Abstract
Aim:
Given recent concerns regarding the influence of different cements and implants on the rate of aseptic failures in total knee arthroplasty (TKA), we wondered if cementation technique could play a role. The primary aim of this review was to collect and compare the surgeon education materials from eight orthopedic implant manufacturers to evaluate the manufacturers’ recommended cementation technique in TKA and identify if there was any consistency in these readily available guides as to the best practices of cementation of their implants.
Materials and Methods:
We reviewed contemporary surgeon education guidelines for all TKA systems available from eight manufacturers. Variables included: cement type, batches prepared, surface preparation prior to application, cleaning and/or drying the bone surface, mixing the cement, the waiting phase after the cement has been mixed prior to application, pressurizing the cement, location of cement application, and the curing time. Data were recorded and organized for qualitative comparisons.
Results:
We identified a total of 43 guides covering 38 implants from eight different manufacturers. There were 41 surgical technique guides and two general brochures regarding cementation techniques available from the manufacturers. Even within the manufacturers’ own guidelines for the different implants, there was a wide variety of differing guidelines on many aspects of the cementation technique.
Conclusion:
There is clearly no consensus for a preferred cementation technique both within and among manufacturers’ surgeon education materials regarding tibial baseplate cementation during TKA. Efforts may be needed to identify a best-practice cementation technique in an effort to reduce the number of TKA failures associated with aseptic loosening.
Introduction
Although total knee arthroplasty (TKA) has historically been an extremely successful operation, the number of revision surgeries is expected to continue to increase over time. Thus, it is essential to understand why TKA operations fail and to take all possible measures to reduce the incidence of failure. Several mechanisms of TKA failure have been identified, with infection and aseptic loosening being among the most common reasons. 1 Recent concerns have been raised regarding the influence of different cements, including high versus low cement viscosity, antibiotic versus nonantibiotic cement, hand mixed versus commercially mixed antibiotics, bowl mixed versus vacuum mixed cement, full versus partially coated implants, as well as implant design and implant materials in the role of aseptic failures in TKA. 2 –8 Many studies that look at implant survivorship come from case series, databases, or registries. One factor that has been less discussed includes potential variability in surgical cementation technique. 4,9,10 While most retrospective reviews and registry level data may be able to look at the implants, cements, or supplies used during high volumes of surgeries, the actual surgical technique of how cement is applied and utilized is not often captured or analyzed. While there may be many factors that influence a surgeon’s technique, including their training and experience, one source of information that could provide recommendations and is readily available to all surgeons using a particular implant includes the implant manufacturers’ technique guides.
Variations exist in the surgical technique that surgeons use to cement the tibial and femoral components during TKA. In vitro testing has shown that key variables such as time of cement application, cementation of the stem and tray surfaces, cement phase, cement being applied to clean and dry surfaces of just the implants, just the prepared bone surfaces, or both, and cement penetration all have vital roles in influencing the implant–cement interface bonding. 2,4,9 –12 Given the increased concern around aseptic TKA failures and variations in cementation technique, we wondered if there was standardization in the recommendations from implant manufacturers regarding best practice cementation of their implants. While we understand full well that surgeons may not have their practice influenced by the industry-published literature, reviewing this at least serves as a starting point for us to understand if there is any currently published source of possible consensus or best practices.
The purpose of this review was to collect and compare the surgeon education materials for the most popular implants from eight orthopedic implant manufacturers to assess the manufacturers’ recommended cementation technique in TKA.
Materials and methods
We systematically reviewed contemporary surgeon education guidelines for total knee systems available in their respective libraries from the following manufacturers: Zimmer Biomet (Warsaw, Indiana, USA), DePuy Synthes (Warsaw, Indiana, USA), Stryker (Kalamazoo, Michigan, USA), Smith & Nephew (Memphis, Tennessee, USA), Aesculap (Center Valley, Pennsylvania, USA), Ortho Development Corporation (Draper, Utah, USA), Medacta International (Castel San Pietro, Switzerland), and Conformis (Billerica, Massachusetts, USA). Based on global sales from 2017, we identified Zimmer Biomet, Depuy Synthes, Stryker, and Smith & Nephew as the four largest orthopedic implant manufacturers. 13 After these four manufacturers, the total combination of all “other companies” sales slightly exceeded the sales of the fourth largest company. To capture guidelines outside of the four major manufacturers, we also included the review of surgical guidelines for knee implant systems from Aesculap, Ortho Development, Medacta International, and Conformis. We identified a total of 43 guides covering 38 implants from eight different manufacturers (Figure 1). They consisted of 41 surgical technique guides and 2 general brochures regarding cementation techniques provided by the manufacturers.
