Abstract
Background:
It is unclear whether antecedent primary malignancies (APMs) have any negative impact on the prognosis of soft tissue sarcoma (STS). We retrospectively reviewed STS patients with APMs (STS-APM) and compared their survival to those of STS only (STS-O).
Methods:
Twenty-one cases of STS-APM from 2008 to 2017 in our institution were analyzed. One hundred and seventy cases of STS-O at the same period were compared as a control group. Overall survival was estimated using Kaplan–Meier survival curves and prognostic factors were analyzed using logistic regression analyses and contingency table analyses.
Results:
As the final status of STS-APM patients, 12 patients were in disease-free survival, 5 were alive with disease, 3 have died of disease, and 1 has died of another disease. There was no case that died of APM. The 5-year overall survival rates were 88% in STS-APM and 78% in STS-O, showing no statistical significant (p = 0.65). The 5-year overall survival rates in each stage of STS-APM and STS-O were 100/100% in stage I, 100/85% in stage II, 86/72% in stage III, and the 3-year overall survival rates were 67/51% in stage IV, with no statistical significance. With regard to prognostic factor, histological grade of STS was the only significant factor. Although antecedent radiotherapy tended to show a high odds ratio, the association was not statistically significant. Antecedent chemotherapy did not show any estimated prognostic risk.
Conclusions:
Our study suggested that APM in STS patient would not be a negative prognostic factor.
Introduction
Soft tissue sarcoma (STS) is reported to account for approximately 1% of all malignant tumors, 1,2 consequently it is thought to be a rare malignant neoplasm. The population of patients with malignant tumors in Japan has been increasing in recent years due to the growth of the elderly population. While the survival of patients with malignant tumors has improved with the recent developments in diagnostic modalities and treatment, there has been an increase in cases involving individual patients with multiple primary malignancies. There have been several reports about the influence of multiple primary malignancies on the prognosis of tumors that are more frequently malignant, for instance breast cancer or prostate cancer. 3 –5 However, there have been very few reports about the relationship between preceding primary malignancy and the prognosis of STS. In the present study, we compared STS patients with/without antecedent malignancy, focusing on their clinical features and prognosis.
Patients and methods
The prospectively collected database of our hospital included 414 patients with STS who were treated from August 2008 to July 2017. Atypical lipomatous tumor/well-differentiated liposarcoma, solitary fibrous tumor, hemangioendothelioma, gastrointestinal stromal tumor, patients with palliative treatment only, and second opinion cases were excluded; thus, 191 patients were included in the analysis. After receiving Institutional Review Board approval, medical records were reviewed to compare the clinicopathological differentiation of patients with STS and antecedent primary malignancies (APMs) (STS-APM), and patients with STS only (STS-O). The demographic factors, STS characteristics, treatment, and prognostic outcome were also reviewed.
STS-specific survival was defined as the duration from the date of the diagnosis of sarcoma to the date of the patient’s death due to STS. There were no cases of death due to APM. Cases in which the patient was alive were censored at the time of the last follow-up examination. Statistical analyses were conducted with the JMP software program (Version 13.0 for Macintosh; SAS Institute Inc., Cary, North Carolina, USA), and p values of <0.05 were considered to indicate statistical significance. Survival was estimated using Kaplan–Meier survival curves and compared using the log-rank and Wilcoxon tests. For further analysis of prognostic factors, logistic regression analyses and contingency table analyses were performed. The significance of differences was calculated using the χ 2 test and Fisher’s exact test.
