Abstract
Background:
Deep infection after routine elective orthopedic procedures can be catastrophic, leading to further surgery, loss of limb, disability, and risk of mortality. Ring-fencing elective orthopedic ward has been shown to significantly reduce the incidence of all postoperative infections especially with methicillin-resistant Staphylococcus aureus (MRSA). Our hospital’s current MRSA screening is a four-site MRSA swabs.
Objectives:
This study evaluates the possibility of reducing the number of MRSA swab sites as part of a quality improvement project.
Study design and methods:
Patients on the waiting list for elective orthopedic procedure in our trust who had an MRSA-positive swab from either four sites were analyzed over the time period from January 2012 to December 2014. Those without swabs from all four areas (nose, throat, axilla, and groin) were excluded. Positive swabs of different regions were recorded and compared.
Results:
There were 138 MRSA-positive patients, giving an incidence of 31 per 10,000 screen/year over that time period. Some patients (n = 31, 22.5%) had a positive swab in more than one site. The positive sites were as follows: nose (69.60%, n = 96), groin (26.10%, n = 36), throat (25.30%, n = 35), and axilla (8.70%, n = 12). In our cohort, we would miss a significant proportion of positive patients if we change it to a two swab screening policy (26.8% for nose and axilla combination; 18.10% for nose and groin combination; and 15.20% for nose and throat). However, we would only miss 2.2% of cases for a nose, groin, and throat three-swab policy. There were also 11 instances, where a previously negative site become positive in the next swab.
Conclusion:
A three-swab combination of nasal, throat, and groin swabs improves pickup rate of MRSA significantly compared to a two-swab policy and misses only 2.2% compared to a four-swab policy. Axilla swabbing does not make a significant difference to the results. Based on this study, the policy has now been changed from a four-swab to three-swab screening in our trust. This has now been audited four times and they were all negative. This has helped to reduce cost in terms of staff time and resources. We would not recommend screening only the previous positive site for the next repeat screening swabs as there is an 8% chance of missing MRSA carrier status.
Keywords
Introduction
Surgical site infection (SSI) has been identified as one of the most important preventable sources of morbidity and mortality associated with any surgical treatment. 1 In orthopedic surgery, a significant amount of resources is invested to prevent SSI, such as custom-built theatre suite, laminar flow, and ring-fenced elective ward. 2 However, SSI is still a common complication after any surgery. 3 Staphylococcus aureus is one of the most common organisms in SSI and up to 25% S. aureus strain isolated from nosocomial infections are methicillin-resistant Staphylococcus aureus (MRSA). 4 MRSA is a significant contributor to prolonged hospital stay, poor clinical outcome, and increased healthcare costs among surgical patients. 5 Any intervention to decrease the prevalence of MRSA infection, therefore, has a huge clinical benefit for patients and reduces the healthcare cost.