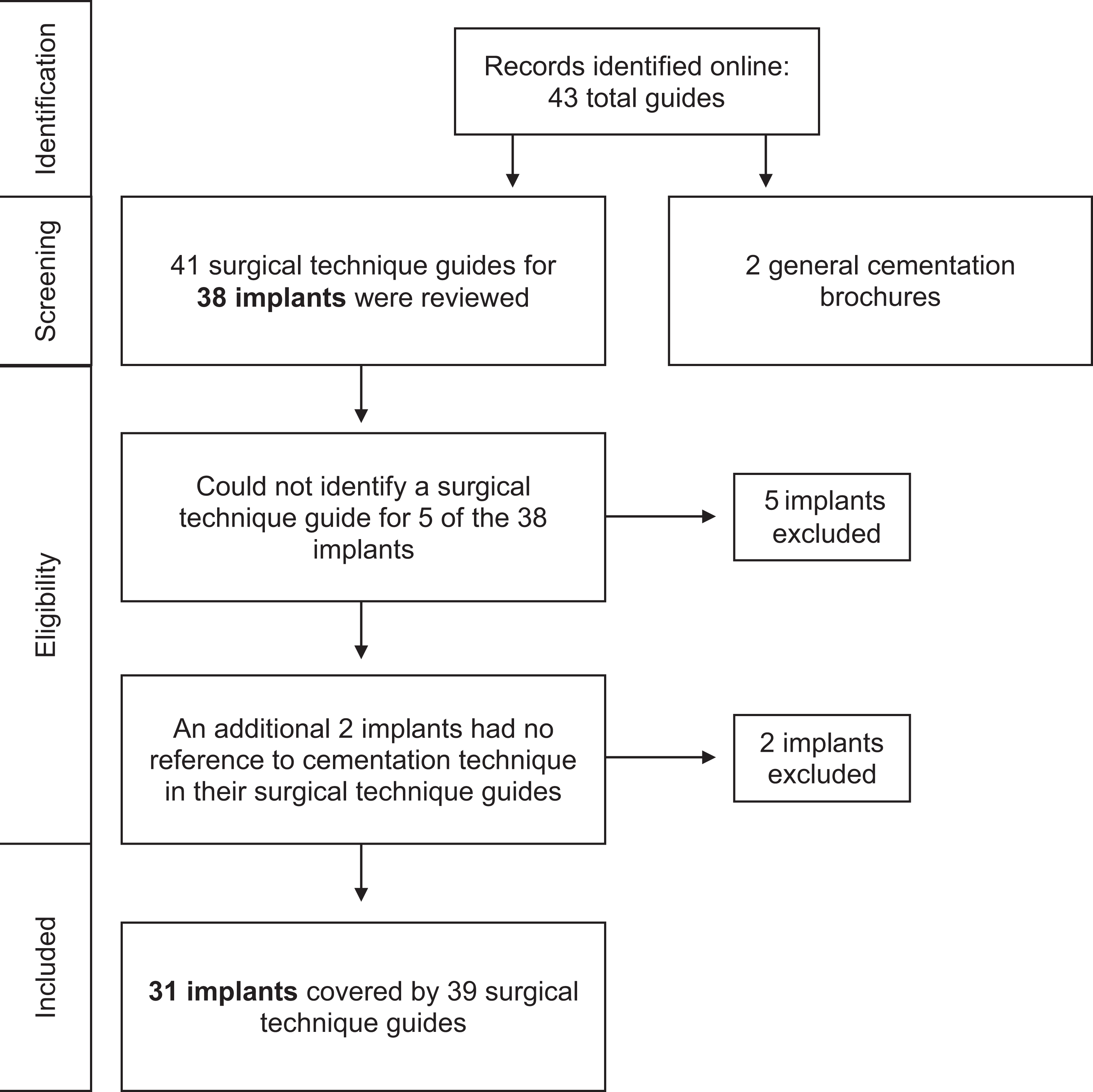
Inclusion criteria flow diagram.