Results
Among the 191 patients, there were 21 (11%) patients with STS-APM and 170 with STS-O. The STS-APM group included 11 male patients and 10 female patients, while the STS-O group included 89 male patients and 81 female patients. The chronology of the multiple malignancies was defined as synchronous if malignancies were detected within 6 months of each other and metachronous if one was detected more than 6 months after the other. Among the 21 STS-APM patients, 3 cases were considered to be synchronous and 18 were considered to be metachronous. The mean age at the time of the diagnosis of STS was 71 years in the STS-APM group (median 70, range 49–89 years) and 58 years (median 62, range 13–101 years) in the STS-O group. The mean age of the STS-APM patients was significantly higher than that of the STS-O patients (p = 0.0026). In the STS-APM group, STS was located in the extremities in 11 patients (52%) and the trunk in 10 patients (48%); in the STS-O group, STS was located in the extremities in 101 patients (59%) and the trunk in 69 patients (41%); there was no significant difference in the locations of the two groups (p = 0.55). The histopathological diagnoses of sarcoma in the STS-APM group were as follows: undifferentiated pleomorphic sarcoma (UPS; n = 7), myxofibrosarcoma (n = 4), angiosarcoma (n = 2), postradiation sarcoma (n = 2), myxoid liposarcoma (n = 1), leiomyosarcoma (n = 1), synovial sarcoma (n = 1), malignant peripheral nerve sheath tumor (MPNST; n = 1), and extraskeletal osteosarcoma (n = 1). The histological diagnoses of sarcoma in the STS-O group were as follows: UPS (n = 37, 22%), myxofibrosarcoma (n = 27, 16%), myxoid liposarcoma (n = 23, 14%), MPNST (n = 20, 12%), dedifferentiated liposarcoma (n = 18, 11%), leiomyosarcoma (n = 11, 6%), synovial sarcoma (n = 9, 5%), angiosarcoma (n = 4, 2%), extraskeletal osteosarcoma (n = 4, 2%), Ewing sarcoma/Primitive neuroectodermal tumor (PNET) (n = 3, 2%), and others (n = 14, 8%). The histopathological diagnoses of the APMs were as follows: breast cancer (n = 7), prostate cancer (n = 5), colon cancer (n = 4), esophageal cancer (n = 2), gastric cancer (n = 1), thyroid cancer (n = 1), uterus cancer (n = 1), bladder cancer (n = 1), and malignant lymphoma (n = 1). Two cases had two individual APMs. The treatments of APMs were as follows: surgery only (n = 11), surgery + chemotherapy (n = 3), surgery + radiotherapy (n = 3), chemotherapy (n = 2), and radiotherapy + chemotherapy (n = 2). With regard to the histological grade of STS, there were 2 low-grade cases and 19 high-grade cases in the STS-APM group and 40 low-grade cases and 130 high-grade cases in the STS-O group. The high- and low-grade distributions were not significantly different between the two groups (p = 0.12).
The stages of STS, according to the Union for International Cancer Control (UICC) system, in the STS-APM group were as follows: stage I (n = 1), stage II (n = 6), stage III (n = 11), and stage IV (n = 3). Those in the STS-O group were stage I (n = 39, 23%), stage II (n = 43, 25%), stage III (n = 70, 41%), and stage IV (n = 18, 11%).
The treatments of STS in the STS-APM group were as follows: surgery alone (n = 15), surgery + chemotherapy (n = 2), surgery + radiotherapy (n = 1), radiotherapy + chemotherapy (n = 1), chemotherapy alone (n = 1), and radiotherapy alone (n = 1). Those in the STS-O group were as follows: surgery alone (n = 119), surgery + chemotherapy (n = 16), surgery + radiotherapy (n = 13), surgery + chemotherapy + radiotherapy (n = 5), radiotherapy (n = 8), chemotherapy (n = 6), and radiotherapy + chemotherapy (n = 3).
The median follow-up period from the date of diagnosis of the STS in the STS-APM group was 35 months (mean 38 months, range 4–87), while that in the STS-O group was 27 months (mean 32 months, range 1–120). The median interval between the dates of the diagnoses of APM and STS was 62 months (mean 117 months, range 1–432). Comparison of STS characteristics between the STS-APM and STS-O groups, patient’s age at the time of the diagnosis of STS only showed statistically significant difference. The final statuses in the STS-APM group were continuous disease-free survival (n = 12), alive with disease (n = 5), dead from disease (n = 3), and dead from another disease (n = 1); there were no cases of disease-specific death due to the preceding primary malignancy. The final statuses in the STS-O group were as follows: disease-free survival (n = 91), no evidence of disease (n = 22), alive with disease (n = 27), dead from disease (n = 29), and dead from another disease (n = 1) (Tables 1 and 2).
