Abstract
Due to accurate preoperative imaging techniques, early diagnosis and effective chemotherapy, many tumors arising in the metaphyseo-diaphyseal regions of long bones can be segmentally resected with joint preservation. The intercalary resection of malignant bone tumor results in a bone defect which can represent a challenging reconstructive problem. The most commonly used surgical reconstructive options for these defects include biologic reconstructions such as allografts, vascularized fibular grafts, autogenous extracorporeally devitalized tumor bearing bone graft, combination of allografts or devitalized autografts with vascularized fibular grafts, segmental bone transport, or induced membrane technique. Nonbiologic reconstructions, on the other hand, use intercalary endoprostheses. Every patient should be carefully evaluated and the reconstructive option should be individually selected. The aim of this article is to discuss the surgical options of reconstruction of bone defects after intercalary resection of malignant bone tumors with reviewing of their indications, advantages, disadvantages and complications.
Keywords
Introduction
If intercalary resection of malignant bone tumors can be performed with preservation of the adjacent joints, the expected function is superior as the patient’s native joints above and below the reconstruction are left undisturbed. 1,2 Due to accurate preoperative imaging techniques, early diagnosis and effective chemotherapy, many tumors arising in the metaphyseo-diaphyseal regions of long bones can be segmentally resected with joint preservation. 3 The surgical reconstructive options for these defects include allografts, vascularized fibular grafts, autogenous extracorporeally devitalized tumor bearing bone graft, combination of allografts or devitalized autografts with vascularized fibular grafts, segmental bone transport, bone cement spacer, or intercalary endoprostheses (Table 1).
Different reconstruction options after intercalary resection of malignant bone tumors.

FVFG: Free vascularized fibular graft.
Allografts
Allografts are commonly used, especially in North America and Europe, to reconstruct the osseous defects after intercalary resection of malignant bone tumors of long bones (Figure 1). It has been reported that intercalary allografts can achieve good limb function with long-term success rates. 4,5 The main advantage of the allografts is the ability to biologically reconstruct an osseous defect while preserving host bone stock without donor site morbidity. 6,7

Showing reconstruction with intercalary allograft after resection of adamantinoma. 7
Previous studies have reported that allografts can survive for several decades. 6,8 However, they are associated with high rates of complications. The reported incidence of complications has varied from 0% to 18% for infection, 0% to 30% for fracture, and 15% to 55% for delayed union or nonunion. Most of these complications usually occur within the first 3–4 years after the index surgery. 4,9 Widespread use may be restricted by their limited availability in some Asian countries due to socio-religious reasons and by the risk of infectious diseases transmission to the host. 4,5,7,10,11 It has been reported that a higher rate of fracture was observed at the sites of screw holes, suggesting that allografts are very sensitive to stress-concentrating defects. So that obtaining extracortical support by spanning the entire allograft with a long plate may lower the risk of fracture. 12,13
Hornicek et al. 14 reported that chemotherapy may negatively influence the healing of allograft–host junctions. The incidence of nonunion was higher in patients receiving chemotherapy. Other studies reported the effect of internal fixation on the healing of massive allografts and showed that achieving stable fixation could diminish the development of nonunion. However, there was no difference of union rates after fixation with a plate or intramedullary fixation. 5,13 The nonunion rate may be higher in diaphyseal than metaphyseal osteotomies and this may be attributed to the better consolidation rate in metaphyseal regions. 5,10
In a multicenter study, Bus et al. 7 observed 17% incidence of graft failure in their series due to either graft-related complications (infection, fracture, nonunion) or local recurrence. Most of these complications (13%) occurred in the first 4 years after the primary surgery. Failure rates were significantly higher in patients who were 18 years of age and older and in patients undergoing reconstructions with an allograft of 15 cm in length or longer. They also found that complications occurred in 76% of the patients with 46% of them had two or more complications and reoperations were performed in 70% of the patients.