All text, figures, and graphics related to cementation technique (both preparation and application) in the surgeon education guidelines for each total knee system were evaluated. Techniques for only the tibial component were included, given the overall lack of descriptiveness for technique regarding the femoral or patellar components in our initial review. The variables of interest included cement type, batches prepared, surface preparation prior to application, cleaning and/or drying the bone surface, mixing the cement, the waiting phase after the cement has been mixed prior to application, pressurizing the cement, location of cement application, and the curing time.
Data were recorded and organized for comparison both within and between individual manufacturers. Qualitative comparisons are reported.
Results
Both the surgical technique guides and the cementation brochures lacked uniformity in regard to the variables of interest analyzed (Tables 1 to 8). Further, as mentioned, there was a lack of description of the technique for components other than the tibial tray, and thus, we organized the systematic description of techniques around just the tibial component.
Zimmer Biomet cementation guidelines.

IM: intramedullary; PFJ: patello-femoral joint.
a Although we did not find any surgical technique guide that addressed cementation for this implant, the brochure states that this implant can be implanted using any Zimmer surgical technique including Zimmer® Minimally Invasive Solutions™ (MIS™) procedures.
Depuy Synthes cementation guidelines.

MV: medium viscosity; HV: high viscosity.
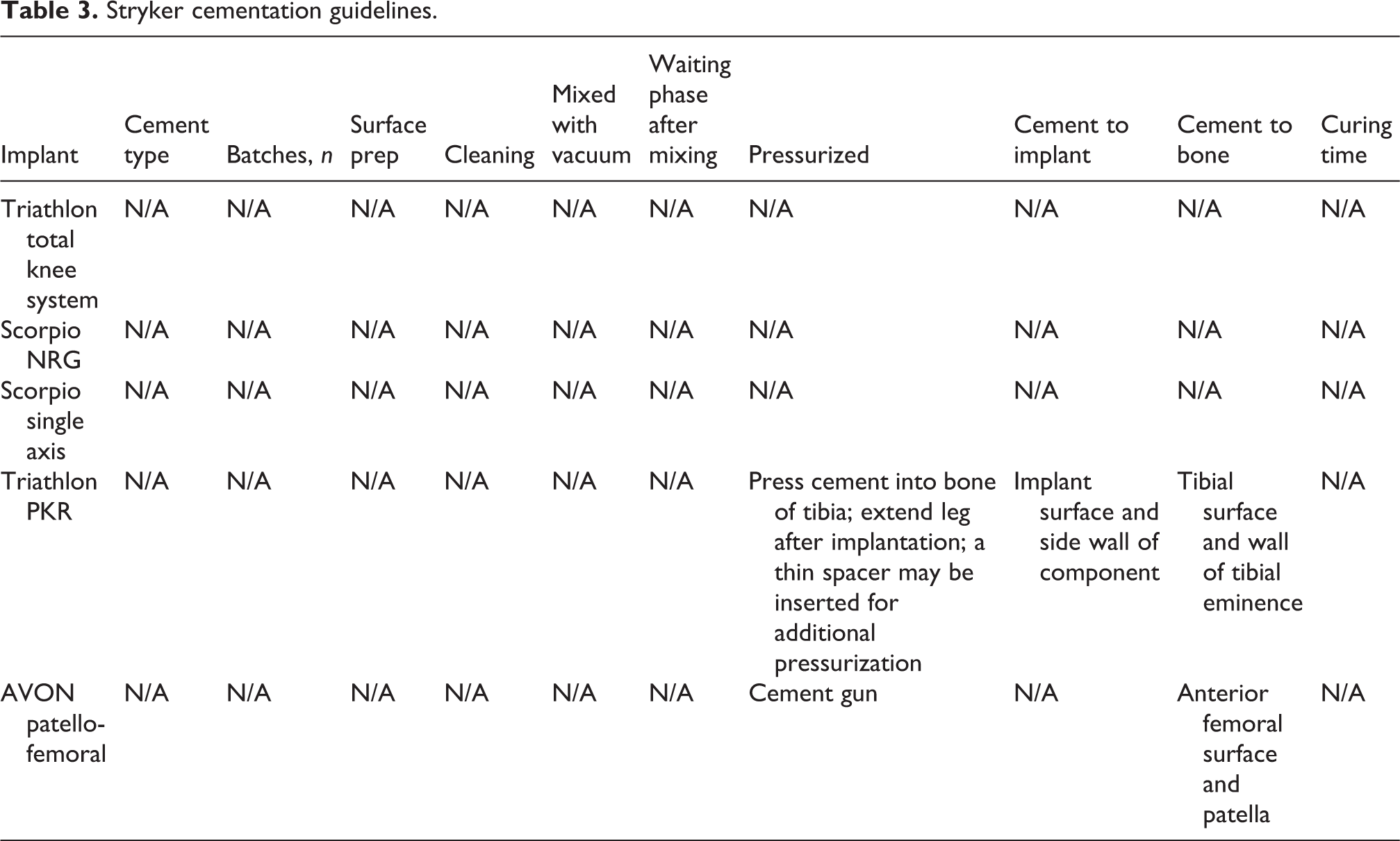
Stryker cementation guidelines.
