Abstract
Aggressive fibromatosis is a benign, locally invasive fibroblastic proliferation that can cause compressive effects on adjacent structures. The primary cure of this disease rests in wide excision of the tumor. Unfortunately even when surgical margins are clear of tumor, recurrence rates are high. Postoperative radiotherapy is indicated following surgical excision. We present a 13-year-old girl who had been operated for the intraspinal mass in upper thoracic spine and paraparesis with thoracic limited laminectomy and excision of the tumor mass elsewhere. The histopathological examination was reported to be aggressive fibromatosis. After 2 years, she presented again with 1-year duration of progressive deformity in the upper thoracic spine and weakness of both lower limbs. Focal kyphosis at T4–T5 was measuring 68°. Magnetic resonance imaging (MRI) showed recurrent tumor involvement of posterior elements of T2–T5 and paravertebral soft tissues with signal changes in the cord at T2–T5 vertebral levels with focal kyphosis and internal gibbus. She underwent posterior spinal revision decompression with internal gibbectomy and instrumented fusion. The histopathology showed features suggestive of aggressive fibromatosis. After wound healing at 2 weeks, she underwent 3-D conformal radiotherapy, based on the preoperative tumor extent on MRI (dose of 45 Gy in 25 fractions over 5 weeks). She had normal neurology at 2-year follow-up and was tumor free on MRI. Hence, aggressive fibromatosis can recur following successful surgical wide excision. Multilevel thoracic laminectomy in growing children can cause progressive spinal deformity and neurological deficits. Operative treatment of recurrent tumor involves en bloc excision with instrumented fusion followed by local radiotherapy. This is the first pediatric recurrent spinal fibromatosis reported with successful treatment as per author’s knowledge.
Introduction
Aggressive fibromatosis are benign, locally invasive fibroblastic proliferation that can cause compressive effects on adjacent structures. They have the capacity for recurrence and may be uncontrollable. They do not metastasize and hence are benign aggressive tumors. 1 They originate from the deep muscle layers or intramuscular planes and manifest clinically as rapidly expanding soft tissue mass that can be uncomfortable and painful because of compressive effects on adjacent muscular, vascular, and neural structures.The primary cure rests in wide excision of the tumor. Unfortunately even when surgical margins are clear of tumor, recurrence rates are high. Postoperative radiotherapy is indicated following surgical excision. We hereby present the first pediatric case report of recurrent aggressive fibromatosis with progressive thoracic kyphosis and neurological deficits treated with radical surgical resection, instrumented fusion, and postoperative radiotherapy.
Case report
A 13-year-old girl was operated for tumor involving the spinous process of upper thoracic vertebra and paraparesis with thoracic laminectomy T2–T5 and excision of spinous process tumor elsewhere (Figure 1(a) and (b)). She recovered neurology completely in 6 months after surgery and was walking independently. Histopathological examination revealed aggressive fibromatosis. After 1 year, she started developing upper back pain, progressive focal thoracic kyphosis, and weakness of both lower limbs. She presented at 15 years of age with progressive kyphosis, spastic paraparesis in lower limbs, and wheelchair bound. Neurological examination demonstrated grade 2–3 strength in both lower limbs and hypoesthesia was noted below T5. There was no history of bowel or bladder dysfunction.
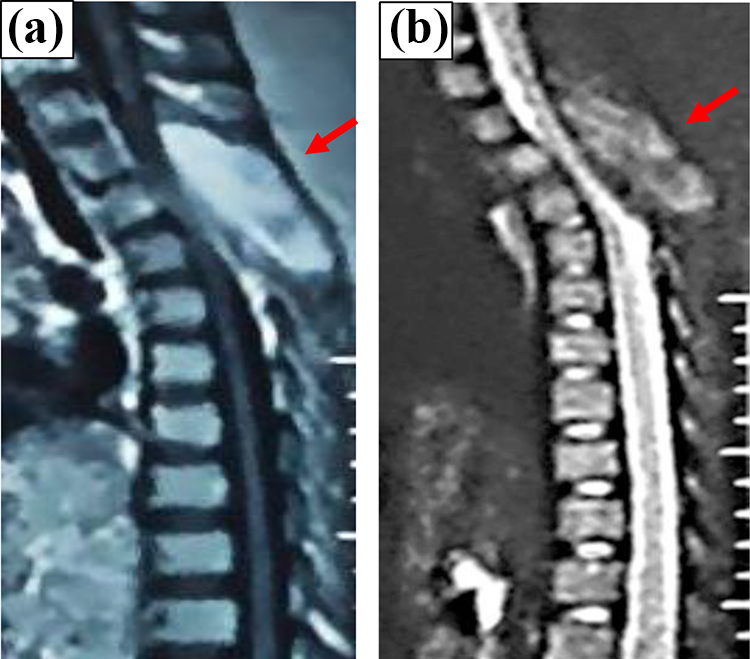
(a) and (b) MRI T1 and T2 images of thoracic spine taken before the primary surgery show tumor involving the spinous process with spinal cord compression. MRI: magnetic resonance imaging.
