Abstract
Objective:
This study aims to examine the iliac vascular space in patients with lumbosacral tuberculosis and to evaluate the feasibility of anterior internal fixation for lumbosacral tuberculosis.
Methods:
A retrospective analysis was performed in 36 patients with lumbosacral tuberculosis. The preoperative three-dimensional computed tomography angiography images were analyzed for anatomical parameters.
Results:
There were large variations among the anterior lumbosacral iliac vessels. Predominantly, the left iliac vein and the right iliac artery formed the two borders of the triangular iliac vascular space in the coronal plane. The mean distance between the iliac vessels straddle point and the inferior endplate of L5 was 15.01 ± 15.08 mm. In the sagittal plane, presacral abscess increased the distance between the posterior iliac vessel and the anterior vertebra. The distances on the left and right sides were 9.94 ± 6.03 and 10.15 ± 5.46 mm, respectively, at the inferior endplate of L5 and were 11.90 ± 6.97 and 11.68 ± 5.52 mm, respectively, at the superior endplate of S1.
Conclusions:
The space on sagittal plane occupied by presacral abscess may push forward the vessels and therefore provide opportunities for anterior internal fixation.
Keywords
Background
Lumbosacral tuberculosis accounts for approximately 2–3% of spinal tuberculosis cases. 1,2 The lumbosacral region has complex anatomy and requires high mechanical stability. Thus, anterior debridement and interbody fusion combined with posterior pedicle screw internal fixation has become the surgical procedure of choice. 3,4 However, this surgical procedure has many disadvantages, such as long operation time, considerable intraoperative blood loss, and major trauma. 5 –7 In recent years, more and more surgeons have chosen one-stage anterior debridement and interbody fusion internal fixation. 8 –10 For example, He and Xu 9 used anterior internal fixation for the treatment of lumbosacral tuberculosis and demonstrated that one-stage anterior surgery was significantly better than combined anterior–posterior surgery in terms of operation time, blood loss, and length of hospital stay. Our group developed the multidirectional self-locking plate (arch plate) for the treatment of lumbosacral spine tuberculosis, and this arch plate was successful used in clinical application. 11
In contrast with degenerative diseases, the inflammation of spinal tuberculosis causes the iliac vessels to adhere to the surrounding tissues, resulting in difficulty in the intraoperative management of dissecting vessels. A triangular iliac vascular space in the anterior lumbosacral spine section, constituted by the iliac vessels crossing from both sides together with the sacral promontory, is commonly recognized as the safe operating region for anterior lumbosacral surgery. 12,13 Therefore, the iliac vascular space is the main region for surgery and placement of fixation in the patients with spinal tuberculosis. Surgeons often select the location of internal fixation from this area by preoperative computed tomography angiography (CTA). However, the size and location of the iliac vascular space in some patients is markedly limited, rendering fixator placement infeasible.
Previous studies on the iliac vascular space only focused on healthy populations and mostly on their anatomical characteristics in the coronal plane. 12 –25 In spinal tuberculosis patients, anterior iliac vessel relaxation can be caused by partial L5 and S1 bone destruction due to tuberculosis along with a decreased lumbosacral angle. 2 The iliac vessels may also be affected by presacral abscess, which may push the vessels forward, possibly forming a space between these vessels and the anterior vertebral body in the sagittal plane. The anterior part of this gap consists of a residual abscess wall segmented from the iliac vessel, which can prevent delayed vascular injury caused by friction between the pulsating iliac vessels and the internal fixator. It can also provide some safe space for internal fixator placement after debridement surgery.
This study retrospectively analyzed preoperative three-dimensional (3-D) CTA data of patients with lumbosacral tuberculosis and measured the relative 3-D anatomical relationship between the local lumbosacral bone and the anterior iliac vascular space. This study may lay an anatomical foundation for the improvement of preoperative CTA assessment for anterior internal fixation in lumbosacral tuberculosis.