Methicillin resistance in S. aureus isolated from patients was first reported in 1961, 6 and since then it has been a growing concern for medical practitioners in both medical and surgical specialties. Resistance to methicillin is the result of the acquisition of mecA gene segment, a mobile genetic element that encodes a modified penicillin-binding protein, PBP 2a, 7 this protein has transpeptidase activity for peptidoglycan synthesis and decreased affinity for β-lactam antibiotics, thereby allowing S. aureus to synthesize peptidoglycans in the presence of such drugs. Once MRSA adheres to an implant, it reduces its metabolic rate and begins to secrete a glycocalyx layer that protects the bacterium from antibiotics and cell-mediated immunity. Bacteria that produce such biofilms are up to 100 times more resistant to antibiotic treatment than their nonbiofilm-producing counterpart. 8
Epidemiologic studies have demonstrated that most cases of SSI are caused by strains of S. aureus that are brought into the hospital environment by patients themselves. 1 MRSA-colonized patients are at higher risk of infection than non-MRSA colonized patients, 9 this was observed by many other studies, such as Kim et al., 1 who found that a significantly higher rate of SSI in MRSA carrier patients compared to noncarrier; similarly, Davis et al. 10 also found that 19–25% of patients colonized by MRSA subsequently developed MRSA infection. Therefore, it makes sense to have a screening program to identify MRSA-colonized patients. Effective screening for MRSA colonization or infection on admission to the hospital could greatly reduce the risk of infection to both colonized and noncolonized patients through timely identification and implementation of targeted infection control measures. 11
Between 1992 and 2003, data from National nosocomial infection surveillance system, United States, show that the prevalence of MRSA increased from 32% to 64% of all isolated nosocomial pathogens found on patients in hospital intensive care unit, representing 3.1% increase in MRSA prevalence per year. 12 During the same period, United Kingdom and Canada also noted a similar increase in the incidence of MRSA bacteremia. However, after the improvement in the infection control protocol, United States, United Kingdom, and Canada have all seen significant reduction in MRSA bacteremia since 2005. 13 UK studies have shown a significant reduction in MRSA incidence as a result of adoption of MRSA screening 14 ; therefore, MRSA screening should be continued, and in the absence of a screening and eradication program, the number of MRSA cases is likely to rise again. MRSA screening is reported to be a cost-effective intervention, and Lee et al. 15 evaluated culture-based screening program and demonstrated that a screening method would be cost-effective even if the MRSA prevalence was as low as 1% and decolonization success as low as 25%.
For patients undergoing surgery requiring a prosthetic implant, nasal colonization with S. aureus is the most important independent risk factor for the development of an SSI. 16 Although nose is the most common site of MRSA colonization, extra-nasal sites of MRSA colonization have been documented, and these sites include inguinal, rectal, axillary areas as well as throat and skin. 17 Therefore, it is possible that nasal carriage may simply be a marker for skin colonization, and extra-nasal sites of colonization would not be identified and decolonized adequately with treatment protocols established based on nasal screening alone.
Most centers in United Kingdom have an MRSA screening policy but the protocol differs from each center. Since the introduction of ring-fenced elective orthopedic wards in our trust, the policy has been screening of four sites (nose, throat, groin, and axilla) for all elective orthopedic patients. The purpose of this project was to evaluate whether we can safely reduce the number of sites swabbed for MRSA screening in elective orthopedic patients. We also wanted to test the hypothesis whether nasal swabbing alone is sufficient for MRSA screening of elective orthopedic patients, which is routine practice in non-UK centers.
Material and methods
Patients on the waiting list for elective orthopedic procedure in our trust who had an MRSA-positive swab from either of four sites were analyzed over the time period from January 2012 to December 2014. Those without swabs from all four areas (nose, throat, groin, and axilla) were excluded. Positive swabs of different regions were recorded and compared. Only first positive swab was used in counting patient numbers, that is, further positive swabs of same patients were only counted once.
Our protocol
All patients on the waiting list for elective surgery in our orthopedic units are required to attend the preassessment clinic, where swabs were taken from nose, throat, axilla, and groin and tested for MRSA. Only patients tested negative on MRSA swabs are admitted to our ring-fenced elective orthopedic ward. Patients who were tested positive were classed as being colonized with MRSA and eradication treatment with mupirocin ointment and chlorhexidine wash was prescribed and they were listed back for surgery only when three sets of consecutive MRSA swabs done at a weekly interval were negative.
The methodology used in our unit for culturing MRSA is by inoculating patient’s swabs onto commercially purchased chromogenic agar. Swabs from different sites were inoculated separately for culture testing. Chromogenic agar used was Oxoid Brilliance 2 (Thermo Fisher Scientific, UK). The agar is then incubated for 18–24 h, after this time, the biomedical scientist will check every plate for every site for distinct and specified colonies of bacteria. If the bacterial colonies are dark blue, then these possible MRSA colonies would undergo further biochemical tests and antibiotic susceptibility tests. Antibiotic susceptibility testing was performed using Kirby–Bauer method according to the European Society of Clinical Microbiology and Infectious Diseases protocol. If the S. aureus colony is methicillin resistant, then a report is issued stating that MRSA is isolated from that specific site. MRSA-positive patients then undergo decolonization with chlorhexidine and mupirocin and culture are then repeated after 7 days.