Radiographs demonstrated focal kyphosis at T4–T5 with angle measuring 68° and magnetic resonance imaging showed recurrent tumor involvement of posterior vertebral elements, costochondral processes, and paravertebral soft tissues with severe spinal cord compression with signal changes from T2 to T5 (Figure 2(a), (b), and (c)).

(a) Plain radiograph showing angular kyphotic deformity of 68°. (b) MRI T2 sagittal image showing recurrent lesion with cord compression and (c) MRI axial image showing the cord pushed toward one side with clear bone involvement. MRI: magnetic resonance imaging.
Radical surgical resection of the tumor with spinal cord decompression and kyphosis correction with instrumented fusion T1–T8 was performed. Postoperative radiograph showed good kyphosis correction and sagittal balance (Figure 3(a) and (b)). Histopathological examination revealed bundles of spindle cells arranged in fascicles with chronic inflammation, necrosis, atypia, and dense fibrocollagenous stroma suggestive of recurrent aggressive fibromatosis (Figure 4). After the complete wound healing and suture removal, she underwent external radiotherapy by 3-D conformal technique to a dose of 45-Gy (25 fractions, 5 days a week, over 5 weeks).
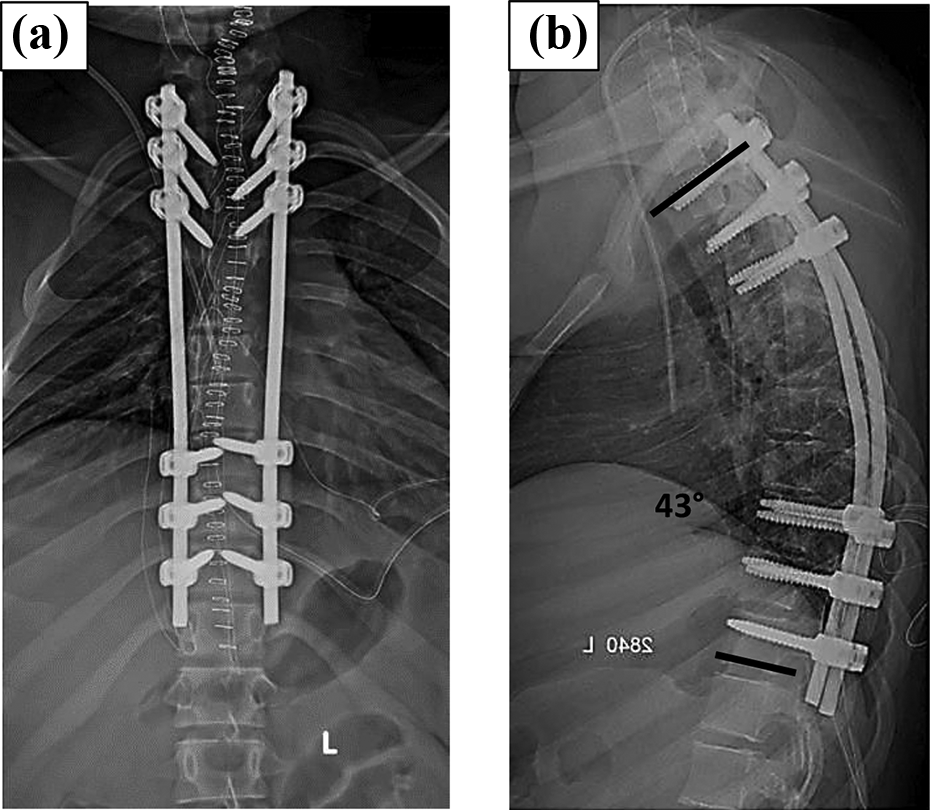
Plain radiograph showing posterior spinal instrumented fusion with kyphosis correction to near normal thoracic kyphosis.

Histopathological picture with bundles of spindle cells arranged in fascicles and dense fibrocollagenous stroma.
At 2-year follow-up, her neurological recovery was complete with aggressive physiotherapy and she could walk independently. Plain radiograph taken showed good maintenance of normal thoracic kyphosis with instrumented fusion and repeat magnetic resonance imaging taken at the last follow-up showed no evidence of tumor recurrence (Figure 5(a), (b), and (c)).