Materials and methods
Patients
Thirty-six patients diagnosed with lumbosacral tuberculosis and underwent surgery between 2010 and 2015, at the Department of Orthopedics, Southwest Hospital, Third Military Medical University, were included in this study. Inclusion criteria: (1) age over 18, no gender restriction; (2) the lesion site is mainly located in the L5-S1 region, including cumulative L4, S2, but no additional fixed segments; (3) spinal tuberculosis was diagnosed by clinical, imaging, and laboratory examination and was confirmed by postoperative pathological examination; (4) surgical indications (obvious bone destruction, large paravertebral abscess, obvious segmental instability or neurological dysfunction, etc.); and (5) preoperative abdominal CT angiography has been completed. Exclusion criteria: (1) other lesions associated with spinal canal or spine, such as spinal deformity, tumor or old fracture, and so on; (2) no operation indication; and (3) history of abdominal surgery, which may cause iliac vascular deformation. This research was approved by the Ethics Committee of the First Affiliated Hospital of the Third Military Medical University, People’s Liberation Army (PLA). Written informed consent to participate was obtained from all of the patients. Written informed consent for publication of their clinical details and clinical images was obtained from the patients.
CTA examination
The dual-source spiral CT (Somatom Definition Flash, Siemens Healthcare, Forchheim, Germany) was used to scan the lumbosacral spine and anterior large blood vessels (scan layer thickness, 5 mm; pitch, 1.15; reconstruction thickness, 1.0 mm; overlapping, 30%). Intravenous injection of 100 ml of omnipaque (iohexol 35 g) was performed at the middle of the right elbow at an injection rate of 4 ml/s. Arterial tracking technology was used to set a trigger point in the abdominal aorta, tracking the CT threshold until it reached 150 Hu. The arterial phase scan was started and then delayed 30–40 s for the venous phase. The scan time was 25–30 s in the arterial phase and 60–70 s in the venous phase. All images were treated with maximum-intensity projection, volume rendering technology, and multiplanar reconstruction so that the anterior large blood vessels could be clearly visualized. A SIEMENS dual-source CT postprocessing station was used for image observation and measurement.
