Abstract
Purpose:
Choosing a therapeutic cell source for osteochondral repair remains a challenge. The present study investigated coculturing mesenchymal stem cells (MSCs) from different sources to provide an improved therapeutic cell option for osteochondral repair.
Methods:
Dutch and Japanese white rabbits were used in this study, the first for isolating MSCs and the second for creating an osteochondral model in the medial femoral condyle. The 26 rabbit knees were divided randomly into four groups: control (n = 6), bone marrow-derived MSCs (BMSCs) (n = 7), synovial tissue MSCs (SMSCs) (n = 7), and cocultured MSCs (n = 6). Tissue repair was assessed using the Fortier scale, and colony-forming assay was performed.
Results:
At different cell densities, cocultured and SMSCs formed larger colonies than BMSCs, indicating their high proliferative potential. After 2 months, complete filling of the defect with smooth surface regularity was detected in the cocultured MSC group, although there was no significant difference among the therapeutic groups macroscopically. Also, tissue repair was histologically better in the cocultured MSC group than in the control and SMSC groups, due to repair of the subchondral bone and coverage with hyaline cartilage. Additionally, toluidine blue and collagen-II staining intensity in the repaired tissue was better in the cocultured MSC group than in the remaining groups.
Conclusion:
Our results suggest that cocultured MSCs are a suitable option for the regeneration capability of osteochondral defects due to their enhanced osteochondrogenic potential.
Introduction
The complexity of osteochondral lesions remains a great challenge for clinicians due to the demand for cartilage, bone, and bone–cartilage interface repair. 1 The results of traditional surgical reparative treatments, such as drilling, microfracture, and grafting, remain unsatisfactory due to inferior fibrous tissue repair, subchondral sclerosis, cysts, and poor integration with surrounding tissue, leading to long-term deterioration of the repaired tissue. 2
Autologous chondrocyte implantation is the first promising cell-based therapy option for addressing cartilage defects, 3 although limitations remain, especially for defects including subchondral bone, which influences the mechanical function of cartilage. Other limitations comprise potential donor site morbidity, dedifferentiation of chondrocytes during expansion, additional surgical intervention for cartilage biopsy, and declining properties of chondrocytes from osteoarthritic joints. 4 Recently, mesenchymal stem cells (MSCs) have been used for their combined ability to reconstruct cartilage and subchondral bone due to their potential for multilineage differentiation and in vitro expansion. These cells are easily accessible from different sources, such as the bone marrow-derived MSCs (BMSCs), synovial tissue MSCs (SMSCs) and adipose tissue, and exhibit different phenotypes. 5
The choice of which MSCs to use for osteochondral repair remains a clinical challenge, due to the variability of the properties of MSCs from different sources and search for an ideal treatment strategy. The bone marrow is the most famous source of MSCs, which represent 0.001–0.01% of the total population of nucleated cells. 6 In 2001, De Bari et al. succeeded in isolating MSCs from human synovial tissue by harvesting them arthroscopically with a relatively low degree of invasiveness. 7 SMSCs have a greater proliferation capacity, limited senescence with superior chondrogenic and inferior osteogenic potentials, compared with BMSCs which revealed high osteogenic and less chondrogenic potentials. 8
Recently, several studies have revealed that the coculture of articular chondrocytes (ACs) with BMSCs can support the redifferentiation of ACs and the production of more cartilage matrix; however, the limited number of available chondrocytes is a disadvantage, especially for large defects. 9,10 Indeed, due to the low number of BMSCs in bone marrow aspirates, adipose-derived stem cells have been cocultured with either normal or osteoarthritic chondrocytes, with the aim of improving the cartilage matrix consistency. 11,12
To compensate for the aforementioned limitations, the combination of MSCs from different sources may improve osteochondrogenic characteristics of the cells for osteochondral repair; this method has not previously studied. Thus, the aim of the current study was to explore the potential of SMSCs with BMSCs in a coculturing system (1:1), rather than to one MSC type independently, as a method to enhance the osteochondrogenic potency for in vivo repair of the osteochondral defect.
