phone-call follow-up. Despite these methods, it is possible that we are underestimating the incidence of MUA in both groups. Further, the etiology of arthrofibrosis and MUA is likely multifactorial. While we have tried to control for the variables of similar thresholds to perform MUA, similar perioperative care pathways, similar patient cohorts in terms of demographics, similar patient preoperative and postoperative pain and function PROs, similar surgical techniques and implants, and other potential confounding variables, we acknowledge that with evolving practices in modern-day health care, some unaccounted for changes or confounding variables could have influenced the rate of MUA in our study, which we have not been able to fully account for despite our best efforts to pick a time point wherein our group had made no other formal changes. Prospective studies that could evaluate the rate of an MUA as a primary endpoint while controlling for confounding variables would be a reasonable next step to help further investigate our findings. While we in no way can suggest causations or correlation, our retrospective review does provide some foundation to help direct such a future study, and we might suggest the outcome of arthrofibrosis and need for MUA be added as an outcome of interest. Lastly, although postoperative inpatient PT was evaluated and not found to be different between the two groups, we were unable to account for actual outpatient therapy visits which may have influenced the rates of arthrofibrosis and ultimately rates of MUA.
Conclusion
In summary, the use of LB was associated with a greater frequency of MUA after TKA compared with a MR cocktail in this study. Given no other identifiable changes to the care pathways or MUA thresholds following TKA, the reasons for these findings are unclear. The etiology of arthrofibrosis and the potential effects of PAI of LB on outcomes following TKA warrants further study.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Several authors report financial interests outside of the scope of this study. Two authors (CEP and CLP) receive royalties, institutional research support, and/or consult for Zimmer Biomet. One author (CLP) has stock options in CoNextions Medical and Muve Health. One author (CEP) is a paid consultant for Total Joint Orthopaedics and Acelity, Inc. One author (MBA) consults for and has equity interest in OrthoGrid Systems, Inc.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