Measurements of anatomical parameters
The following anatomical parameters were measured. The level of abdominal aortic bifurcation and inferior vena cava confluence was observed on the 3-D reconstruction image along with the iliac vessel straddle point (triangular iliac vascular space vertex) level. Vessel types were identified on both sides of the L5-S1 areas (Figure 1(a)).
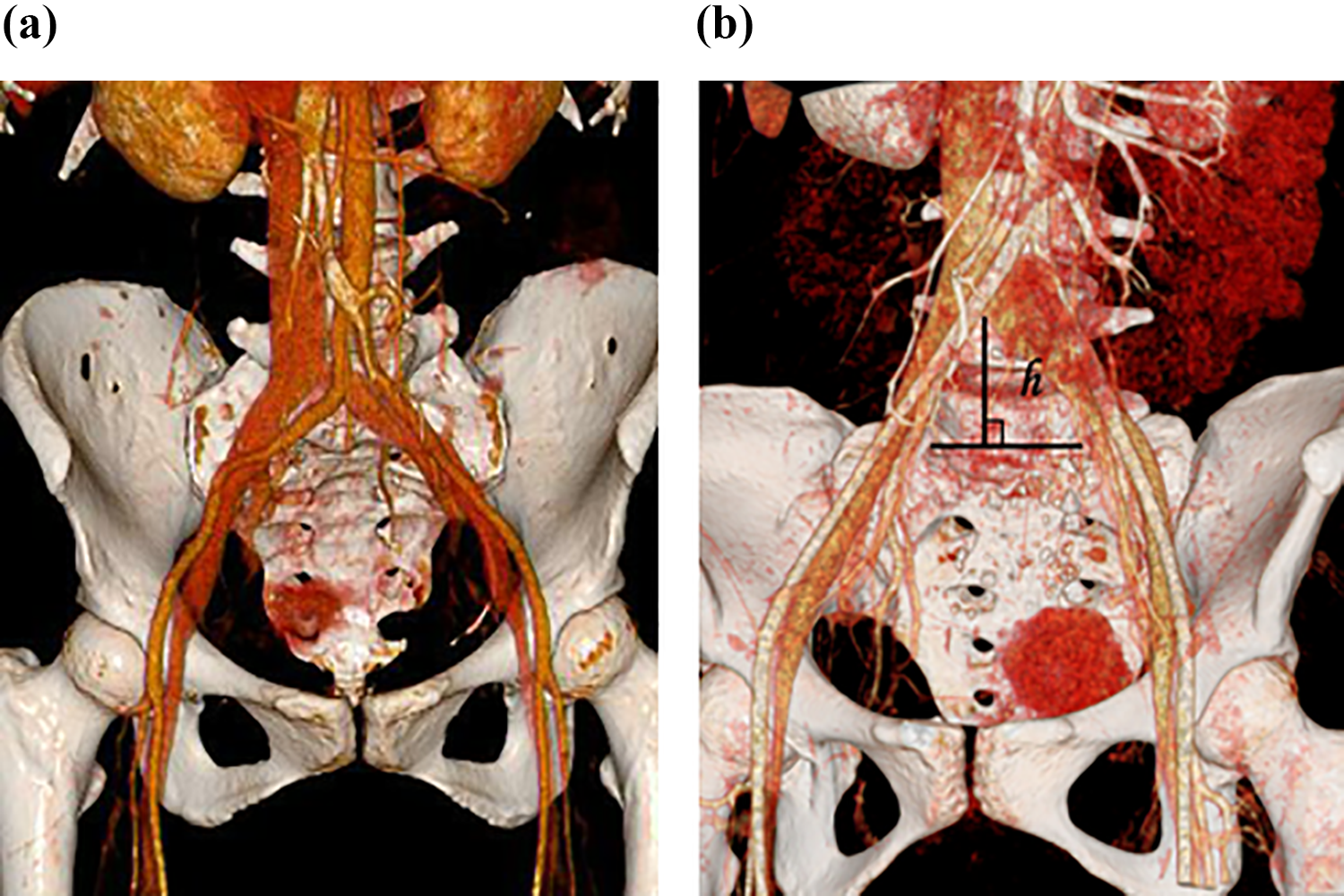
Three-dimensional computed tomography angiography image of lumbosacral tuberculosis. (a) The vessel types in the iliac vascular space and the locations of the vessel bifurcation and confluence and (b) the distance from the iliac vessel straddle point to the inferior endplate of L5 (h).
In the 3-D coronal reconstruction image, the distance (h) was measured from the iliac vessel straddle point to the inferior endplate of L5 (Figure 1(b)). We also measured the distance (l a and l b) from the left and right sides of the iliac vessels to the positive sagittal plane at the inferior endplate of L5 and the superior endplate of S1, respectively (Figure 2(a)), along with the distances (l ab) between the inner edges of both sides of the iliac vessels (Figure 2(b)).

The anatomical parameters of the iliac vascular space (measured on axial native slices). (a) The distance from the left and right sides of the inner edge of outermost iliac vessels to the positive sagittal plane on the coronal plane (l a, l b). (b) The distance between the inner edges of both sides of the outermost iliac vessels on the coronal plane (l ab). (c) The distance between the posterior iliac vessel wall and the anterior vertebral body on the left and right sides on the sagittal plane (L A1, L A2). (d) The sagittal diameter and maximum sagittal diameter of abscesses (the thickness of abscesses, L B and L C).
In the 3-D sagittal reconstruction image, the distances (L A1 and L A2) were measured between the posterior iliac vessel wall on the left and right sides and the anterior vertebral body at the inferior endplate of L5 and the superior endplate of S1, respectively (Figure 2(c)). The sagittal diameter and maximum sagittal diameter of abscesses (the thickness of abscesses, L B and L C) were measured in the sagittal plane at the same level (Figure 2(d)).
