Abstract
Purpose:
The purpose of this study was to evaluate local and systemic pathology in a murine model of ischemia–reperfusion (I/R) injury induced by long-term application of a tourniquet on the hind limbs and to assess the protective effects of edaravone, a potent systemic scavenger of free radicals, using this model.
Methods:
Sixty C57BL6 mice were divided in two groups, with one group receiving a 3 mg/kg intraperitoneal injection of edaravone and the other group receiving an identical amount of saline 30 min before ischemia under deep anesthesia. The left thigh of each animal was constricted for 4 h with a 4.5-oz. orthodontic rubber band to induce ischemia; 4 h was the critical duration for skeletal muscles. After ischemia, specimens of skeletal muscles, both kidneys, and plasma were collected at 0, 2, 12, 24, 48, and 72 h. Injury to the skeletal muscles and vacuolar degeneration of the kidneys were histologically assessed. Additionally, apoptosis of skeletal muscle cells was assessed by analysis of caspase 3/7 activity and TUNEL staining. Plasma tumor necrosis factor (TNF)-α levels were measured using an enzyme-linked immunosorbent assay kit.
Results:
Skeletal muscles exhibited prominent injury of myofibers at 12 h after I/R injury, with clear upregulation of plasma TNF-α expression and histologic evidence of tubular dysfunction of the kidneys. Plasma TNF-α levels declined and histologic renal damage was ameliorated in edaravone-treated mice, but treatment did not protect skeletal muscle following ischemia for 4 h. Nonetheless, compared with group S, expression of the apoptosis marker caspase 3/7 was significantly inhibited in the skeletal hind limb muscles of Ed-group mice affected by reperfusion injury following ischemia for 4 h.
Conclusion:
The present study demonstrated that edaravone is a potentially useful drug for systemic or local treatment of reperfusion injury resulting from long-term ischemia.
Introduction
In ischemia–reperfusion (I/R) injury, anaerobic metabolism products that accumulate during ischemia induce the migration of leukocytes and release of inflammatory factors in the reperfusion phase, ultimately leading to the destruction of tissues and organs, with serious general complications. 1,2 Although the pathophysiology of I/R injury is unclear, several reports have suggested that proinflammatory cytokines such as tumor necrosis factor (TNF)-α and chemical mediators such as free radicals are involved, and these chemical mediators not only accelerate injury of local tissue in I/R injury, they also damage other vital organs. 2 –5 Skeletal muscles are frequently exposed to I/R injury in a variety of clinical settings, including limb trauma, surgery involving the extremities, and revascularization for the peripheral arterial disease. 6 –8 As a result of I/R injury (especially in the lower extremities, which contain a large amount of skeletal muscle), serious systemic conditions including the acute renal failure can be caused by secreted chemical mediators derived from ischemic skeletal muscles, as represented by crush syndrome. 3,7
Although the pathophysiologic details of I/R injury have not been fully elucidated due to the complex interactions between inflammatory and immunologic signaling pathways, free radicals are thought to be one of the leading mediators.
2,6,9
Free radicals, which are produced during the early phases of ischemia, cause direct injury to cells via peroxidation of the cell membrane; indirect injury can result from the migration of inflammatory cells and expression of proinflammatory cytokines.
8,10
It is reasonable to hypothesize that the blockade of free radicals could be an effective treatment for I/R injury in a variety of tissues, including skeletal muscle. Researchers have reported that antioxidants such as vitamin E,
Our previous study showed that edaravone (3-methy-1-pheny1-2-pyrazolin-5-one), a potent systemic scavenger of free radicals, protects the hind limb skeletal muscles of mice against I/R injury induced by application of a tourniquet for 1.5 h. 15 Edaravone inhibits both nonenzymatic lipid peroxidation and the lipoxygenase pathway, in addition to exhibiting potent antioxidant effects against I/R injury-induced vascular endothelial cell injury, delayed neuronal death, brain edema, and concomitant neurologic deficits. Edaravone was approved for use in treating acute brain infarctions in Japan in 2002; in 2015, it was approved for treating amyotrophic lateral sclerosis based on its neuroprotective effects. 16 –18 Cumulative evidence gained from I/R injury models of various organs suggests that apoptosis is the major contributor to I/R injury-induced cell death, rather than necrosis caused by ischemia. Also, apoptosis as well as necrosis have been associated with the mechanism of cell death in the pathophysiology of I/R injury of skeletal muscle. 19,20
The purposes of present study were as follows: (1) to evaluate local and systemic pathology in a mouse model of I/R injury induced by long-term application of a tourniquet on the hind limbs; (2) to evaluate the effectiveness of edaravone for treating reperfusion injury following 4 h of ischemia, as edaravone demonstrated a protective effect against I/R injury following 1.5 h of ischemia 15 ; and (3) to assess the role of apoptosis in the mechanism of I/R injury in skeletal muscle and the effect of edaravone in preventing apoptosis in injured tissues.