Vascularized fibular graft
Free vascularized fibular graft (FVFG) was firstly reported in 1977 by Weiland et al. 15 for reconstruction of long bone defect after resection of bone tumor. Since then, the FVFG has become a popular biological reconstructive option for limb-sparing surgery after resection of the malignant bone tumors. 16 Vascularized bone grafts don’t undergo resorption by creeping substitution due to their intact vascularity. FVFG can incorporate into the adjacent host bone directly via bone union similar to the process of healing of fractures. Due to the viability of FVFG, the structural integrity is well preserved until the bony union is achieved. Furthermore, soft tissue structures like skin, fascia, and muscle may be harvested concomitantly with the fibula and used for more complex soft tissue reconstruction. 17
FVFG can be used in three forms of reconstructive options: (1) Single vascularized fibular graft which is mainly indicated for reconstruction of areas with lighter stress loads as in upper extremity reconstruction, segmental defects of the mid-tibia, and intercalary defects in pediatric patients. (2) Vascularized double-barreled fibula which can be indicated for areas with intermediate stress loads such as femur and pelvis. This technique can provide the double volume of a fibular graft for the same length of the defect with the same number of the microvascular anastomosis. (3) In combination with an allograft or devitalized autograft such as pasteurized or irradiated autograft. 16,18 –20
Despite these advantages, the fibula has small cross-sectional area and is weaker than the originally resected long bone, particularly the femur. The sufficient fibular thickness to allow full weight-bearing is achieved several years after the surgery as the fibula needs a long time to undergo hypertrophy through processes of pressure transport, microfractures, and callus formation. Therefore, a long period of non or partial weight-bearing is necessary. 21,22
The reported complications include recipient site complications (fatigue fractures of the fibular graft, delayed union/nonunion, infection, loss of flap, and hardware failures) and donor site complications (bleeding, peroneal nerve palsy, contracture of flexor hallucis longus, ankle pain, instability, and/or progressive valgus deformity commonly in children). 17,23 –25
Combined vascularized fibula and allograft reconstruction
Capanna et al. 26,27 introduced the procedure of combining an allograft with a FVFG for large bony defects after tumor resection. Each of allograft and FVFG has its own specific complications when used alone for reconstruction of intercalary defects. To overcome these complications, it is better to combine their use. The rational of this approach is to combine the biological activity of the FVFG with the initial mechanical strength of the allograft. Also, the vascularized graft facilitates the host–allograft union and minimizes allograft failures. 27 –29 During the first postoperative years, the allograft supports the weak vascularized fibula structurally, without depriving it completely from weight-bearing. This exposure to weight-bearing stresses induces a progressive concentric fibular hypertrophy of the fibular graft. 1,29,30
Allograft can be augmented with the vascularized fibular graft using either an intramedullary or an on-lay techniques. The intramedullary technique is considered when the vascularized fibula is inserted through a trough opened in the anterolateral cortex of the allograft. The intramedullary technique is more commonly indicated for femoral and tibial reconstructions. 31 Ideally, the length of the vascularized fibula should be 5 to 6 cm longer than the intercalary defect, so that at least 2 cm of vascularized bone can be inserted into both host ends. The on-lay technique may be considered if the intramedullary canal of the allograft does not allow the fibula to be inserted. This technique is more easily performed in the femur than in the tibia because of more soft tissue coverage. The intramedullary technique is usually preferred over the on-lay technique because the irregular contour of the allograft and the host bone doesn’t allow the optimal bone-to-bone contact to be achieved with the on-lay technique. 32
