Abstract
Purpose:
To report the clinical, functional, and magnetic resonance imaging (MRI)-based outcomes of a novel autologous-made matrix consisting of hyaline cartilage chips combined with mixed plasma poor rich in platelets clot and plasma rich in growth factors (PRGF) for the treatment of knee full-thickness cartilage or osteochondral defects.
Methods:
Between July 2015 and January 2018, all patients with full-thickness cartilage or osteochondral defects undergoing this novel cartilage restoration surgical technique were approached for eligibility. Indications for this procedure included traumatic or atraumatic full-thickness knee cartilage defects or osteochondritis dissecans. Patients were included if they had no concomitant use of stem cells, previous ipsilateral cartilage repair procedure, or follow-up was less than 10 months. The outcomes included data on current symptoms, physical exam, patient-reported, and functional outcomes (visual analogue scale (VAS) for pain, Lysholm score, Tegner activity scale, International Knee Documentation Committee (IKDC) subjective form, Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) score, Lequesne index, and short form-12 (SF-12)) and the magnetic resonance observation of cartilage repair tissue (MOCART) score. These outcomes were compared to preoperative values, except for the MOCART score.
Results:
Fifteen patients were included in this preliminary study: mean (standard deviation (SD), range) follow-up 15.9 months (7.2, 10–32), age 26.8 years (12.1, 16–58), and body mass index 23.2 (2.1, 19.3–26.9). There were 14 men (93%) and 1 woman (7%). There was a statistically significant improvement between pre- and postoperative periods for VAS for pain (p = 0.003), Lysholm score (p = 0.002), IKDC subjective form (p = 0.003), WOMAC for pain (p = 0.005), WOMAC for stiffness (p = 0.01), WOMAC for function (p = 0.002), Lequesne Index (p = 0.002), and SF-12 physical component summary (p = 0.007). The postoperative mean (SD; range) MOCART score was 70 (12.4; 40–85).
Conclusions:
The use of this novel cartilage restoration surgical technique provides excellent clinical, functional, and MRI-based outcomes in young, active individuals with full-thickness cartilage or osteochondral defects.
Level of evidence:
Level IV—Therapeutic case series.
Keywords
Introduction
Articular cartilage injuries are a very common cause of knee pain and dysfunction. In addition, these injuries may lead to early-onset osteoarthritis and have a huge negative impact on the patients’ function and quality of life. Articular cartilage injuries are challenging to treat due to the inherent poor healing and regenerative potential of hyaline cartilage.
There are many options for orthopedic surgeons to treat knee articular cartilage injuries: chondroplasty, microfractures, mosaicplasty (osteochondral autograft transfer), osteochondral allograft transplantation, scaffold-based repair (with or without cell therapy), autologous chondrocyte implantation (ACI), or matrix-induced ACI. 1 –4 Some pitfalls of these techniques include fibrocartilage formation without long-lasting improvement (especially in high-level athletes), expensive treatments, need for two surgical procedures, long recovery time, or unpredictable results in the athletic population. 1,2,5 These pitfalls drive the need for continuous research and improvements in the treatment of cartilage injuries.
It is well-known that the platelets have a major role in hemostasis, inflammation, and proliferation for remodeling and tissue healing. In addition, platelets have an angiogenic power to deliver molecules into the damaged tissue. This inherent healing potential has made some authors to use plasma rich in growth factors (PRGF) for the treatment of cartilage injuries. In particular, Cugat et al. published two cases of full-thickness knee cartilage injuries treated with an autologous-made scaffold consisting of hyaline cartilage chips combined with a clot of mixed plasma poor rich in platelets. 6 This method has been recently named the CN-Biomatrix. 7 They reported successful outcomes with good function and ability to return to high-level soccer. Moreover, the same group performed a study in sheep where the same technique was successfully applied. 8 An histological and immunohistochemical analysis demonstrated that this technique was able to restore hyaline cartilage with adequate presence of type II collagen and minimum of type I collagen. 8 Despite the promising future of this technique given the high-quality cartilage tissue formation observed in sheep, this novel treatment option for articular cartilage injuries in humans has only been published as a case report. 6
The purpose of this study was to report the preliminary clinical, functional, and magnetic resonance imaging (MRI)-based outcomes of a novel autologous-made matrix consisting of hyaline cartilage chips combined with mixed plasma poor rich in platelets clot and PRGF in liquid stage for the treatment of full-thickness cartilage and osteochondral knee defects.