Materials and methods
All procedures were performed according to the guidelines for animal experimentation and were approved by the ethical committee of research facilities for laboratory animal science (Institute of Biomedical and Health Sciences, Hiroshima University, Japan).
Isolation and expansion of BMSCs and SMSCs
Briefly, according to a previously established protocol for the isolation of BMSCs, 13 a 5 ml blood sample was harvested with 1 ml of heparin (1000 units/1 ml) (Mochida Pharmaceutical Co. Ltd, Japan) from the bone marrow of Dutch rabbits (18–21 weeks old) by penetrating the cortex of the posterior surface of the iliac crest using an 18-gauge needle. The sample was centrifuged for 5 min at 420 g, and the resulting supernatant, including heparin and fat, was discarded. The extract was resuspended in 2 ml of Dulbecco’s Modified Eagle Medium (DMEM) (Wako Pure Chemical Industries Ltd, Osaka, Japan) containing 10% deactivated fetal bovine serum (FBS) (Sigma-Aldrich, St. Louis, Missouri, USA) and 1% antibiotics (penicillin, streptomycin, and Amphotericin B) (Nacalai Tesque, Inc., Kyoto, Japan). Then, 2 ml of the extract suspension was suspended in 8 ml of culture medium and seeded on 10-cm2 tissue culture dishes (BD Falcon™; BD Biosciences, Franklin Lakes, New Jersey, USA). The dishes were incubated for 4 weeks in a humidified atmosphere with 5% Co2 at 37°C. The medium was changed after 7 days to remove nonadherent cells and changed every 2 days thereafter. The cells proliferated in isolated colonies over a period of approximately 2 weeks. After the cells were detached by treatment with 0.25% trypsin (Gibco®; Thermo Fisher Scientific, Waltham, Massachusetts, USA), the cells were subcultured three times under the same conditions to maintain as homogeneity as possible. These adherent cells are referred to as BMSCs. Third passage BMSCs were used in this experiment. While, SMSCs were isolated by the explant method from the synovial tissues of the outer medial region of the knee joint. These SMSCs were cultured in DMEM containing FBS and antibiotics; cells migrated from periphery of the tissue and strongly adhered to the dishes. Then, the tissue was removed after 1 week. 14 At passage 3, an equal number of SMSCs and BMSCs were cocultured at a 1:1 ratio, and then harvested blindly upon reaching confluency after 5 days to be used in the coculture group. The cells were counted by an automated cell counter (Bio-Rad TC10™, Singapore).
Colony-forming assay
After different cell densities (1, 2, 3, and 5 × 102) were cultured on six-well plates for 2 weeks, 0.5% crystal violet solution (Sigma-Aldrich, St. Louis, Missouri, USA) was applied and incubated with the cells for 30 min at room temperature (RT). The size of the colony was used as a possible indicator of the proliferative potential.
Intra-articular injection of MSCs into the knee joint
Thirteen male Japanese white rabbits aged ≥8 months (Kitayama Labes Co. Ltd, Ina, Japan) weighing 3.0–3.49 kg were used in this study and were maintained freely in separate stainless-steel cages. Anesthesia was induced by an intravenous injection of pentobarbital (30 mg/kg body weight) (Somnopentyl; Kyoritsu Seiyaku Ltd, Tokyo, Japan). To expose the knee joint, the medial parapatellar approach was used, and lateral dislocation of the patella was performed. Using a drill, an osteochondral defect (Ø = 3 mm) was created bilaterally on the weight-bearing surface of the medial femoral condyle. Then, the 26 rabbits’ knees were randomly divided into four groups: the control group, without treatment (n = 6); the BMSC group (1.5 × 106 BMSCs) (n = 7); the SMSC group (1.5 × 106 SMSCs) (n = 7); and the cocultured group (1.5 × 106 cocultured MSCs) (n = 6). Then, an intra-articular injection of MSCs was administered after routine closure of the joint capsule, after which the skin was closed using a nonabsorbable suture material.