Capanna et al.27 reported a 93.3% graft survival after an average follow-up period of 9 years. Previous reports suggested that FVFG is more resistant to infection due to its own vascularity. 27,33 The osteogenic potential of the vascularized fibular graft lowers the risk of nonunion. A review of literature reveals that nonunion rates for combined allograft and vascularized fibula reconstructions are less compared to the allografts alone (up to 31% and 46%, respectively). 27,30,33 –35 Also, hypertrophy of the vascularized fibula can compensate the resorptive weakening of the allograft due to creeping substitution leading to lower fracture risk. 29,30,32
This technique may be associated with complications such as infection, fracture, nonunion, or donor site complications. Rabitsch et al.29 reported that 50% of the patients of their series had additional surgical interventions to treat such complications. The event-free survival rate was 64% after 2 years and 39% after 3 and 5 years. The reported fracture rates were 0% to 33.3%, nonunion rates were 4% to 33.3%, and infection rates were 0% to 16.6%. 27,29,30,33,34 Also, the risk of anastomosis failure by thrombosis should be kept in mind.29
Segmental transport
Limb-sparing surgery using distraction osteogenesis after resection of bone tumors was firstly introduced by Tsuchiya et al. 36 in the management of 19 patients using three different procedures: type I: conventional bone transport, type II: shortening distraction, and type III: bone transport or shortening distraction combined with an intramedullary nail to reduce the total period of external fixation. Gradual distraction was started 7–14 days after the operation with a rate of 1 mm per day. Distraction osteogenesis has the ability to regenerate living bone which can remain permanent once it is found (Figure 2). 36 –38
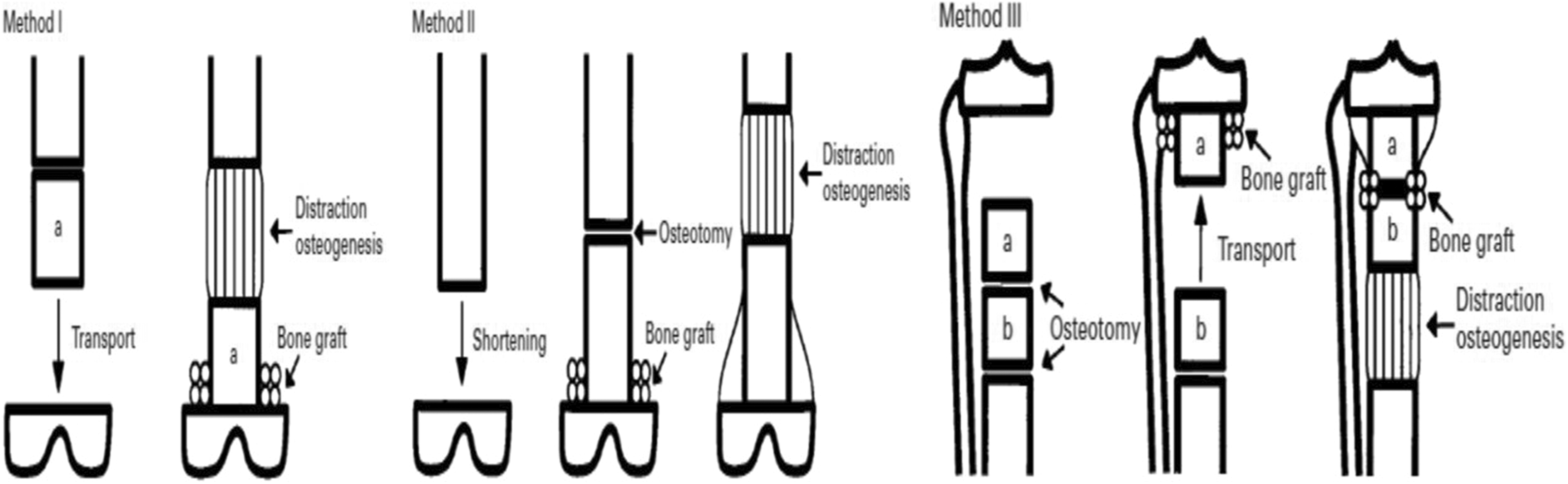
Diagram showing three different methods of intercalary reconstruction by bone transport (distraction osteogenesis). 38
Previous studies reported the effects of common chemotherapeutic agents as doxorubicin, methotrexate, cyclophosphamide, dactinomycin, bleomycin, and caffeine-potentiated chemotherapy on distraction osteogenesis. They observed that these agents didn’t have any significant adverse effects on bone regeneration. 36,38 –40 Furthermore, Minematsu et al. reported that distraction osteogenesis allowed the regional blood flow to the area of tumor to be within the normal range or higher. 41
However, this technique is time-consuming and needs the patient’s cooperation. Also, it has some complications including pin tract infection, delayed consolidation, union problems at the docking site, and may have malalignment especially in patients with larger bone defects. Because of these complications, this treatment option should be preferred for the management of bone tumors in children and patients with a long-life expectancy. 22,42
Autogenous extracorporeally devitalized tumor bearing bone graft
Biological reconstruction performed via reuse of the devitalized resected tumor-bearing bone is common in certain Asian countries as the concept of bone donation is not widely accepted. The techniques of devitalizing procedures include irradiation, autoclaving, pasteurization, or freezing with liquid nitrogen. Reconstruction is performed by reimplantation of the devitalized autograft and stabilization by suitable osteosynthesis. Major advantages of these procedures are that the reconstruction surgery can be performed more easily as the dimensions of the devitalized autograft precisely match the host bone. There is no risk of disease transmission or graft rejection due to immunological reaction. 43 –46 Moreover, devitalized autograft acts as a scaffold for creeping substitution as the graft is invaded by viable bone tissue with progressive substitution from peripheral adjacent host bone. 44,45
Potential complications in extracorporeal devitalized autografts include nonunion/delayed union, infection, fracture, epiphyseal slip in younger patients, bone resorption, and graft necrosis. 1,2