Characteristics of the 21 STSs with APMs.

UPS: undifferentiated pleomorphic sarcoma; MPNST: malignant peripheral nerve sheath tumor; OS: osteosarcoma; S: surgery; R: radiotherapy; C: chemotherapy; CDF: continuous disease free; AWD: alive with disease; DOD: dead of disease; DOA: dead of another disease; STS: soft tissue sarcoma; APM: antecedent primary malignancy.
Comparison of STS characteristics between the STS-APM and STS-O groups.
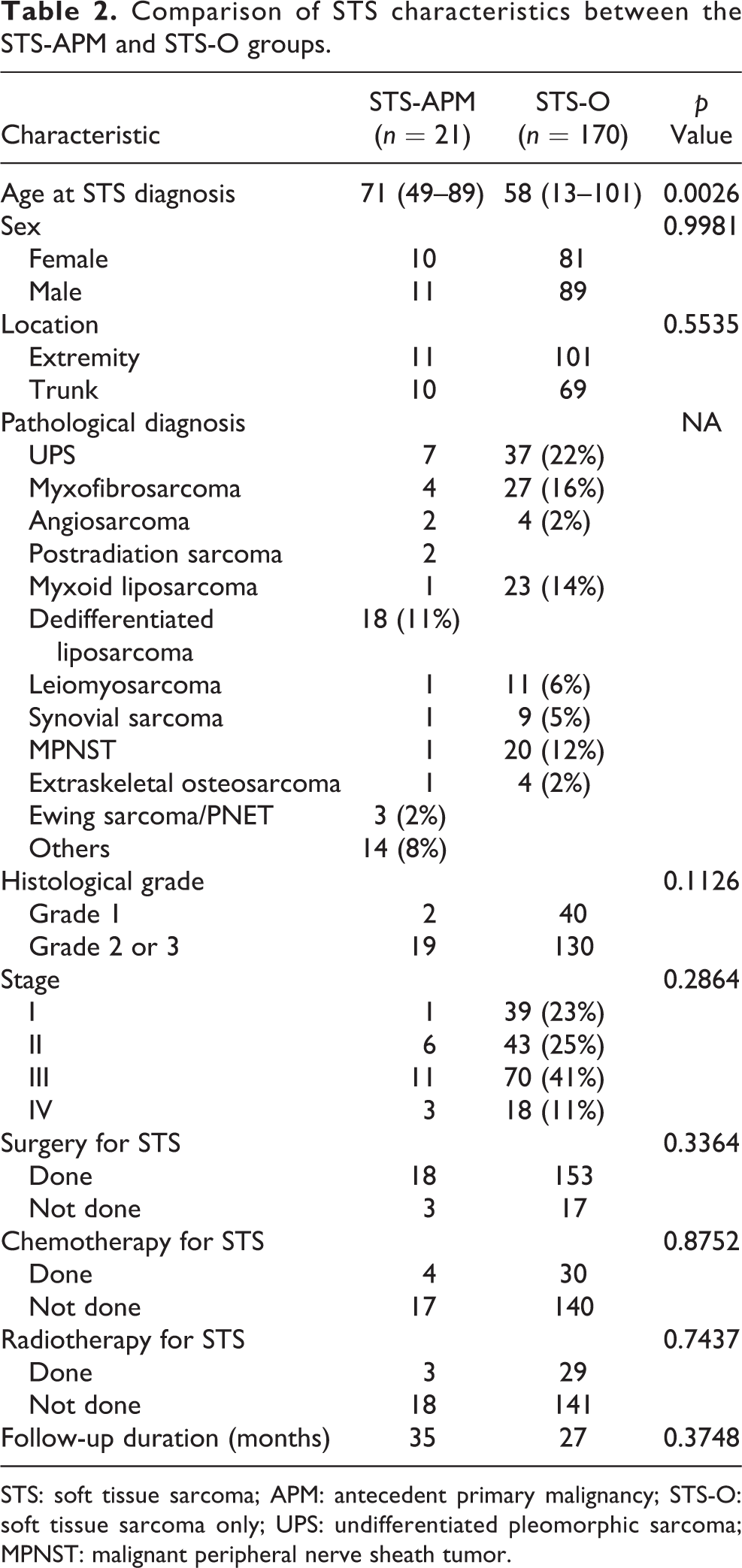
STS: soft tissue sarcoma; APM: antecedent primary malignancy; STS-O: soft tissue sarcoma only; UPS: undifferentiated pleomorphic sarcoma; MPNST: malignant peripheral nerve sheath tumor.