Macroscopic and histological assessment of the repaired tissue
At 2 months after the treatment, an intravenous injection of a lethal dose of pentobarbital was used to sacrifice the rabbits. The femoral condyles were evaluated macroscopically using a macroscopic scoring system described by Goebel et al. 15 (Table 1), including five major parameters with 0 points representing excellent results, and 20 points representing the worst results. The condyles were then fixed in a 4% paraformaldehyde phosphate buffered solution (Wako Pure Chemical Industries Ltd) for 48 h, after which the samples were decalcified with 10% ethylenediaminetetraacetic acid solution (Nacalai Tesque, Inc.) for 8–10 weeks, dehydrated using graded alcohols and embedded in paraffin blocks. The samples were cut into 5-µm-thick sagittal sections. For histological evaluation of the repaired tissue, sections were stained with toluidine blue (Muto Pure Chemicals Co. Ltd, Tokyo, Japan) to enable histological scoring on the Fortier scale 16 (Table 2).
Macroscopic scoring system.

Histological Fortier’s scale.

Immunostaining using collagen type II
In brief, paraffin sections were evaluated immunohistochemically using collagen type II to determine the quality of the repaired tissue. According to a previous protocol established by Harada et al. 17 the sections were covered with 3% H2O2 (Wako Pure Chemical Industries Ltd) at RT for 10 min to block nonspecific reactions. Subsequently, the sections were incubated in anti-human collagen type II antibody diluted with PBS 1:250 at RT for 1 h. After being washed with PBS three times, the samples were incubated in a working dilution of a few drops of anti-mouse secondary antibody diluted with PBS 1:200 (VECTASTAIN® Elite ABC Mouse IgG kit (pk-6102), Vector laboratories Inc., Burlingame, California, USA) at RT for 30 min. Finally, the samples were incubated in a 3,3′-diaminobenzidine tetrahydrochloride (DAB) dilution (Vector® DAB substrate) (0.5 mg/ml, with 0.01% H2O2 and a buffer stock solution as a solvent, pH 7.6, Vector Laboratories, Inc.) for 1–5 min, depending upon the degree of color change (brown stain) indicating collagen II.
