Abstract
Aim:
To determine whether a mechanical, high-frequency vibration device (Tenease™) can improve pain and function for the treatment of tennis elbow (TE), compared with standard treatment.
Methods:
Adults presenting to an elbow clinic with a clinical diagnosis of TE were randomized to standard treatment with physiotherapy, activity modification and analgesia or standard treatment plus Tenease therapy. Tenease therapy consisted of a 6-week period of treatment using the Tenease device with three 10-min episodes each day. The primary outcome measure was the quick Disabilities of the Arm, Shoulder and Hand score at 6 months, with scores also taken at 6 weeks. Secondary outcome measures were the Patient Rated Tennis Elbow Evaluation Score and EuroQol 5-Dimension Visual Analogue Scale at the same time points.
Results:
Fifty-four patients were recruited into the study. Following randomization and initial dropout, 18 patients were included in the standard group and 27 in the Tenease group. Both groups reported improvements in primary outcome measure scores. The control group had a mean score of 44.3 (standard deviation (SD) = 18.8) at baseline, which dropped to 31.2 (SD = 17.2) at 6 months (p = 0.002). The Tenease group had a mean score of 43.2 (SD = 22.7) at baseline, which dropped to 23.4 (SD = 15.0) at 6 months (p = 0.064). Similar improvements were seen in secondary outcome measures with none reaching statistical significance. There were no statistically significant differences seen between the primary outcome scores at 6 weeks (p = 0.9) or 6 months (p = 0.5). No complications were noted in either group.
Conclusions:
Vibration therapy did not result in any statistically significant improvement in functional outcome scores compared to standard treatment for TE. It is important to note that this was a relatively small cohort and a high dropout rate was observed.
Introduction
Tennis elbow (TE) or lateral epicondylopathy is a common condition usually affecting working people with an estimated annual UK incidence of between 2.4 and 4.5 per 1000. 1 It is a significant cause of absenteeism, with the annual costs in 2012 estimated at £27 million in the United Kingdom. 2 The pathogenesis of TE has been hypothesized to involve repetitive microtrauma and degeneration of the tendinous origin of extensor carpi radialis brevis (ECRB) and extensor digitorum communis. 3 This results in a loss of the normal collagen architecture and angiofibroblastic hyperplasia. 4 Various pain factors have been identified in the ECRB tendon, including substance P, tumour necrosis factor and calcitonin gene-related peptide, implying that neurogenic inflammation plays an important role in the condition. 5 –7
A large number of treatment options have been described for TE with varying success rates reported in the literature. Pharmacological treatment in the form of topical or oral non-steroidal anti-inflammatory agents has been shown to have short-term efficacy. 8,9 Physiotherapy, particularly involving progressive eccentric loading of the extensor mechanism, can also be beneficial. 10,11 Injections of corticosteroid, both intralesional and intramuscular, have been routinely used, again with short-term benefits reported. 12,13 However, there is now strong evidence that outcomes following steroid injections are worse in the medium and long terms, with higher recurrence rates seen compared to physiotherapy alone. 14,15 More recently, injections of autologous blood products such as platelet-rich plasma (PRP) have shown promising early results. 16–17 Other techniques such as shock wave therapy, botulinum toxin injections and laser therapy have been trialled with varying success rates. 18 –24 The fact that there is such an array of treatment options described in the literature demonstrates equipoise in the treatment of TE.
The Tenease™ unit is a portable device that vibrates at a high frequency and has been postulated to work through the gate control theory of pain, 25 where afferent nociceptors are modulated by the activation of convergent mechanoreceptors in the dorsal horn of the spinal cord. 25 –27 Various studies have reported the benefits of vibration therapy for acute or chronic musculoskeletal pain, neurogenic pain and osteoarthritis in the hand and knee. 28 –31
A single study has investigated the effects of vibratory stimulation in TE and reported an increase in pain thresholds by 1.2–2.3 times in the affected elbow following treatment. These thresholds returned to normal 45 min after cessation, although the subjective pain relief lasted up to 7 h in 12 of the 18 patients involved. 32
The aim of this study is to determine whether treatment with the Tenease device results in a greater improvement in patient reported outcome scores at 6 months compared with standard treatment alone.
