Abstract
Purpose:
Various animal models have been proposed to mimic the pathophysiologic process of intervertebral disc degeneration, a leading cause of back pain. The purpose of this study is to describe a minimally invasive technique via percutaneous needle puncture of the annulus fibrosus in New Zealand white rabbits.
Methods:
Under fluoroscopic guidance, an 18-gauge spinal needle was inserted 2 cm lateral to the midline spinous process. The needle was slowly advanced at approximately 45° angle until it was adjacent to the L5/L6 disc space. Lateral and anteroposterior views were used to verify correct needle position before advancing into the nucleus pulposus. The rabbits underwent weekly X-rays for 4 weeks to assess disc height index. MRI T2 relaxation was evaluated at week four to assess morphological changes. Discs were histologically graded on a 12-point scale to assess degeneration and compared to discs obtained from uninjured rabbits.
Results:
There were no complications associated with the percutaneous needle puncture procedure. All animals survived the duration of the experiment. Four weeks after injury, the disc height had progressively narrowed to approximately 50% of baseline. MRI assessment at the 4-week time point demonstrated a mean T2 relaxation time at the L5/L6 level that was 20.9% of the T2 relaxation time at the uninjured L4/L5 disc level (p < 0.001). Histological analysis demonstrated lamellar disorganization of the annulus and decreased cellularity and proteoglycan content within the injured nucleus compared to uninjured control discs.
Conclusion:
The present study demonstrated a reliable technique of inducing an annular tear via a percutaneous needle puncture. Compared to open surgical approaches, the percutaneous model produces similar progressive disc degeneration while minimizing harm to the animal subjects.
Clinical Relevance:
The present study establishes a technique for the introduction of novel therapeutic agents to treat disc degeneration that may translate to future clinical trials.
Introduction
Intervertebral disc (IVD) degeneration is one of the leading causes of back pain, accounting for approximately 20% of all cases, 1 and is often associated with other common etiologies of back pain, including spinal stenosis, disc herniation, segment instability, and radiculopathy. 2 Both conservative and surgical treatment strategies are directed at relieving the symptoms of disc disease rather than preserving disc integrity. Recently, the paradigm in the research emphasis of disc degeneration has shifted to understanding and reversing the degenerative process. 3 –9
The disc is composed of three parts: an inner gel-like nucleus pulposus (NP), a peripheral ring of cartilage called the annulus fibrosus (AF), and cartilaginous endplates. The extracellular matrix of the NP contains hydrated proteoglycan gel in a loose network of type-II collagen. 10 –12 The outer AF is composed of primarily type-I collagen and therefore provides structural support and withstands the tensile strain of the spine. 3,13 Degeneration is characterized by a progressive loss of NP cells and degradation of the surrounding extracellular matrix content, of which the most noteworthy is the reduction of proteoglycans. 14 –16 A previous longitudinal study suggested an association between tears of the AF and subsequent disc degeneration. 17
Various animal models have been proposed to mimic the pathophysiologic process of disc degeneration in humans, from instrumented compression 18,19 or torsion 20 to surgical removal of the facets and transverse processes, 21 and direct stab injury to the disc using a scalpel. 22 Masuda et al. 23 noted the inconsistency of injury and irreproducibility of these models and described a technique using needle puncture through the annulus via an open retroperitoneal approach to the lumbar disc spaces. Although their technique introduced a reproducible progressive degeneration, the large surgical incision may present substantial risk and morbidity to the recovery of the animal subjects. The purpose of this study was to describe a minimally invasive technique via percutaneous needle puncture of the AF in a rabbit model of IVD degeneration. We hypothesized that this technique will effectively produce progressive disc degeneration with minimal harm to the animal subjects.