Smith & Nephew cementation guidelines.

Aesculap cementation guidelines.a

a We were unable to secure a copy of the surgical technique guides for the implants to review, although the implant manuals refer to it and say that it is available on request.
Ortho Development cementation guidelines.
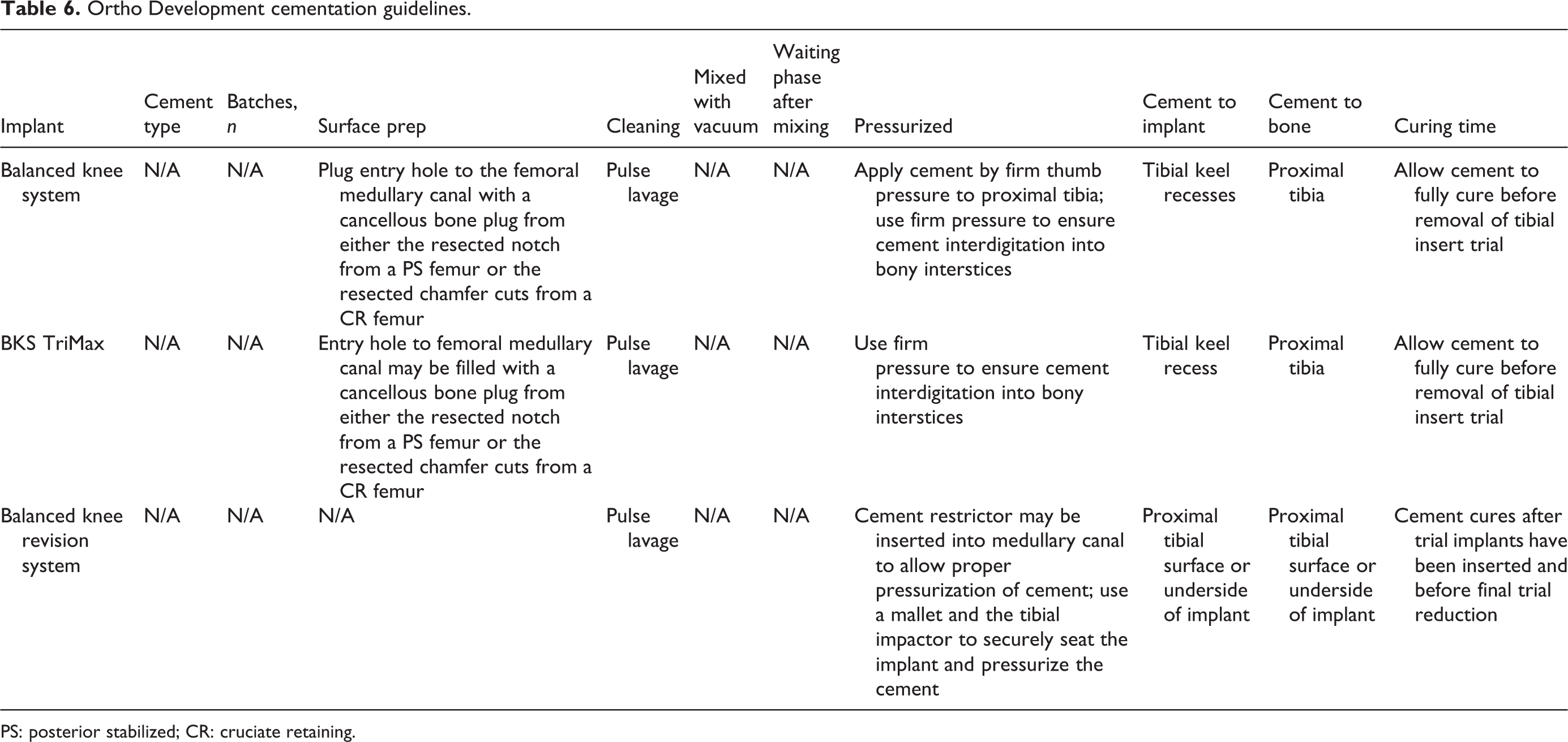
PS: posterior stabilized; CR: cruciate retaining.