Results
Patients’ demographics and clinical characteristics
Our study included 36 patients with lumbosacral tuberculosis, including 14 males and 22 females (average age of 37 years, ranging from 19 years to 68 years). Clinical symptoms mainly included lumbosacral pain and associated neurological disorders. There were 31 cases with presacral abscess and 12 cases with psoas abscess. There were 3 cases with tuberculosis-damaged L5 vertebral body, 1 case with tuberculosis-damaged S1 vertebral body, 32 cases with tuberculosis-damaged L5-S1 region, and 8 cases with multiple vertebral tuberculosis. None of the patients had congenital spinal deformities. Tuberculosis was confirmed by postoperative pathology analysis.
Vessel types in the iliac vascular space
The vessel types in the iliac vascular space were evaluated in patients with lumbosacral tuberculosis. The vessels on either side of the iliac vascular triangle space in the anterior lumbosacral section were the left iliac vein and the right iliac artery (86.11% of cases), while both iliac veins and both iliac arteries accounted for 5.56% and 8.33%, respectively. No left iliac artery or right iliac vein types were found.
Locations of the vessel bifurcation and confluence
The vessel bifurcation and confluence were analyzed in patients with lumbosacral tuberculosis. Abdominal aortic bifurcation was present in 5.56% (2 of 36) of the patients at L3, 5.56% (2 of 36) at L3-L4, 61.11% (22 of 36) at L4, 19.44% (7 of 36) at L4-L5, and 8.33% (3 of 36) at L5. Iliac veins and inferior vena cava confluence occurred in 2.78% (1 of 36) of patients at L3-L4, 16.67% (6 of 36) at L4, 38.89% (14 of 36) at L4-L5, and 41.67% (15 of 36) at L5. There were 8.33% (3 of 36) of patients with the iliac vessel straddle points at L4, 16.67% (6 of 36) at L4-L5, 58.33% (21 of 36) at L5, and 16.67% (6 of 36) at L5-S1 (Figure 3).

Vessel distribution at different levels of the lumbar vertebrae. The abdominal aortic (AA) bifurcation, the inferior vena cava (IVC) confluence, and the iliac vessel straddle points in relation to the level of the lumbar vertebrae were analyzed.
Analysis of anatomical parameters in the L5-S1 region on the coronal and sagittal planes
On the coronal plane, the distance between the iliac vessels straddle point and the inferior endplate of L5 was 15.01 ± 15.08 mm. The anatomical measurement parameters at the inferior endplate of L5 and the superior endplate of S1 (L5-S1 region) are listed in Table 1. At the inferior endplate of L5, the width of the inner edges of the iliac vessels was <15 mm in 16.67% (6 of 36), 15–25 mm in 16.67% (6 of 36), 25–45 mm in 41.67% (15 of 36), and >45 mm in 25.00% (9 of 36) of patients. At the superior endplate of S1, the width of the inner edges of the iliac vessels was <15 mm in 8.33% (3 of 36), 15–25 mm in 8.33% (3 of 36), 25–45 mm in 50.00% (18 of 36), and >45 mm in 33.33% (12 of 36) (Figure 4).
Anatomical parameters in the L5-S1 region on the coronal plane (mm).


The percentages of the widths of the inner edges of the iliac vessels at different levels.
The anatomical parameters of the sagittal plane in the L5-S1 region are listed in Table 2. For the inferior endplate of L5 and the superior endplate of S1, there were 13.89% and 8.33% of patients with a distance <5 mm between the posterior wall of the iliac vasculature and the anterior vertebral body, respectively; 44.44% and 30.56% with a distance of 5–10 mm, respectively; and 41.67% and 61.11% with a distance of >10 mm, respectively (Figure 5). At the same measured level, the sagittal diameter of the abscess was <5 mm in 19.44% and 11.11% of patients, respectively; 5–10 mm in 47.22% and 41.67%, respectively; and >10 mm in 33.33% and 47.22%, respectively. The maximum sagittal diameter of the abscess was <5 mm in 8.33% and 8.33%, respectively; 5–10 mm in 27.78% and 16.67%, respectively; and >10 mm in 63.89% and 75.00% of patients, respectively (Figure 6).
