Abstract
Objective
The aim of this study was to evaluate an intraarticular injection of different doses of autologous mesenchymal stem cells (MSCs) for improving repair of midterm osteochondral defect.
Design
At 4 weeks postoperative marrow stimulation model bilaterally (3 mm diameter; 4 mm depth) in the medial femoral condyle, autologous MSCs were injected into knee joint. Twenty-four Japanese rabbits aged 6 months were divided randomly into 4 groups (n = 6 per group): the control group and and MSC groups including 0.125, 1.25, and 6.25 million MSCs. Repaired tissue was assessed macroscopically and histologically at 4 and 12 weeks after intraarticular injection of MSCs.
Results
At 12 weeks, there was no repair tissue in the control group. The gross appearance of the 1.25 and 6.25 million MSC groups revealed complete repair of the defect with white to pink tissue at 12 weeks. An osteochondral repair was histologically significantly better in the 1.25 and 6.25 million MSC groups than in the control and 0.125 million MSC groups at 4 and 12 weeks, due to presence of hyaline-like tissue in the deep layer at 4 weeks, and at 12 weeks hyaline cartilage formation at the periphery and fibrous tissue containing some chondrocytes in the deep layer of the center of the defect. Subchondral bone was restructured in the 1.25 and 6.25 million MSC groups, although it did not resemble the normal bone.
Conclusion
An intraarticular injection of 1.25 or 6.25 million MSCs could promote the repair of subchondral bone, even in the case of midterm osteochondral defect.
Introduction
Osteochondral defects are much more complex and present a far greater surgical and therapeutic challenge, because damaged tissue does not only include the superficial cartilage layer but also subchondral bone. 1 The weight-loaded area of the medial femoral condyle is the most common affected area, and it is the predominant location of severe full-thickness lesions. 2 Although most therapeutic options for osteochondral defects are widely used, such as marrow-simulating techniques (MST) and osteochondral graft, clinical success is limited. 3 MST aims to recruit marrow elements through penetration of the subchondral plate, differentiating into fibrocartilaginous tissue of mainly low inferior quality. 4 Recent reports confirmed potential changes of the subchondral bone associated with MST, such as thickening of the subchondral bone, subchondral cysts, and intralesional osteophytes. 5 Also, either autograft or allograft osteochondral grafts recorded some serious drawbacks as well as incomplete integration and degradation of the graft tissue. 6 Mesenchymal stem cells (MSCs) hold great promise as a new option for the treatment of musculoskeletal regeneration. 7 MSCs have a high expansion capacity and the potential to differentiate along all mesenchymal lineages. 8 On the other hand, chondrocytes are not suitable for osteochondral defects, because they are highly differentiated cells with a limited proliferation capacity to obtain even small numbers of autologous chondrocytes through cartilage biopsy, also causing additional damage. 9 MSCs can be injected directly into diseased tissue where cells might function through the secretion of bioactive factors and eventually integrate into any receptive cells and surfaces. 10
Most studies have used growth factors, such as transforming growth factor-β, to stimulate differentiation of MSCs into chondrocytes in vitro, but in vivo their application has been unsuccessful and expensive, increasing the overall cost. 11 In addition, using transforming growth factor-β for tissue regeneration has the potential for ectopic tissue formation in vivo. 12 The limitations of natural scaffolds due to extensive purification protocols, and potential pathogen contamination when harvested from an animal or human source, epitomize the disadvantages of using natural materials. 7 The degradation products of synthetic materials are highly concentrated acids and particulates, which can have the adverse effect of causing localized inflammation and cell death, 13 poor cell attachment, and an inability to direct cellular differentiation. 14
Transplanted uninduced MSCs have been reported to differentiate into both bone and cartilage depending on the surrounding environment, 15 although the exact mechanisms that control the tissue homing of MSCs are mysterious. Obviously, MSCs themselves secrete a broad spectrum of bioactive molecules that have immunoregulatory 16 and/or regenerative effects. 17 Therefore, MSCs may be beneficial for the repair of osteochondral defects prior to bone marrow stimulation and osteochondral graft, especially for subchondral bone. However, injection of too many MSCs (1 × 107) intraarticularly in the rat knee can generate free bodies of scar tissue. 18 Subsequently, the aim of the study was to evaluate whether distinct numbers of autologous MSCs can alter midterm osteochondral repair tissue quality as postoperative intraarticular injections.
Materials and Methods
This study was approved by the guide for animal experimentation and by the ethical committee of laboratory animal science research (Graduate School of Biomedical & Health Sciences, Hiroshima University, Japan).
Isolation and Expansion of Autologous BM-MSCs
Bone marrow–derived MSCs (BM-MSCs) were isolated and expanded in vitro as described previously. 19 After creation of osteochondral defect, immediately 5 mL of bone marrow blood from the iliac crest of male Japanese white rabbits was aspirated with 1 mL of heparin (1000 units/1 mL) (Mochida Pharmaceutical Co. Ltd., Tokyo, Japan). The samples were centrifuged for 5 minutes at 1500 rpm, and the resultant supernatant including heparin sodium and fat was discarded. The extract was resuspended in 2 mL of culture medium, composed of Dulbecco’s Modified Eagle Medium (Gibco, Life Technologies, Warrington, UK) with 10% fetal bovine serum (Sigma-Aldrich Corp, St Louis, MO) and 1% antibiotics (penicillin, streptomycin, and Amphotericin B) (Nacalai Tesque, Inc., Kyoto, Japan). Two milliliters of the suspension was then seeded onto 100-mm tissue culture dishes (Falcon; BD Biosciences, Franklin Lakes, NJ), and 8 mL of culture medium was added to each dish. The dishes were incubated for 4 weeks under a humidified atmosphere with 5% CO2 at 37°C. The medium was not changed for the first 7 days but was then washed twice with phosphate buffer saline (PBS) to remove nonadherent cells, after which the medium was changed every 3 days. The cells proliferated and reached confluence approximately 2 weeks after seeding. The cells were harvested by treating them with 0.25% trypsin (Gibco, Life Technologies), washed off, and then seeded onto culture dishes (3.5 × 105 cells/10 cm culture dish). After a confluent cell layer was formed again, the cells were reseeded onto 100 mm culture dishes with the same density under the same conditions. These adherent cells are referred to as MSCs. As a result, cells at P3 were used in the current experiment, kept in ice tank for 1 to 1.5 hours between resuspension of MSCs in PBS and injection into the joint of both knees.