Materials and methods
Subjects
Sixty C57BL6 mice (8–10 weeks old, male, 22–28 g; SLC, Hamamatsu, Japan) were used for all experiments in this study. The animals were housed in a temperature-controlled environment and maintained on a 12-h light–dark cycle, with food and water provided ad libitum. The experimental protocol was approved by the Mie University Committee of Animal Research.
Experimental model
All 60 mice were deeply anesthetized with an intraperitoneal injection of pentobarbital sodium (0.05 mg/g body weight). The left proximal thigh of each mouse was bound with 4.5-oz. orthodontic rubber bands (ORBs; American Orthodontics, Sheboygan, Wisconsin, USA), which were introduced by Crawford et al. for inducing reproducible I/R injury in murine hind limbs. 21 The period of ischemia was 4 h to induce critical damage to the skeletal muscles, after which the rubber bands were removed for reperfusion at 0, 2, 12, 24, 48, or 72 h (n = 5 in each group). Under deep anesthesia, the mice were fixed on a surgical table, and blood was then collected by cardiac puncture for isolation of plasma. Thereafter, specimens of skeletal muscle (including the gastrocnemius (GC) and tibialis anterior (TA) muscles) and both kidneys were carefully removed using a surgical microscope.
Drug administration
Edaravone was provided by Mitsubishi Tanabe Pharma Corporation (Tokyo, Japan) and dissolved in 1 N NaOH that was titrated to pH 7.4 with 1 N HCL to prepare a final concentration of 0.3 mg/ml. The mice were randomly assigned into two groups, with one group receiving a 3 mg/kg edaravone (group Ed) in the peritoneal cavity 30 min before application of the rubber band. The other 30 mice received the same amount of saline (group S).
Histology
Samples of GC and TA skeletal muscle and kidneys harvested from each animal were immediately fixed in 4% paraformaldehyde for 24 h at 4°C overnight and then embedded in paraffin. The specimens were cut transversely at 4 µm using a microtome and then stained with hematoxylin and eosin (HE). HE staining was performed after dewaxing in xylene and rehydration in graded methanol (99–70% v/v) followed by distilled water. The stained sections were examined on a microscope equipped with a digital camera (BX50; Olympus, Tokyo, Japan).
Muscle histologic injury severity score
The absolute injury score for each muscle sample was determined using a method similar to that reported by McCormack et al. 22 Every myocyte in 15 (GC) or 10 (TA) photographic fields was scored. The muscle injury score was expressed as a percentage, obtained by dividing the number of injured myocytes by the total number of myocytes scored within all fields.
Histologic evaluation of kidney injury
Cortical areas of the kidneys were evaluated for vacuolar degeneration of the tubular cells. A percentage was calculated by dividing the number of tubular cells with the vacuolar degeneration by the total number of tubular cells. 23
Terminal deoxynucleotidyltransferase-mediated 2′-deoxyuridine-5′-triphosphate nick-end labeling (TUNEL) assay
TUNEL was performed to detect nicking of DNA in skeletal muscles subjected to I/R injury for 12 h. Briefly, sections prepared on slides were treated with proteinase K and hydrogen peroxide and then incubated with terminal deoxynucleotidyltransferase and 5-bromo-2′-deoxyuridine-5′-triphosphate. The sections were counterstained with hematoxylin. Apoptosis was assessed by determining the number of TUNEL-positive cells in 10 random fields at 400× magnification.