Methods
This study was undertaken with full ethical approval from our local Research Ethics Committee (reference number 11/SW/0190). Patients were recruited between March 2014 and May 2016 from the orthopaedic outpatients department of a university hospital.
Study design and population
Patients were included if they were English speaking, over the age of 18 and had a diagnosis of TE made by a fellowship-trained consultant elbow surgeon. All patients had the following features: – lateral elbow pain, possibly radiating down the forearm; – tenderness within 2.5 cm of the common extensor origin; – pain on resisted extension of the wrist; and – pain on resisted extension of the middle finger.
Patients were excluded if they had a diagnosis of inflammatory arthritis, had a structural abnormality of the affected elbow, had undergone surgery, shock wave therapy or PRP injections for TE or were unable to apply or understand how to use the device correctly.
Study protocol
Patients who were identified as potentially suitable for the trial underwent a screening process with a research nurse and received an information sheet outlining the study, its aims and their right to withdraw at any point. They were shown the Tenease device and allowed to trial it during screening. If they agreed to take part in the study, patients were consented and randomized at a ratio of 1:1 into either of the two treatment groups using the permuted block method. Patient demographics were recorded and data regarding past medical history obtained from the notes and a patient questionnaire.
Treatment groups
Patients randomized into the standard group received an information leaflet and education regarding the self-limiting nature of TE, activities to avoid and exercises to undertake. Simple analgesia was recommended, but steroid injections or other treatments were not allowed unless the patient wished to end their involvement in the study.
Patients randomized into the Tenease group received the same treatment as above plus a Tenease device and education on how to use it. Treatment with the Tenease device was as per the manufacturer’s guidelines, with three continuous periods of use for 10 min recommended each day, for a minimum of 6 weeks unless symptoms had fully resolved before then. Simple analgesia was permitted, but again steroid injections or other treatments were not allowed unless the patient wished cease participation.
Following the initial consultation, patients in both groups were then invited back to clinic and reviewed at 6 weeks and 6 months.
Outcomes
In keeping with the UK National Institute of Clinical Excellence recommendations for health technology appraisal, 33 Patient-Reported Outcome Measures (PROMs) were selected as the study outcomes. The quick Disabilities of the Arm, Shoulder and Hand (qDASH) score was selected as the primary outcome measure. 34 This PROM has been extensively validated in upper limb pathology, 35 has been formally adapted to UK English participants 36 and has a significant history of use in TE studies. 37 In line with the national collection of PROMs material and good practice guidance, a generic health measure was also collected in the form of the EuroQol 5-Dimensions Visual Analogue Score (EQ5-D VAS). This tool has been validated for the UK English population and frequently collected in UK interventional trials. 38 –40 The Patient-Rated Tennis Elbow Evaluation (PRTEE) was also utilized as a secondary outcome measure. This condition-specific PROM has a history of validation in TE patients and is also commonly used in TE research. 41,38 It was not used as the primary outcome measure as it has not been formally validated without alteration in UK English populations and, therefore, falls short of the recommended primary outcome PROM selection criteria. It was selected primarily to serve as a comparator to other previously published studies. 42,43
Statistical analysis was undertaken using STATA Software (StataCorp. 2015. Stata Statistical Software: Release 14. College Station, TX: StataCorp LP). Student’s paired t-test was used to compare pre- and post-intervention data within groups. Data between groups were compared using the unpaired t-test. Data are reported as mean (standard deviation). Significance was set at p < 0.05. Analysis was undertaken on an intention-to-treat basis.
Results
A total of 54 patients were recruited into the study. The flow through the study is shown in Figure 1.