Establishment of Midterm Osteochondral Defect in a Rabbit Model
Twenty-four 6-month-old male Japanese white rabbits (48 knees) (Kitayama Labes Co. Ltd., Nagano, Japan), ranging 2.5 to 2.99 kg in body weight, were used in this study. Anesthesia was induced by intravenous injection of pentobarbital sodium (30 mg/kg body weight) (Somnopentyl, Kyoritsu Seiyaku Ltd, Tokyo, Japan). The medial parapatellar approach was used, and dislocation of the patella was performed laterally to expose the knee joint. Using a drill, an osteochondral defect (diameter: 3 mm; depth: 4 mm) was created at the loaded area of the medial femoral condyle in both knees. Four weeks after creation of the defect, an intraarticular injection of autologous MSCs was administered into the knee joint. The rabbits were randomly divided into 4 groups and each group contained 6 rabbits receiving identical treatment: control group: PBS injection; 0.125 million MSC group: 1.25 × 105 MSCs; 1.25 million MSC group: 1.25 × 106 MSCs; and 6.25 million MSC group: 6.25 × 106 MSCs, where MSCs at P3 were suspended in 100 µL PBS ( Table 1 ). The rabbits were then returned to their cages and were left free to exercise. In each group, 3 rabbits were sacrificed at 4 weeks and a further 3 rabbits at 12 weeks after intraarticular injection of MSCs by an intravenous injection of a lethal dose of pentobarbital sodium (100 mg/kg body weight).
Study Design.
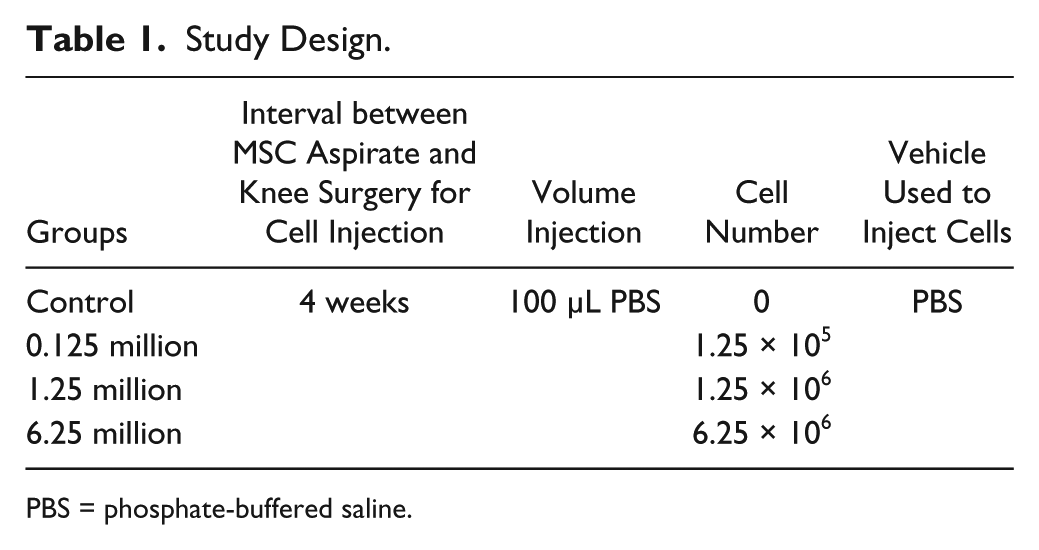
PBS = phosphate-buffered saline.