Caspase 3/7 activity assay
Caspase 3/7 activity in GC samples was assessed using the end-point apo-one homogeneous caspase-3/7 assay (Promega KK, Madison, Wisconsin, USA). Frozen GC samples were pulverized in liquid nitrogen using a Cryopress (Microtech, Chiba, Japan) and then homogenized in radioimmunoprecipitation assay buffer. The entire volume of homogenate was centrifuged at 13,000 r/min for 10 min at 4°C, and the supernatant was used for the assay. A 1:1 ratio of caspase reagent to sample supernatant was used for the assay, and the samples were incubated at room temperature for 4 h. Fluorescence was measured at an excitation wavelength of 485 ± 20 nm and emission wavelength of 530 ± 20 nm on a FluoStar Galaxy fluorescence spectrometer (BMG LABTECH GmbH, Ortenberg, Germany). 24
Quantitation of plasma TNF-α
Collected blood was immediately centrifuged at 1500 r/min for 10 min at 4°C to separate the plasma, which was then preserved at −80°C. TNF-α levels were measured using an enzyme-linked immunosorbent assay kit (BD Bioscience, Franklin Lakes, New Jersey, USA).
Statistical analyses
Stat View 5.0 for Windows (SAS Institute, Cary, North Carolina, USA) was used for statistical analyses. Data were analyzed using the Mann–Whitney U-test or by calculating Spearman’s coefficient of rank correlation. Values of p < 0.05 were deemed statistically significant.
Results
Histology revealed prominent injury of skeletal muscle myofibers 4 h after I/R injury with systemic inflammation and renal dysfunction
No mice died during the experimental procedures of this study, despite exposure to reperfusion following 4 h of ischemia, which was the critical period of time for skeletal muscle injury. Histologically, the muscle fibers from immediately to 2 h after I/R injury were normal in the mice of group S, with well-defined borders, consistent texture, and uniformity throughout the fibers, without holes or breaks. By contrast, the myofibers of both the GC and TA samples exhibited prominent destruction of muscle fibers, with edema and inflammatory cell infiltration at 12 h after reperfusion (Figure 1, Table 1). These pathologic findings increased in severity over time and were significantly more pronounced at 3 days. Indeed, few myofibers with normal architecture could be found at that time (Figure 1, Table 1). Furthermore, plasma TNF-α levels tended to be high with 8.0 ± 0.9 pg/ml at 2 h and were predominantly higher with 1152.2 ± 557.4 pg/ml at 12 h compared with 5.3 ± 3.2 pg/ml immediately after I/R injury. The levels declined by 48 h after reperfusion but were still higher at 72 h after I/R injury compared with immediately after injury (Figure 2). Moreover, histologic analyses of the kidneys demonstrated tubular dysfunction at 12 h after I/R injury induced by tourniquet application to the hind limbs (Figure 3).
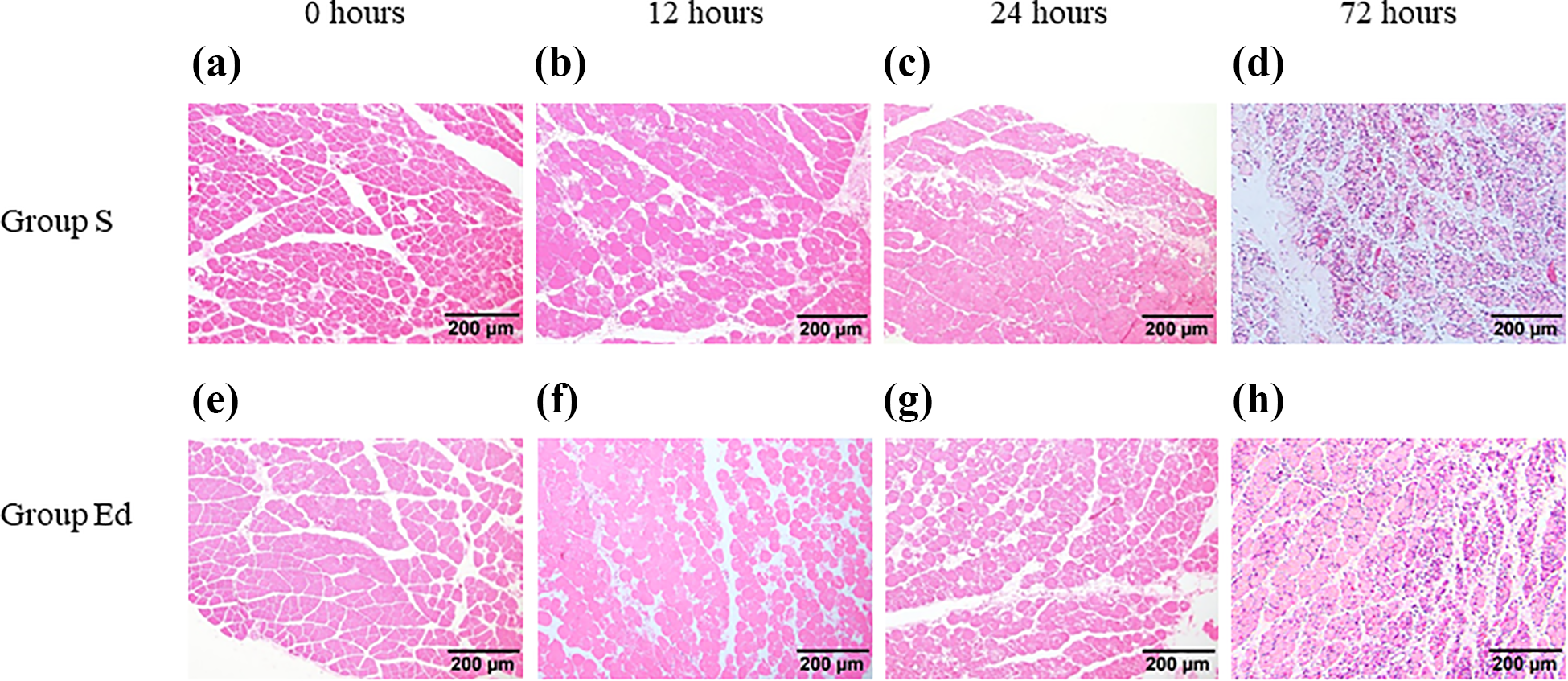
