Abstract
Background:
Deficient osteogenic capacity of bone marrow stem cells plays a critical role in the pathophysiology of osteoporosis. Adipose-derived stem cells (ADSCs) have emerged as a promising source of skeletal progenitor cells. The capacity of ADSCs to undergo osteogenic differentiation and induce mineralized tissue formation may be beneficial in the treatment of osteoporosis. We question whether administration of autologous ADSCs into the proximal femur of osteoporotic rats will induce osteogenesis and enhance bone quality and strength.
Materials and Methods:
Thirty ovariectomized female rats were randomly assigned to one of the two treatment groups: (1) percutanous implantation of autogenous ADSCs-seeded scaffold into the proximal femur and (2) percutanous implantation of non-seeded scaffold. The contralateral untreated femur served as control. The effect of treatment on bone characteristics was assessed at 12-week follow-up by micro-computed tomography analysis, mechanical testing, and histological analysis.
Results:
The mean cortical thickness, total bone volume density, and bone load to failure in femora injected with autologous ADSCs-seeded scaffold was significantly higher compared to femora injected with non-seeded scaffold and compared to the untreated control femora (p < 0.01). Histological examination of the injected specimens revealed complete osseo-integration of the scaffolds with direct conversion of the ADSCs into osteoblasts and no inflammatory response.
Conclusions:
Autogenous ADSCs implantation into the proximal femur of rats with ovariectomy-related osteoporosis promoted bone regeneration and increased bone strength at short-term follow-up. These findings highlight the potential benefit of autogenous ADSCs in the treatment of osteoporosis.
Level of Evidence:
Level I, randomized controlled trial, animal study.
Keywords
Introduction
Femoral neck fractures in elderly patients are serious consequence of osteoporosis and are associated with considerable morbidity, mortality, and economical burden. 1,2 Current prevention methods focus on patient education, exercise programs, and antiresorptive pharmacotherapy. 3,4 Cement reinforcement of the femoral neck has also been suggested as prevention for osteoporotic fractures. 5,6 Despite the benefits of these prevention methods, they are associated with many disadvantages, which highlight the need for further treatment options. 7,8
The use of mesenchymal stem cells as a regenerative therapy for osteoporosis is a topic of current interest. The in vitro and in vivo osteogenetic capability makes stem cells a promising source of skeletal progenitor cells. 9,10 Stem cells therapy has shown promising results in the treatment of various musculoskeletal pathologies, for example, articular cartilage defects, 11,12 bone defects, 13 meniscal injuries, 14 and degenerative disk disease 15 in numerous studies. Nevertheless, only a few studies investigated the effect of stem cells therapy for the treatment of osteoporosis. 16 –19
Autologous adipose-derived stem cells (ADSCs) have emerged as an attractive alternative to the traditional bone marrow-derived stem cells because of their availability and characteristics (e.g. relatively easy to cultivate, pluripotency, lack of immunogenicity). 18–19 The use of ADSCs has gained popularity over the past decade in bone and cartilage tissue engineering. 20 Nevertheless, further research is required before ADSCs therapy could be implemented for the treatment of osteoporosis in humans. 10,20
We hypothesized that local percutaneous implantation of autologous ADSCs to the proximal femur of osteoporotic rats will induce osteogenesis and enhance bone quality and strength. To evaluate the osteogenic potential of ADSCs implantation, we used micro-computed tomography (CT), mechanical, and histological analyses.
Materials and methods
Experimental animals and procedure
This randomized controlled trial was conducted at Pharmaseed Ltd Laboratories (Ness-Ziona, Israel). We used thirty 5-month-old bilateral ovariectomized female rats of the Sprague-Dawley strain weighing 260–320 g. The animals were kept at 23°C with a 12-h light cycle and free access to standard laboratory food and water. The animals were selected and treated by the specialized laboratory team, who performed the ovariectomy and followed up the animals during the experiment. The ovariectomy was performed using a 3-cm long midline dorsal skin incision halfway between the middle of the back and the base of the tail according to the technique described by Lasota and Danowska-Klonowska. 21 Osteoporosis was confirmed by the presence of extensive trabecular deterioration in high-resolution micro-CT analysis. 22
The animals were randomly assigned to one of the two treatment groups (15 animals per group): (1) percutanous implantation of autologous ADSCs seeded in hyaluronic acid scaffold into the proximal femur metaphysis and (2) percutanous implantation of non-seeded hyaluronic acid scaffold into the proximal femur metaphysis. The contralateral untreated femur served as control in all the cases. The study was conducted in accordance with the Israel Animal Welfare Act (1994) and the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines.
