Abstract
Purpose:
Interleukin 6 (IL-6) and the acute phase C-reactive protein (CRP) blood concentrations after lumbar spine fusion may be affected by age. The purpose of this prospective observational study was to assess postoperative serum levels of pro-inflammatory IL-6 and CRP after instrumented lumbar spine fusion surgery. We hypothesized that older patients would have increased levels of IL-6 and CRP after surgery.
Methods:
IL-6 and high-sensitive CRP biochemical marker levels were measured before instrumented spinal fusion, and postoperatively at 1 and 3 days, 6 weeks, and 3 months. The 49 patients in this sample were divided into two groups: age ≤ 60 years (n = 23) and age > 60 years (n = 26).
Results:
Acute changes in IL-6 high-sensitivity and CRP from preoperative levels to postoperative day (POD) 1 increased with age. Mean (95% CI) difference between the age-groups in changes of IL-6 at PODs 1 and 3 was 45 pg/ml (10–83, p = 0.014) and 20 pg/ml (5–36, p = 0.021), respectively. Mean (95% CI) difference between groups in changes of CRP at PODs 1 and 3 was 9.6 mg/l (−3.5 to 22.7, p = 0.47) and 24.8 mg/l (−17 to 67, p = 0.33), respectively. Both groups had decreased IL-6 and CRP levels at 6 weeks after surgery compared to the preoperative level.
Conclusions:
Elevation of IL-6 and CRP is stronger in patients over 60 years old after instrumented lumbar spinal fusion. The CRP and IL-6 are sensitive markers for acute postoperative inflammation. Even high acute CRP values do not necessarily indicate postoperative infection.
Introduction
Spinal pathologies can cause significant pain and impairment. In severe lumbar spine pathologies and cases where conservative treatment has failed, spinal fusion in combination with decompression can decrease the level of disability and pain as well as increase patients’ health-related quality of life. 1 –3 However, surgical trauma activates endocrine, metabolic, and inflammatory responses. 4 Local infection control and healing process after spinal fusion is achieved through the inflammatory response where pro-inflammatory cytokines promote healing. One of these cytokines is interleukin 6 (IL-6), which is produced by cells in the immune system. 5,6 IL-6 functions in mediating systemic inflammation that induces the production of acute phase C-reactive protein (CRP) in liver hepatocytes. 7 –10 A rise in serum IL-6 and CRP concentration following spinal surgery correlates with the magnitude of tissue trauma caused by the procedure. 11 –15 Biochemical marker concentrations are higher in patients after instrumented spinal fusion than they are in patients without instrumentation. 9,10 CRP levels are widely used also for detecting surgical site infection after spinal fusion. 10,16 –18
In addition to surgical trauma and infection, the patient’s age may influence the intensity of the inflammatory response. Increased levels of CRP, IL-6, and tumor necrosis factor alpha can be found in healthy older individuals compared with younger persons. 19,20 IL-6 response after hip surgery increases with older age. 21 Age-related pro-inflammatory state can be caused by cardiovascular risk factors and increased morbidity with aging. 20 Furthermore, accumulation of intra-abdominal adipose tissue elevates IL-6 and CRP levels in older people. 22 However, no data have been available on the interactions between IL-6 and CRP levels with age in patients who have undergone instrumented lumbar spinal fusion. After lumbar spinal fusion, these markers might be affected by age.
The aim of this study was to assess whether increased postoperative IL-6 and CRP serum levels after instrumented lumbar spinal fusion surgery are related to age.
Materials and methods
This study hypothesized that older patients would have increased levels of CRP and high-sensitivity C-reactive protein (hs-CRP) after instrumented lumbar fusion surgery compared to those of younger patients. Patients aged 18 or more with successful blood samples were included. Patients with inflammatory diseases or severe cardiovascular, psychiatric, social, or general health disorders were excluded from this study. Results are reported adhering to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. The ethics committee of Tampere University Hospital approved the study protocol.