Histological Examination of Repair Tissue
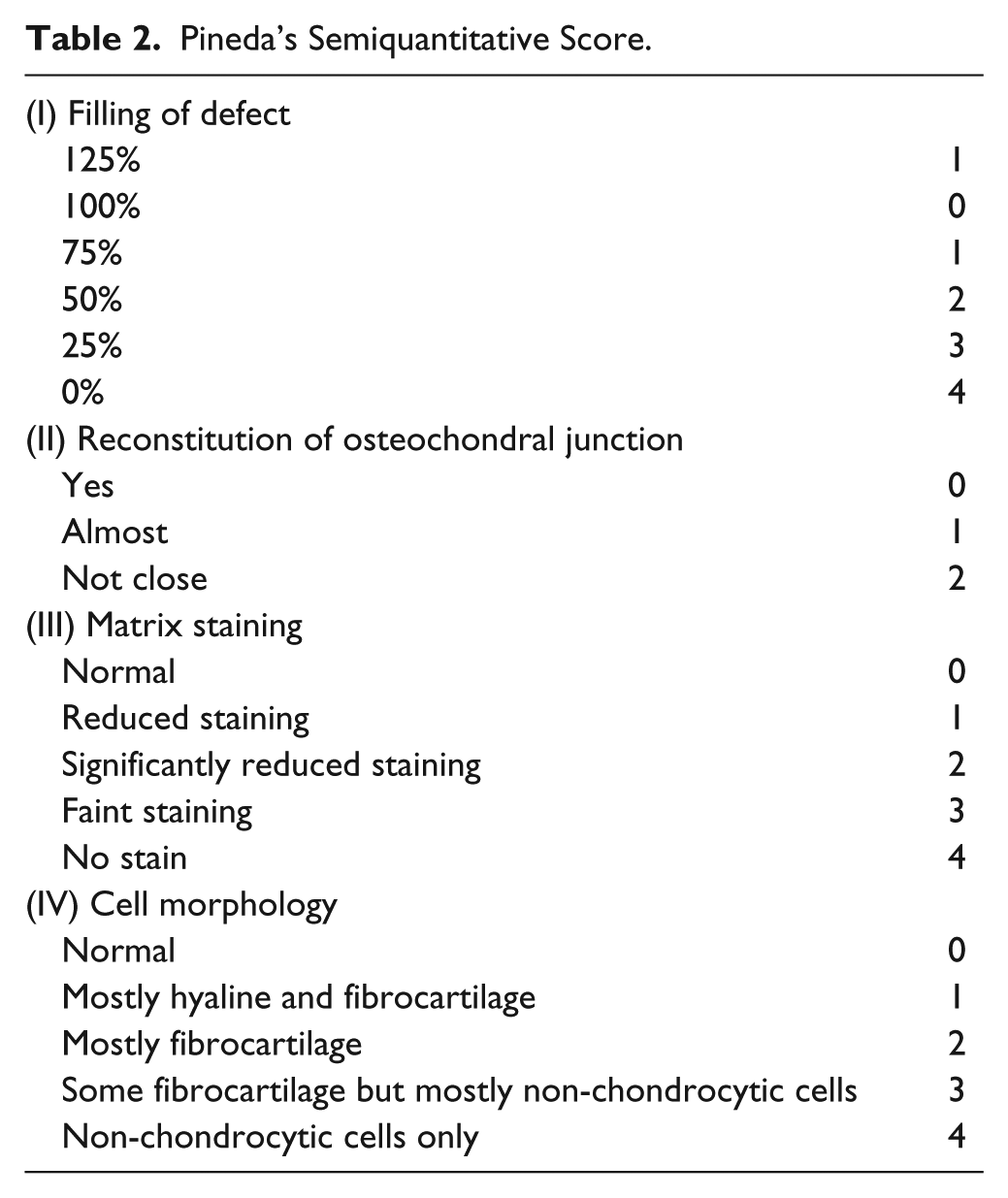
The distal part of the femur was excised and fixed in paraformaldehyde PBS 4% (Wako Pure Chemical Industries Ltd., Osaka, Japan) for 2 days, then decalcified in EDTA 10% at pH 8.0 for 8 weeks, dehydrated in a series of graded alcohols, and embedded in paraffin blocks. The samples were cut into 5-µm sections sagittally. For histological evaluation, the sections were deparaffinized and stained with Safranin O/Fast green stain for assessment of hyaline-like cartilage using Pineda’s semiquantitative scale ( Table 2 ). 20
Pineda’s Semiquantitative Score.
Immunohistochemistry
Other sections were used for the immunohistochemical staining of collagen type II by collagen type II antibody (Anti-hCL II, purified IgG, Fine Chemical Company, Tokyo, Japan). The sections were covered with 3% H2O2 at room temperature for 10 minutes to block nonspecific reactions. Subsequently, the sections were incubated in anti-human collagen type II antibody diluted with PBS 1:250, and we used PBS instead of primary antibody for collagen II as a negative control at room temperature for 1 hour. After being washed with PBS 3 times, the sections were incubated in a working dilution of anti-mouse secondary antibody (VECTASTAIN Elite ABC Mouse IgG, Vector Laboratories, Burlingame, CA) at room temperature for 30 minutes. Finally, the samples were incubated in 3,3′-diaminobenzidine tetrahydrochloride (DAB) dilution (Vector DAB substrate) (0.5 mg/mL, with 0.01% H2O2 and buffer stock solution as solvent, pH 7.6, Vector Laboratories, Inc.) for 1 to 5 minutes depending on the degree of color change. Collagen type II was stained brown.
Statistical Analysis
Statistical analysis was performed for overall histological scoring using the Kruskal-Wallis test and for comparison between the groups using the Steel-Dwass test with a 95% confidence interval. For all statistical analyses, SPSS version 20 was used (IBM, Armonk, NY). Values of P < 0.05 were considered significant in both tests.
Results
Macroscopic Findings
At 4 weeks, there was little fibrous tissue in the control and 0.125 million MSC groups, while in the 1.25 and 6.25 million MSC groups partially repaired tissue formed in the defect. At 12 weeks, there was little fibrous tissue in the control group, and partially repaired tissue in the 0.125 million MSC group, while in the 1.25 and 6.25 million MSC groups the defect was fully filled with white to pink repaired tissue to the same level as normal tissue ( Fig. 1 ).