The procedure was carried out under ether anesthesia. Hyaluronic acid-based scaffold (0.2 ml) with or without ADSCs (6 × 105 cells), according to the group assignment, were injected percutaneously into the proximal femur metaphysis of the rats via 18-gauge needle connected to a 1-ml syringe under fluoroscopic control. The animals were euthanized at 12 weeks. The femora were excised and underwent micro-CT scanning, mechanical testing, and histological analysis. The results of the micro-CT scanning and histological analysis were reviewed by a senior radiologist and histopathologist, respectively, who were blinded to the treatment group.
ADSCs cultivation
Autologous ADSCs were isolated from the abdominal fat of the animals. Approximately 2 ml of abdominal fat were aseptically removed from the abdomen of each rat. The samples were washed twice with 10 ml phosphate-buffered saline (PBS), digested with 1.5 mg/ml collagenase, and then filtered through a 250-μm nylon mesh. Red blood cells were removed with erythrocytes lysis buffer. The remaining cells were plated in culture flasks (i.e. F-12/Dulbecoo’s modified Eagle’s medium containing 10% fetal calf serum and 1% antibiotics) at 37°C in a humidified atmosphere and 5% carbon dioxide. The cells were allowed to reach 80% of confluence and were then detached from the culture plate using trypsin–ethylenediaminetetraacetic acid solution, washed with PBS, and plated again. After three passages, the multipotent ADSCs were seeded on 3-D hyaluronic acid-based spongy scaffold with pore size up to 400 μm. 23
Outcome measures
To assess the effect of ADSCs implantation on bone quality and strength, we used the following assessment tools: High-resolution micro-CT analysis of bone morphology:
24
Cortical bone thickness and bone volume to total sample volume ratio (BV/TV) were assessed using high-resolution micro-CT analysis (μCT40, Scanco Medical, Bruttisellen, Switzerland). Since the rats’ femoral cortex has a noncircular profile with uneven wall thickness, we determined the cortical thickness as the mean of six measurements evenly spaced around the cortex on transverse micro-CT views at the level of the diaphysis center. Mechanical testing of bone strength: This was assessed by a static three-point axial compression bending test until bone failure. The femur specimens were mounted to the testing device (Instron-2810, Instron Worldwide, Waltham, Massachusetts, USA) at the upper and lower ends of the bone, while bending force was applied at the center of the specimen with the actuator displaced at 2 mm/min in the posterior–anterior axis until failure. Histological analysis: The specimens were stained with hematoxylin and eosin and were examined under standard light microscope.
Data analysis
Parameters are described as mean ± standard deviation. The means of cortical thickness, BT/TV, and load to failure were compared between the ADSCs-seeded and non-seeded groups for the injected and control sides using unpaired two-tailed t-test. The means of the injected and control sides within each treatment group were further compared using paired two-tailed t-test. Statistical analysis was performed using MedCalc Statistical Software version 17.5.3 (MedCalc Software bvba, Ostend, Belgium). With the α level set at 0.05, we determined prospectively (based on preliminary results) that 12 specimens per group would give 90% power to identify a 10% difference in load to failure, cortical thickness, and BV/TV ratio between the intervention groups.
Results
Comparisons between the ADSCs-seeded and non-seeded groups and between the injected and untreated contralateral (control) femora within each group are summarized in Table 1. The mean cortical thickness and BV/TV in rats’ femora injected with autogenous ADSCs-seeded scaffold was significantly higher compared with rats’ femora injected with non-seeded scaffold (by 11% and 13% respectively; p < 0.01) and compared with untreated control femora (both by 12%; p < 0.001) (Figures 1 and 2). The mean bone load to failure was also significantly higher in femora injected with autogenous ADSCs-seeded scaffold compared with non-seeded scaffold and control femora (by 8% and 9%, respectively; p < 0.01). The mean cortical thickness, BV/TV, and bone load to failure values of femora injected with non-seeded scaffold were similar to those of the untreated control femora. Similar values of all outcome measures were found in untreated (control) femora of both treatment groups. Histological examination of the injected specimens revealed complete osseo-integration of both ADSCs-seeded and non-seeded scaffolds with direct conversion into osteoblasts and no inflammatory response (Figure 3).
Summary of outcome measures.a

a Values are presented as mean ± standard deviation.
b Comparison between the seeded and non-seeded groups for the injected and control sides. All parameters were compared using unpaired two-tailed t-test.
c Comparison between the injected and control sides within each treatment group. All parameters were compared using paired two-tailed t-test.