Materials and methods
Subjects and surgical technique
All aspects of the study were approved by our Institutional Animal Care and Use Committee. Fourteen New Zealand white rabbits weighing approximately 3–3.5 kg were used. Under isoflurane anesthesia, the rabbit was shaved in the lumbar back area and prepared using betadine swabs. The rabbit was then placed in a prone and slightly lateral position with a bump in the midsection to widen the disc space of interest. Initial lateral images were obtained on a mini c-arm to locate the L5/L6 disc level. Rabbits have seven lumbar spine segments compared to five in humans. 24 The correct disc level was ascertained by locating the sacrum under fluoroscopic guidance and counting two segments cephalad. This area can be palpated approximately 3 cm cephalad from the iliac crest. Under fluoroscopic guidance using lateral views, an 18-gauge spinal needle was inserted 2 cm to the right of the midline spinous process (Figure 1(a)). The needle was slowly advanced at approximately 45° angle until it was adjacent to the L5/L6 disc space (Figure 1(b)). The mini c-arm was rotated to obtain anteroposterior views to verify correct needle position before the needle was advanced through the AF into the NP (Figure 1(c)). A syringe was then attached to the needle, and 10 mL of negative pressure was withdrawn to injure the disc. No closing sutures were required. The rabbits were carefully observed during recovery from anesthesia. They were housed singly or in pairs and were allowed food, water, and activity ad libitum. After surgery, the rabbits received cefazolin (30–35 mg/kg intramuscularly) for perioperative antibiotic and buprenorphine (0.01–0.05 mg/kg subcutaneously every 12 h for 3 days) for analgesia.

(a) Under isoflurane anesthesia, the rabbit was shaved in the lumbar back area and prepared using betadine swabs. The rabbit was then placed in a prone and slightly lateral position with a bump in the midsection to widen the disc space of interest. Under fluoroscopic guidance using lateral views, an 18-gauge spinal needle was inserted 2 cm to the right of the midline spinous process. (b) Using lateral views of the lumbar spine, the 18-gauge spinal needle was slowly advanced at approximately 45° angle until it was adjacent to the L5/L6 disc space. The tip of the spinal needle may be bent to facilitate needle passage past the transverse process, which protrudes in a cranial and ventral direction in the rabbit. (c) To ascertain the correct needle position, the fluoroscopic imaging must be switched from lateral to anteroposterior view before advancing the needle through the annulus fibrosus into the center of the nucleus pulposus.
Radiographic evaluation
Under isoflurane anesthesia, X-rays were obtained before surgery and then at weekly intervals for 4 weeks to compare the disc height index (DHI), calculated as a ratio of the injured disc height to the sum of the height of the two adjacent vertebral bodies, as previously described by Masuda et al. (Figure 2). 23 The % DHI is therefore represented as DHI normalized to the baseline preoperative measurement (postoperative DHI/baseline DHI × 100).

The DHI is calculated as a ratio of the injured disc height to the sum of the height of the two adjacent vertebral bodies. DHI = 2 × (A + B + C)/(D + E + F + G + H + I). DHI: disc height index.
In vivo MRI of the lumbar spine was performed at 4 weeks after disc puncture using a 3T MRI system (MAGNETOM Skyra, Siemens Healthcare, Erlangen, Germany), which was connected to inhalation anesthesia apparatus outside the MRI suite. Sagittal T2-weighted images were obtained for analysis. The T2 relaxation time of the IVDs was quantified using ImageJ processing software (Version 1.50b, National Institutes of Health, USA) to quantify the amount of collagen degradation and hydration status, which was measured within the injured (L5/L6) and adjacent uninjured discs (L4/L5). Starting with the midsagittal section, an elliptical voxel was placed in the center of the disc. The size of the voxel was modified based on the disc size and specified for each image series to minimize contamination from adjacent tissues. 25 The voxel intensity of the midsagittal section and the three image slices to the left and right of the midsagittal section were measured. The intensity measurements of these seven sections were averaged for analysis and repeated for adjacent uninjured disc levels.