Statistical analysis
Histological scoring was analyzed by the Kruskal–Wallis and Mann–Whitney U tests, with a 95% confidence interval. Values of p < 0.05 were considered significant.
Results
At several densities, the colony-forming assay showed that the SMSCs and cocultured MSCs formed large visible colonies after 2 weeks, with colony–colony contact in the case of SMSCs, while the BMSCs just formed one small colony at the 500-cell density (Figure 1).
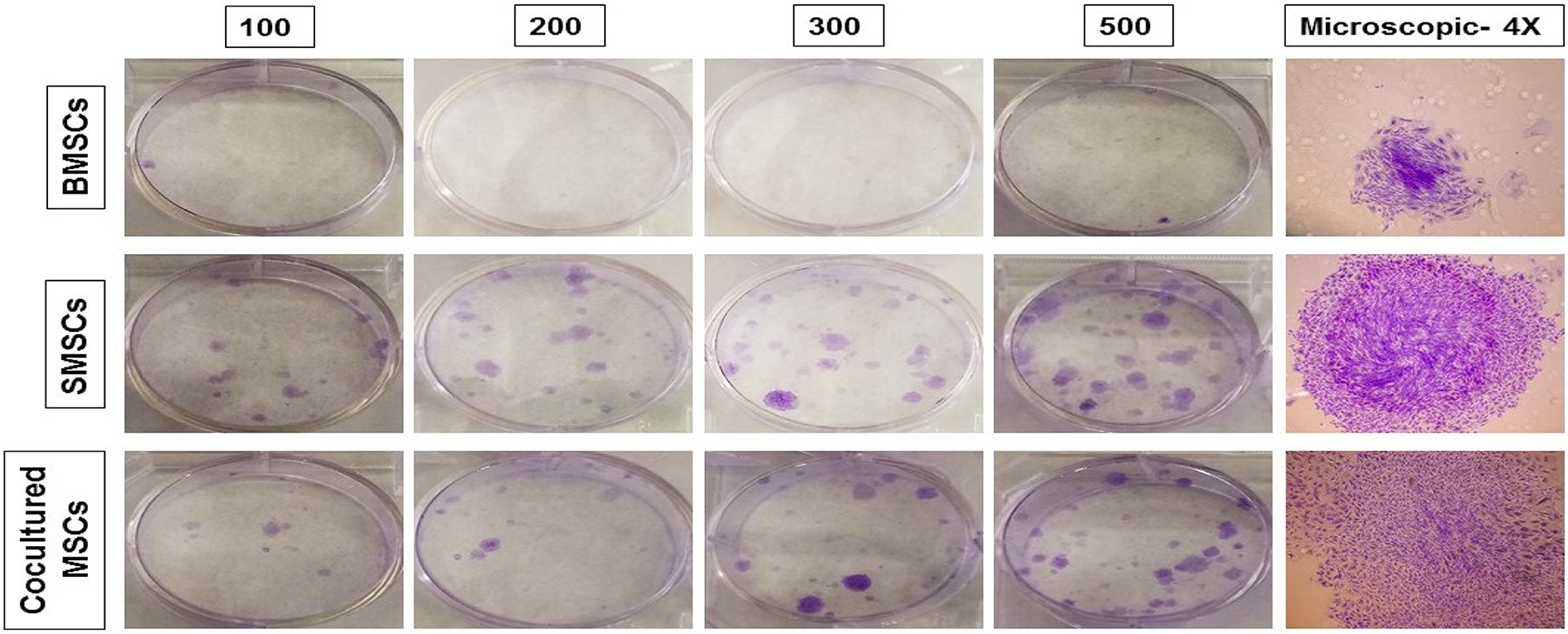
Results of the colony-forming unit assay at different cell densities (1, 2, 3, 5 × 102). After 2 weeks, the numbers of MSC colonies per well/six-well plate; large visible colonies of SMSCs and cocultured MSCs were observed. Additionally, microscopic detection of spindle-shaped MSCs per colony (×4). MSC: mesenchymal stem cell.
Macroscopically, there was no tissue repair in the defect site of the control group. Filling of the defect with white tissue was detected more in the cocultured MSC and BMSC groups than in the SMSC group, with recognizable defect margins and good surface regularity in the cocultured group compared with the other groups (Figure 2). The macroscopic scoring results for the different groups were as follows: control: 19.5 ± 0.8; BMSCs: 6.6 ± 3.9; SMSCs: 13.1 ± 6.1; cocultured MSCs: 5 ± 3.7. There was no significant difference among the therapeutic groups, while there was a significant difference between the control and either the BMSC or cocultured group (p = 0.0039) (Figure 3).

Representative figures showing the macroscopic findings; good surface regularity of the repaired tissue at the level of the surrounding tissue was observed for the cocultured MSCs. MSC: mesenchymal stem cell.

Macroscopic assessment of the repaired tissue using new developed macroscopic scoring. There was no significant difference between BMSCs, SMSCs, and cocultured therapeutic groups, while the score of BMSCs and cocultured MSCs groups were significantly better than the control group. *p Value was less than 0.05. MSC: mesenchymal stem cell; BMSC: bone marrow-derived mesenchymal stem cell; SMSC: synovial tissue mesenchymal stem cell.