Material and methods
Procedures
Between July 2015 and January 2018, all patients with full-thickness cartilage defects undergoing a novel cartilage restoration procedure consisting of an autologous-made matrix using hyaline cartilage chips combined with platelet-rich growth factors (PRGF) in a semisolid state (named CN-Biomatrix) 6 were approached for eligibility. Indications for this procedure included traumatic or atraumatic full-thickness cartilage defects or osteochondritis dissecans of the medial or lateral femoral condyles, femoral trochlea, or patella. Patients were excluded if the surgery was conducted with concomitant use of adipose or bone marrow-derived stem cells, if the surgery involved a joint other than the knee, if a previous ipsilateral cartilage repair procedure was used, or if the follow-up was less than 10 months. Included patients were scheduled for a follow-up appointment to obtain data from current symptoms, physical exam, patient-reported outcomes, and MRI. These outcomes were compared to preoperative values. The follow-up appointment was conducted by the same experienced researcher, who was a fellowship-trained orthopedic specialist. The MRI of all patients was read by one independent radiologist specialized in knee injuries.
Surgical technique
The surgical technique for the CN-Biomatrix has been previously described. 6 The procedure was performed using a combination of arthroscopy and mini-open (arthrotomy) techniques (Figure 1). The patient was placed in supine position with knee extension and no leg support. The cartilage lesion was confirmed and the defect was prepared. Samples of healthy hyaline cartilage from defect edges were obtained using a curette. Care was taken to create a perpendicular or vertical rim on the cartilage defect so that it may better contain the autologous matrix (Figure 1). A debridement of the lesion was carried out using a combination of shaver and curette down to the subchondral bone by either an arthroscopic or mini-open technique. In the meantime, the cartilage samples were particulated in the back table so that cartilage chips were created to act as a framework or scaffold for the PRGF-based semisolid matrix (Figure 2). Then, a 3-cm longitudinal paramedial or paralateral (depending on the defect location) skin incision was created (Figure 1). In cases of patellar defects, the patella must have been averted. In the other cases, the knee was flexed until the defect was completely exposed. Currently, our preferred method for cartilage harvesting is using a shaver connected to an aspiration filter so that obtaining the cartilage can be achieved arthroscopically and time is saved because there is no need to use the blade to create the particulated cartilage chips. The arthrotomy is also used because it is helpful to create the perpendicular edges of the lesion in a shorter period of time and will be needed to introduce the matrix into the joint anyway.

CN-Biomatrix surgical technique. (a) Dry arthroscopy view of the osteochondral defect after the lesion is debrided up to the subchondral bone and the edges of the surrounding cartilage are perpendicularly created. (b) Demonstration of the semisolid CN-Biomatrix state and intra-articular placement once the mini-arthrotomy is created. (c) Dry arthroscopy view of the osteochondral defect once the CN-Biomatrix has been placement. Note the needle at the top of the image used to create a film above the defect using the supernatant obtained from Figure 2(c). (d) Second-look arthroscopy 10 months after surgery demonstrating the adequate coverage of the defect and integration of the CN-Biomatrix with the surrounding healthy cartilage.

Preparation method of the CN-Biomatrix. (a) The healthy hyaline cartilage harvested from the edges of the defect are particulated with a blade or directly obtained with a shaver connected to an aspiration filter. (b) The platelet-rich growth factors preparation is mixed with the hyaline cartilage chips. (c) The mixture is left at room temperature for about 30 min until the semisolid state is reached. (d) The supernatant is extracted from the autologous cartilage mesh. This supernatant will be used above the mesh once it is placed in the defect.