Histologic evaluation
The animal subjects were euthanized at 12 weeks after needle puncture. Two healthy rabbits of similar age and weight were obtained from another study to harvest tissue to serve as control. The lumbar spine was excised en bloc for histologic analysis. The spine tissue was first fixed in 10% buffered formalin, then decalcified in Immunocal (StatLab Medical Products, McKinney, Texas, USA). Discs were paraffin-embedded and sectioned at a thickness of 5 µm. Sections were stained with hematoxylin and eosin and imaged using a Hamamatsu NanoZoomer 2.0-HT digital slide scanner (Hamamatsu Photonics, Hamamatsu, Japan). Histology was scored using a previously described grading scale based on four categories of degenerative changes: AF, border between AF and NP, cellularity of the NP, and matrix of the NP (three points in each category, minimum four points, and maximum 12 points). 23 Normal pattern is denoted by one point, while severe disruption, hypocellularity, or degeneration is denoted by three points. The histology samples were randomized and scored by a blinded investigator (ZKZ).
Statistical analysis
Weekly changes in DHI were analyzed using repeated measures analysis of variance (ANOVA) with Bonferroni post hoc test. Differences in T2 relaxation between the injured and uninjured discs were compared using a paired t-test. All data were reported in means ± SE. Statistical analyses were performed using SPSS 20 software (IBM, Armonk, New York, USA). Significance level was set at p < 0.05. A priori power analysis for two-tailed paired t-test for comparing two matched pairs with the following parameters: power of 0.8 and α ≤ 0.05. Effect size was determined based on MRI outcomes at week 4 from our initial pilot study of four rabbits, which yielded an average normalized T2 relaxation time of 0.2. It was determined that a clinically significant improvement would be indicated by a large effect size of 0.9. Using these values, a sample size of 12 subjects was sufficient to achieve an actual power of 0.81. 26
Results