Anatomical parameters in the L5-S1 region on the sagittal plane (mm).


The percentages of the distance between the posterior wall of the iliac vessels and the anterior vertebral body at different levels.
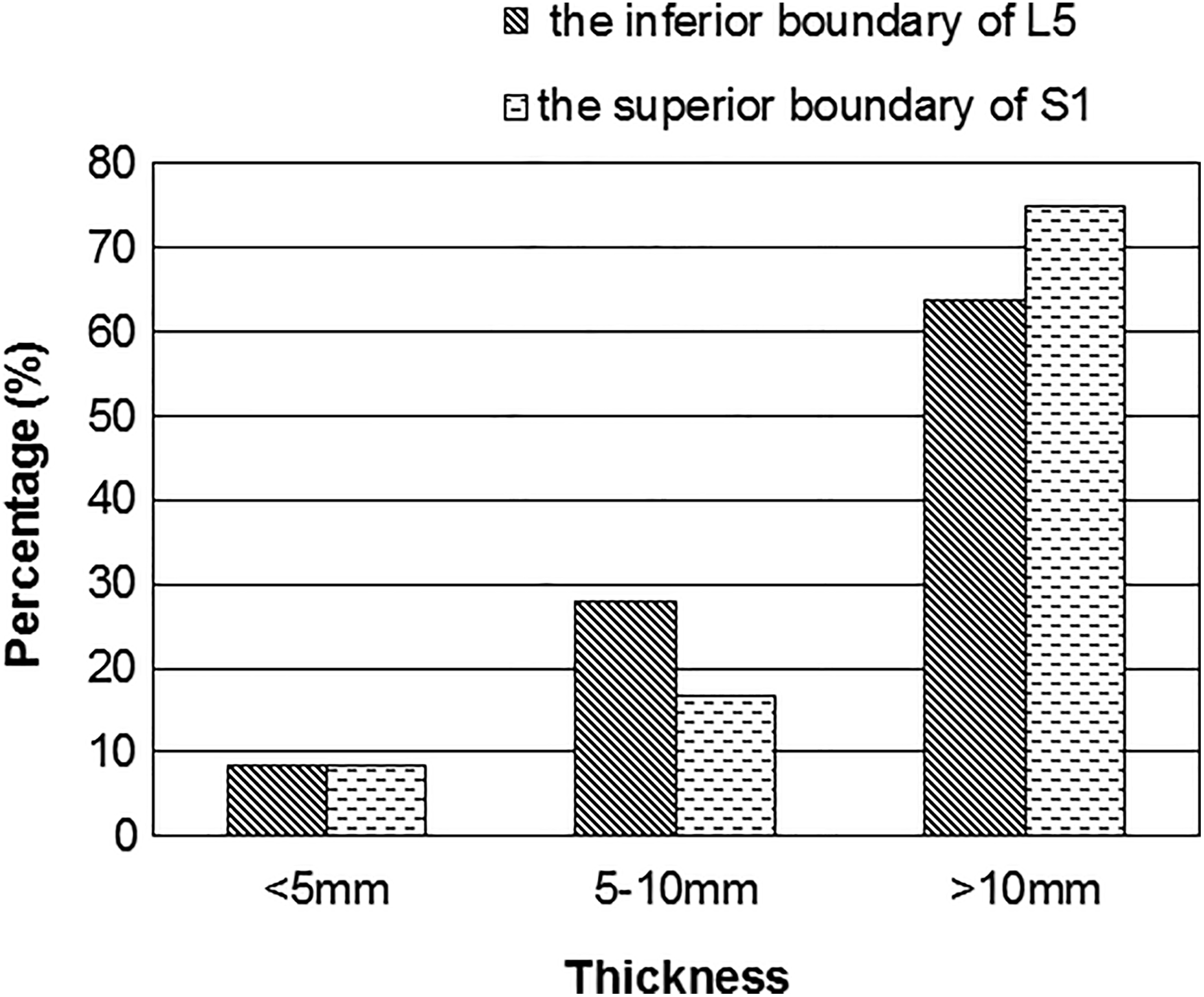
The percentages of the maximum thickness of the abscess at different levels.