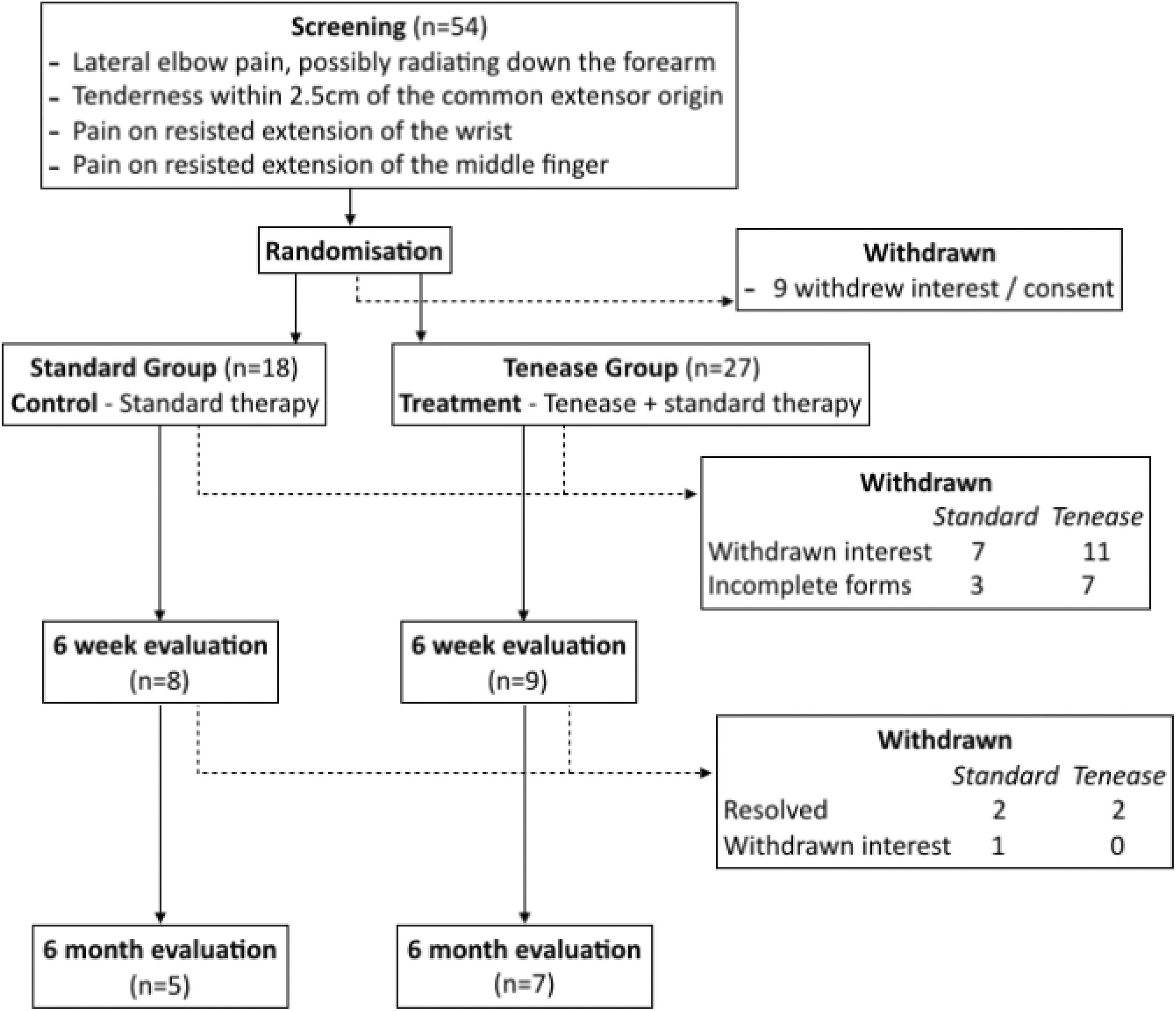
CONSORT study flow diagram.