There were no complications associated with the percutaneous needle puncture procedure. All animals resumed normal diet and activities after the procedure and survived the duration of the experiment. There were no prolonged bleeding or infections noted at the needle insertion site. During the entire experimental period, the rabbits were healthy, gained weight, and did not demonstrate behavior associated with pain.
Radiographic evaluation of disc height
Narrowing of the IVD space was observed beginning 1 week after the needle puncture procedure. Mean DHI was 0.079 ± 0.003 at baseline, which decreased to 0.054 ± 0.003 at 1 week, equating to a % DHI of 68%. Repeated measures ANOVA demonstrated a significant decrease over time between baseline and the 4-week time point (p < 0.001, Figure 3). At the 2-, 3-, and 4-week time points, % DHI were 62%, 50%, and 49%, respectively. Post hoc analysis demonstrated a statistically significant difference in DHI between baseline and each of the subsequent weekly measurements.
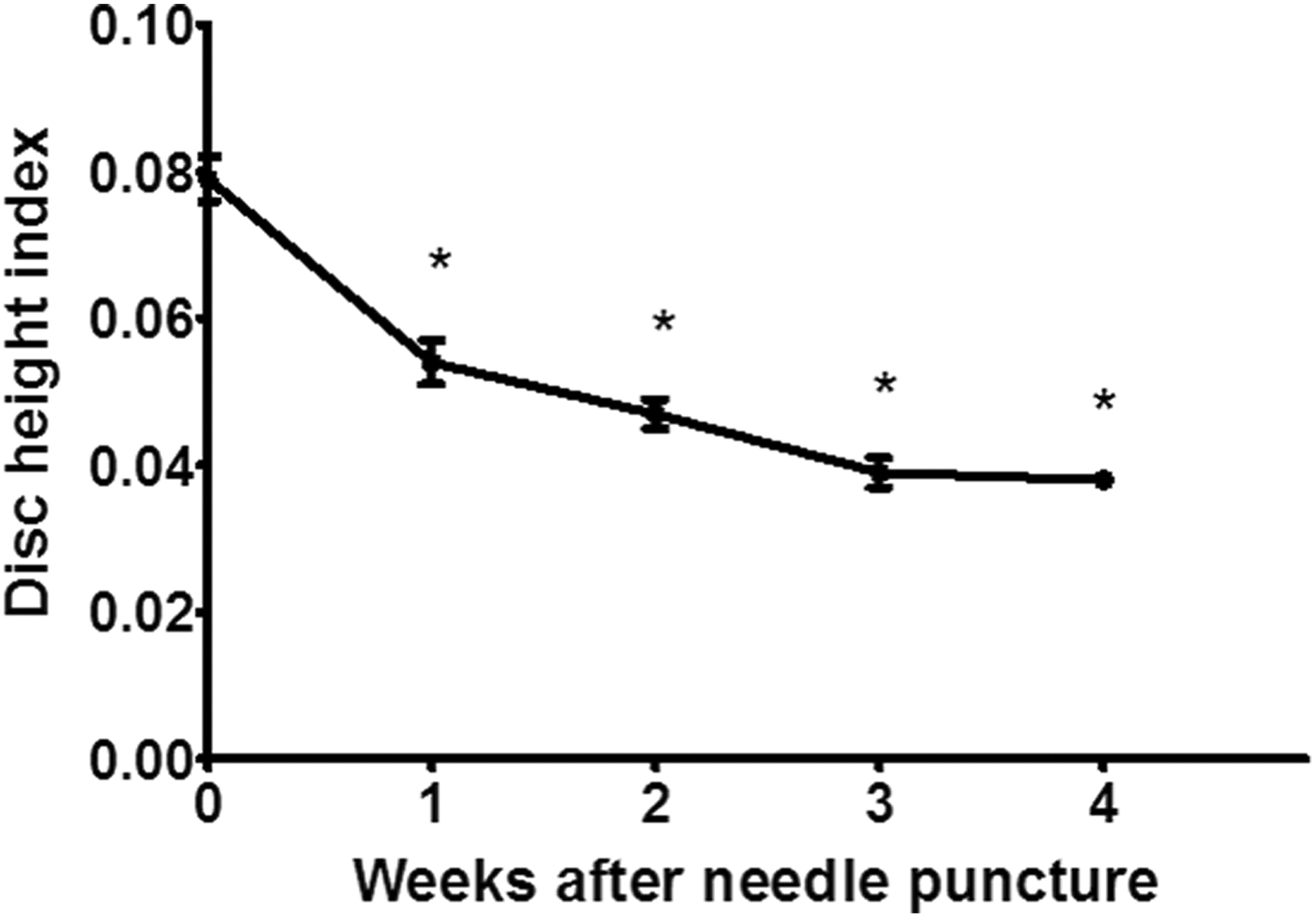
DHI progressively decreased over time between baseline and the 4-week time point (p < 0.001). % DHI post hoc analysis demonstrated a statistically significant difference in DHI between baseline and each of the subsequent weekly measurements (*p < 0.001), which decreased to 0.054 ± 0.003 at 1 week, equating to 68% of baseline DHI. DHI: disc height index.
MRI evaluation of disc degeneration
MRI obtained at the 4-week time point demonstrated greater signal changes within the L5/L6 IVD compared to the adjacent uninjured disc levels (Figure 4(a) and (b)). Signal intensity within the disc, as quantified using ImageJ software, demonstrated a mean T2 relaxation time at the L5/L6 level that was 20.9% of the T2 relaxation time at the uninjured L4/L5 disc level (Figure 5, p < 0.001).

(a) and (b) Signal intensity changes were easily discernible within the injured L5/L6 intervertebral disc (red arrow) compared to the adjacent uninjured disc levels (white arrows: L4/L5 and L6/L7).

Quantification of the MRI signal intensity demonstrated a significantly reduced T2 relaxation time at the L5/L6 level in comparison with the uninjured L4/L5 disc level (*p < 0.001).