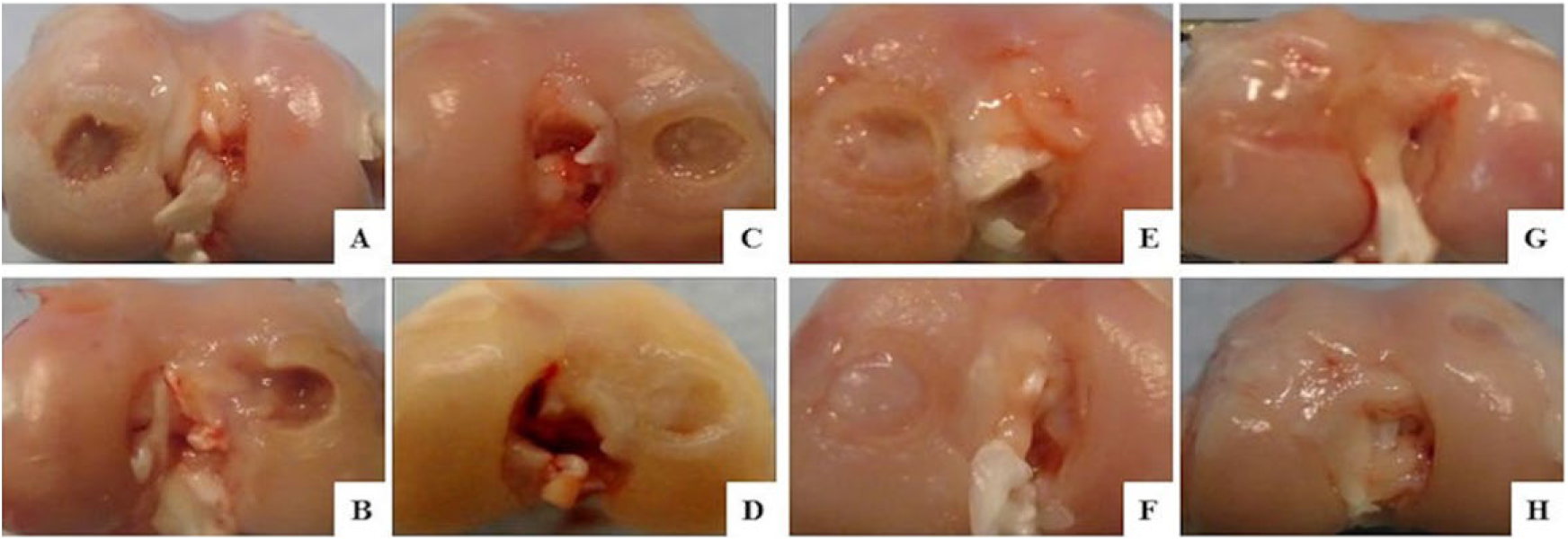
Macroscopic findings at 4 (upper row) and 12 (lower row) weeks after an intraarticular injection of MSCs inside the joint. (
Histological Evaluation and Scoring
At 4 weeks in the control group, there was no repair tissue for subchondral bone and cartilage; there was just a small amount of replacement fibrous tissue and poor integration with the adjacent normal tissue. In the 0.125 million MSC group, the result resembled that of the control group, but with slight integration. In the 1.25 and 6.25 million MSC groups, there was formation of hyaline-like tissue in the deep layer of the defect with incomplete integration with the normal cartilage and partial repair of the subchondral bone. At 12 weeks, histological findings in the control group were quite similar as well as at 4 weeks. In the 0.125 million