Discussion
In recent years, one-stage anterior internal fixation surgery for the treatment of lumbosacral tuberculosis has been investigated. 9,10 Compared with other surgical treatment strategies, the one-stage anterior surgery is advantageous because of clearer operative field, thorough debridement, single incision, less trauma, shorter operation time, less bleeding, shorter hospital stay and recovery time, reduced fusion segments, earlier ambulation, and functional exercise after the surgery with brace protection. 9 However, the anatomical complexity of the lumbosacral region may increase the surgical risk of anterior fixation, especially when important structures such as anterior large vessels are present. The reported anterior lumbosacral surgical complications mainly include vascular injury, retroperitoneal injury, retroperitoneal hematoma, ureteral injury, retrograde ejaculation, and erectile dysfunction. 26,27 Among these complications, the incidence of large vascular injury can be as high as 18.1% 28 –32 with fatal consequences. The anatomical structures of the abdominal iliac vessels may differ between patients with lumbosacral tuberculosis and healthy individuals. Therefore, preoperative evaluation of large abdominal vessels is helpful for understanding the vascular anatomy in the anterior lumbosacral region, thus reducing the risk of vascular injury in patients with lumbosacral tuberculosis.
Many researchers have studied the anatomical structures of large anterior lumbosacral vessels and have proposed the concept of the iliac vascular space. For instance, Tribus and Belanger 19 evaluated this space directly by measuring cadavers. However, because of measurement errors caused by vascular collapse, measurements from cadavers may not truly indicate the prevertebral vascular anatomy. Barrey et al. 12 proposed the concept of vascular window and measured its distance using CTA. In his study, the vascular window was measured in the middle of the L5-S1 disc, without the coverage of L5 and S1 vertebral body for anterior internal fixation. Liu et al. 15 used more detailed anatomical parameters to evaluate the iliac vascular space, such as the distance between the left and right iliac vessels on the inferior endplate of L5 and the superior endplate of S1, and the distance from the vertex of the triangular space to the inferior endplate of L5. In 2005, Inamasu et al. 13 used 3-D-CTA to acquire an anatomical model of large abdominal anterior vessels for the first time. Because of the simultaneous, clear display of the vertebral body positions and their relative anatomical relationships with adjacent large vessels, this method is widely used in preoperative evaluations.
In this study, through comparison between the coronal anatomical parameters of the iliac vascular space of the lumbosacral tuberculosis patients and the published data of the healthy population, we found that the iliac vessel straddle point of the patients with the lumbosacral tuberculosis tended to go down, and the width of the iliac vascular space tended to become smaller (Table 3). This may be affected by vertebral bone destruction, collapse, and iliac vascular relaxation. Our study also showed that the distance from the inner edge of the right iliac vessel on sagittal plane was slightly larger than that of the left side, suggesting that when placing the internal fixator, surgeons may not necessarily need to place the fixator in the middle. For example, positioning slightly to the right side can result in better space, but comprehensive evaluation of bone condition is still needed. In this study, one patient had the iliac vessel straddle point (vertex of the triangle space) at the level of the L5-S1 intervertebral disc, which increases the risk during anterior internal fixation. This patient had combined anterior–posterior surgery. The other 35 patients had iliac vessel straddle points at the L5 vertebrate body or above. There was enough operative space in the L5-S1 region, and thus one-stage anterior debridement and interbody fusion internal fixation surgery was successfully performed.
Comparison of anatomical parameters in the L5-S1 region between lumbosacral tuberculosis patients and the published data of the healthy population on the coronal plane (mm).

a This author measured the L5-S1 vascular window not separately at the inferior boundary of L5 and at the superior boundary of S1.