Histological grading
The sagittal histologic sections depicted the cartilaginous endplates, adjacent vertebral bone, the AF, and the NP of the discs. Comparison of the sections between the injured (n = 4, Figure 6(a) and (b)) and uninjured (n = 2, Figure 6(c) and (d)), discs clearly revealed greater disorganization of the AF lamellae and loss of the gel-like nature of the NP in the injured, degenerated discs. Hematoxylin and eosin (H&E) staining of the degenerated disc demonstrated lamellar disorganization, with a wavy appearance and ruptured pattern fibers of the AF (Figure 6(e)). This contrasted with the well-organized, intact AF with concentric lamellae in uninjured, healthy discs (Figure 6(f)). Degenerated discs demonstrated a loss of a distinct border between the AF and NP (Figure 6(g)) in contrast to uninjured discs (Figure 6(h)). Within the NP of degenerated discs, there was loss of vacuolated cells on H&E staining (Figure 6(i)). The uninjured NP demonstrated numerous large, vacuolated cells within normal extracellular matrix (Figure 6(j)). On Toluidine blue (TB)-staining, degenerated discs showed increased open cavity with severe loss of proteoglycan content (Figure 6(k)). TB-stained sections of uninjured discs showed minimal disruption of the proteoglycan gel within the NP (Figure 6(l)). Mean histologic scores for the injured and uninjured discs were 10.8 ± 1.5 and 5.5 ± 0.7, respectively, with the higher score signifying greater degeneration.
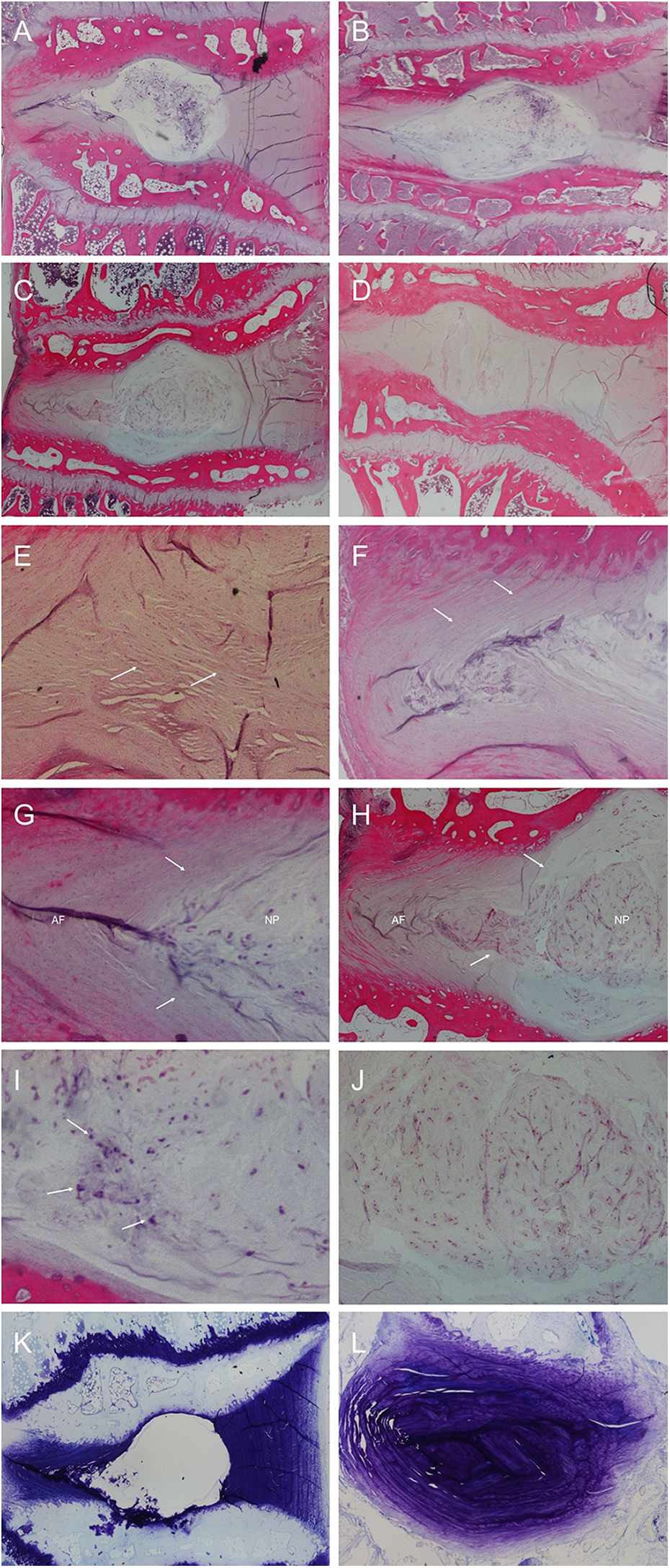
Histologic comparison of the injured, degenerated disc (a) and (b) and the uninjured, healthy disc (c) and (d) on H&E stain. The degenerated disc demonstrated high-grade changes, including lamellar disorganization of the AF (e), loss of a distinct border between the AF and NP (g), and loss of normal vacuolated cells within the NP (i). In contrast, the normal, healthy discs demonstrated concentric, organized lamellae in the AF (f), clear borders between the AF and NP (h), and the presence of numerous large, vacuolated cells within normal extracellular matrix within the nucleus (j). On Toluidine blue (TB) staining, degenerated discs showed increased open cavity with severe loss of proteoglycan content (k) compared to minimal disruption of the proteoglycan gel within the NP of healthy discs (l). AF: annulus fibrosus; NP: nucleus pulposus.