Patients
Altogether 52 consecutive patients who underwent an elective instrumented lumbar spine fusion and decompression at Tampere University Hospital were recruited for the study. All participants signed an informed consent. Blood samples were collected successfully from 49 of the 52 patients: 3 spinal stenosis patients, 11 isthmic spondylolisthesis patients, and 35 degenerative spondylolisthesis patients. Diagnosis was based on magnetic resonance imaging obtained of patients in a supine position and plain radiographs obtained in a standing position. The main indication for surgery was radicular lower extremity pain. All patients underwent treatment with instrumented open posterolateral lumbar spine fusion using a midline approach. The length of the fusion was one or two level fusion for 35 patients and more than two levels for 14 patients. In addition, transforaminal lumbar interbody fusion was used in 12 patients. Patients were divided into two groups: age ≤ 60 years and age > 60 years (Table 1). Patients did not receive glucocorticoid or immunomodulatory medication during the follow-up. There were no postoperative infections in the study group during the 3 months’ follow-up time based on clinical observations. All operations were performed under general anesthesia.
Participants’ clinical and sociodemographic data.
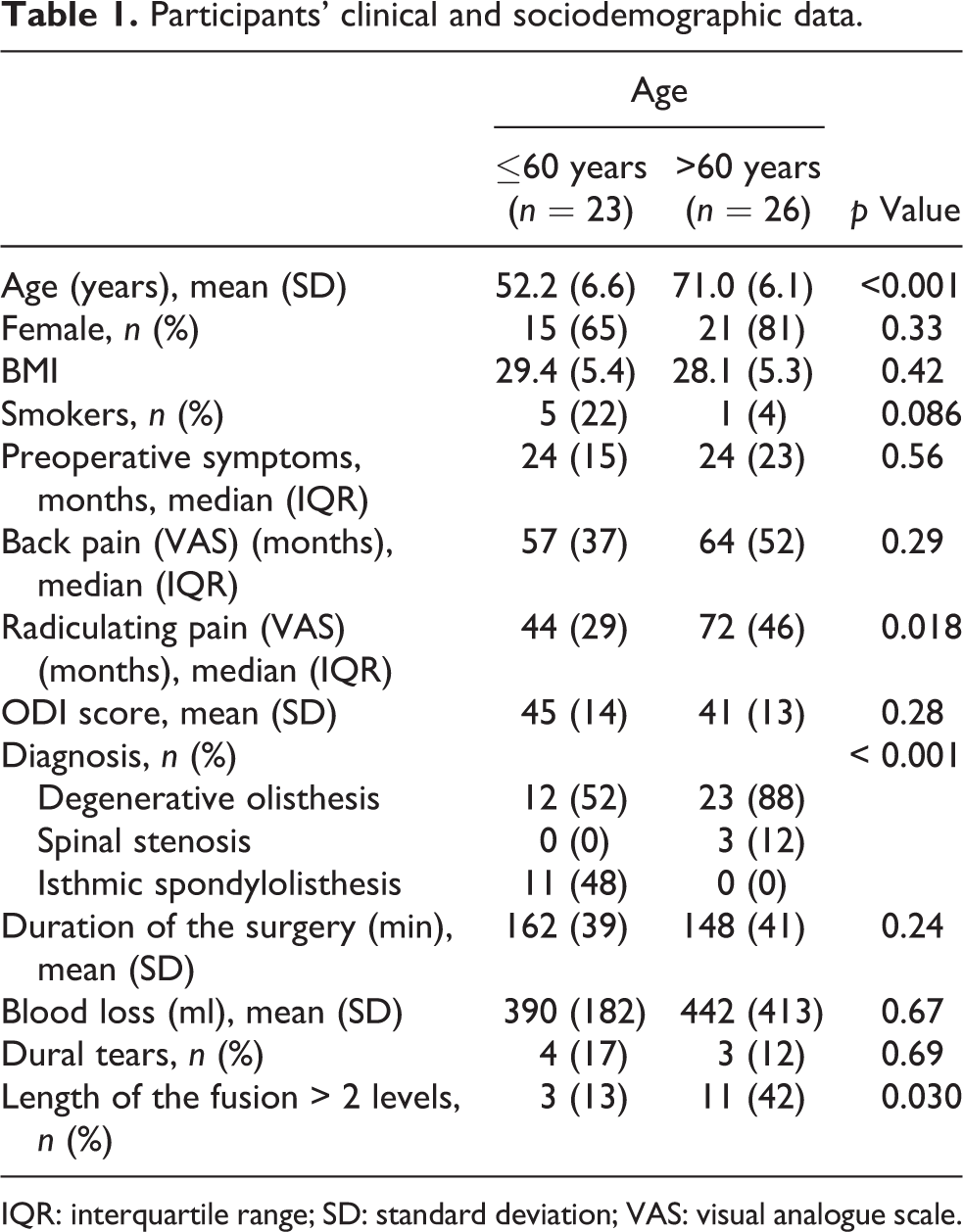
IQR: interquartile range; SD: standard deviation; VAS: visual analogue scale.