Results
There were 6076 patients (24,304 swabs, 4 for each patient) who had MRSA swabs taken during the 3-year time period and among them, we had 138 MRSA-positive patients (52 males and 86 females), giving an incidence of 31 per 10,000 screen/year over that time period. Patient demographics are shown in Table 1.
Patients demographics.

THR: total hip replacement; TKR: total knee replacement.
The positive sites were as follows: nose (n = 96, 69.60%), groin ( n = 36, 26.10%), throat (n = 35, 25.30%), and axilla (n = 12, 8.70%), of which 31 patients (22.5%) had positive swab in more than one site. The lowest number of cases missed would be 2.2%, which is based on a combination of nose, groin, and throat three-swab policy (Table 2).
Sensitivity of different screening sites and combinations for detection of MRSA.
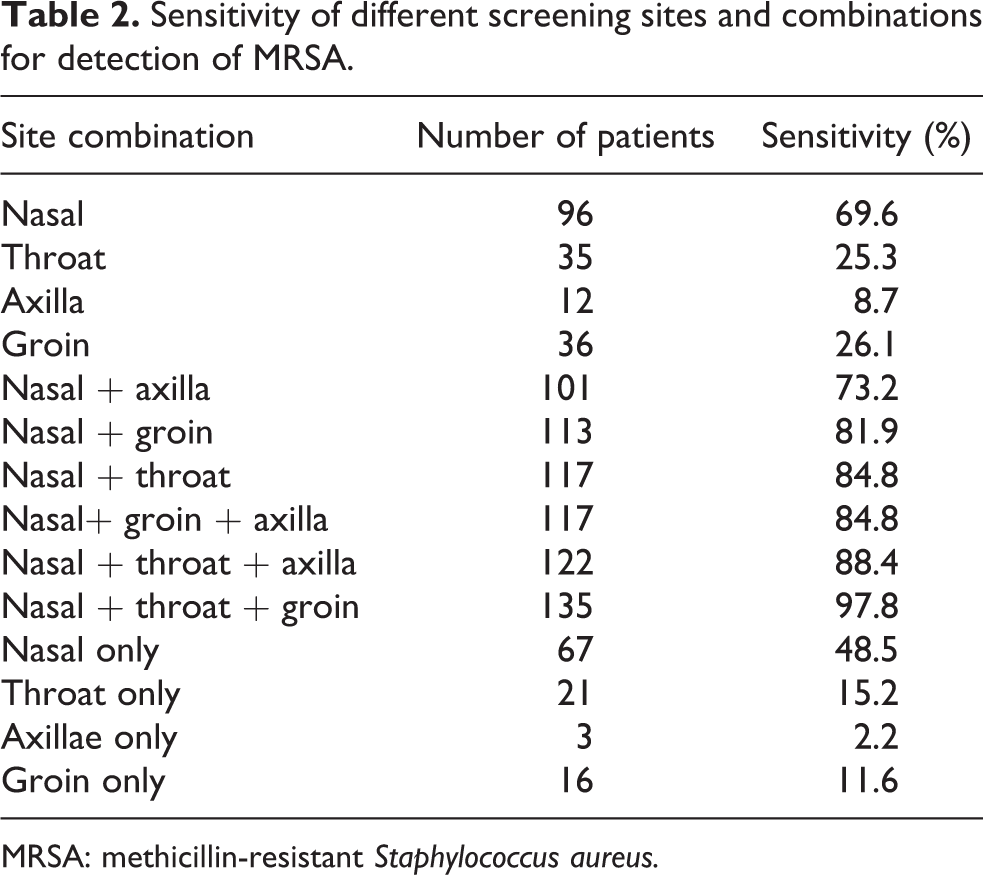
MRSA: methicillin-resistant Staphylococcus aureus.