Several points were considered in the histological evaluation of the osteochondral defect repair, including the cartilage and subchondral bone repair quality, tide mark formation, integration with the surroundings, defect filling, and cellular distribution. Generally, no tissue was formed in the control group. In the BMSC group, repair of the subchondral bone was confirmed. However, no repair tissue was observed in the SMSC group, with just one case revealing repair of the subchondral bone and another case revealing evidence of cartilage inside the defect without repair of the subchondral bone. In the cocultured group, 50% of the cases showed repaired, with subchondral bone covered by hyaline cartilage and with good toluidine blue staining intensity, reflecting proteoglycan content and the presence of chondrocytes, especially in the deep layers of cartilage (Figure 4). The histological scoring results for the different groups on the Fortier scale were as follows: control: 18.8 ± 1; BMSCs: 15.7 ± 2.3; SMSCs: 17.7 ± 0.5; cocultured MSCs: 11.8 ± 4.4. The cocultured group was significantly different from the control (p = 0.0039) and SMSCs (p = 0.0027) groups. However, there was no significant difference between the BMSC and cocultured MSC groups (Figure 5). Additionally, chondrocytes embedded in collagen type II were observed in the cocultured MSC group (Figure 6).

Representative figures showing the histological findings of osteochondral formation at 2 months, with good repair of the subchondral bone covered with cartilage matrix, especially in the deep layer, in the case of cocultured BMSCs and SMSCs compared with BMSCs or SMSCs used alone. In addition, integration of the repaired tissue with the surrounding tissue was confirmed, especially in the subchondral region. BMSC: bone marrow-derived mesenchymal stem cell; SMSC: synovial tissue mesenchymal stem cell.
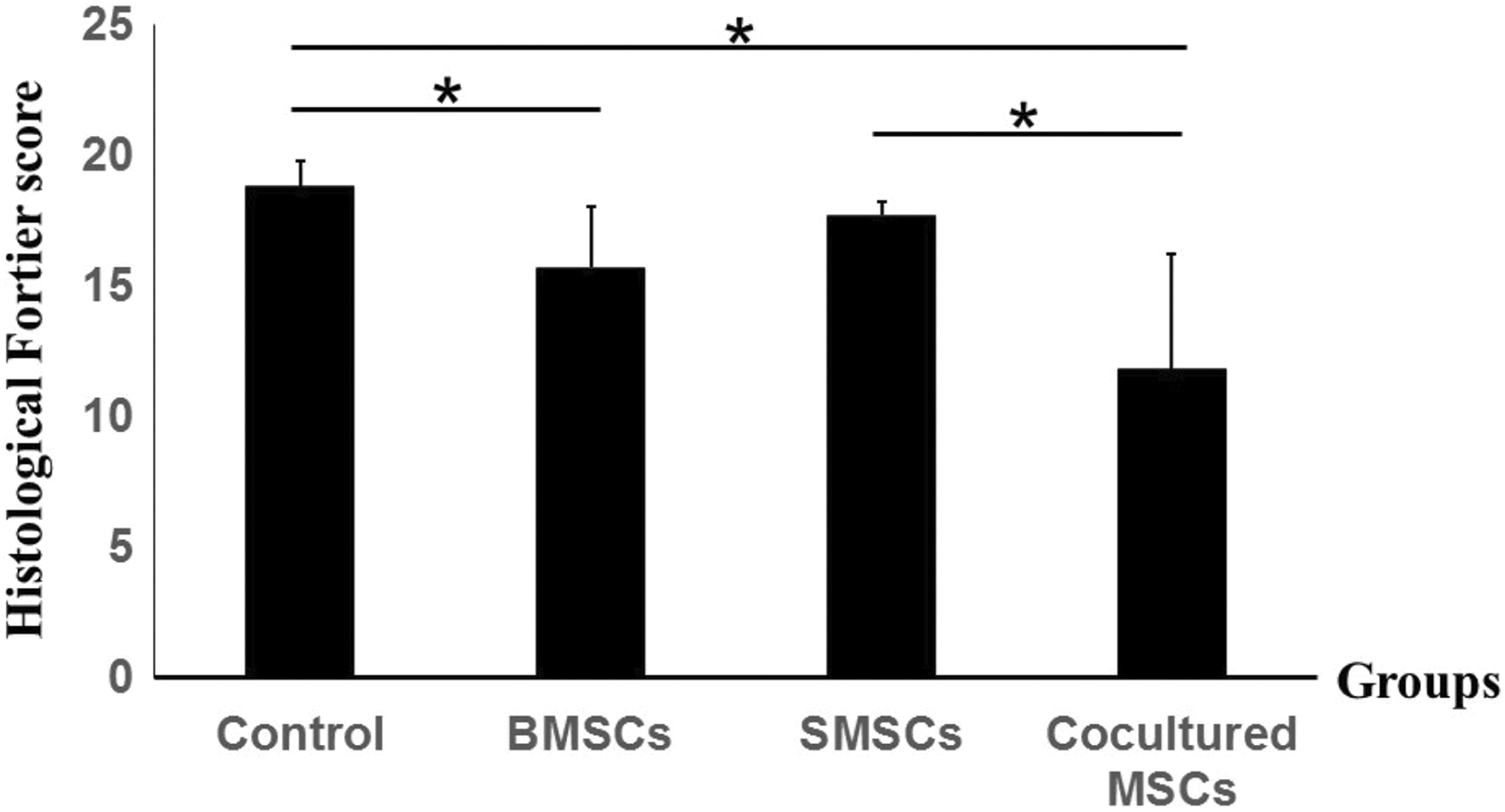
Histological assessment using the Fortier scale: better histological results were observed in the cocultured MSC group than in the control and SMSC groups, and the BMSCs group showed significant better histological results than the control group. *p Value was less than 0.05. MSC: mesenchymal stem cell; BMSC: bone marrow-derived mesenchymal stem cell; SMSC: synovial tissue mesenchymal stem cell.
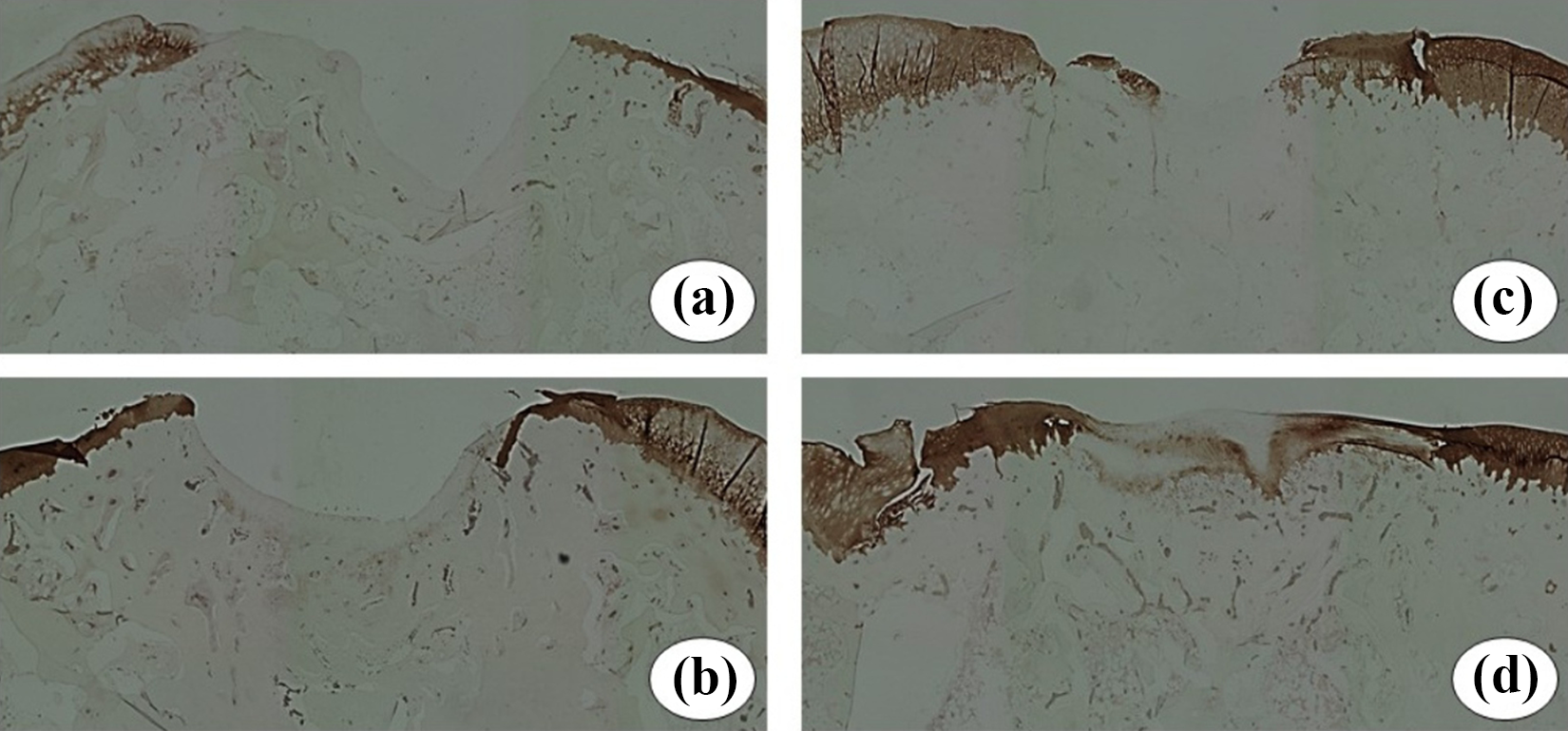