MSC group, subchondral bone was detected at a lower level than normal and little fibrous tissue replaced cartilage. In the 1.25 and 6.25 million MSC groups, there was formation of hyaline-like cartilage in the periphery and fibrous tissue containing some chondrocytes in the deep layer of the center of the defect. Additionally, reconstruction of the subchondral bone was observed in the 1.25 and 6.25 million MSC groups compared with the control and 0.125 million MSC groups, although it did not seem to be repaired as a normal subchondral bone (Figs. 2-4).
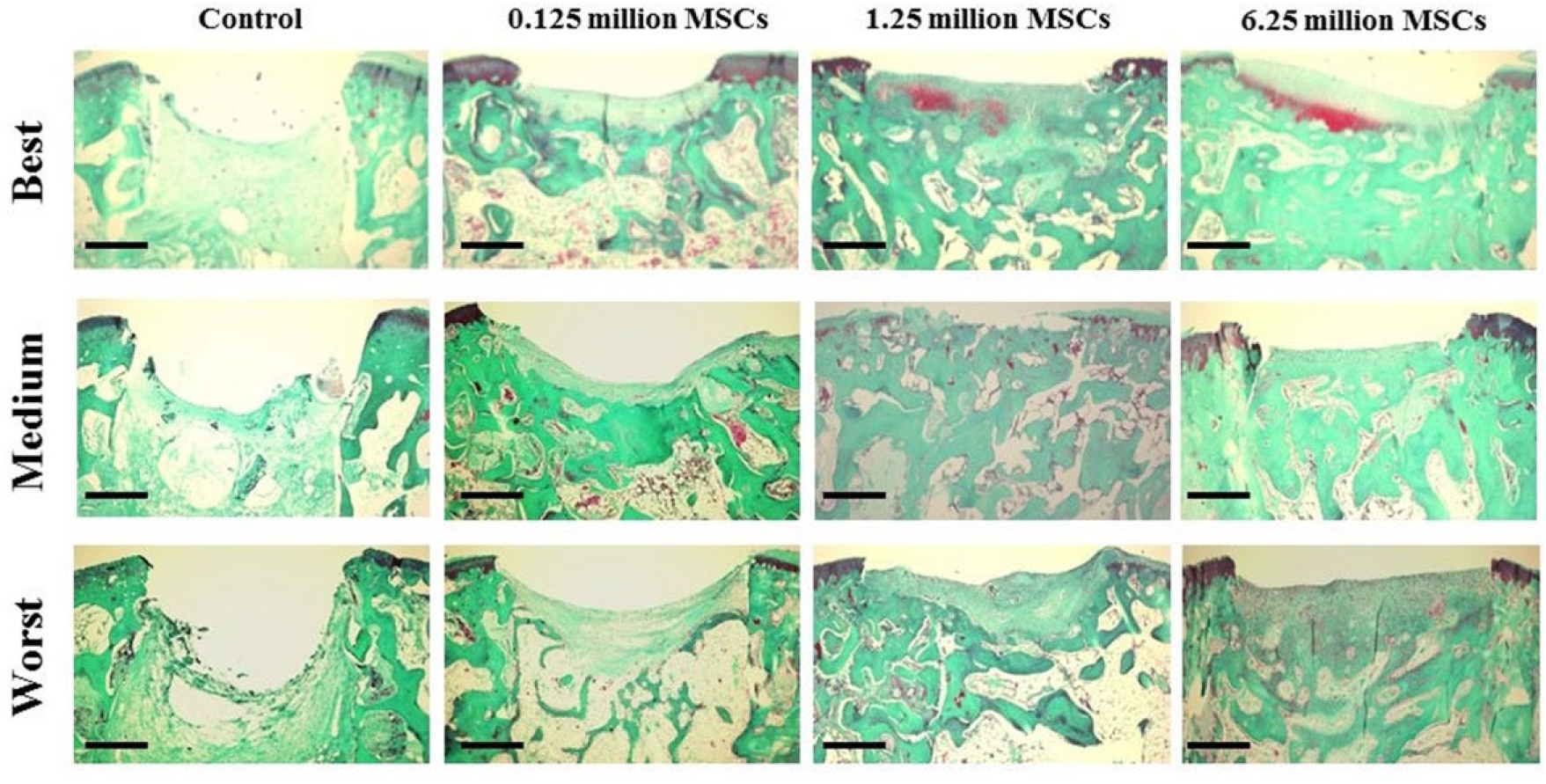
Representative histological findings at 4 weeks after an intraarticular injection of MSCs using Safranin O/Fast green stain, showing presence of hyaline-like tissue in the deep layer of the defect of the 1.25 million and 6.25 million MSC groups. Scale bars = 500 µm.