We also evaluated whether it was safe to just swab the previous positive site after decolonization. There were 11 instances (8%), where a previously negative site had become positive in the next swab.
Analyzing the 138 MRSA-positive patients retrospectively in our pathology database, we found that 107 of these patients had previous MRSA swabs taken during the previous hospitalization and the time period varied widely from 16 years to 2 months. Forty of these patients had at least one positive MRSA swab and 67 patients were MRSA negative.
The result of decolonization and eventual outcome of MRSA-positive patients is depicted in Figures 1 and 2. Nine patients did not have any MRSA-negative swabs, and in those nine patients, three were lost to follow-up, two patients had surgery despite being MRSA positive, one patient died, and three patients are still on the waiting list. Three patients who had only one negative MRSA swabs are also awaiting surgery. Thirty patients did not undergo surgery, among them, 3 were classed as not fit, 7 decided not to have surgery, and 20 patients are on waiting list. Fifty-six patients who did not had surgery within 1 month of three sets of negative swabs, seven among them had a further positive swab while waiting for surgery, mean duration until further positive swab was 107 days (n = 21, 63, 73, 90, 100, 157, 245). Our study also showed that it took from 21 days to 11 months to have a set of three negative swabs.

Result and outcome after decolonization in our cohort.

Surgery after decolonization in our cohort.
Based on our result, we changed our screening policy in October 2016 from four swabs to three swabs (nose, throat, and groin) and we audited to make sure that we are not missing any MRSA-positive patient. During the audit period, all patients admitted in our elective ward who were MRSA negative based on three swabs were swabbed again with four site swabs (nose, throat, groin, and axilla) for MRSA, and we did not identify any positive MRSA in our audit period. To increase the validity of our assumption, we repeated the audit three more times (four cycle in total), and during each of this period, there were no positive MRSA patients on repeat four swab screening; therefore, we conclude with confidence that our three-swab policy is robust and safe enough to implement a change in our standard policy. There was no change in the SSI rate in our unit during the study period.
The cost of MRSA screening via culture route per patients in our trust was £23.2 for four swabs and £17.4 for three swabs, by successfully changing the policy from four swabs to three swabs per patient, the lab processing charge has decreased. In terms of saving, we estimate that it would save £5.80 × (6076 patients/3 years) = £11,747 per year.
Discussion
Our study has shown that in the absence of a screening program, a significant number of our patients would have been MRSA colonized risking both themselves and other patients in our ward to MRSA infection. Mortality and morbidity of MRSA bacteremia is significantly higher compared to methicillin-sensitive S. aureus bacteremia. 18 The study by Nixon et al. 19 has shown that the morbidity of MRSA-infected patients is considerable, and it resulted in extended hospital stay of additional 50 days for proximal femur fracture patients and additional surgical and medical treatment. The cost of care for such patients greatly exceeds the cost of introducing a policy to prevent MRSA infection and introducing a surveillance and treatment policy is a cost-effective way of reducing its incidence. Nirvani et al. 20 in their study also found that MRSA infection or colonization contributed to an increased length of hospital stay; 88 days compared to 11 days on average for non-MRSA patients, therefore significantly increasing the healthcare cost of managing such patient.