Collagen-II staining intensity was clearly evident in the repaired tissue in the cocultured MSC group (d), whereas there was only evidence of collagen II at the periphery of the defect in the BMSC group (c). Additionally, collagen-II was not detected in the control (a) or SMSC group (b). MSC: mesenchymal stem cell; BMSC: bone marrow-derived mesenchymal stem cell; SMSC: synovial tissue mesenchymal stem cell.
Discussion
Our findings demonstrated that the intra-articular injection of cocultured MSCs yielded improved repair of osteochondral defect with good surface regularity macroscopically, repair of subchondral bone covered by hyaline cartilage histologically, and presence of collagen II containing chondrocytes immunohistochemically.
For proliferation potential, one small colony of BMSCs was formed at the 500-cell density in comparison with large colonies which were formed by SMSCs and cocultured MSCs. These data result in great expansion potential of SMSCs and lower expansion potential of rabbit BMSCs. Our results were consistent with previous report which revealed that rat SMSCs have a greater proliferation potential than of rat BMSCs. 8 This means that colony size is an indicator for expansion rate of cells.
Regarding the numerous tissue sources, MSCs have variable multipotency toward chondrocytes, osteoblasts, and adipocytes. 18 Most previous experimental and clinical studies have used BMSCs, although 39% of cases have reported chronic pain at the donor site. 19 On the other hand, it is easy to obtain SMSCs arthroscopically from the synovial tissue of rabbits and horses with minimal complications at the donor site due to the strong regenerative effect for self-repair after synovectomy 20,21 ; SMSCs have the greatest chondrogenic and lowest osteogenic potential among MSCs. 22 For this reason, the present study aimed to enhance the osteochondrogenic potential for osteochondral repair using cocultured MSCs from different sources, that is, BMSCs and SMSCs. Both the macroscopic and histological results of the cocultured MSCs were consistent with our hypothesis on the beneficial synergistic effect of a variety of MSCs via coculturing compared with using either cell type alone. In the cocultured MSC group, chondrocytes were embedded in the cartilage matrix and proteoglycans, and tide mark formation and repaired cartilage integration with the subchondral bone were observed; these results were significantly different from those of the control and SMSC