Extracorporeal irradiated autograft
Reimplantation with extracorporeally irradiated autograft was firstly described by Spira and Lubin 47 in 1968 for the management of bone sarcoma. After resection of the tumor-bearing bone, the soft tissue part of the tumor was removed and the resected bone was devitalized by extracorporeal irradiation and then reimplanted and fixed to host bone with suitable osteosynthesis. 48
Vascularized fibular graft was recommended in some studies to augment the reconstruction with irradiated autograft (Figure 3). 35,49 –52 While the vascularized fibular graft can improve the vascularity of the recycled autograft, the surgery time is much prolonged and there is an associated risk of donor site complications. The fibula also does not significantly improve the structural strength of the devitalized bone. 49 Nonunion rate of 7% to 31% has been reported in intercalary reconstructions using irradiated autografts supplemented with vascularized fibular grafts. 35,50,52

Showing intercalary reconstruction of osteosarcoma of distal femur with vascularized fibular graft augmented with irradiated autograft. 18
Other studies 48,53 recommended filling the medullary cavity of the irradiated bone with bone cement or gentamicin containing bone cement to improve the mechanical strength of the bone graft. They believed that the cement augmented autografts has many advantages including improvement of mechanical strength of the recycled autograft, and better screw hold and pull out strength of the screws fixed to bone cement.
Extracorporeal autoclaved autograft
Autoclaved autografts are usually indicated in osteoblastic tumors, where more than two-thirds of the cortex were preserved. Although osteoinduction property of autograft is lost by autoclaving procedure, the osteoconduction is preserved. Incorporation at the junctions between the graft and host bones is a result of the periosteum of the host bone and forms around the autoclaved graft. 54,55
During the surgical procedure, wide excision of the tumor-bearing segment is performed, and the soft-tissue components are grossly removed. Then the tumor-bearing bone is heated in an autoclave at 120°C for 10 min after removal from the autoclave; the remaining soft tissue can be easily scraped off from the surface of the bone; and the specimen is then cleaned with normal saline and prepared for reinsertion. The whole procedure should be performed under sterile conditions, with sterile wraps used for transport between the surgical field and the autoclave. 56
The disadvantages of the autoclaved bone graft are that it takes a long time for revascularization and incorporation into the surrounding bone. The autoclaved bone itself is brittle, and there is possibility of graft resorption or fracture. 22,57 Khattak et al. 58 reported mean union time of 24.2 months and infection rate of 41.6%. Pan et al. 56 reported that the rates of nonunion, infection, and fracture were 30%, 20%, and 20%, respectively.
Extracorporeal pasteurized autograft
Pasteurization was proven to be an effective method for devitalizing the tumor-bearing bone. Pasteurization can kill the malignant cells while the mechanical strength and bone-inducing property of the graft can be preserved. Pasteurization is the technique of heating the resected tumor bone segment at a low temperature (60°C) for 30 min. 44 –46,59,60
The superiority of pasteurization technique over other methods of heat treatment such as autoclaving or boiling was proved by previous reports of the literature. 61 Urist et al. observed that in heat treatment with the temperature greater than 70°C, the biologic activity of the bone morphogenetic protein (BMP), which is essential for bone induction, is completely destroyed. 62
Potential complications of pasteurized autograft include infection, nonunion, fracture, and bone resorption. Jeon et al.60 reported that the rates of deep infection, bone nonunion, and fracture were 14.3%, 23.8%, and 9.5%, respectively. Han et al. 63 reported infection rate of 15%, nonunion rate of 42%, and fracture rate of 23%.