Medacta International cementation guidelines.

Conformis cementation guidelines.
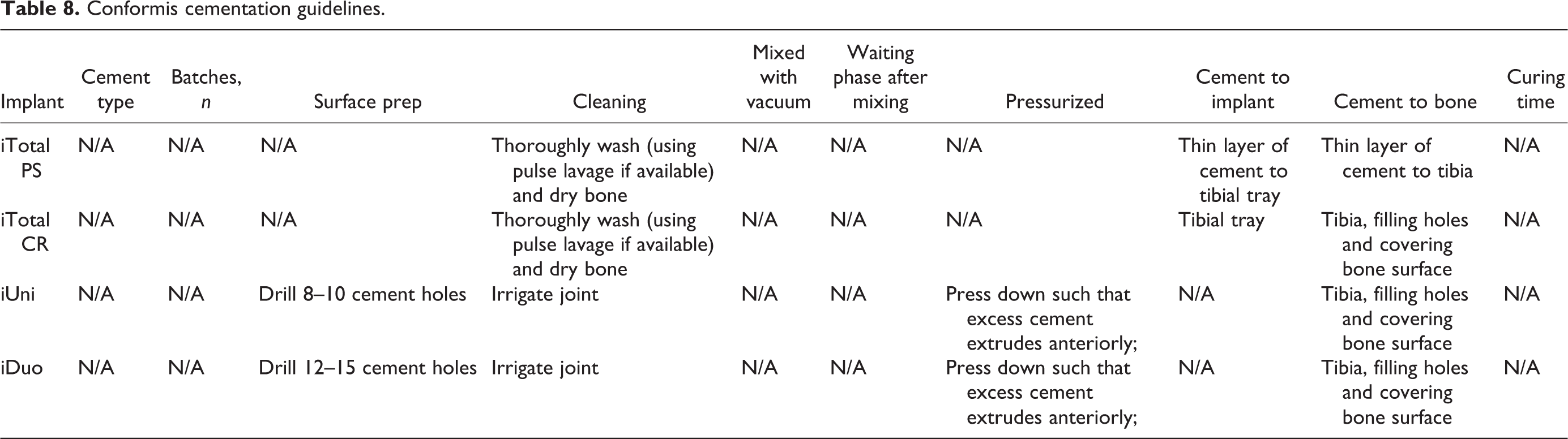
Six implants lacked guides with any information on cementation technique. Ultimately, only 5 implants had guides that addressed the type of cement to be used, 2 implants had guides that addressed the number of suggested cement batches, surface preparation prior to cementation was provided for nine implants, 13 had guides that addressed cleaning and/or drying the bone surface, 6 had guides that addressed mixing the cement, 9 had guides that addressed the waiting phase after the cement has been mixed, 32 had guides that addressed pressurization of the cement, 24 had guides that addressed location of the cement application, and 10 had guides that addressed the curing of the cement.