The STS-specific 5-year overall survival rate was 88% in the STS-APM group and 78% in the STS-O group. The prognoses of the two groups did not differ to a statistically significant extent (p = 0.65) (Figure 1).

Comparison of the overall survival between the STS-APM group and STS-O group. No significant difference was observed between the two groups (p = 0.65). STS: soft tissue sarcoma; APM: antecedent primary malignancy; STS-O: soft tissue sarcoma only.
The overall survival rates in each stage of STS-APM/ STS-O were as follows: stage I, 5-year 100/100%; stage II, 5-year 100/85%; stage III, 5-year 86/72%; and stage IV, 3-year 67/51%. The prognoses of the two groups did not differ to a statistically significant extent (Figure 2 (a) to (d)). With regard to the 3-year overall survival rates according to the tumor chronology, the 3-year overall survival in the STS-APM group was 87% in patients with metachronous disease and 100% in patients with synchronous disease; the difference was not statistically significant (p = 0.56). With regard to the 5-year overall survival rates according to the tumor location, the 5-year overall survival rates in the STS-APM group were 100% in patients with tumors located on an extremity and 72% in patients with tumors located on the trunk; the difference did not reach statistical significance (p = 0.051). Logistic regression analyses were performed to investigate the prognostic factors in STS-AMP and STS-O patients. The histological grade of STS was a significant prognostic factor (p = 0.02), while an older age (>65 years) was not significant (Table 3). However, the percentage of high-grade sarcoma in the STS-APM group was 90%, which was higher than the value of 76% in the STS-O group, although as previously mentioned, the high- and low-grade distributions were not significantly different between the two groups. The proportion of high- and low-grade lesions was determined not to have affected the difference in the 5-year survival between the groups. Contingency table analyses were performed to further analyze other associated prognostic factors in the STS-AMP patients. Distant metastasis at the diagnosis of STS and nonsurgical treatment for STS were significantly associated with the prognosis (with a high odds ratio). Radiotherapy for APMs showed a high odds ratio; however, the result was not statistically significant. Chemotherapy for APMs was not associated with any prognostic risk (Table 4).

A comparison of the overall survival in each stage between the STS-APM group and STS-O group. (a) In stage I, both STS-APM and STS-O cases showed a 100% 5-year survival rate. (b) In stage II, STS-APM and STS-O cases showed 100 and 85% 5-year survival rates, respectively (p = 0.43). (c) In stage III, STS-APM and STS-O cases showed 86 and 72% 5-year survival rates, respectively (p = 0.41). (d) In stage IV, STS-APM and STS-O cases showed 67 and 51% 3-year survival rates, respectively (p = 0.51). No significant difference was observed between the two groups. STS: soft tissue sarcoma; APM: antecedent primary malignancy; STS-O: soft tissue sarcoma only.
Associated factors for survival between STS-AMP and STS-O.