PRGF preparation was conducted using the Endoret© PRGF© system (BTI Biotechnology Institute, Álava, Spain). A BTI System IV© (BTI Biotechnology Institute) was used to centrifugate eight 9 ml tubes (each tube with 3.8% citrate solution) containing the patients’ blood extracted before surgery. The centrifugation process lasted 8 min at 580 g, obtaining the sedimentation of red and white cells at the bottom and platelets with plasma on the top part of the tubes. 6 The centrifugation process created two fractions: fraction 1 was the upper part of the supernatant and was the plasma poor in platelets (PPP), which was placed in BTI 9 ml, sterile-fractionation tubes; fraction 2 was the plasma rich in platelets (PRP) which was obtained by extracting the layer just over the white cells and placed in other BTI 9 ml, sterile-fractionation tubes. It was paramount to avoid aspiration of white cells when obtaining fraction 2 so that no inflammatory reaction was elicited. The growth factors were activated using calcium chloride (CaCl2) at a rate of 0.02 ml per milliliter of plasma, maintaining the tubes at room temperature. The activated PPP-PRP at a 50/50 ratio was combined with the hyaline cartilage chips and left for 30 min, until the semisolid CN-Biomatrix was formed (Figure 2). The time needed to form the Biomatrix varies according to the room temperature and patients’ blood. The autologous matrix was then placed in the patient’s chondral defect slightly below the level of the healthy surrounding cartilage to avoid overgrowth (Figure 1). The Biomatrix was homogeneously distributed along the defect and left in place for 5 min for adequate adhesion. Then, gentle flexion–extension movements were performed under direct visualization to ensure adequate stability of the construct. Next, the arthrotomy and portals were closed and the remaining of the fraction 2 intra-articularly injected after activation with CaCl2.
Rehabilitation protocol
Patients with chondral defects in the weight-bearing area of the femoral condyles were placed in a non-weight-bearing regimen for 2 months, with progression to full weight-bearing using crutches during the following months. Patients with chondral defects in other locations were allowed touchdown weight-bearing using crutches for 1 month. All patients were immobilized using two (medial and lateral) cast splints including the entire lower extremity for 1 week. Then, gentle passive flexion–extension exercises were conducted once a day to avoid stiffness for 1 week, progressing to twice a day for the following 2 weeks. Aquatic-based exercises were allowed as soon as skin incisions were healed. Stationary bicycle was typically allowed after 2–3 months, running at 4–5 months, and return-to-sports no earlier than 7 months, depending on the type of sport, injury characteristics, and status of the patient.
Outcome measurements
The clinical and functional outcomes (obtained preoperatively and at last follow-up) included the visual analogue scale (VAS) for pain, the Tegner activity scale, the Lysholm score, the International Knee Documentation Committee (IKDC) subjective evaluation form, the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) score, the Lequesne index, and the short form-12 (SF-12). The VAS is a 0–10 scale, where 10 represents the highest possible pain. The Tegner activity scale is a 0–10 scale where each number corresponds to a certain intensity of physical activity (10 being the highest intensity). The Lysholm knee scoring system consists of eight items measuring pain (25 points), instability (25 points), locking (15 points), swelling (10 points), limp (5 points), stair climbing (10 points), squatting (5 points), and need for support (5 points). The total score ranges from 0 to 100, where 100 indicates a better outcome with fewer symptoms or disability. The IKDC subjective evaluation form is a knee-specific, patient-reported outcome measurement tool evaluating symptoms, sports activity, and knee function. A total score of 0 to 100 is obtained, where higher scores represent higher levels of function. The WOMAC score contains 42 items related to five dimensions: pain (9 items), symptoms (7 items), daily activities (17 items), sports and recreational activities (5 items), and quality of life related to the knee (4 items). Each subscale is assigned a score ranging from 0 to 100, where 100 indicates good knee function. The Lequesne index is a 10-question survey including five questions related to pain or discomfort, one question dealing with maximum distance walked, and four questions about activities of daily living. The index is reported in a 0–24 scale where 0 is no functional limitation. The SF-12 Health Survey is a general health status questionnaire with 12 questions producing 2 summary scores, the physical component summary (PCS-12) and the Mental Component Summary (MCS-12). The SF-12 is scored so that higher scores represent better physical and emotional function.