Discussion
The percutaneous needle puncture technique described herein resulted in a progressive narrowing of the lumbar IVD space over 4 weeks. Greater than 50% loss of the disc height was evident by week 4. The signal intensity changes on MRI indicated the loss of water and proteoglycan content within the NP as part of the degenerative process. High grade histological changes within the NP further supported the functionality and utility of the needle-puncture, annular-tear technique to reproduce intervertebral disc degeneration (IDD) in a rabbit model.
Masuda and colleagues 23 introduced the needle puncture technique via a posterolateral retroperitoneal approach, which induced an injury more representative of the slow, progressive disc disease process compared to the previous stab model using a scalpel blade. In their comparison study, the stab technique resulted in significant decrease in DHI as early as 2 weeks after injury. The authors further delineated the effect of needle size (16-, 18-, and 21-gauge) on the reliability and reproducibility of disc degeneration and observed slower progressive narrowing of disc height with the 16- and 18-gauge needles.
Thus, in the present study, 18-gauge spinal needles were chosen for its reliability in the rabbit model. Instead using an open surgical approach, however, we demonstrated the feasibility of a percutaneous approach that offers a minimally invasive method of inducing a consistent annular tear. The resulting changes in DHI followed a linear pattern of regression (Figure 3) similar to the needle puncture results of Masuda et al. 23 The use of fluoroscopic guidance allowed for the accurate placement of the spinal needle, which obviated the need for surgically exposing the spine of the rabbits. Although the percutaneous technique requires an equally high level of technical skill to precisely pinpoint the disc of interest in a three-dimensional space, there may be substantial savings in surgical time and supply cost compared to an open procedure. After induction of anesthesia and preparation of the lumbar skin surface, each percutaneous procedure took approximately 10 min, with the majority of time spent on positioning the needle adjacent to the disc space on the lateral fluoroscopic view. As Masuda et al. 23 noted in their study, caution must be taken to avoid irritation of the surrounding bony and ligamentous structures to prevent vertebral osteophyte formation. The tip of the spinal needle may be bent to facilitate needle passage past the transverse process, which protrudes in a cranial and ventral direction in the rabbit. Another anatomical consideration in this rabbit model is the seven lumbar vertebrae 24 compared to the five lumbar segments in humans. Recognition of the correct disc level can be achieved by locating the sacral spine at the sacroiliac junction of the hip and counting the lumbar vertebral bodies up the spine.