Patients in the standard group had a mean age of 54.0 (range 37.8–66.3; standard deviation (SD) = 6.1), and there were six men. The mean age of patients in the Tenease group was 54.4 (range 37.9–77.2 (SD = 11.0); p = 0.92), and there were four men. There were improvements in qDASH scores from baseline to 6 months in both groups, although this improvement was statistically significant only in the standard group (range 44.3 (SD = 18.8) to 31.2 (SD = 17.2); p = 0.003 vs. range 43.2 (SD = 22.7) to 23.6 (SD = 15); p = 0.064). Similar improvements were noted in both groups for EQ5-D VAS and PRTEE scores, although these again failed to reach statistical significance. There were also no statistically significant differences seen when the individual scores for each outcome measure were compared at each time point.
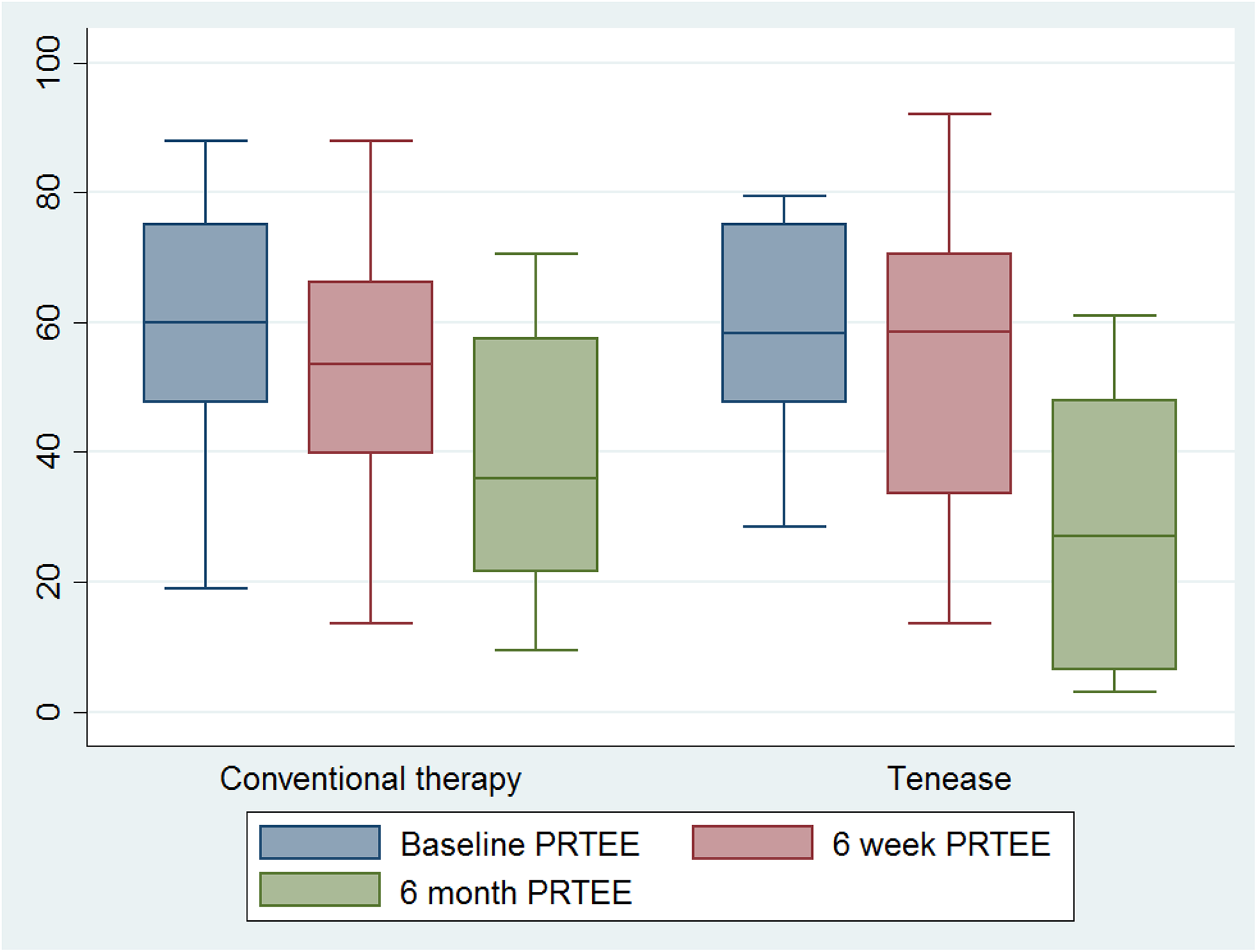
The outcome measures are presented in Table 1 and Figures 2 to 4. There were no complications or adverse reactions recorded in either group.