The quantity and quality of the cartilage repair tissue was evaluated using the magnetic resonance observation of cartilage repair tissue (MOCART) score. 9 This score contains nine items related to the degree of defect repair and defect filling, integration to the border zone, surface of the repair tissue, structure of the repair tissue, signal intensity of the repair tissue, subchondral lamina, subchondral bone, presence of adhesions, and presence of effusion. The score ranges from 0 to 100, where 100 represents the best cartilage repair tissue.
Statistical analysis
Descriptive statistics were used to summarize demographic and injury characteristics and the outcome. Central tendency values were reported using the mean, and dispersion values reported as standard deviation and range. A Kolmogorov–Smirnov test of normality was performed. Parametric tests were applied for variables with normal distribution, whereas nonparametric tests were applied for variables without normal distribution. The paired t-test or Wilcoxon tests were used to compare preoperative and postoperative outcomes. The α level was set at 0.05. The statistical analysis was conducted using the SPSS v20 (SPSS Inc., Chicago, Illinois, USA).
Results
As of January 2019, 92 surgical procedures using this technique have been performed. During the study period (July 2015 to January 2018), a total of 30 cartilage procedures had been conducted. Of these, two were excluded because stem cell therapy was used, nine because surgery was not conducted for the knee, and three because the follow-up was less than 10 months. A total of 15 patients could be included in this preliminary study: mean (standard deviation (SD), range) follow-up 15.9 months (7.2, 10–32), age 26.8 years (12.1, 16–58), height 1.7 m (0.1, 1.67–1.87), weight 73.1 kg (8.6, 57–90), and body mass index 23.2 (2.1, 19.3–26.9). There were 14 (93%) men and 1 (7%) woman, and 10 (66%) procedures were performed for the right knee and 5 (34%) for the left knee. There were nine (60%) patients playing soccer at a competitive level, three (20%) recreational athletes, two (13%) sedentary individuals, and one (7%) recreational basketball players. Comorbidities included low back pain in four (27%), obesity in two (13%) patients, diabetes mellitus in one (7%) patient, depression in one (7%) patient, and inflammatory bowel disease in one (7%) patient. There were two (13%) active smokers in the sample. There were 5 (34%) patients with and 10 (66%) patients without previous surgery. Previous surgery included isolated anterior cruciate ligament reconstruction in one patient (7%), combined anterior cruciate ligament reconstruction plus meniscectomy in one patient (7%), partial meniscectomy in one patient (7%), combined patellofemoral bone and soft-tissue realignment procedure in one patient (7%), and revision anterior cruciate ligament reconstruction in one patient (7%). This last patient underwent a total of four revision anterior cruciate ligament reconstructions and one medial collateral ligament reconstruction after the primary anterior cruciate ligament reconstruction procedure. The characteristics of the injury are summarized in Table 1.
Injury characteristics in the sample.