The majority of information available was regarding pressurization and cement placement. Thirty-one implants had guides that addressed pressurization of the cement. Ultimately, two main types of pressurization were described including the use of a mechanical device versus manual pressurization. Mechanical devices included the use of a cement gun (3) or a syringe (1). Manual pressurization techniques included extending the leg after implantation with a spacer (8), using mallet blows (4), using a tibial impactor (29), or “other” manual means including digital impaction (10). One guide discussed the use of a restrictor to aid in pressurization. The majority of guides suggested multiple means of pressurization.
Twenty-four implants had guides that addressed cement location, either on the implant (22) and/or on the bone (24). Cement location on the implant varied from the general underside of the baseplate (6/22), a specific location on the underside of the baseplate (2/22), the tibial tray (2/22), the surface of the implant (1/22), the sidewall of the implant (1/22), the keel (3/22), and the keel recesses (2/22). Seven implants had guides that directed the surgeon to apply cement to the implant but lacked more specific instructions. Seven implants had guides that stated that it is optional to put cement on the implant, with four specifically stating that it is based on the surgeon’s preference. The locations of cement application on the bone were also inconsistent with locations including the general bone (7/24), general bone with specific cement penetration dimensions (1/24), resected surface holes (4/24), resected tibial surface (6/24), wall of tibial eminence (1/24), tibial intramedullary canal (3/24), and the proximal tibia (6/26). Five implants had guides, which stated that it is optional to put cement on the bone, and three implants had guides, which indicated that the application of cement to the bone was based on the surgeon’s preference.
Notably, the cement type, batches prepared, mixing the cement, the waiting phase after the cement has been mixed prior to application, and curing time all lacked consistency in the published guides.
It is of particular interest that both the surface preparation and cleaning of the bony surfaces were also very limited in information as these are thought to be very important variables in cementation technique. Nine implants had guides that addressed surface preparation before cementation. While most implants (7/9) had guides that recommended drilling holes into the bone to improve cement interdigitation, only two of them specified how many holes to drill, and only one specified the exact dimensions of how big, deep, and how spaced apart the holes should be. Thirteen implants had guides that suggested cleaning the bone before cement application. Most of those that suggested cleaning specifically recommended pulse lavage (10/13), but only five directed the surgeon to dry the bone after cleaning.
Even within the manufacturers’ own guidelines for the different implants that they provided, there was a wide variety of differing guidelines on many aspects of the cementation technique (Tables 1 to 8). Finally, it should be noted that tibial impaction was not included in the data tables under the pressurization column as it was found in 29/31 implants reviewed.
Discussion
This study confirms our suspicion that currently available orthopedic implant manufacturer product literature lacks consistency in recommended cementation techniques for TKA. There was a wide variety of differing and inconsistent guidelines in surgeon education material both within and between manufacturers with no clear consensus of best practice. Given that aseptic loosening remains among the top two most common causes for failure in TKA, it is possible that the underlying causes relating to this failure should be further understood. 1 Infection has been identified as the primary reason for revision with rates generally ranging from 25% to 36% of all revisions, whereas aseptic loosening generally comprises 18–27% of revisions. 14 –16 There has been, and continues to be, a great deal of orthopedic research dedicated toward understanding and lowering infection rates following TKA through patient optimization, efficient surgery, controlling intraoperative conditions, and decreasing postoperative complications. 17,18 Reasons for aseptic loosening, however, are not completely understood and are likely multifactorial in nature but may have room for additional investigation. This includes inquiring about best practices in cementation technique. Etiologies for TKA aseptic failure have included polyethylene wear with subsequent osteolysis, postoperative alignment, and body mass index (BMI). 8,19 –25 As implant design and polyethylene production have improved, we have seen a decline in failure due to polyethylene wear and subsequent osteolysis. 26,27 As such, other factors of implant fixation that have gained recent scrutiny and include factors related to the cement or implant include: (1) implant selection with considerations for surface roughness, 28 implant material, 28,29 implant pattern, 28 and placement; 24,30 (2) surface preparation including perforation of sclerotic bone 3,31 and pulsed lavage 32 –34 ; (3) viscosity of the cement 6,11,35 ; and (4) cementation technique 4,7,9,34,36 to include the set phase, 9 penetration, and coverage 7,36 (surface, bisurface, or fully cemented keels). The recognition that cementation technique is a potential contributing factor to aseptic loosening may be of importance, particularly if there is a lack of consensus on best practice, standardization in technique, and documentation thereof. Given that registry or database studies often look at implant, cement, or supplies, these studies may not be able to detect the influence of technique on outcomes or survivorship. 37