Questionnaire compilation
Participants were asked about the duration of their preoperative symptoms, back pain, and radicular pain at the time of admission. In addition to participants’ sociodemographic data, the questionnaire addressed height, weight, medical conditions, current medication, tobacco use, and received treatments. The Finnish version of the Oswestry disability index (ODI) 23 was used to assess participants’ back-specific disability level.
Blood samples
Blood samples were collected pre- and postoperatively between April 2010 and November 2011 at Tampere University Hospital. All samples were collected in the morning before breakfast (fasting blood tests): before surgery as well as after surgery on PODs 1 and 3 at the hospital ward, and 6 weeks and 3 months postoperatively in a follow-up at the outpatient clinic. Blood was drawn from the antebrachial vein into serum vacuum tubes. Serum samples were stored at −80°C until assayed with the Immulite1000 analyzer (Siemens Healthineers Global, Siemens Healthcare GmbH, Erlangen, Germany). Levels of IL-6 and hs-CRP were measured. The hs-CRP accurately measures low concentrations of CRP. It can, therefore, be used for accurate assessment of inflammation level. Blood samples were analyzed using Immulite1000 High Sensitivity CRP test® and Immulite1000 IL6 test®. Measuring ranges were 0.1–60,000 pg/ml for IL-6 and 0.1–3780 mg/l for hs-CRP. The detection limits were 2 pg/ml for IL-6 and 0.1 mg/l for hs-CRP.
Statistics
Data are presented as means with standard deviations (SD), medians with interquartile ranges (IQR), 95% confidence intervals (95% CI), or as counts with percentages. Repeated measures for continuous outcomes (IL-6 and CRP) were analyzed using a generalized estimating equations (GEE) model with the unstructured correlation structure. A possible nonlinear relationship between age and the IL-6 or the hs-CRP values were assessed using 5-knot-restricted cubic spline regression. Models included baseline value, sex, and duration of the surgery as covariates. In cases where the assumptions were violated (e.g. non-normality), a bootstrap-type method was used (10,000 replications) to estimate the standard error. The normality of variables was evaluated by the Shapiro–Wilk W test. Difference of one and at least two-level fusion was assessed using the Mann–Whitney U test. All analyses were performed using STATA 14.1.
Results
Patient details
The patients’ mean age was 71.8 years (range 49–93). Thirteen patients were male and 36 female. The mean (SD) body mass index (BMI) was 28.7 (5.3). There was no significant difference in BMI, gender, or the amount of smokers between the age-groups (Table 1). However, patients older than 60 years had longer periods of radiating leg pain before operation than did patients younger than 60 years (Table 1). Concerning the operation, there were no significant differences between the two groups in the duration of the surgery, blood loss, or amount of dural tears (Table 1).
Biochemical markers
Acute changes in IL-6 from the preoperative level to POD 1 increased with age (Figure 1(a)). The level of IL-6 increased in both groups from the preoperative level to its highest value at POD 1 and thereafter started to decrease, though it was still elevated at POD 3. The increase was higher in the older age-group. Mean (95% CI) difference between the age-groups in changes of IL-6 at PODs 1 and 3 was 45 pg/ml (10 to 83, p = 0.014) and 20 pg/ml (5 to 36, p = 0.021), respectively (Figure 1(a)). At 6 weeks after the operation, IL-6 levels were at a normal (equal to preoperative) level in both groups.