Three-dimensional reconstructions of the distal femur using high-resolution micro-CT system (μCT40; Scanco Medical, Bruttisellen, Switzerland) with 20 μm voxel size. (a) A femur specimen injected with non-seeded scaffold. (b) A femur specimen injected with autologous ADSCs-seeded scaffold. CT: computed tomography; ADSC: adipose-derived stem cells.

Two-dimensional coronal and transverse sections of femur specimens using high-resolution micro-CT (μCT40; Scanco Medical, Bruttisellen, Switzerland) with 20 μm voxel size. (a) Non-injected (control) femur specimen. (b) A femur specimen injected with non-seeded scaffold. (c) A femur specimen injected with ADSCs-seeded scaffold. CT: computed tomography; ADSC: adipose-derived stem cells.
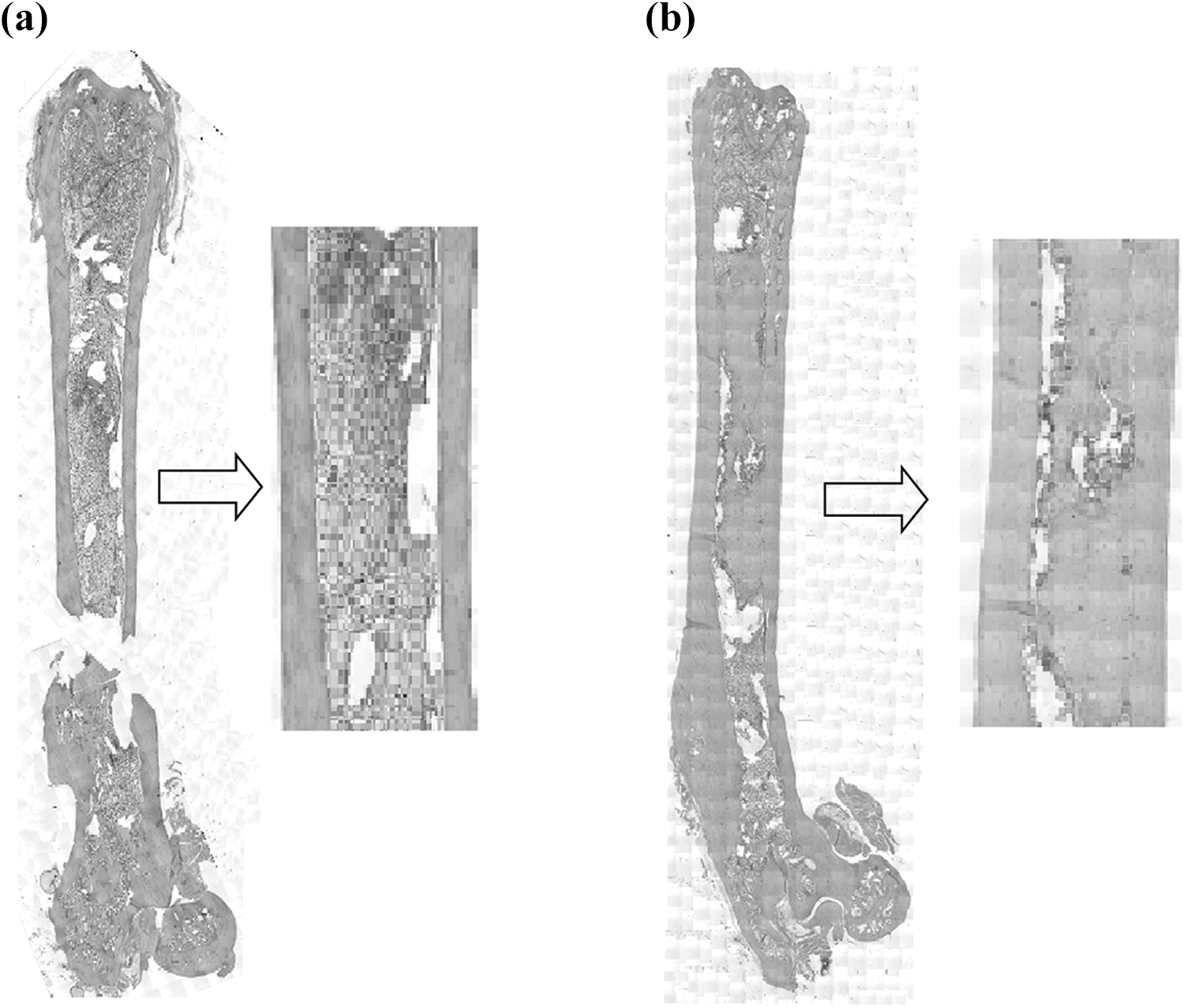
Histological specimens stained with hematoxylin and eosin. (a) A femur specimen injected with non-seeded scaffold showing complete osseo-integration of the scaffold and no inflammatory response. (b) A femur specimen injected with ADSCs-seeded scaffold showing complete osseo-integration of the scaffold with direct conversion of the ADSCs into osteoblasts and no inflammatory response. ADSC: adipose-derived stem cells.