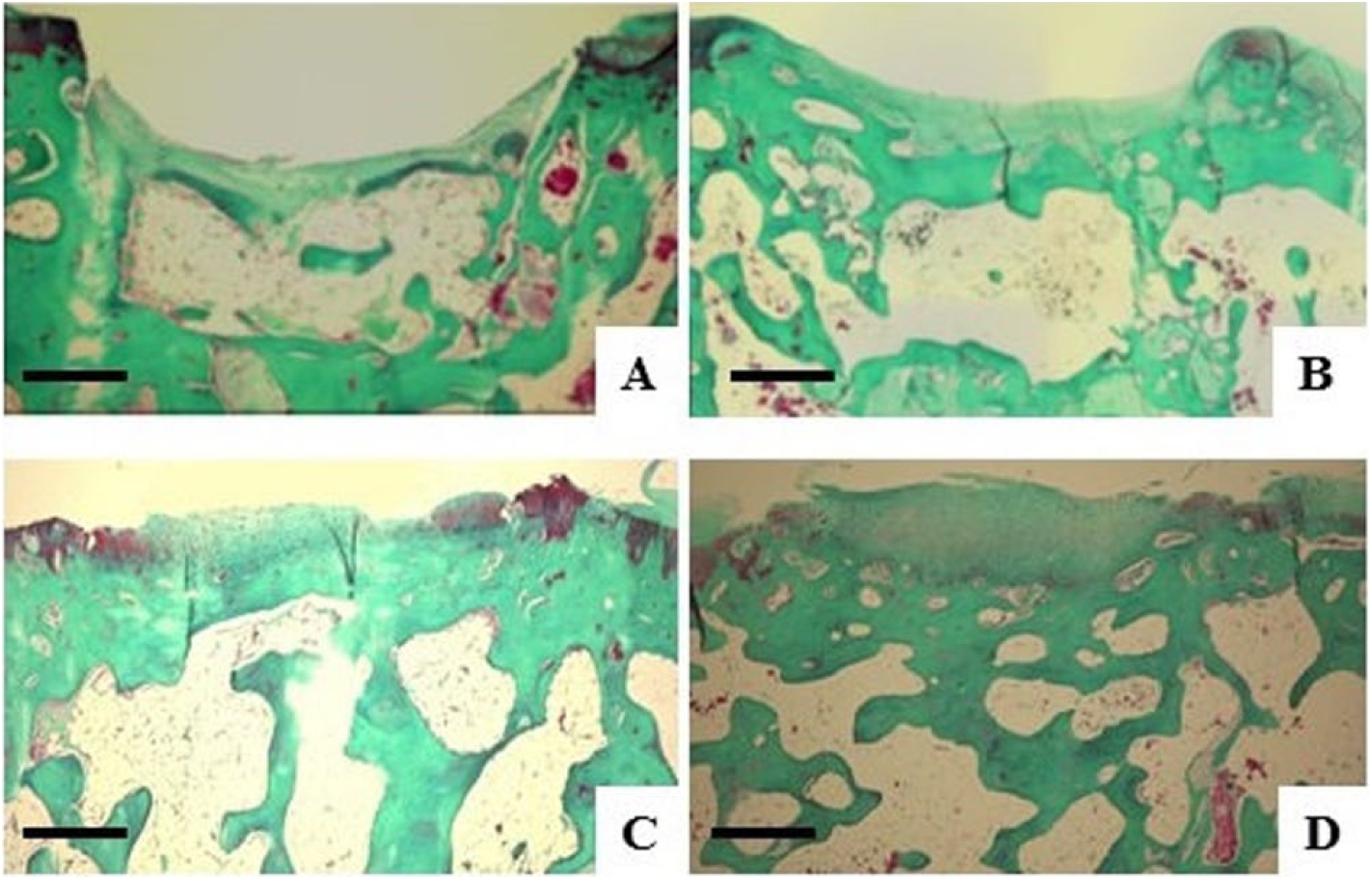
Microscopic findings at 12 weeks after an intraarticular injection of MSCs using Safranin O/Fast green stain. (
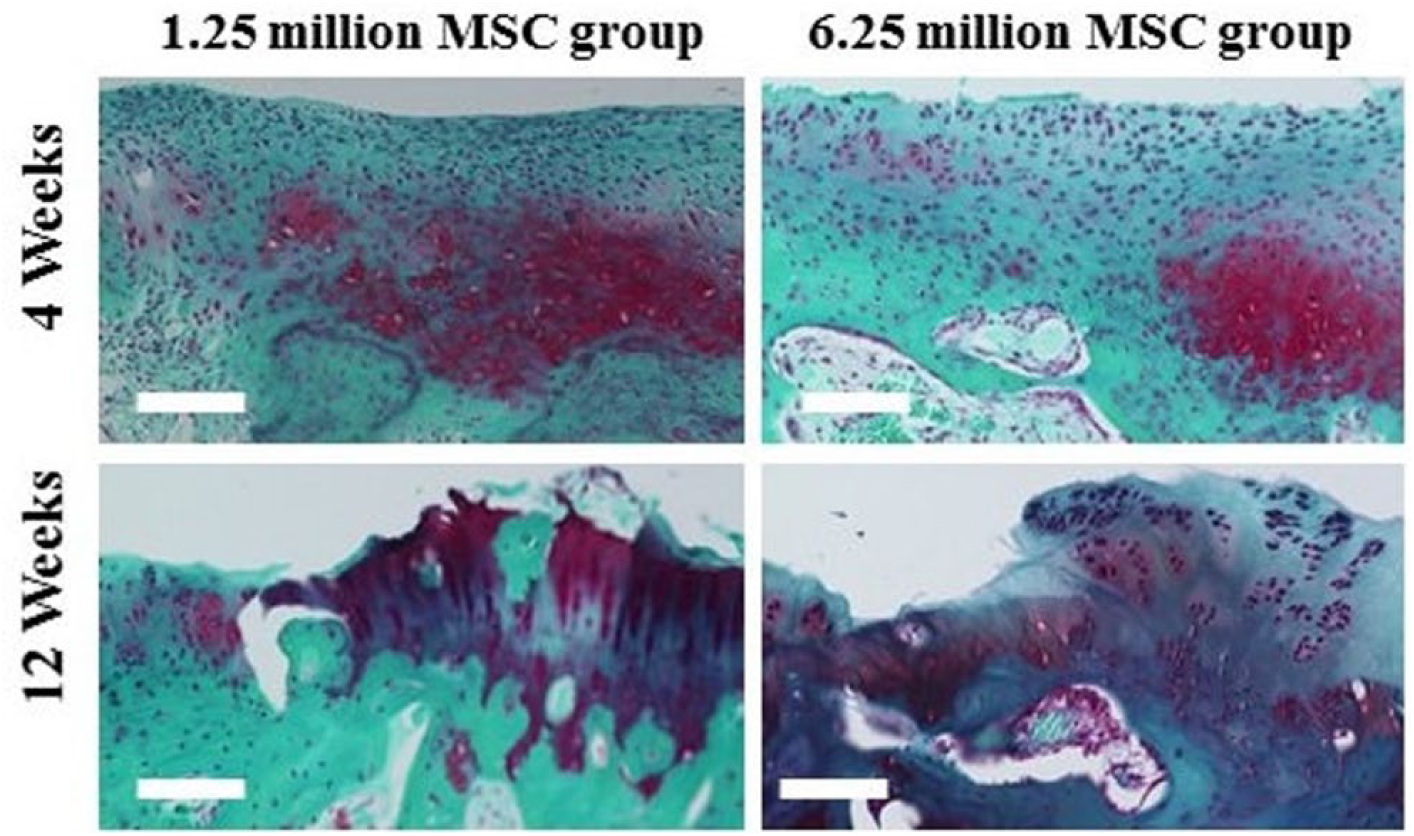
Higher magnification for the 1.25 and 6.25 million MSCs groups at 4 and 12 weeks for Safranin O/Fast green. Scale bars = 100 µm.
At 4 and 12 weeks, histological assessment using Pineda’s semiquantitative scale showed results of 13 ± 0.6 and 10.6 ± 2.3 in the control group, 11.2 ± 0.8 and 10 ± 1.8 in the 0.125 million MSC group, 8.2 ± 2.1 and 5.5 ± 2.3 in the 1.25 million MSC group, and 8.7 ± 2 and 5.7 ± 1.2 in the 6.25 million MSC group. According to these results, repaired tissue was histologically significantly better in the 1.25 and 6.25 million MSC groups than in the control and 0.125 million MSC groups at 4 and 12 weeks ( Fig. 5 ).