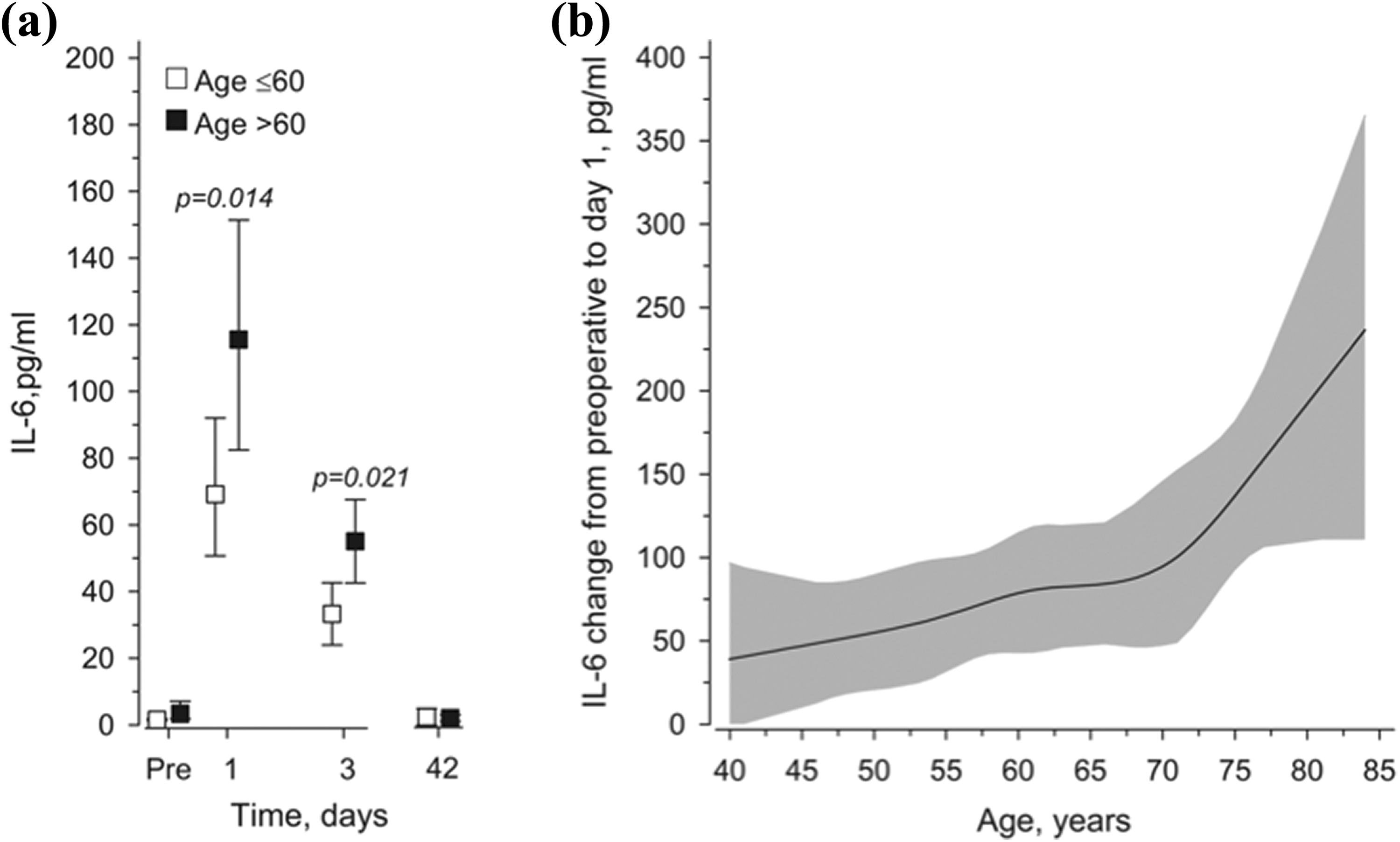
(a) Mean IL-6 values in lumbar spinal fusion surgery patients from preoperative to 6 weeks postoperatively. Adjusted for age, leisure-time physical activity, body fat percentage, and smoking. Whiskers show 95% confidence intervals. Significant differences were found between the age-groups in changes of IL-6 at POD 1 (p = 0.014) and POD 3 (p = 0.021). (b) Relationships of acute changes in IL-6 from preoperative to POD 1 as the function of age. The curves were derived from 5-knot restricted cubic splines regression models. The models were adjusted for leisure-time physical activity, body fat percentage, and smoking. Gray area represents 95% confidence intervals. IL-6: interleukin-6; POD: postoperative day.