Histologic findings of the gastrocnemius muscles at (a) and (e) 0 h, (b) and (f) 12 h, (c) and (g) 24 h, and (d) and (h) 72 h after the ischemia for 4 h. (a) to (d): group injected saline (group S) and (e) to (h): group injected with edaravone (group Ed). Histologic findings showed no morphologic changes at 0 h, but prominent broken myocytes and inflammatory cell infiltration were observed at 12 and 24 h in both group S and Ed. In addition, almost myofibers were broken with the inflammatory cell infiltration at 72 h, regardless of the administration of edaravone. Scale bar = 200 μm.

Plasma TNF-α levels were significantly higher at 24 h after reperfusion, after which the levels gradually decreased. At 72 h after reperfusion, the levels were still significantly higher in control mice, however. Group S: group injected with saline and group E: group injected with edaravone. *p < 0.05 (group S vs. group Ed at each time point). TNF-α: tumor necrosis factor-α.

(a) Histologic analyses showed vacuolar degeneration of kidney tubular cells (arrows) occurring from 24 h after reperfusion. The tubular degeneration was predominantly 48 h later in both groups injected with saline (group S) and with edaravone (group Ed). (b) Beginning at 48 h, significantly less tubular degeneration of the kidneys was observed in mice of group Ed compared with group S. *p < 0.05 (group S vs. group Ed at each time point). Scale bar = 100 μm.
Histological assessment of percentage of the injured myofibers in the muscles of the hindlimb at each time point after the ischemia–reperfusion injury for 4 h.a

GC: gastrocnemius; TA: tibialis anterior; group S: group injected with saline; group Ed: group injected with edaravone.
a The values represent the mean ± standard deviation of muscle injury score at each time point after ischemia–reperfusion injury. There existed no significant differences of muscle injury score between group S and Ed in both GC and TA at each time point.
Edaravone significantly inhibited systemic inflammation and renal damage but did not protect skeletal muscle exposed to reperfusion injury following 4 h of ischemia
Levels of TNF-α in plasma were significantly higher with 200.6 ± 255.2 pg/ml in the mice of group Ed at 12 h compared with 4.7 ± 1.6 and 3.1 ± 4.3 pg/ml at 0 and 2 h after I/R injury, respectively, like those in group S. There were no differences in plasma TNF-α levels between group S and group Ed at 0 and 2 h. However, plasma TNF-α levels were significantly lower in mice injected with edaravone at each time point from 12 h onward, compared with mice injected with saline. The suppression of plasma TNF-α levels was maintained for at least 72 h after I/R injury (Figure 2). Simultaneously, vacuolar degeneration of kidney tubular cells was observed in both groups 12 h after I/R injury (Figure 3); edaravone did not prevent this degeneration, although the percentage of tubular cells exhibiting sings of vacuolar degeneration began to decline at 24 h after I/R injury in edaravone-treated mice. Similarly, analyses of samples of myofibers of both GC and TA muscles subjected to ischemia indicated that application of edaravone did not provide significant protection against reperfusion injury. Indeed, there were no significant differences of the muscle injury score between group S and group Ed in the samples from both GC and TA at each time point (Figure 1, Table 1).