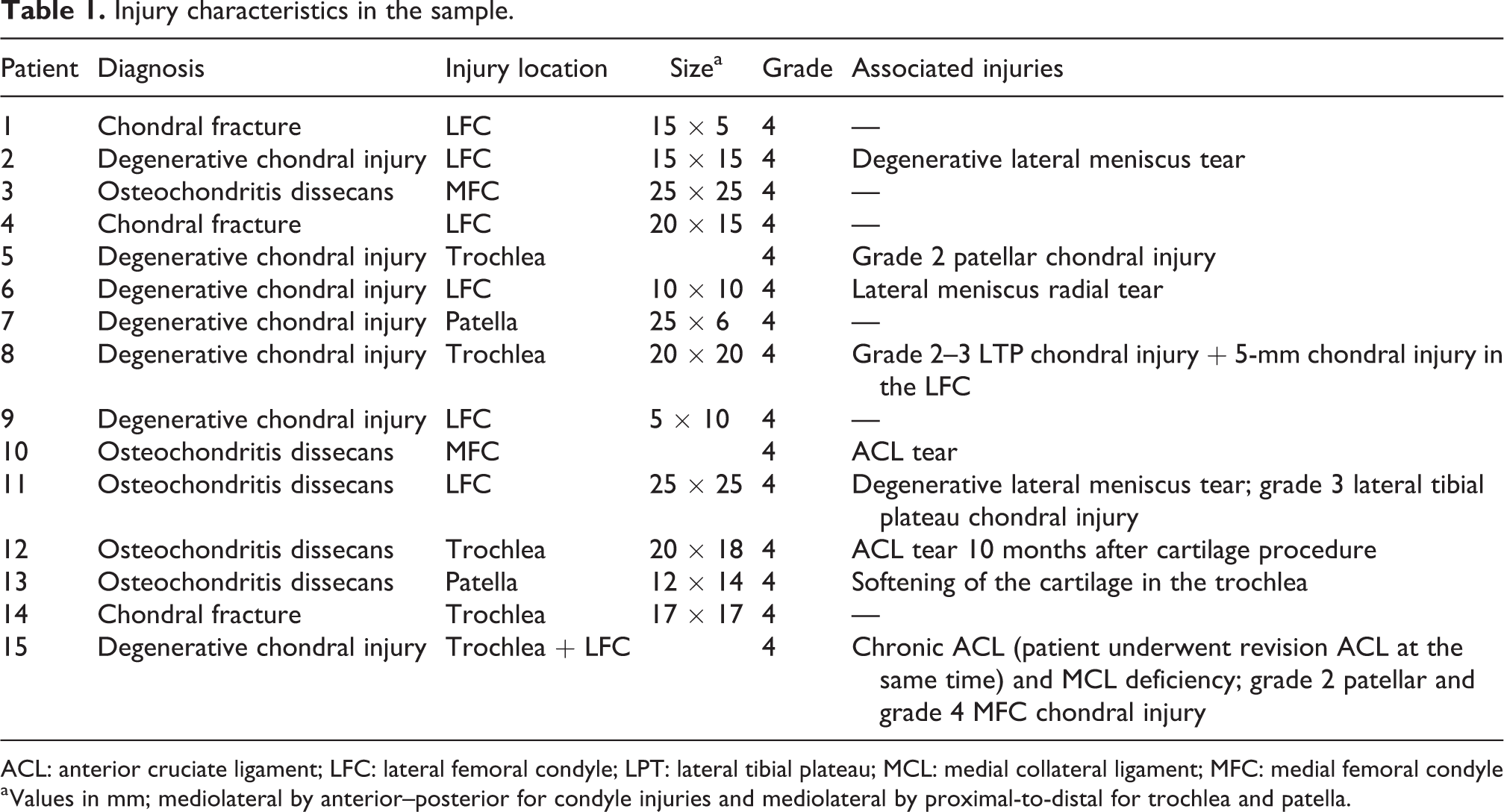
ACL: anterior cruciate ligament; LFC: lateral femoral condyle; LPT: lateral tibial plateau; MCL: medial collateral ligament; MFC: medial femoral condyle
a Values in mm; mediolateral by anterior–posterior for condyle injuries and mediolateral by proximal-to-distal for trochlea and patella.
There was a statistically significant difference in all clinical and functional outcomes (except for the Tegner activity scale and SF-12 MCS) when comparing the preoperative and postoperative periods (Table 2). The mean (SD; range) MOCART score was 70 (12.4; 40–85). The number (percentage) of patients distributed in each of the MOCART subscales has been reported in Table 3. The MOCART score could not be obtained for one of the patients.
Comparison of preoperative and postoperative outcomes.