Rao et al. 21 reported using universal screening and selective decolonization of S. aureus in patients undergoing elective total joint replacement and found that the rate of SSI decreased from 2.6% to 1.5%. Robotham et al. 22 used a mathematical model to calculate cost-effectiveness of admission MRSA screening policy in English hospital and found to be cost-effective in a high-risk specialty, such as trauma and orthopedics. Kavanagh et al. 23 concluded based on their study that surveillance followed by targeted intervention is effective in reducing SSIs. Thus, the literature suggests that the implementation of an institution’s wide-screening program using a screening test such as polymerase chain reaction (PCR) or culture would lead to a significant reduction in postoperative SSI. Similarly, Goyal et al. 24 did a literature review and suggested that patients should be screened and MRSA-positive patients should be treated before surgical admission in order to reduce the risk of SSI. In conclusion, there is an overall unanimity that there should be a screening program to identify MRSA colonized patients but where different institution differs is what sort of screening protocol should be implemented.
Our result concurs with the finding of NHS Scotland MRSA screening pathfinder program, which found that nasal swabbing alone on the standard routine screening method only detected 69% of true positive case as assessed against the gold standard measure (all body site swabs on chromogenic agar plus broth enrichment combined). 11 The Scottish study recommends clinical risk assessment (CRA) based screening; however, our study (we would have missed 62% of patients if only high-risk group screened) as well as other studies have highlighted that MRSA is not restricted to high-risk group only and selective screening will miss a high number of cases. 25 Although CRA-based screening appears appealing in reducing the cost of screening program but studies have shown that epidemiology of MRSA has changed over a period of time. The last two decades have shown an increase in community-acquired MRSA, a subpopulation of MRSA with unique antibiotic resistance, virulence, and pathogenic capability. 26 This subset of MRSA tends to affect young and otherwise healthy patients and is increasingly found in athletes, children in day care, homeless people, and military recruit 27 and is likely that these subgroups of patients will be missed on CRA-based screening.
For screening program, there should be an effective remedy for the problem identified and as shown in our study (Figure 1), 129 patients (94%) become MRSA negative after having treatment with mupirocin nasal ointment and chlorhexidine wash. Harbarth et al. 28 also showed that the most effective treatment regimen for MRSA colonization is a combination of intranasal muporicin and chlorhexidine soap baths, which led to 86% reduction of SSI compared to control group.
We believe based on our study that three sites should be swabbed for MRSA screening, nose, throat, and groin. We realize that this method will only provide a snapshot of patient MRSA status at that time on a particular site, for example, a patient could have MRSA on their foot/in their ear, and the usual nose, throat, and groin site could be negative, so one could only conclusively rule out MRSA if a patient has a full body screen but a sensible balance has to be reached between identifying MRSA colonization and healthcare resources employed to achieve this. We do not recommend nasal swabbing on its own as it will miss significant proportion of colonized patients and our recommendation is also supported by the study done by Senn et al. 29 who showed that only 62% of MRSA will be identified by nasal PCR but including nose, throat, and groin increases sensitivity to 99%, and therefore, they recommend three site swabs.
There can be an argument that we should have a universal decolonization of all our patients, and the study by Huang et al. 30 has shown the benefit in reducing MRSA bloodstream infection compared to screening and decolonization but it is worth noticing that their screening protocol had only nasal swabbing, thus potentially missing a good number of carrier and such benefits are unlikely to be realized with units having three-site screening protocol for MRSA, and therefore, we believe it is unnecessary to subject all patients to a medical intervention (MRSA decolonization), which perhaps they did not need in first place.
The value of our study is in the fact that based on our finding, our hospital has changed the MRSA screening protocol and axilla swabs are no longer taken. This has been audited four times for quality control and we did not find any MRSA positives missed through our three-swab policy. This has resulted in reducing the cost of MRSA screening by approximately £11,747 per year based on an average number of patients screened in orthopedic department. It has also streamlined the care by reducing unnecessary additional swabbing and reduced the time and error of nursing staff but those cost could not be quantified in real term. We have also noticed in our study that a previously negative site can become positive even after decolonization treatment, and therefore, we will strongly recommend that repeat screening should be of all three sites, as just screening only the previous positive site for the next repeat screening swabs, there is an 8% chance of missing MRSA carrier status.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