There was an increase in hs-CRP from the preoperative level to POD 1 according to age (Figure 2(a)). The hs-CRP level started to increase at POD 1 in both groups and achieved its highest value on POD 3. Mean (95% CI) difference between groups in changes of hs-CRP on PODs 1 and 3 were 9.6 mg/l (−3.5 to 22.7 mg/l, p = 0.47) and 24.8 mg/l (−17 to 67 mg/l, p = 0.33), respectively. Thirteen of the 49 patients had an hs-CRP level over 200 mg/l level measured at POD 3. Furthermore, 11 of these patients were over 60 years. At 6 weeks, hs-CRP levels had decreased to the preoperative level in both groups (Figure 2(b)).
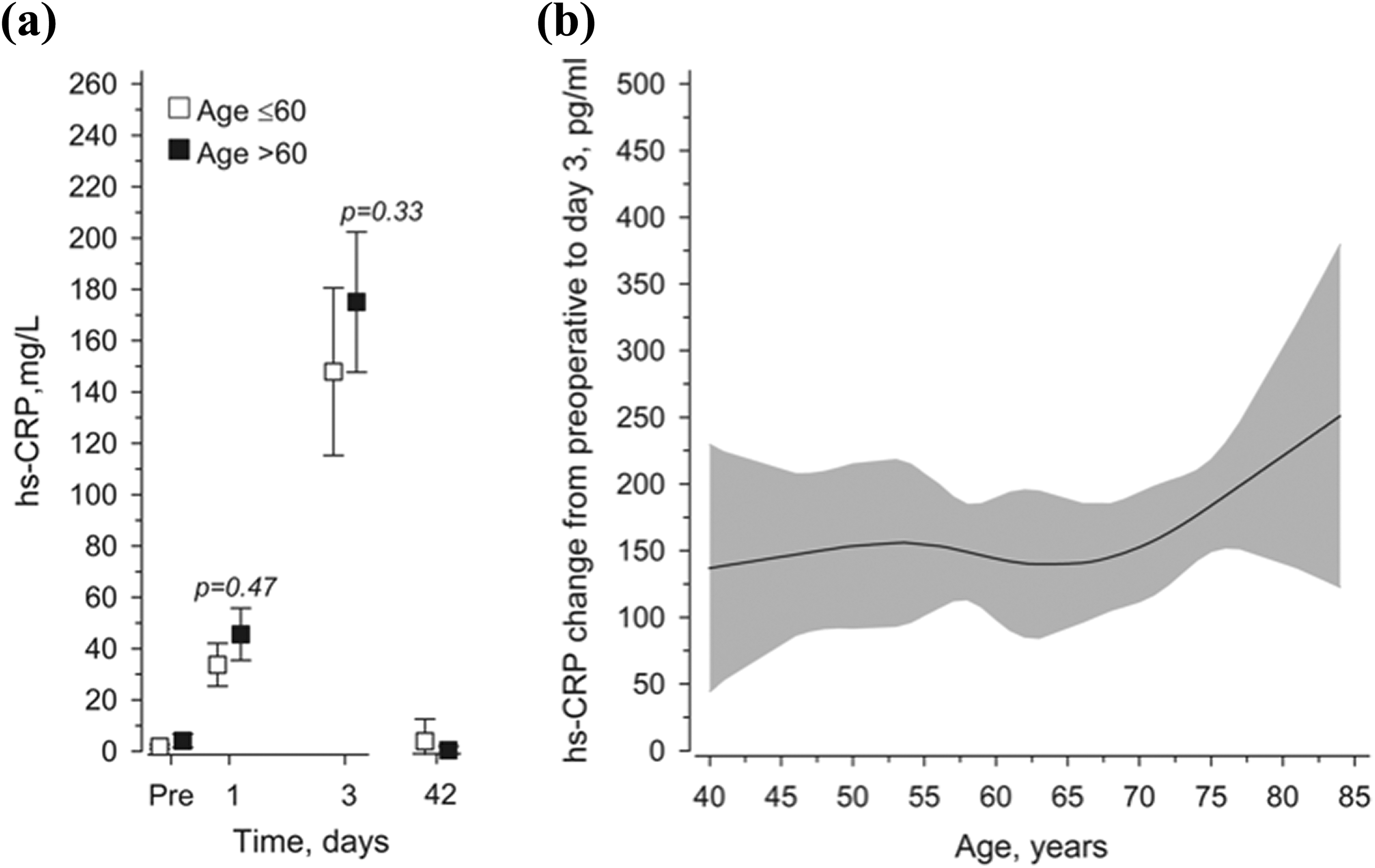
(a) Mean CRP values in lumbar spinal fusion surgery patients from preoperative to 6 weeks postoperatively. Adjusted for age, leisure-time physical activity, body fat percentage, and smoking. Whiskers show 95% confidence intervals. There were no significant differences noted between the age-groups in changes of hs-CRP on PODs 1 (p = 0.47) and POD 3 (p = 0.33). (b) Relationships of acute changes in CRP from preoperative to POD 1 as the function of the age. The curves were derived from 5-knot restricted cubic splines regression models. The models were adjusted for leisure-time physical activity, body fat percentage, and smoking. Gray area represents 95% confidence intervals. hs-CRP: high-sensitivity C-reactive protein; IL-6: interleukin-6; POD: postoperative day.