Implant design and materials may be a contributing factor in aseptic cemented TKA failures. Pittman et al. 28 found that metal–cement interface strength generally increases with increasing surface roughness, showing that the type of metal substrate and surface preparation of contemporary tibial baseplates may influence the strength of the metal–cement interface. They reported that almost all of the surface finishes attained stronger bonds with cement when the sample prostheses were constructed from titanium bar stock (as opposed to cobalt-chrome). They also found that a macrosurfaced implant may be more vulnerable to rotational forces, which may be partially responsible for the failures with associated implant-cement debonding reported by Mikulak et al. 29 with the press-fit condylar modular total knee prosthesis. Finally, some reports have suggested that implant placement, specifically a mechanical alignment error more than 3°, increases the risk for aseptic loosening and that this alignment error is amplified in patients with an increased BMI. 24,30
Drilling or perforation of the sclerotic bone should be performed prior to pulsed lavage to allow for complete removal of the bone debris. 3 Perforation of the sclerotic bone has been shown to improve cement penetration and decrease radiolucent lines at 1- and 2-year follow-up when larger diameter (4.5 mm) bits are used. 31 Final preparation of the bone–cement interface is best performed using pulsed lavage. In separate studies, Schlegel et al. 32,33 confirmed the importance of bone preparation and pulsed lavage in enhancing fixation strength and recommended that it is a mandatory step in surface preparation along with copious drying of the tibial surface when cementing tibial components in TKA.
Cement viscosity is another factor that has gained recent attention, particularly as high viscosity cement (HVC) has seen increasing utilization. 5 Cement viscosity contributing to TKA loosening was brought to attention by Kopinski et al. 35 when they detailed a case series that showed the association between the use of HVC and debonding of the tibial implant–cement interface. Kopec et al. 6 found evidence to support this claim in a study, where cement penetration into the tibial plateau was significantly reduced with the use of HVC. Conversely, Crawford et al. 11 showed that HVC and a standard 40-mm I-beam tibial tray has a very low incidence of tibial aseptic loosening even in a high-risk group of obese patients. It is also interesting to note that the percentage of TKAs performed using HVC has steadily increased over the most recent 7 years for which the American Joint Replacement Registry has data. 5 While Kopinski et al. 35 proposed a potential for tibial baseplate debonding with the use of HVC, they noted several limitations to their case series. Primarily, they note that due to all of the procedures occurring elsewhere, they (1) lacked the ability to provide a description of the baseplate material (e.g. cobalt chromium vs. titanium baseplates) and (2) are unable to describe the surgical technique (e.g. how was the cement mixed, was cement applied to the implant and prepared bone, or one or the other). The findings in their publication may need to be reassessed with all of these factors accounted for.
There are several factors associated with the preparation and application of the cement itself, including the set phase of the cement. Silverman et al. 9 compared cement penetration of different cement phases in TKA and found that cement penetration was greatest in the dough phase. They suggest that this application of the cement in this set phase may decrease the likelihood of aseptic loosening. When considering the surface location of the cement, a cadaveric study performed by Peters et al. 7 found no difference in micromotion between surface cementing TKA and fully cemented TKA and suggest that this may be due to the cement penetration of 3–5 mm in both groups. Using a sawbone model, Vanlommel et al. 38 concluded that a penetration depth of 3–5 mm is optimal and recommended bisurface application of cement on the underside of the tibial baseplate as well as on the cut surface of the proximal tibia using digital impaction or impaction with a spatula. Pelt et al. 34 confirmed adequate results of implant stability using a surface cementation technique of the tibial component (as compared to the fully cemented keel technique). Finally, Han et al. 4 found that pressurizing and using a bisurface cementation technique significantly reduce the occurrence of early loosening around the femoral component. They claim that this is due to the paramount importance of the initial fixation of cement. Interestingly, Galasso et al. 36 suggest that cementing the keel had no additional advantage for fixation.