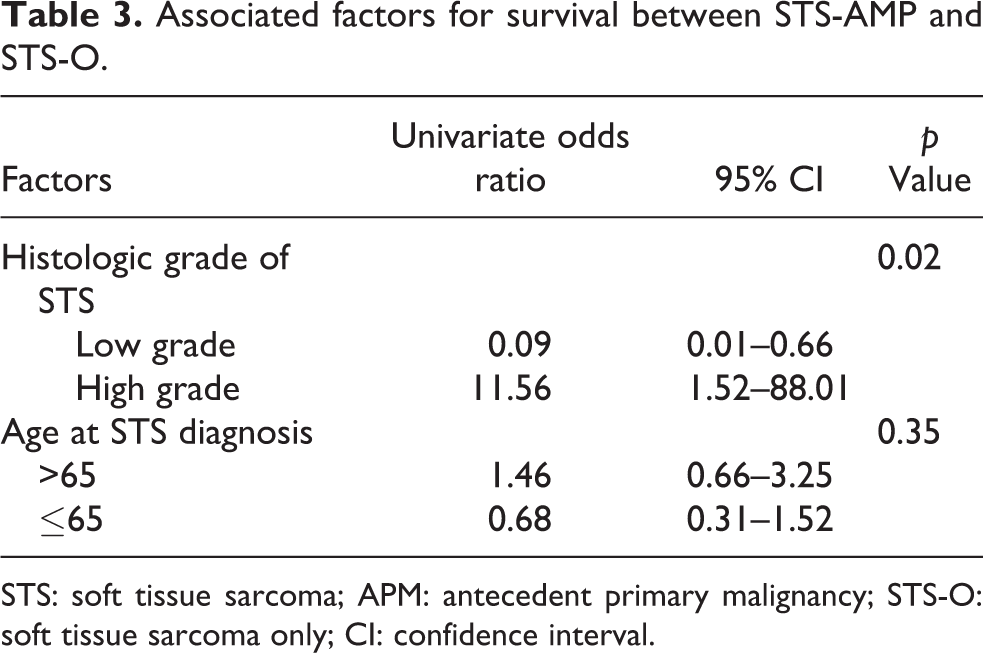
STS: soft tissue sarcoma; APM: antecedent primary malignancy; STS-O: soft tissue sarcoma only; CI: confidence interval.
Associated factors for survival in STS-AMP.

STS: soft tissue sarcoma; APM: antecedent primary malignancy.
Discussion
The first report about multiple primary malignant tumors (MPMs) in an individual was published by Billroth and Winiwarter. 6 Their definition of MPM included the following criteria: different histopathological features were identified in each tumor, the tumors were not in contact and each tumor had individual distant metastasis. However, this definition was considered to be impractical. Warren and Gates later established a more practical definition: each tumor must present a definite picture of malignancy, each must be distinct, and the probability of one being metastasis of the other must be excluded. 7 Although an accurate resolution of the timing of the occurrence of a malignant tumor is difficult to determine in the clinical setting or based on histopathological examinations, the chronology of MPM was defined as synchronous and metachronous. The definition of chronology varied. For example, the diagnostic period between multiple malignancies in synchronous cases ranged from 2 months to 12 months, depending on the surveyor. In the present study, we classified a diagnostic period of <6 months as synchronous, and ≥6 months as metachronous, according to the definition of Moertel et al., 8 because we thought that this was appropriate for the present study.
According to data from Cancer Registry and Statistics, and National Cancer Center Registry in Japan, the estimated number of cancer cases was 532,233 in 2000 and 862,452 in 2013. In this way, the number of cancer cases is considered to be increasing year by year. Previous reports indicated that MPM accounted for 2.4–5.2% of all primary malignancies. 9,10 On the other hand, the 5-year relative survival rate of all cancer cases in 2006–2008 was 62.1%, while that in 1997–1999 was 54.3%. Because the number of cancer survivors has been increasing, it is estimated that the number of patients with MPM has also been increasing. According to the National Cancer Center Registry and Statistics in Japan, the annual incidence of STS was estimated to be 3.6 per 100,000 people and corresponded to the definition of so-called rare cancer, in that the annual incidence rate was <6 per 100,000 people. There have been few reports about MPM with STS. The reported incidence rates of MPM either before or after STS were 2.3, 6, 7.5, 8.6, and 21%. 11 –14 Tateishi et al. reported that the total incidence of MPM in patients with STS as 8.6%, while the incidence rates before and after STS were 3.7 and 4.9%, respectively. 14 In the present study, the incidence of STS-APM was 11%, which was similar to previous reports. Although STS is a rare cancer, MPM in STS is not considered to be rare.