Outcome scores for standard and Tenease™ groups at baseline, 6 weeks and 6 months.

SD: standard deviation; EQ5-D VAS: EuroQol 5-Dimensions Visual Analogue Score; qDASH: quick Disabilities of the Arm, Shoulder and Hand; PRTEE: Patient-Rated Tennis Elbow Evaluation.

Box plot of qDASH scores comparing standard and Tenease™ therapy at baseline, 6 weeks and 6 months. qDASH: quick Disabilities of the Arm, Shoulder and Hand.
Box plot of EQ5-D VAS scores comparing standard and Tenease™ therapy at baseline, 6 weeks and 6 months. EQ5-D VAS: EuroQol 5-Dimensions Visual Analogue Score.
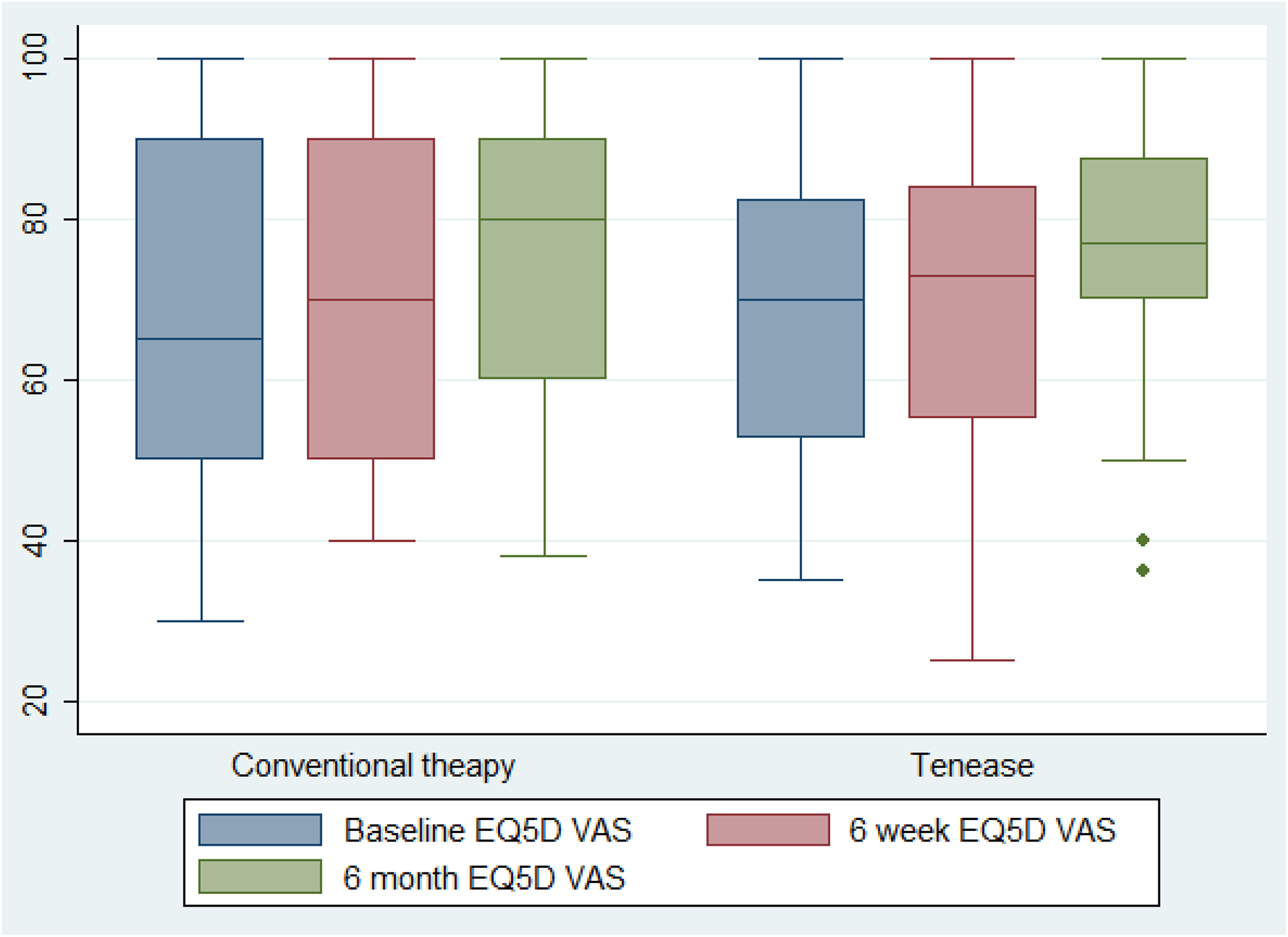
Box plot of EQ5-D VAS scores comparing standard and Tenease™ therapy at baseline, 6 weeks and 6 months. EQ5-D VAS: EuroQol 5-Dimensions Visual Analogue Score.
Discussion
This represents the first randomized controlled study comparing vibration therapy with standard treatment for TE. It was hypothesized that vibration therapy would result in a greater improvement in functional outcome scores than standard treatment alone, but this was not born out from these results.
This study reports a statistically significant improvement in qDASH scores from baseline to 6 months in the standard treatment group only. Owing to the small sample size, no inference has been made on either the level of smallest detectable change beyond measurement error or whether this change could be categorized as clinically meaningful.
A statistical difference was not found between the EQ5-D or PRTEE results. Although the lack of responsiveness in the general health measure may be expected, the condition-specific score (which one might expect to mirror the qDASH) was not found to have changed. An assessment of comparative responsiveness between the PRTEE and qDASH has not been formally conducted; however, against the non-abbreviated DASH score, it has been found to be equivalent. 44 It may be that the high dropout rate and consequential low numbers have resulted in a large score spread (seen in the high standard deviation) and, therefore, an inability of this study to truly measure a significant difference. However, the lack of statistical difference between the baseline and 6 months scores when comparing groups suggests that neither treatment arm was superior.