In the percutaneous technique described by Kim and colleagues, 1 the authors noted that the L5/L6 and L6/L7 disc spaces can only be accessed through an open anterior approach, while the more cephalad discs can be percutaneously injured. In our experience, the elongated height of the vertebral bodies in the rabbit results in the L5/L6 disc level situated cephalad to the iliac crest and safely allows the passage of a needle into all disc spaces from L5/L6 and above. While Kim et al. 1 subjectively reported the MRI signal changes from disc degeneration, we quantified an approximate fivefold decrease in T2 relaxation time compared to uninjured discs. The MRI results were corroborated by our histological grading of the injured tissues. MRI outcomes have been previously shown to correlate with total histological score and histological grading of NP characteristics (cellularity, matrix, and border with AF). 23
Various animal models of degenerative disc disease have been developed, beginning with the disc stab model first introduced in 1948. 22 Rapid degeneration from this model served as the standard and was ideal for investigating factors associated with disc regeneration. Conversely, a progressive injury pattern is necessary to study the effects of therapeutic interventions. Animals ranging in size from mice 27 and rats 28 to sheep, 29 pigs, 30 and cows 31 have been used. Although quadruped animal models have been criticized for their translational validity to bipedal human spines, biomechanical studies have demonstrated that quadruped spines are loaded primarily in axial compression, similar to humans. 32,33 Strong bending and torsional moments experienced by the quadruped spine are counterbalanced by paraspinal muscles and ligaments exerting considerable tensile forces along the long axis. 32 Smaller quadrupeds make the procedure more technically challenging, while larger quadrupeds are costly and more difficult to obtain given the ethical and practical considerations, making rabbits an ideal animal model for IDD research. Drawbacks of the rabbit model include the persistence of notochordal cells, 34 which produce hyaluronan, affect proteoglycan metabolism, and ultimately respond differently compared to cells in the adult human NP. 35,36 Most other animal models commonly used in IDD research (mouse, rat, cat, dog, and pig) share this limitation, and all retain notochordal cells in the NP into adulthood. 36 Anatomically, rabbits’ discs have a high degree of homology to human discs due to the presence of facet joints, paraspinous muscles, and ligaments. 37
In addition to animal selection, the ideal model should reproducibly mimic the pathophysiology of IDD. The ideal animal to accomplish this remains up for debate, as most animal models fail to completely recreate the inciting factors documented in human observational studies. 38 The rabbit model, however, demonstrates MRI outcomes similar to that of IDD in human subjects. 39 Masuda et al. 23 laid the groundwork for a progressive disc degeneration model to allow for future studies of therapeutic interventions. We sought to modify their technique by further minimizing the morbidity and mortality to the subject. Open surgeries via paraspinal, 40 retroperitoneal, 23 and anterior transabdominal approaches 41 have been described but may require greater time, surgical supplies, and therefore cost compared to the percutaneous technique. The large incisions and dissections involved in the open approaches may further place the subject at increased risk for infection, elevated postoperative pain, and stress; however, given the nature of our study, we were unable to directly compare these outcomes between the percutaneous and open techniques. Furthermore, in studies in which a second surgery is required for introduction of a novel therapeutic agent, the index surgery may result in excessive adhesions, especially via the transabdominal approach, making subsequent surgeries more difficult.
The present study is limited by the small number of rabbits available for histologic analysis. This was part of a larger study to evaluate needle injection of a therapeutic treatment 4 weeks after initial injury. As such, all subjects were evaluated at 4 weeks, which allowed for an adequate amount of time to demonstrate injury progression. Radiographic parameters were obtained in all subjects. Although the assessment of DHI and histology was inherently subjective, we minimized bias by blinding the observer for histologic analysis. Our results were strengthened by objective quantification of the MRI signal differences between the injured and uninjured discs, which had not been previously performed after percutaneous annular puncture.
Conclusion
We have demonstrated a feasible and reliable technique of inducing an annular tear via a percutaneous needle puncture. The model simulates disc herniation and the ensuing progressive degeneration similar to previous more invasive surgical techniques while minimizing harm to the animal subjects. More importantly, the model further establishes a technique for the introduction of novel therapeutic agents to treat degeneration and optimizes the subsequent treatment surgery free from potential adhesions after an open surgery.
Footnotes
Authors’ note
The study was performed at the Wake Forest School of Medicine.
Acknowledgements
The authors thank Ms Eileen Elsner, Ms Debra Fuller, Ms Jiaozhong Cai, Ms Rebecca Sharp, and Ms Brandi Bickford for their contributions to this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by a research grant from NuTech Medical, Inc. (Birmingham, AL).