Extracorporeal frozen autograft
Yamamoto et al.64 have reported that the treatment of osteosarcoma cells with liquid nitrogen is effective both in vitro and in vivo. Freezing procedure with liquid nitrogen destroys tumor cells through the process of ice crystal formation and cell dehydration. Also, the freezing technique can cause thrombosis of the microcirculation leading to ischemic infarction of the tumor cells. 65 Takata et al. reported that bone morphogenetic activity of the frozen autograft was better preserved than autoclaved or pasteurized bone grafts. Additionally, biomechanical properties of frozen autografts were maintained adequately. 64
The use of frozen autografts has multiple advantages; osteoinduction and osteoconduction properties are well preserved, 66 biomechanical strength doesn’t deteriorated, 64 early revitalization with possible cryoimmunological effects and they don’t contain harmful denatured substances. The freezing procedure requires less equipment than other recycling techniques. 43,65 Also, frozen autograft does not require augmentation with vascularized fibular graft as in allograft or pasteurized autograft. 67,68
The freezing procedure protocol is performed according to the following steps: the tumor-bearing segment is frozen with liquid nitrogen at −196°C for 20-min followed by thawing at room temperature for 15 min and finally rinsed with distilled water for 10 to 15 min. Two main types of freezing procedures were prescribed according to the number and sites of osteotomies. The first procedure is the free-freezing procedure. In this type of freezing procedure, either two osteotomies or a hemicortical resection is done. Thereafter, the tumor-bearing segment is immersed in liquid nitrogen, without anatomical continuity between the resected and healthy host bone. The second type is the pedicle-freezing procedure in which a single osteotomy is performed at one end of the tumor-bearing segment. The tumor-bearing bone was rotated and frozen in liquid nitrogen while the anatomical continuity of the tumor-bearing bone with the healthy host bone is maintained at one of its two ends (Figure 4). 3,69
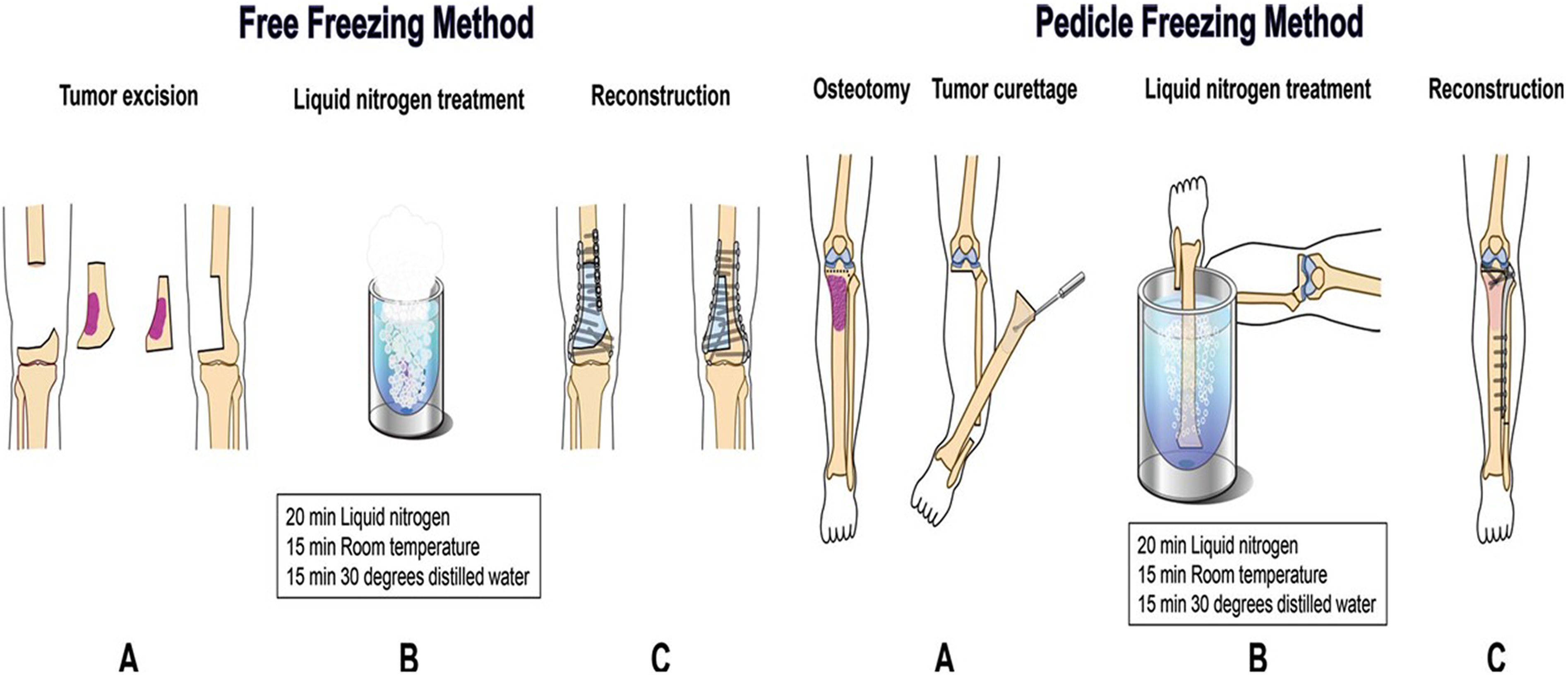
Illustration showing the free-freezing method and the pedicle freezing method. 3
Segmental prosthesis
An intercalary prosthesis is an option to provide immediate stability, early weight-bearing, and rapid rehabilitation, but complications such as mechanical loosening, implant breakage, and periprosthetic fracture may be higher than that of the biological reconstructions. Aseptic loosening has been considered the major complication of intercalary prosthesis occurring in up to 33% of the patients. Other complications as infection and fracture have been reported to occur in a range of 0% to 3.6% and from 0% to 16%. 70,71,72 –74