In this study, 18 patients had withdrawn from the Tenease group by the 6-week follow-up. Many patients volunteered that the device was either uncomfortable to use, occasionally painful or simply ineffective at treating their symptoms. A previous study using vibratory therapy for 37 patients with lateral or medial epicondylitis also showed a high withdrawal rate, possibly in relation to the physical effect of the treatment. 29
A significant limitation in this study is the large dropout rate, with nine patients withdrawing immediately after being randomized to the standard group. The resultant small number prohibits a definitive statement on this device, with a significant risk of both type I and II error present. Patients reported that they were disappointed that they had not been randomized to the Tenease treatment and felt they had effectively been randomized to no intervention, as many had already undertaken physiotherapy prior to their consultation in the orthopaedic outpatient department.
The high rate of withdrawal from studies comparing treatment versus no treatment is well recognized. In a recent national multicentre randomized trial, 427 of 740 patients assessed to be eligible for inclusion withdrew following the initial consultation and prior to randomization. Furthermore, only 78 from 104 patients randomized to the no-treatment arm in the study actually received no treatment. 45
Conclusions
In this study, no evidence was found that vibration therapy offers any benefit over a standard management protocol in the treatment of TE, with only the standard treatment group showing an improvement at 6 months in the primary outcome measure. There were no complications in either study arm, suggesting that the Tenease device is safe to use.
A high dropout rate was noted in both groups and patients randomized to the Tenease group voiced negative opinions regarding the comfort and efficacy of the device. This dissatisfaction combined with a lack of a statistically significant difference should be taken into consideration when offering this treatment to patients and designing future studies into the treatment of TE.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