Discussion
Deficient osteogenic capacity of bone marrow stem cells plays a critical role in the pathophysiology of osteoporosis 25 and is inadequately addressed by the common prescribed medication for osteoporosis which focuses mainly on inhibiting bone resorption. 26 The use of multipotent stem cells may enhance bone osteogenic potential and may reduce the risk of undesirable osteoporotic fractures. 16 –19 ADSCs offer many advantages over other sources of mesenchymal stem cells and have emerged as a promising source of skeletal progenitor cells. 18,20 We hypothesized that implantation of autologous ADSCs into the femora of osteoporotic rats will induce osteogenesis and enhance bone quality and strength. This study was designed to investigate this hypothesis.
Our findings support the study hypothesis and show that local implantation of autogenous ADSCs seeded on hyaluronic acid-based scaffold significantly improved bone morphological characteristics (cortical thickness and BT/TV) and increased bone strength (assessed by three-point bending test) compared with implantation of non-seeded scaffold and untreated control femora. Improved bone morphomeric characteristics after local implantation of ADSCs was found in other osteoporotic animal model studies. Mirsaidi et al. 18 administered autologous ADSCs to tibiae of osteoporotic mice and found a significant increase in trabecular bone volume density and trabecular number compared with the untreated tibiae at 6 weeks analysis. Ye et al. 27 investigated the effect of ADSCs implantation into the distal femur of osteoporotic rabbits. Twelve-week postimplantation analysis revealed significantly more bone formation in femora treated with ADSCs compared with control femora. The average bone density and bone thickness in the ADSCs treated femora were approximately 30% higher than in control femora. This increase in bone morphometric parameters seems higher compared with approximately 10% increase found in our study; however, the differences in the animal model and outcome assessment tools preclude comparisons between the studies.
Monitoring the efficacy of antiosteoporotic therapy is essential for successful osteoporosis management. Change in bone mineral density and bone turnover markers are considered as valid indicators of fracture risk reduction in humans. 28 Whether the 10% increase in bone strength found in our animal model would be meaningful clinically in fracture risk reduction and not only statistically significant remains questionable. Furthermore, despite similarities in pathophysiologic responses between human and rat skeleton, which make the rat a valuable model in osteoporosis research, the correlation between increased bone strength in rats treated with ADSCs and fracture prevention in humans should be further studied. 29
Histological examination of the specimens treated with ADSCs-seeded scaffold in our study showed more mineralized bone formation compared with non-seeded scaffold and untreated specimens with complete osseo-integration of the scaffold and direct conversion of the autogenous ADSCs into osteoblasts. No inflammatory response or necrosis was observed. These findings support the reports of previous studies 18,27 and highlight the favorable characteristics of autogenous ADSCs (e.g. osteogenetic capability and low immunogenicity). On the other hand, Pei et al. 30 reported that ADSCs harvested from osteoporotic rats exhibited relatively higher adipogenic potential and lower osteogenic potential compared with ADSCs from non-osteoporotic rats. This may suggest that allogenic ADSCs from a non-osteoporotic donor may offer an advantage in terms of osteogenic potential (over autologous ADSCs) in the treatment of osteoporosis. Nevertheless, the high immunogenicity of allogenic stem cells should be taken into consideration.
Our study has a number of limitations. First, we analyzed the outcome of ADSCs implantation at a short-term follow-up of 12 weeks. Although our post-intervention follow-up is longer compared with 6-week follow-up reported by Mirsaidi et al., 18 the long-lasting effect of the treatment cannot be determined. Second, our osteoporotic animal model consisted of ovariectomized female rats. Whether osteoporosis of other etiologies, especially age-related osteoporosis, would respond similarly to ADSCs therapy should be further studied. Finally, bone strength in this study was assessed only by a static bending test to failure. Performing additional mechanical tests could add to our findings, nevertheless we believe the data presented are of scientific value.
In conclusion, our results show that autogenous ADSCs injected into the proximal femur of rats with ovariectomy-induced osteporosis induced mineralized bone formation and promoted bone regeneration and strength at short-term follow-up. These findings highlight the potential benefit of autogenous ADSCs in the treatment of osteoporosis. The long-term results of ADSCs implantation and its effect on age-related osteoporosis should be further studied.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