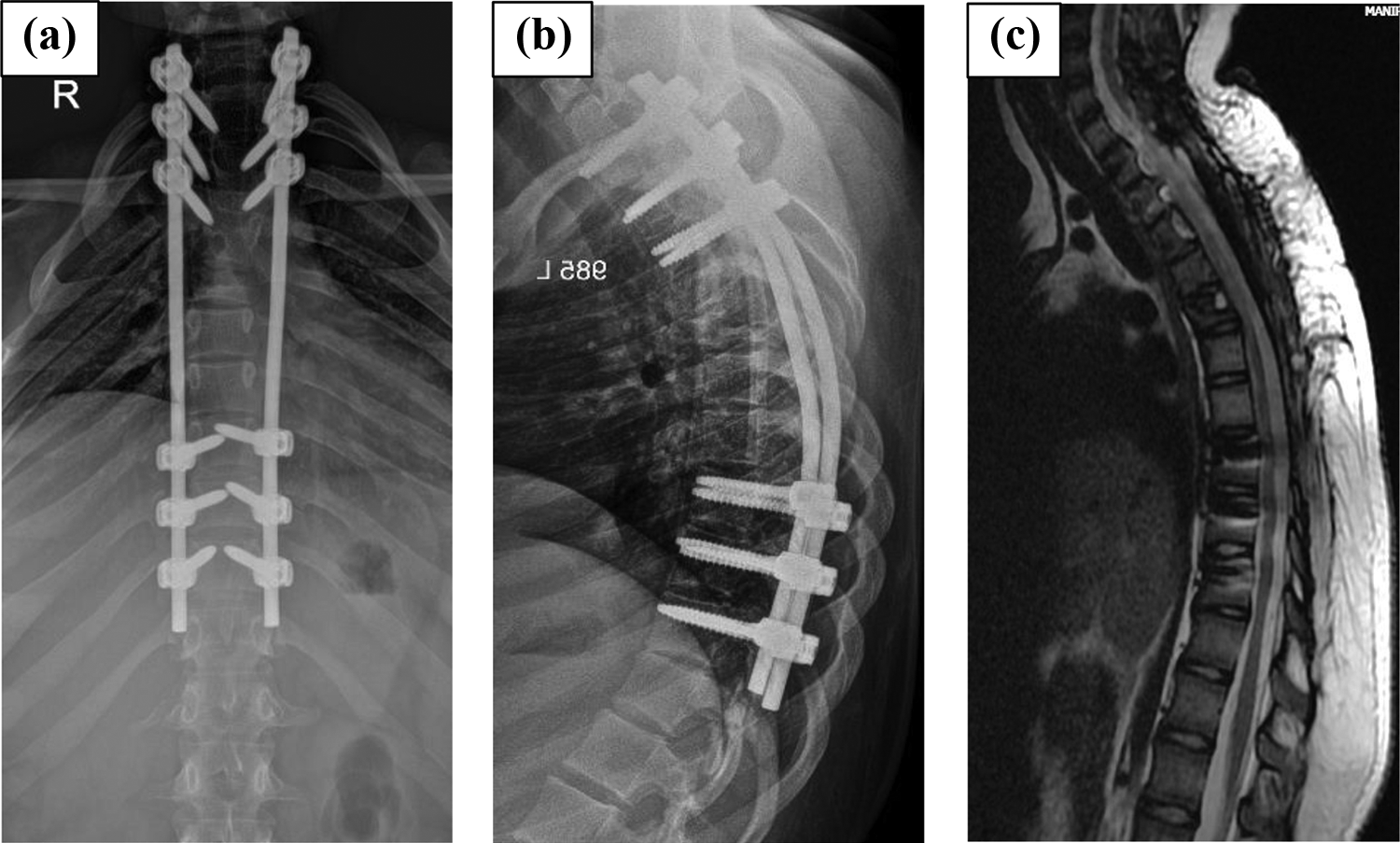
At the end of 2-year follow-up, (a) and (b) plain radiographs showing good maintenance of normal thoracic kyphosis with instrumented fusion and (c) MRI with no recurrence of tumor. MRI: magnetic resonance imaging.
Ethics and consent
Institute ethical committee approval was sought and informed consent taken from the patient.