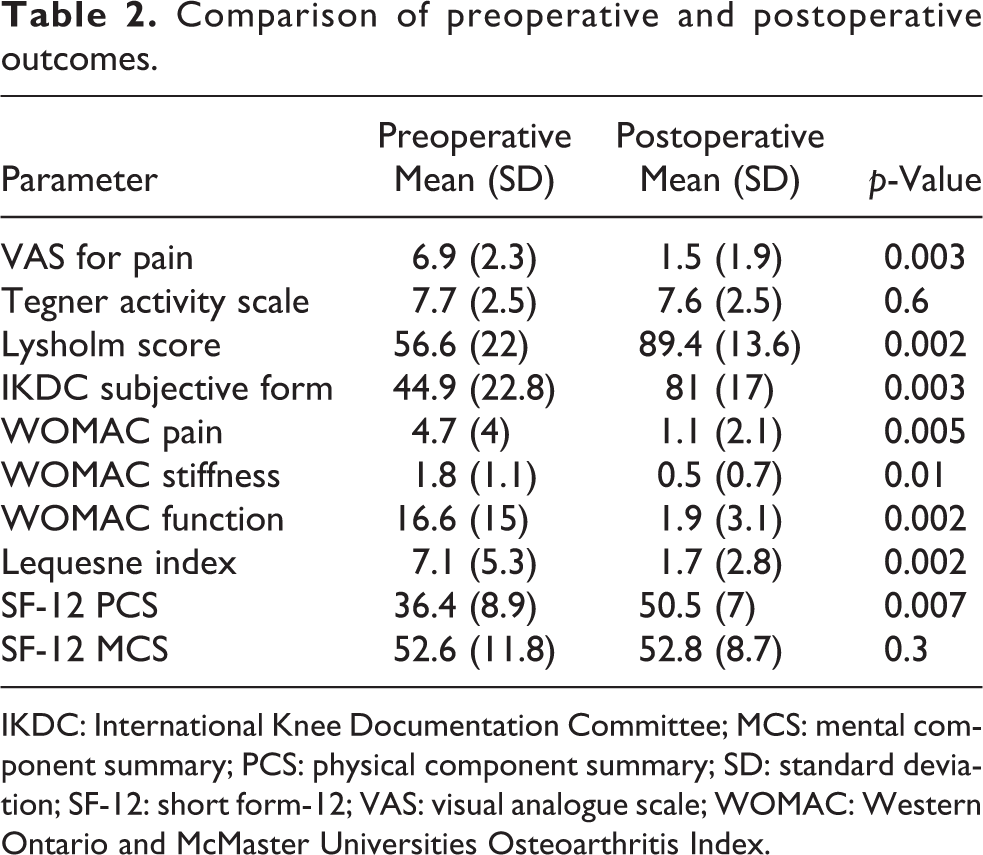
IKDC: International Knee Documentation Committee; MCS: mental component summary; PCS: physical component summary; SD: standard deviation; SF-12: short form-12; VAS: visual analogue scale; WOMAC: Western Ontario and McMaster Universities Osteoarthritis Index.
MOCART score.a

MOCART: magnetic resonance observation of cartilage repair tissue; FSE: fast spin echo.
a One missing value.
Discussion
The principal finding of this study was the excellent outcomes with a novel autologous-made matrix using hyaline cartilage chips and platelet-rich growth factors (the CN-Biomatrix) for the treatment of full-thickness cartilage defects in a sample of young active individuals. The present surgical technique provided excellent pain relief and high functional outcomes, with very good quantity and quality of the cartilage repair tissue.
Cartilage lesions are one of the most common injuries in the general and athletic population. 10 –12 These are challenging, severe, and worrisome injuries because of the limited healing and regenerative potential of hyaline cartilage. The downsides of the previous surgical techniques for cartilage repair have been the formation of fibrocartilage without long-lasting improvement (especially in high-level athletes), expensive treatments, need for two surgical procedures, long recovery time, or unpredictable results in the athletic population. 1,2,5 Of particular interest is the fact that most of the previous cartilage repair procedures have not been able to produce a normal cartilage. 2
Domínguez-Pérez et al. reported a macroscopic, histological, and immunohistochemical study on sheep with full-thickness knee cartilage defects using the presented technique. 8 At 6 months, the macroscopic evaluation consisted of nearly normal International Cartilage Repair Society score. The histological analysis demonstrated a mature cartilage tissue in the defect with similar structure than the healthy hyaline cartilage. Interestingly, the immunohistochemical analysis revealed that 75% of the collagen was type II, with minimal (8%) type I, in the newly formed cartilage. However, a clinical study was needed to investigate whether the high-quality articular cartilage observed with this surgical technique in the animal study implied adequate clinical and functional outcomes in humans.