The correlation was the strongest between the changes from baseline to POD 1 in IL-6 and hs-CRP levels (r = 0.65, 95% CI 0.46 to 0.85). There was a moderate correlation between IL-6 change in POD 3 and hs-CRP change in POD 3 (r = 0.57, 95% CI 0.33 to 0.80), between IL-6 change at POD 1 and hs-CRP change in 3 days (r = 0.40, 95% CI 0.14 to 0.66). No correlation was found between the changes from baseline to POD 3 for IL-6 and to POD 1 for hs-CRP levels (r = 0.16, 95% CI −0.14 to 0.47).
There was a statistically significant difference (p = 0.043) in the POD 3 IL-6 values patients who had undergone one level fusion and those who had undergone at least two level fusion. Other tested inflammation markers in the predefine time points were non-significant.
Discussion
Spinal fusion surgery causes tissue trauma leading to an acute inflammation reaction in all age-groups. In the present study, the inflammatory response was stronger in patients over 60 years, although the duration of the surgery and blood loss were comparable between the two age-groups. To the authors’ knowledge, the present study was the first to investigate whether the levels of inflammatory markers vary between older and younger patients after instrumented lumbar spine fusion. Pro-inflammatory IL-6 and the acute-phase protein CRP are at present the key components in the inflammatory response. In spinal surgery, CRP values are used to evaluate the healing process and the occurrence of postoperative complications such as infection. In the present study, the peak value of IL-6 was highest at POD 1 and the hs-CRP was the highest at POD 3 after the spinal fusion procedure, results which are in line with previously published data. 9,10 This was probably due to the fact that the production of CRP is mediated by the IL-6, a process that takes time. Overall, the inflammatory response of older patients appeared to show higher levels of IL-6 and CRP production.
The timing and quantity of the IL-6 and hs-CRP peak concentrations depend on the extent of damaged tissue 11 –15 and the strength of the inflammatory response. 4,19 In general IL-6 and CRP levels are higher after open surgery than in minimally invasive lumbar spine fusion. 11 –15 Furthermore, the CRP and IL-6 values after instrumented spinal fusion are higher than those after non-instrumented spinal fusion or decompression-only surgery. 9,10 In the present study, all patients underwent open instrumented surgery with midline approach and detachment and retraction of extensor muscles, which results in high IL-6 and CRP levels. In addition, there were 11 patients over 60 years on whom more than two level fusions were performed. These additional levels of fusion instrumentation could partly explain the stronger inflammatory response.
In the present study, changes in serum concentrations of CRP adhered to the IL-6 levels. CRP levels increase during the first three PODs in patients who have undergone spinal surgery. 8,9 Interestingly, statistical significance was found between the two age groups in the IL-6 levels but not in the CRP levels. The absence of a rapid decline or secondary rise in CRP levels after the acute phase indicates postoperative infection. 10,16 –18 Serum IL-6 concentration increases rapidly within 1 day after spinal surgery and decreases within a week to the preoperative level. 8 Thelander and Larsson reported that CRP value reached its peak on POD 3 and decreased to a normal level over 14 days after instrumented posterior lumbar interbody fusion in patients with uncomplicated surgery. 12 IL-6 mediates the production of CRP. This process can take time. Aono et al. reported that 19 of the 143 patients who had undergone posterior lumbar interbody fusion in treatment of degenerative spinal disease had a CRP level of 100 mg/l on POD 4. 18 In the present data, no postoperative infections were observed. At the 6-week follow-up, the hs-CRP values of all patients had decreased back to the preoperative level.