Discussion
Aggressive fibromatosis is a locally infiltrative tumor that originates from facial planes of soft tissues such as muscle, subcutaneous tissue, and neurovascular structures but does not metastasize. 1 Its locally destructive and invasive behavior was similar to that of low-grade fibrosarcomas. Their overall incidence is 0.4–1%. The reported incidence of fibromatosis is approximately two to four individuals per 1 million per year. 2 Although it often occurs sporadically, trauma secondary to surgical procedures has been implicated as an etiology. 3 Reitamo et al. showed that 80% of fibromatosis tumors occur in females, of whom 50% are within 30–50 years of age. 4
The prefix “aggressive” was preferred because it gives better impression of tumor biology with its extraordinary local morbid behavior. 1 Pathologically, these tumors show a characteristic mass of interwoven spindle cells in bundles with varying amounts of collagen found within their matrix, with occasional sarcolemmic giant cells. 5
Fibromatosis tumors can often be morphologically indistinguishable from various growths such as fibrosarcoma, neurofibroma, lymphoma, granulomatous inflammation, reactive fibroblastic proliferations, and desmoplastic fibroma of bone, myxoma, and fibrosarcomatous transformation of fibromatosis. 6 Although fibromatosis demonstrates rapid invasiveness likened to that of fibrosarcoma, the lack of high mitotic counts, atypical nuclei, and predominance of collagen are some distinguishing features. Confusion still remains and some consider the aggressive form to be a low-grade fibrosarcoma. 7 When compared with reactive fibrosis, fibromatosis exhibits a less variable growth pattern. A key diagnostic characteristic of fibromatosis tumors is the presence of somatic β-catenin or Adenomatous polyposis coli (APC) gene mutations leading to intranuclear accumulation of β-catenin. 6 Somatic β-catenin or APC gene mutations have been reported in more than 74% of the deep fibromatosis. 1
The symptoms associated with these tumors are few. Nearly all patients present with soft tissue mass. Pain is less frequent and occurs approximately in 30% of patients from the mass, which usually occurs when the tumor compromises joint or muscle mobility. 8,9 The propensity for slow growth and invasion of contiguous structures makes the tumor less morbid unless they encroach on vital structures. 5,10 It was noticed that desmoid tumors show varying rates of growth, however, the growth rate of desmoids seems to be independent of the site of the tumor, and the hormonal status of involved women affects the rate of growth. 11 Also, recurrence has been shown not to be related to age, gender, or site. 12
The management of desmoid tumors remains controversial. Management of aggressive fibromatosis must be planned based on a multidisciplinary approach. Surgical management is considered the preferred treatment method. 13 The overall recurrence rate with surgical excision alone was 40%. 5 Dahn et al. reported recurrence rates of 70% with marginal excision and 8% with radical excision. 8 Although surgery remains the mainstay of therapy, options include observation, radiotherapy, chemotherapy, hormonal and anti-inflammatory therapy, or a combination of all. Because of their rarity, high-level evidence of efficacy does not exist, and treatment recommendations are based on retrospective reviews and expert opinions. Decision-making is further complicated by the seemingly variable natural history of this disease. 14
The treatment of recurrent disease is repeat resection because it results in cure rates similar to those achieved with primary resection. 5,15 Aggressive fibromatosis tends to infiltrate adjacent muscles microscopically, which cannot be determined in the operative field. Rock et al. 9 reported that a higher tendency for recurrence was evident in females over 30 years of age. Recurrent tumors are known to have a more aggressive nature, and hence, our patient had rapidly progressive kyphosis and neurological deficits. Moreover, in a revision surgery, it is very difficult to distinguish between scar tissue and tumor, which may result in inadequate safety margin for excision. 16 Nuyttens et al. 17 reported that revision surgery supplemented with radiation therapy results in better local control than surgery alone.
Aggressive fibromatosis is a radio responsive tumor. 16 Most experts suggest that when a wide surgical margin is not achieved or when the tumor is unresectable, or in revision surgery for recurrent tumors postoperative radiotherapy should be administered. Greenberg et al. 18 reported an 89% rate of relapse-free survival for radiotherapy combined with surgery. Goy et al. 19 reported that patients with positive margins who underwent adjuvant radiotherapy and surgery improved local control to 78%, in contrast to 32% with surgery alone. Jelinek et al. 20 reported an 81% local control rate at 5 years for patients who received postoperative radiotherapy in comparison with 53% who were treated with surgery alone.
In our case, we delivered external radiotherapy immediately following surgical wound healing because of the known aggressive recurrent behavior of tumor and difficulty in delineating the tumor margin intraoperatively. Many authors have reported that doses in the range of 50–60 Gy would be adequate to control microscopic disease without causing much morbidity. In the reported patient, compromised spinal cord function preoperatively was a limiting factor to deliver doses ≥45 Gy, for the fear of radiation myelopathy. 21 It is reported that doses ≥45 Gy are sufficient to achieve long-term disease control with least risk for toxicity. 22 We delivered a dose of 45 Gy (25 fractions, 5 days a week for 5 weeks). Postirradiation myelopathy and fibrosis at operative site is a possible complication if doses exceed > 45 Gy near spinal cord. 21
Conclusion
We herewith present the first reported pediatric case in the literature of recurrent aggressive fibromatosis of spine presenting with progressive deformity and neurological deficits. Surgical excision with negative surgical margins and no functional impairment is highly difficult in revision spinal surgeries, and hence, it is recommended for adjuvant external radiotherapy. The dose of radiotherapy needs to be titrated to be curative to the tumor and within the tolerance limits of the spinal cord.
Limitations
Strong conclusion cannot be done with single case.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