Therefore, the intercalary prosthesis may be limited to metastatic, lymphoma and multiple myeloma patients. 1,29,70,71 They are ideally indicated for elderly patients due to poor healing capacity or patients with limited life expectancy in whom immediate restoration of function is important than durability and to provide immediate stability by cementation, without having to wait for healing. 75,76 The use of segmental prostheses in young patients was complicated by low implant survival rates with high potential for late failure. While biological reconstructions achieve a stable construct after the first years of surgical interference, segmental prostheses continue to fail. 70,71,73 Aldlyami et al.72 reported that the cumulative overall survival for the prosthetic reconstruction was 63% at 10 years without achieving a stable plateau.
Bone cement spacer and induced membrane technique
In this technique, after resection of the tumor, the intercalary defect is filled with a bone cement spacer. Masquelet et al. 77 observed that, when a segmental bone defect is temporarily occupied by a polymethylmethacrylate spacer, a reactive inflammatory membrane is created around the spacer in 6 weeks. In a second step, when the bone cement is removed and replaced by cancellous bone graft, this membrane acts as a periosteum, prevents resorption, and secretes growth factors. Apard et al. 78 modified the above procedure and used internal fixation with intramedullary nails instead of external fixation, so that their patients resumed weight-bearing more quickly. However, some surgeons considered that the Masquelet technique is suitable for reconstruction of upper limb defect as the slow bone integration and remodeling might lead to mechanical failure in the lower limb reconstruction. 79
In some studies, bone cement is used as spacer for mechanical augmentation of the internal fixation as interlocking intramedullary nails or combined nails and plates after resection of metastatic lesions of long bones especially for the upper limb reconstruction. 80
Conclusion
Many tumors of the metaphyseo-diaphyseal regions of long bones can be resected with joint preservation resulting in intercalary bone defects. There are many options for reconstruction of these defects including biological and nonbiological techniques. Each of these techniques has its advantages and disadvantages so that every patient should be carefully evaluated and the reconstructive option should be individually selected.Declaration of conflicting interests
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