Expression of the apoptosis marker caspase 3/7 was upregulated during the early phase of I/R injury and significantly inhibited in the skeletal muscles of murine hind limbs subjected to reperfusion injury following 4 h of ischemia
Caspase 3/7 activity increased in both groups immediately after I/R injury, peaking at 24 h in group S and 48 h in group Ed (Figure 4). However, histologic evidence of damage to the skeletal muscles was not apparent until 12 h after reperfusion (Figure 1). TUNEL was performed on the sections of skeletal muscle at 12 h after reperfusion to determine the localization of programmed cells. TUNEL-positive skeletal muscle cells with multiple peripheral nuclei and intermyofiber cells were observed. Additionally, compared with untreated controls, pretreatment with edaravone resulted in significantly lower caspase 3/7 activity at 12 and 24 h (Figure 4) as well as fewer number of TUNEL-positive cells at 12 h after I/R injury (40.0 ± 8.0 and 15.0 ± 5.3 in group S and group Ed, respectively).

(a) Expression of the apoptosis marker caspase 3/7 in the gastrocunemius muscles increased in the early stage of reperfusion following 4 h of ischemia in the hind limbs of both group injected with saline (group S) and edaravone (group Ed); the activity was significantly lower in edaravone-treated mice beginning at 12 h after reperfusion. *p < 0.05 (group S vs. group Ed at each time point). (b) TUNEL-positive skeletal muscle cells with multiple peripheral nuclei and intermyofiber cells (arrows) at 12 h after the ischemia–reperfusion injury. Scale bar = 100 μm.
Discussion
The extent and degree of I/R injury are multifactorial but depend primarily on the critical ischemia time, which is defined as the maximum time period that a tissue can tolerate ischemia and still remain viable. The critical time is reported to be between 3 h and 4 h for skeletal muscles, depending on the tissue type. 25 After ischemia, anaerobic metabolism products and breakdown products begin to accumulate in the tissues, and the release of these products can lead to both serious systemic complications and further local tissue damage. Most research in this field has focused on the heart, kidney, liver, brain, and intestines, as skeletal muscle is not considered a vital organ. 2,4,5,26,27 However, interest in skeletal muscle I/R injury resulting in serious complications subsequent to significant extremity injuries sustained in war or natural disasters has recently increased. Crush syndrome is a well-known serious complication with the acute renal failure resulting from I/R injury to skeletal muscles, and new methods to treat this condition have been expected. 3,7 Accordingly, we initially evaluated the local and systemic effects in skeletal muscles subjected to reperfusion following 4 h of ischemia, which was the critical ischemia time. Furthermore, we used the ORBs for the long-term ischemia to the skeletal muscles of the murine hind limb, because the tension of ORBs was shown to be more continuous than the hemorrhoid alligator band, which has been widely used for studying the tourniquet-induced ischemia in the mice. 15 As expected, significant damage to local tissue and adverse systemic effects were observed with a little differences, such as progressive morphologic destruction of the skeletal muscles, prominent increases in plasma TNF-α levels, and predominant increases in tubular dysfunction 12 h after reperfusion.