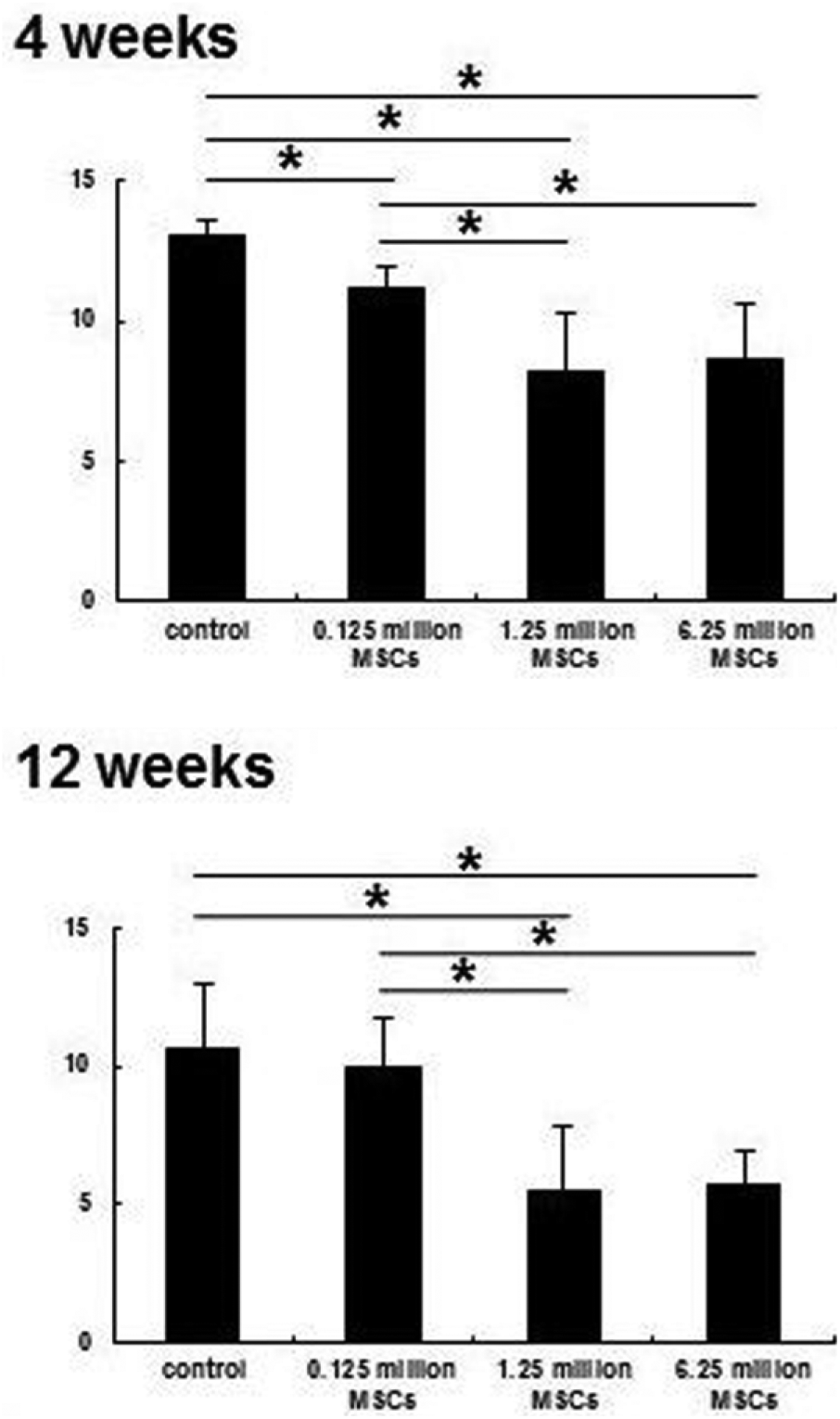
Pineda scale for histological assessment at 4 and 12 weeks. The 1.25 million and 6.25 million MSC groups were significantly better than control and 0.125 million MSC groups at 4 and 12 weeks. *P < 0.05.
Immunohistochemical Findings
There was no evidence of collagen type II in the control and 0.125 million MSC groups, either at 4 or 12 weeks. However, there was slight evidence of collagen type II in the deep layer of the defect of the 6.25 million MSC group at 12 weeks ( Fig. 6 ).