Previous studies on the iliac vascular space are all based on healthy or degenerative disease groups with iliac vessels adjacent to the vertebral body. 12 –14 However, none of these studies focused on the relative anatomical relationship between the iliac vascular space and the lumbosacral spine on the sagittal plane. Tuberculosis destroys the L5 and S1 bone and decreases the lumbosacral angle. Thus, the anterior iliac vessels can be relaxed. In addition, lumbosacral tuberculosis is usually accompanied by prevertebral abscess. With the development of the disease, abscess enlarged, pushing the anterior soft tissue, ligaments, and blood vessels forward, and forming a space in front of the anterior vertebral body in the sagittal position. This lacuna on the sagittal plane separates the vertebral body from the iliac vessels. During surgery, the abscess can be directly accessed to perform debridement, which provides a satisfactory surgical field and a placement space for internal fixation without the need to pull and peel the iliac vessels excessively. The reserved fibrous connective tissue abscess wall after debridement can also form a type of soft tissue shield between the internal fixation and the iliac vessels.
Our study evaluated the size and location of the lacuna by measuring the distance between the posterior wall of the iliac vasculature and the anterior vertebral body as well as measuring the thickness of the abscess in the L5-S1 region. We found that over 80% patients have more than 5 mm of the thickness of the abscess, over 60% of the patients had more than 10 mm of the distance between the posterior wall of the iliac vasculature and the anterior vertebral body at the superior endplate of S1, and 75% of the patients had more than 10 mm of the maximum thickness of the abscess at the superior endplate of S1. These results suggest that more than 80% patients have enough space to accommodate the internal fixation device on the sagittal plane after anterior surgery, which can avoid possible delayed injury caused by direct friction between pulsating vessels and the device.
Anterior debridement and interbody fusion internal fixation surgery is feasible in most patients and is determined by the triangular iliac vascular space formed by traveling iliac vessels on both sides and the remnant bone position. Because of the large variations in the height of the traveling iliac vessels coupled with the impact of tuberculous lesions, we suggest that for all cases in which one-stage anterior internal fixation for lumbosacral spinal tuberculosis is planned, preoperative 3-D-CTA should be conducted to evaluate the safety of surgery.
Measurements were performed based on CTA images of patients with lumbosacral tuberculosis. The criteria for anterior internal fixation surgery are suggested as follows. (1) The vertex of the triangular iliac vascular space should not be lower than the middle level of L5 and the width of iliac vascular space should be larger than the width of the internal fixator. (2) Patients who met the above criteria can safely receive anterior internal fixation surgery; while for those who do not meet the above criteria, the space on the sagittal plane should be monitored. If this space is significantly greater than the internal fixator’s thickness, anterior internal fixation can be performed.
This study has some limitations. Firstly, the sample size is limited. Secondly, the multidirectional self-locking plate is developed by our group and there may be a certain bias in the choice of surgical approach.
Conclusion
Preoperative 3-D-CTA examination can clearly evaluate the relative anatomical relationship between the iliac vessels and vertebral body. The lumbosacral tuberculosis can affect the iliac vascular space and the space occupied by cold abscess in the sagittal plane may provide more opportunities for internal fixation. Considering the iliac vascular space on the coronal plane and the sagittal plane along with the vertebral body residue, one-stage anterior debridement and interbody fusion internal fixation is preferable as it can reduce surgical trauma in lumbosacral spinal tuberculosis.
Footnotes
Acknowledgment
We thank the data collectors and the study participants of radiology department.
Author contributions
The datasets used and analyzed during the current study are available from FL. WW carried out the study, collected data, and drafted the manuscript. SY collected data and drafted the manuscript. CW drafted the manuscript and helped to polish the language. JL helped to polish the language and collected data. XL collected data. ZZ participated in its design and surgery and helped to draft the manuscript. JX participated in its design and surgery. FL conceived of the study, participated in its design, and provided theoretical and technical support. All authors read and approved the final manuscript. WW, SY, and CW contributed equally to this work and should be considered first coauthors. Other authors contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grant from Clinical Innovation Found of the Third Military Medical University (no. swh2011015).