None of the several clinical strategies currently available have been broadly successful in attenuating the detrimental effects of I/R injury. Although the pathophysiology of I/R injury has not been fully elucidated due to the complex interactions between inflammatory and immunologic signaling pathways, it is clear that the accumulation of free radicals and anaerobic metabolites during ischemia induces the migration of white blood cells and the release of inflammatory factors such as interleukins and free radicals in the reperfusion phase. 28 –30 Therefore, one strategy for treating I/R injury that is likely to be considered is scavenging of free radicals, because I/R injury can activate pathways that generate reactive oxygen species capable of degrading the cell membrane. Several reports have demonstrated that scavenging of free radicals is effective at reducing the severity of I/R injury. Cheng et al. reported that pterostilbene, which was an analog of resveratrol and previously shown to exhibit free radical-scavenging and antioxidant properties in various tissues, also protects skeletal muscles against I/R injury. 31 Ozyurt et al. described how caffeic acid phenethyl ester (CAPE) protects skeletal muscles from reperfusion injury and how this protective effect is likely associated with CAPE’s free radical-scavenging activity. 32
Edaravone should also be useful for minimizing the severity of I/R injury to skeletal muscles, as our previous study showed that edaravone protects skeletal muscles against reperfusion injury after 1.5 h of ischemia. 15 Unfortunately, a protective effect of edaravone against reperfusion injury to skeletal muscles was not observed in the present study following 4 h of ischemia, which is the critical ischemia time for skeletal muscles. However, the mice in group Ed exhibited a significant decrease in plasma TNF-α levels and a histologically lower grade of renal degeneration compared with the mice in group S. Edaravone treatment induced scavenging of free radicals but did not directly suppress production of inflammatory cytokines. However, several reports have demonstrated that administration of edaravone inhibits systemic or local inflammation associated with I/R injury in various tissues, suggesting that edaravone indirectly suppresses inflammation by protecting against tissue damage via scavenging of free radicals. 16,33 –35 In the present study, pretreatment with edaravone suppressed systemic inflammation due to I/R injury induced by tourniquet application on the hind limbs for 4 h, although it did not protect the skeletal muscles exposed to ischemia for the critical time period against damage. These data suggest that edaravone secondarily inhibits systemic inflammation in skeletal muscle subjected to ischemia for long periods by scavenging free radicals from injured vital organs (including the kidneys) that have accumulated due to anaerobic metabolism.
Although edaravone with the dose of 3 mg/kg did not exhibit a protective effect against reperfusion injury in skeletal muscle subjected to 4 h of ischemia, we believe that edaravone does have some effect, as our previous study demonstrated that edaravone at 3 mg/kg protects skeletal muscles subjected to 1.5 h of ischemia. Accordingly, we focused on and evaluated the mechanism of cell death in skeletal muscle with I/R injury. Evidence from various I/R model studies conducted over the last decade indicates that necrosis is not solely responsible for I/R injury-induced cell death and that apoptosis might be the initial cause of cell death. Other researchers have examined the mechanism of cell death resulting from I/R injury to skeletal muscles. Wang et al. reported that both necrosis and apoptosis occurred in I/R injured gracilis muscles in rats subjected to 24 h of reperfusion following 4 h of ischemia, 19 although necrosis was previously considered to be a solo mechanism of cell death in the skeletal muscles after the ischemia. The present study also found apoptotic cells in the skeletal muscles beginning in the early stage of reperfusion, and furthermore, edaravone retarded the development of TUNEL-positive nuclei and significantly inhibited caspase 3/7 activity. On the other hand, the apoptosis was likely to be a main factor in the cell death at the late phase of the I/R injury to the skeletal muscles, since caspase 3/7 activity was predominantly decreased in the group S and not inhibited by the administration of the edaravone at 48 h later after the I/R injury. These findings are important because apoptosis could be inhibited by specific agents to produce a therapeutic benefit. Indeed, edaravone significantly lowered levels of plasma TNF-α, which is a well-known death ligand, even though only one dose was used for pretreatment in this study.
It is a limitation of the present study that only one pretreatment of edaravone with the dose of 3 mg/kg was evaluated for protective effects. Previous studies showed that the edaravone at high dose (10 mg/kg) were daily administered with result in the protective effect for muscular tissues in murine models of cardiac hypertrophy or motor neuron disease. 36,37 In addition, the other organs except the kidneys and the contents in the plasma derived from the skeletal muscles were not evaluated in this study. Nonetheless, it is conceivable that multiple administrations or increased dose of this drug would provide a greater protective effect against systemic inflammation and damage to local tissues after the ischemia for long periods.
In conclusion, although edaravone did not protect against I/R injury to skeletal muscles subjected to 4 h of ischemia, it was protective against I/R injury following 1.5 h of ischemia in our previous study. 15 Edaravone pretreatment did however result in significantly reduced plasma TNF-α levels and lower kidney histologic damage, indicating that scavenging of free radicals is useful for minimizing systemic inflammation after I/R injury to hind limb skeletal muscle tissue. Additionally, this study showed that apoptosis is involved in the mechanism of cell death in I/R injury of skeletal muscles. Administration of edaravone significantly suppressed the activity of caspase 3/7 in skeletal muscles, although the muscles were ultimately destroyed despite the one-dose pretreatment with edaravone.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