Cugat et al. recently published the first two cases of soccer players with full-thickness knee chondral or osteochondral defects treated with the CN-Biomatrix. 6 The authors observed that both players returned at the pre-injury level and demonstrated excellent knee function. The follow-up MRI of these patients showed that there was an adequate filling of the defect with cartilage of similar signal and absence of significant bone edema. Remarkably, a second-look arthroscopy evidenced similar appearance and consistency on palpation of the newly formed cartilage compared to the surrounding healthy hyaline cartilage. The present study confirmed the excellent clinical, functional, and MRI-based outcomes in a larger sample (preliminary results of the first 15 patients). The CN-Biomatrix preparation has slightly evolved with increasing experience. In the original preparation method used in the animal study, 8 only the PPP (fraction 1) was used to create the growth factor-based matrix. It was initially recommended that fibrin membranes should be elaborated using this fraction, 13 but we felt that this method led to a too rigid matrix that could not be easily adapted to the chondral defect and also reached the semisolid state quicker. In addition, the same authors recommended to use PRP (fraction 2) to create growth factor-based clots. 13 Therefore, we modified the original clot preparation method by using a combination of fraction 1 and 2 (50% each) to create the growth factor-based clot. This preparation method has been used in the present study and the previous case report. 6
Previous surgical techniques have demonstrated a variety of MRI-based cartilage repair characteristics. The present surgical technique has demonstrated higher MOCART scores compared to microfracture and collagen membrane matrix, 14 microfracture and a chitosan-based polymer scaffolding biomaterial, 15 mesenchymal stem cells, 16 and cell-free scaffold with collagen and hydroxiapatite. 17 Interestingly, Christensen et al. reported the outcomes of a dual-tissue transplantation technique for osteochondritis dissecans in which autologous bone and cartilage tissue were used. 18 The defect was filled with autologous bone to the base of the surrounding cartilage tissue, and then biopsies of cartilage from the intercondylar area were obtained, chipped, and embedded with fibrin glue and placed in the defect area. The authors reported a postoperative MOCART score of 52.5 at 12 months, as opposed to the 70 obtained in the present study. Both techniques clearly differ from the application of PRGF, so it might be argued that the application of growth factors may make a significant difference in the quality of the cartilage repair tissue. Trattnig et al. reported higher MOCART scores at 24 months (84.4) after applying a cell-free biosynthetic, biodegradable hydrogel implant. 19 The authors observed that the MOCART score progressively increased over time from 6 to 24 months after surgery. It might be possible that a longer follow-up would elicit higher MOCART scores using the CN-Biomatrix, although this needs to be demonstrated in subsequent studies.
The present study has some limitations. Most importantly, this was a preliminary study with a short sample size, short-term follow-up, and absence of a comparative group. In addition, no histological and immunohistochemical study was available for the cartilage repair area. However, this is the first clinical study reporting the outcomes of this technique in young active individuals. This study was needed after the excellent quality of the newly formed cartilage evidenced in the preceding histological and immunohistochemical study in sheep. 8 Further research is needed with high-quality comparative studies with larger sample size and longer follow-up.
Conclusions
This novel autologous-made matrix consisting of hyaline cartilage chips combined with mixed plasma poor rich in platelets clot, and PRGF in liquid stage (the so-called CN-Biomatrix) provided excellent clinical, functional, and MRI-based (cartilage repair quality and quantity) outcomes in young, active individuals with full-thickness cartilage or osteochondral defects. This technique seems an excellent alternative for cartilage injuries, as it can be performed in very active, young individuals as a single surgery, is cheap, has no intolerance or rejection potential, and has demonstrated histological and immunohistochemical characteristics very similar to healthy articular cartilage in animal studies.
Footnotes
Acknowledgements
The authors would like to give a special thanks to Dr Martínez for his excellent contributions to the MRI readings, to Dr Muñoz for his assistance in data collection, and to all patients for their willingness to participate in this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