groups. Recent studies have tried to improve the chondrogenesis of BMSCs via coculture with chondrocytes with the aim of producing more cartilage matrix. However, this may not be clinically applicable due to the restricted availability of chondrocytes from clinical cartilage biopsies with a low chondrocyte density, and the down-regulation of collagen expression in passaged chondrocytes. 23 Similar studies have revealed that SMSCs cocultured with meniscal cells lead to good cell survival, chondrogenic differentiation with collagen II, Sox9, and glycosaminoglycan expression, with a low level of collagen I. 24
Although there were no significant differences among the therapeutic groups regarding the macroscopic score, tissue repaired by cocultured MSCs exhibited good surface regularity at the level of the normal tissue. This result explains why new subchondral bone formation needs to be substantial to achieve good cartilage repair. It is well known that subchondral bone repair is important for the long-term repair and stability of newly formed cartilage. 25 However, newly formed tissue traditionally lacks stability due to poor subchondral bone formation with inferior surface regularity. 26 Additionally, Dahlin et al. revealed the regenerative ability of chondrocytes or MSCs cocultured with chondrocytes, which stimulated cartilage repair without evidence of subchondral bone healing. This leads to the worst surface regularity outcome of the chondrocyte treatment and poor integration of the newly formed cartilage with the surrounding tissue, 9 resulting in deterioration of the repaired cartilage over time with mechanical compression. 27 Additionally, most experimental studies have focused on articular cartilage defects, although subchondral bone is crucial to the development of efficient therapeutics for osteochondral repair. 28 In most cases in the BMSC group, the subchondral bone repair provided convincing evidence of the superiority of osteogenic rather than chondrogenic potential. On the other hand, SMSCs revealed superior chondrogenic and inferior osteogenic potential, 29 which may reduce the potential of SMSCs as a therapeutic option for osteochondral defects.