With regard to carcinoma preceding STS in previous reports, breast cancer was the most frequent, followed by prostate cancer. In the present study, 7 breast cancer patients and 5 prostate cancer patients were observed among 21 patients with STS-APM; the results were consistent with the findings of previous reports. According to data from Cancer Registry and Statistics, National Cancer Center in Japan 2016, breast cancer and prostate cancer were the most frequent causes of major cancer, accounting for 19% of the cancer in female patients and 16% of the cancer in male patients; the 5-year relative survival rate was more than 90%. We considered that these cancers were frequently observed in STS-APM due to their high incidence and the long survival of patients with these cancers. Radiotherapy and chemotherapy are associated with an increased risk of developing a second malignant neoplasm. 15 –17
In the present study, the preceding malignancy was treated by surgery alone in 11 of 21 cases. Five of 21 cases were treated by radiotherapy with or without surgery and chemotherapy. This study included two cases of postradiation sarcoma within the irradiated fields (uterine carcinoma, n = 1; malignant lymphoma, n = 1). With the exception of postradiation sarcoma, the relationship between the treatment of preceding malignancy and sarcoma was still unclear.
In previous reports, the most popular diagnosis of patients with MPM with STS was malignant fibrous histiocytoma (MFH) or UPS, followed by myxofibrosarcoma. 11 –14 Seven UPS cases, 4 myxofibrosarcoma cases and 1 myxoid liposarcoma case were observed among the 21 STS-APM cases in the present study; the tendency was in line with previous reports. According to the soft tissue tumor registry in Japan 2014, liposarcoma was the most frequent STS, with an incidence of 36%, followed by MFH (UPS; 15%), and myxofibrosarcoma (9%). This was considered to be caused by the fact that UPS and myxofibrosarcoma showed a high incidence rate in STS-APM due to the relatively large population of these sarcomas.
There were very few reports that discussed the prognostic difference between patients with STS-AMP and STS-O.. They have reported that the sarcoma-specific prognosis in MPM-STS was similar to that of the STS-O. The effect of the chronology on the prognosis was also reported to be equivalent in synchronous and metachronous cases of STS-APM, which was in line with the findings of the present. 11
It has been suggested that postradiation sarcoma may represent a subgroup of sarcomas with a poor prognosis. 18 –20 However, recent studies did not find an inferior prognosis in patients with postradiation sarcoma. 21,22 The prognosis of postradiation sarcoma is still controversial. In the present study, although cases of postradiation sarcoma tended to be associated with a poor prognosis in comparison to other STS-APM cases, the association was not statistically significant (p = 0.079). However, there were 18 cases treated with surgery and 3 cases treated with radiotherapy and/or chemotherapy. The nonsurgically treated cases of STS-APM showed a higher odds ratio for surviving than the surgically treated cases. Two cases of postradiation sarcoma were given nonsurgical treatment, mainly due to the site of occurrence. We suspect that the selection of treatment may have influenced the prognosis, although whether or not antecedent radiotherapy itself was an independent prognostic factor is unclear. Although there are many reports about the risk of developing secondary malignancy after chemotherapy, post-chemotherapy STS has not been outside of pediatric malignancies. 23 –25 In our study, there were no STS-AMP cases with a past history of pediatric malignant tumors as a preceding malignancy and no cases were considered to be chemotherapy-induced STS. Furthermore, chemotherapy for APMs did not show any prognostic risk.
Our study is associated with several limitations. First, this was a retrospective study, with the selection biases that are inherent in such studies. Secondly, the number of patients was relatively small. Finally, the prognosis might have been improved because patients receiving palliative treatment were excluded. The prognosis might have been worse if we included cases who received palliative treatment alone.
Conclusions
In this study, the incidence of MPM in patients with STS was 11%, and was not considered to be a rare phenomenon. There was no statistical evidence to suggest that APM was associated with a worse prognosis in STS patients.
Footnotes
Authors’ note
The authors state that the manuscript has been read and approved by all the authors and that the requirements for authorship as stated earlier in this document have been met and that each author believes that the manuscript represents honest work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