A proper inflammatory reaction is essential for tissue healing and infection control after surgery. 24 However, imbalance between pro- and anti-inflammatory mediators increases the risk of postoperative infection. 25 In addition to weaker preoperative physical condition, the stronger inflammatory response is one reason for slower functional recovery. 26 Aging is also connected with chronic low-grade inflammation. 27 Furthermore, visceral fat and intermuscular thigh fat has been shown to increase IL-6 and CRP concentrations. 28 The present study showed that in addition to chronic inflammation, older patients had stronger acute inflammatory reaction after lumbar spine fusion. The IL-6 concentration increased from the preoperative level to POD 1 along with the age of the patient. The same tendency was noted in the hs-CRP concentrations during PODs 1–3. Muscle mass decreases in the aging process. Thus, hypothetically, trauma caused by tissue stretching in the operation should also decrease. It seems that, in addition to the extent of the tissue trauma, sensitivity to surgical trauma, blood loss, and perioperative stress might cause a stronger inflammatory response in older patients who undergo instrumented spinal fusion. In the present study, the strongest correlation in inflammatory markers was in the changes from baseline to POD 1 in IL-6 and hs-CRP levels.
An unexperienced physician may consider the rise of CRP levels as an indication for infection. To the authors’ clinical experience, when the CRP values have risen above 150, an unnecessary antibiotic treatment may have had initiated during the postoperative recovery phase after lumbar spine fusion surgery. CRP values reach their tip on the POD 3 according to the report by Thelander and Larsson. 12 The present study showed that the decrease in the IL-6 value was already noticeable on the POD 3 after decompression and lumbar spine fusion. Considering these findings, the rise of the CRP values should continue after POD 3 to indicate an underlying infection. The diagnostics of an infection might be postponed as CRP should be measured repeatedly. Based on the results presented here, rise in the IL-6 level on POD 4 would indicate infection. Measuring both the IL-6 and the CRP could give insight for absence or presence of infection.
One of the strengths of this study was its prospective setting, which allowed IL-6 and hs-CRP measurements at distinct time points. Further, the study assessed pro-inflammatory cytokine and acute phase protein as raw laboratory values to bring deeper insight to the inflammation response after instrumented lumbar spine fusion. The study used a comprehensive questionnaire compilation to assess patients’ baseline health state. One could speculate that patients’ visceral or body fat percentage could have been assessed to eliminate bias. However, this was not the scope of the study, and the two groups were comparable in BMI values. The timing of taking the blood samples was based on normal clinical practices just before hospital discharge, and at 6-week and 3-month follow-ups. The blood samples were obtained, for ethical as well as practical reasons, in real clinical settings to guarantee that patients would be inconvenienced as little as possible. The timing was similar as in previously published studies, 9 –18 though this study had a significantly longer follow-up for the final blood sample than did studies with a similar scope. Three of the 52 patients had to be excluded from the analyses as blood samples were not obtained at every time point or patients had already eaten their breakfast. The results are generalizable to patients who have undergone instrumented lumbar spine fusion and decompression. Nonetheless, patients’ general condition, 29 length of the fusion area, or invasiveness of the surgery might have an impact to the postoperative CRP values. As these factors were not specifically investigated in this study, future studies could address this particular question.
Conclusions
The increase in IL-6 and hs-CRP concentrations after the surgery is stronger in patients over 60 years of age. The IL-6 and hs-CRP proved to be sensitive markers for acute postoperative inflammation. Their concentrations returned to normal level 6 weeks after lumbar spine fusion in patients without postoperative infection. Even high CRP values do not necessarily indicate postoperative infection.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study is funded by the Competitive State Financing of the Expert Responsibility Area of Tampere University Hospital.