Immunohistochemical staining using collagen type II at 4 (upper row) and 12 (lower row) weeks after an intraarticular injection of MSCs. (
Discussion
MSCs hold great promise as a cell source for the repair of several tissue lineages such as bone, cartilage, and muscle. Although MSCs are limited in number, their potential in regenerative medicine and immunomodulatory properties through the secretion of bioactive molecules has been promising. Direct injection of MSCs is the most immediate and convenient method of cell delivery. 7 MSCs can mobilize to an injured site through the production of inflammatory signals from the injured site which attract MSCs. 21 Recently, Chen et al. revealed that stromal cell derived factor-1 facilitates MSCs homing for osteochondral repair. 22
In the present study, we have demonstrated that a less invasive intraarticular injection of 1.25 or 6.25 million MSCs enhanced repair of midterm osteochondral defect in the rabbit model (diameter 3 mm, depth 4 mm) without using a scaffold, growth factors, or MST. Although cartilage repair was not perfect, repair of the subchondral bone was observed in the 1.25 and 6.25 million MSC groups better than that in the control and 0.125 million MSC groups. Subchondral bone plays a critical role in cartilage repair, and its potential changes are responsible for the worst outcomes both in chronic defects as well as in cartilage defects previously treated with MST. 3 One study revealed that the failure rate of autologous chondrocyte implantation in previous bone marrow–stimulated defects is 3 times higher than in not previously treated defects due to known potential complications of MST. 23
Our study focused on clinical relevance by investigating the different doses of MSCs for repair of midterm osteochondral defects at the weight-bearing surface of the medial femoral condyle, because this is considered to be the most predominant location for knee lesions. Osteochondral defects pose a major challenge for clinicians and researchers due to the inclusion of cartilage and subchondral bone. Previous studies have aimed to find a treatment for cartilage defect models, although most cartilage problems extend deeply into the underlying subchondral bone, which hasnot received much attention until now. Therefore, further elucidation of osteochondral defects is necessary. Effective treatment comprises the repair of cartilage, subchondral bone, and bone-cartilage interface. In our study, we found that injection of 1.25 million MSCs enhances osteochondral repair (3 mm in diameter) in the weight-bearing area of a rabbit model. In the equine model, McIlwraith et al. compared microfracture versus microfracture combined with (20 × 106) MSCs and 22 mg hyaluronic acid (HA) in the healing of 1 cm2 defects in the medial condyle. This study reported that arthroscopic and gross evaluation confirmed a significant improvement in the repair tissue firmness, although there was no evidence of significant clinical and histological improvement in the joints injected with MSCs. 24 We suggest that direct injection of MSCs is less invasive and is therefore the most convenient method for osteochondral repair, because most surgical treatments offer limited success in addition to some draw backs such as thickening of subchondral bone, formation of a subchondral cyst, and intralesional osteophytes. 5 In addition, HA is highly biocompatible and fully resorbable in 3 months. However, degradation products of HA can cause chondrolysis. 25
Wakitani et al. reported on the complete repair of a large full-thickness articular cartilage defect in rabbits after 24 weeks by implanting cultured mesenchymal osteochondral progenitors cells suspended in collagen gel isolated from the periosteum and bone marrow. 26 Also, Im et al. reported that implantation of cultured autologous MSCs (1 × 106) for repair of cartilage defects (3 mm diameter and 2 mm in depth) in the patellar groove of rabbits promotes but does not guarantee cartilaginous healing. 27 Nishimori et al. reported that combination therapy of the bone marrow–stimulating procedure and injection of BM-MSCs (1 × 106) for repair of osteochondral defects in the patellar groove (2 mm × 2.5 mm and 1 mm depth) of rats had a significantly better outcome than that of the drilling group, although the repair was not perfect. 28 In our study, repair tissue in the 1.25 and 6.25 million MSC groups was better than in the control and 0.125 million MSC groups histologically, although at 4 weeks the repair was not typical. There was partial repair of subchondral bone and formation of hyaline cartilage in the deep layers. At 12 weeks, repair of the subchondral bone was progressed in the 1.25 and 6.25 million MSC groups. Although it did not seem to be normal subchondral bone, it was much better than that in the control and 0.125 million MSC groups. In addition, formation of hyaline cartilage at the periphery with fibrous tissue containing some chondrocytes at the center of the defect in the 1.25 and 6.25 million MSC groups. However, cartilage repair was not perfect in our study, complete repair of subchondral bone was achieved histologically. We can hypothesize that BM-MSCs secrete growth factors which promote osteogenesis rather than chondrogenesis and that the depth of the defect may be a stimulation mechanism of MSCs toward osteogenesis rather than chondrogenesis. In addition, MSCs properties might be negatively affected with using PBS as a vehicle. Although several previous reports displayed contradictory results using PBS, dextrose 5%, or saline 0.9% at different temperatures and storage time.29-31 Hence, this study suggests that different aqueous carriers should be used to inject cells in the hopes of improving repair tissue quality.
Challenges faced made us to obtain good osteochondral defect repair. First, integration between repaired and surrounding tissue was very important. In our study, the integration between repaired cartilage tissue and around normal cartilage was unfortunately incomplete even in the 1.25 and 6.25 million MSC groups, especially at 12 weeks. Failure to integrate led to deterioration of the repaired cartilage, which was reported over a long-term period, especially in the case of the osteochondral graft. 32 Second, the loading compression must be taken into consideration according to the topographical location of the defect, since compressive overloads can cause cartilage degeneration through inhibition of synthesis of the important components of the cartilage matrix such as collagen II and aggrecan. 33 The location of the defect in most previous studies was the patellar groove, although Orth et al. found that assessment of cartilage and osteochondral repair were better in the trochlea than in the condyle. Compared to chondral defects, osteochondral defects were found to cause more osteoarthritic degeneration of the adjacent cartilage. 34
Potential limitations of our study must be mentioned: creation of bilateral defects in the same rabbit was more stressful than unilateral defects. Short-term follow-up was not sufficient to assess the durability of the repaired tissue. Long-term follow-up will be required for future studies. The intraarticular injected cells were not labeled for in vivo tracking cell fate. It is uncertain whether the cells survived following intraarticular injection, and if MSCs survived postinjection, it is uncertain where MSCs resided after injection.
Conclusion
This study focused on subchondral bone as a bed for cartilage repair, with an intraarticular injection of 1.25 or 6.25 million MSCs enhancing midterm osteochondral defect repair. An intraarticular injection of 1.25 or 6.25 million MSCs promoted the repair of subchondral bone and cartilage even though it was not perfect.
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by Highway Program for Realization of Regenerative Medicine to Prof. Mitsuo Ochi from Japan Agency for Medical Research and Development (AMED). It was also supported by the Egyptian Joint Supervision Scholarship from the Egyptian Cultural Affairs and Missions Sector. No benefits in any form have been received from commercial organizations related directly or indirectly to this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for this study was obtained from laboratory animal science research (Graduate School of Biomedical & Health Sciences, Hiroshima University, Japan) (APPROVAL NUMBER: A13-128).
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