When evaluating our own series of TKA patients from 2003 to 2008, we identified only 4/468 (<1%) TKA cases with aseptic loosening. 39 Similarly, when assessing those with the only surface cementing of the tibial baseplate, we reported <1% aseptic loosening rates at a minimum 5-year follow-up. 7 Our cementation technique remained relatively stable over the course of these studies and even to the present time. We feel this may provide some explanation as to the low rates of aseptic loosening as we follow a strict cementation regimen. Our technique has consisted of perforation of any sclerotic bone with a drill bit in multiple drill holes to allow for adequate penetration. The bone is then thoroughly irrigated with pulse-lavage and thoroughly dried. Then, in its most viscous state, the cement is mixed and applied to the clean dry implant prior to any contamination. Following this, cement is applied to the prepared bone with the use of a cement gun or wide osteotome with several pressurizing passes made with the osteotome and thumb pressurization with significant focus of maximizing cement interdigitation to the prepared bone, with the goal of 3–5 mm of cement penetration. Meticulous cleaning of excess cement is performed. The tibia is cemented first, followed by the femoral component using the same technique under a single batch of cement. With the final bearing in place, the knee is then taken to extension and held still with slight constant pressure until the cement has fully cured in the bowl. Only then is the knee moved, inspected, and cleaned of any residual extruded cement.
While many factors may independently influence implant loosening, ultimately the underlying consideration to all of these factors is the cement application to the bone and implant. We feel that this is likely a factor that is less considered by surgeons and manufacturers, maybe under-assessed in survivorship studies given the inherent lack of discrete data on technique available to registry in database studies, and likely requires further investigation.
This study provides an analysis of just one potential source of information that could be referenced by practicing surgeons in regards to best practices in cementation. We fully recognize that the techniques that surgeons may actually employ during surgery are likely rarely influenced by product literature and can be assumed to be more influenced by their background, training, practices of their mentors, and on their own knowledge and experience. However, if there is an absence of best practice consensus in the published product literature, it is quite likely that there are few other published sources of consensus on best practice cementation techniques in TKA. Our findings represent, perhaps more than a scientific revelation, a potential call to ourselves and our organizations to consider making the topic of cementation technique a priority to gather additional data and provide a consensus agreement on best practices. Given the unaccounted-for variables that could be introduced by surgical technique, we feel that case series, database, or registry reviews raising concerns over implant designs or cement types may fall short of understanding the entire picture surrounding aseptic loosening and failure in TKA in modern practice.
This report has several imitations. Many of the surgical technique guides either had very little information or no information at all on cementation technique. Even after contacting representatives at the respective manufacturers, it was difficult to find any additional information or guidelines on cementation technique. We feel that part of the obstacle in obtaining clear guidelines for cementation could possibly be due to manufacturers’ efforts to purposefully limit information on cementation technique in their brochures and surgical technique guides to avoid off-label use. By doing so, they are not responsible for getting approval from governmental oversite agencies on the technique or device (polymethylmethacrylate, bone cement is an FDA class II device) and ultimately leave the cementation technique up to the cement manufacturers or surgeon’s preference. Further, we did not survey orthopedic surgeons to determine adherence to recommended cementation techniques found in the respective surgical technique guides, nor on their current practices. Knowing how surgeons learned their cementation technique and what cementation regimen they actually incorporate into their practice for TKA procedures might lead to greater insight on this topic. Many surgeons’ practices may be primarily influenced by the teachings of their mentors and their own clinical experience, as opposed to product guides or published recommended practices for cementation. Further research may be done to better understand the influence of surgical technique on the outcomes of aseptic loosening and failure following TKA.
Conclusion
There is clearly no consensus for a preferred cementation technique both within and among manufacturers’ surgeon education materials regarding tibial baseplate cementation during TKA. Efforts may be needed to identify a best-practice cementation technique in an effort to reduce the number of TKA failures associated with aseptic loosening. We recommend urgent action be taken to develop a working group to establish a consensus on the best practices in TKA cementation. At a minimum, acceptable standards and guidelines must be identified. Further research is needed to determine if different cementation techniques may account for differences in outcomes. To date, national registry data and most case series on outcomes focus on the implants rather than techniques. Standardization of all TKA cementation technique instructions should be forthcoming to improve the efficacy of TKAs.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Appendix 1
Websites and date accessed for surgical guides reported.