The results of our study illustrate the promise of a variety of cocultured different allogeneic MSCs for clinical applications. The observed outcome would improve the resulting quality of life, especially regarding the immunosuppressive characteristics of both types of MSCs, due to their ability to inhibit T-cell proliferation in a mixed lymphocyte reaction. 30 Several studies have used allogeneic chondrocytes and MSCs for articular cartilage and meniscal repair, 31 –33 and xenogeneic chondrocytes in the case of chondral and osteochondral defects, without evidence of immune response. 34 Trials of chondrocytes cocultured with MSCs have been using up to 75% chondrocytes, achieving equal or greater chondrogenic potential compared with the sue of 100% chondrocytes. 35 Hence, further studies will be necessary to evaluate the best SMSCs/BMSCs ratio in the coculture system either for in vitro and in vivo assessment of osteochondrogenic potential with long-term evaluations.
Several limitations of our study were recorded. First, transplantation of BMSCs and in particular SMSCs did not result in a clear significant improvement in the histological findings as therapeutic option for osteochondral defects, may be due to low cell density and absence of scaffold. Concerning our results of SMSCs is matching with previous report which found that transplantation of SMSCs/gel composites with 5 × 107cells/ml was successful for osteochondral defect, whereas transplantation of SMSCs/gel composites containing 1 × 106 cells/ml results in failure. Higher cell density promote the healing of a cartilage defect more effectively, although the preparation of a sufficient number of cells for chondrogenic potential is not always possible, 22 especially in elder age. Secondly, lack of in vitro assessment of chondrogenic, osteogenic, even adipogenic differentiation capacity for BMSCs, SMSCs, and cocultured MSCs. Third, the lack of cell tracking to determine which cell type participates in osteochondral repair via differentiation into either osteoblasts or chondrocytes.
Conclusion
Although macroscopic and histological scores do not show a significant improvement between BMSCs and cocultured MSCs, the coculture of MSCs from different sources could be a suitable cell therapeutic option for producing a good clinical outcome in osteochondral lesion repair.
Footnotes
Acknowledgments
The authors would like to thank Takako Miyata for her technical support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by a grant-in-aid to Prof. Mitsuo Ochi for scientific research from the Ministry of Education, Culture, Sports, Science and Technology, Japan (no. 25253089).
